Abstract
An understanding of the processes that occur during development of the intervertebral disk can help inform therapeutic strategies for discogenic pain. This article reviews the literature to identify candidates that are found in or derived from the notochord or notochordal cells and evaluates the theory that such factors could be isolated and used as biologics to target the structural disruption, inflammation, and neurovascular ingrowth often associated with discogenic back pain. A systematic review using PubMed was performed with a primary search using keywords “(notochordal OR notochord) And (nerves OR blood vessels OR SHH OR chondroitin sulfate OR notch OR CTGF) NOT chordoma.” Secondary searches involved keywords associated with the intervertebral disk and pain. Several potential therapeutic candidates from the notochord and their possible targets were identified. Studies are needed to further identify candidates, explore mechanisms for effect, and to validate the theory that these candidates can promote structural restoration and limit or inhibit neurovascular ingrowth using in vivo studies.
Keywords
Concepts and factors important during development of the spine may be useful as therapeutic agents to address intervertebral disk (IVD) degeneration (IDD). The notochord patterns all regions of the IVD during development secreting several ligands (e.g., sonic hedgehog [SHH], connective tissue growth factor [CTGF], chondroitin sulfate [CS], Noggin, Chordin, and Semaphorin 3A) with the capacity to influence cell differentiation, inflammation, angiogenesis, and axonal growth. 1 , 2 , 3 , 4 A shift in nucleus pulposus (NP) cellular phenotype from predominantly notochordal cells (NCs) to predominantly small nucleus pulposus cells (SNPCs) is associated with maturation, and the loss of NCs has been speculated to initiate the onset of IDD in humans. 5 , 6 NCs secrete soluble factors that are essential for patterning and maintaining the healthy avascular and aneural IVD; hence loss of such factors during growth and aging may contribute to disease progression. Seminal work by Aguiar et al demonstrated increased proteoglycan synthesis of NP cells when cocultured with NCs, as a first demonstration of the therapeutic potential of NC-derived factors, and this was followed by studies examining NC-derived factors on mature human NP and mesenchymal stem cells (MSCs) in vitro. 7 , 8 , 9 NCs also influence annulus fibrosus (AF) cells and may have potential to suppress the expression of proinflammatory cytokines. 10 Therefore, NCs may secrete “candidate” factors that could be harnessed for therapeutic effect to limit or reduce the symptom-causing pathologies of IDD. Structural disruption of the IVD is commonly reported morphologically and with imaging. 11 , 12 Ingrowth of nociceptive nerve fibers deep within the IVD has also been reported in the painful degenerate human IVD and has been suggested to be one of the sources of discogenic pain. 13 , 14 “Targets” for these factors therefore include structural disruption, inflammation, and neurovascular ingrowth, which may all interact at different stages of degeneration.
The purpose of this systematic literature review is to identify possible candidates and explore the hypothesis that candidates found in or derived from the notochord or NCs can address targets in IDD. We also highlight the concept of a cocktail treatment that focuses on both structural restoration and also symptoms associated with discogenic back pain and a need for screening in in vivo animal models for validation of such candidates.
Methodology for Literature Search
PubMed was used as the principal database with a primary search of “(notochordal OR notochord) And (nerves OR blood vessels OR SHH OR chondroitin sulfate OR notch OR CTGF) NOT chordoma.” Keywords for this systematic search were chosen based on the main theme of the study, which includes the notochord- and NC-derived therapeutic candidates and pathways (e.g., SHH, CS, CTGF, and Notch) and their potential targets in IDD associated with discogenic pain (nerves and blood vessels; Fig. 1). We define “therapeutic candidates” as factors from NCs that are essential in development and homeostasis of the immature IVD with proanabolic/proliferative, anti-inflammatory, or antineurovascular properties. We define “targets” as aspects of IDD that require structural regeneration or a reduction in symptoms associated with pain. A total of 390 articles were identified and of those 389 met the inclusion criterion of being in the English language. The titles and contents of the publications were then reviewed based on relevance to: (1) notochord and NCs, (2) development of the IVD and notochordal signaling pathways, (3) therapeutic candidates, and (4) targets of IDD (nerves, blood vessels, pain, and catabolism/anabolism). We identified 37 key publications of interest from the primary search (Appendix; Table 1). To ensure key areas were not excluded from the review, secondary searches were performed, which yielded 49 additional publications within the above inclusion criteria. Human and animal, in vivo, ex vivo, and in vitro studies were all included. Studies on chordoma or unrelated to the IVD were excluded from the review.
Notochordal therapeutic candidates and targets associated with IDD
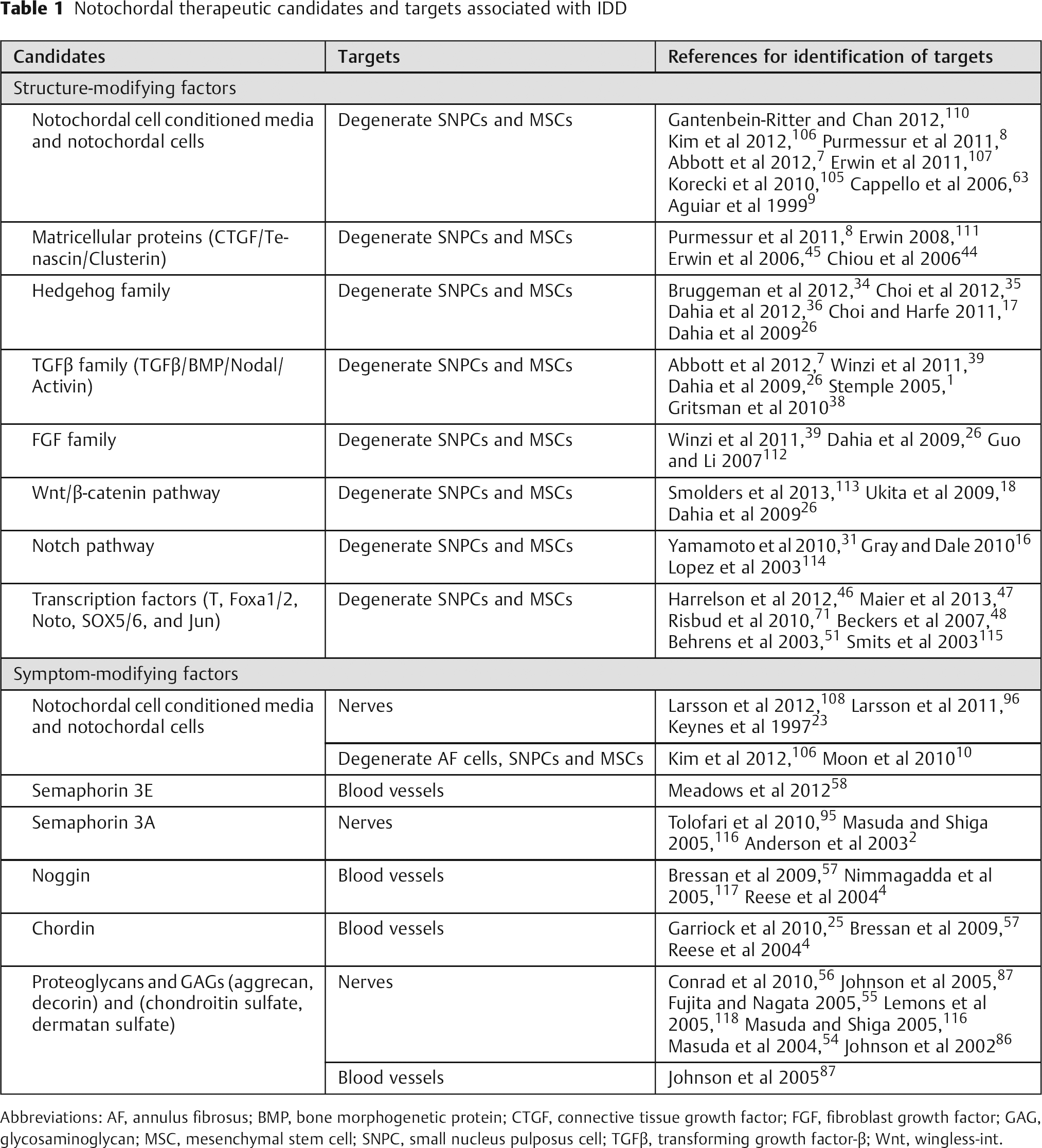
Abbreviations: AF, annulus fibrosus; BMP, bone morphogenetic protein; CTGF, connective tissue growth factor; FGF, fibroblast growth factor; GAG, glycosaminoglycan; MSC, mesenchymal stem cell; SNPC, small nucleus pulposus cell; TGFβ, transforming growth factor-β; Wnt, wingless-int.
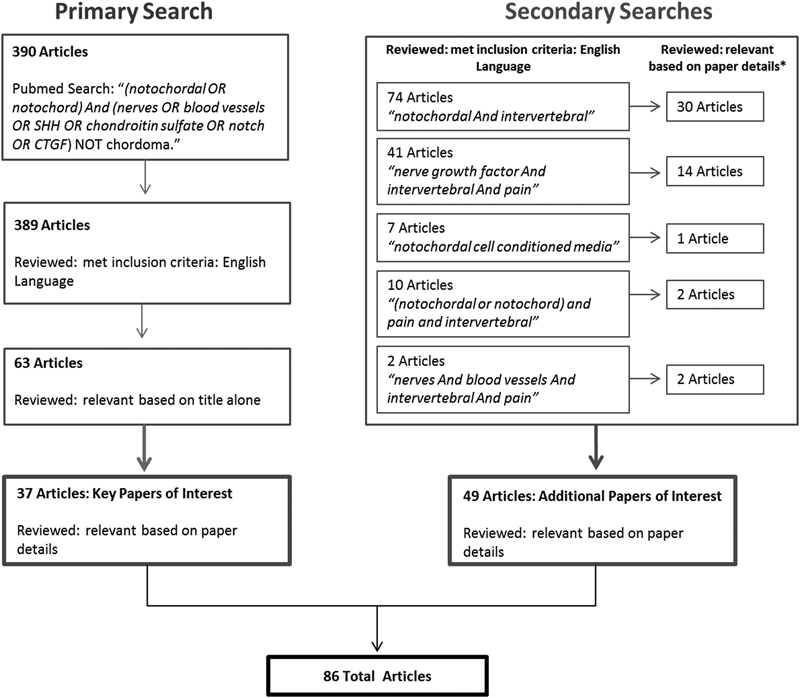
Schematic of primary and secondary systematic searches used to identify key publications with PubMed as the principal search engine. A primary search of “(notochordal OR notochord) And (nerves OR blood vessels OR SHH OR chondroitin sulfate OR notch OR CTGF) NOT chordoma” identified 390 articles. Of these publications, 37 were considered relevant to the review and were written in the English language. Secondary searches were performed to ensure key areas were not excluded, yielding 49 addition publications.
The Notochord: A Multicue Signaling Center That Patterns the IVD
The notochord is a rodlike midline structure of mesodermal origin that rises from the embryonic dorsal organizer. 1 The function of the notochord is regulated by several growth factors, signaling pathways, and transcription factors, which include transforming growth factor-β (TGFβ), Notch, Hedgehog, Wnt/β-catenin, Brachyury (T), Noto, Fox (hepatocyte nuclear factor [HNF]-3/Foxa2/FoxD), and SRY-like box (SOX). 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 The notochord functions as a signaling center during embryonic development and secretes a diverse array of ligands, which pattern surrounding tissues and specify cell fate for the central nervous system, blood vessels, somites, and formation of the IVD. 1 , 4 , 23 , 24 , 25 , 26 , 27 , 28 , 29 However, studies have shown that the floor plate is sufficient for development of the sclerotome or vertebrae without the notochord. 30 The notochord has a mechanical role and provides the embryo with a rigid axial skeleton until the vertebrae and spinal column are formed. The notochord is composed of large vacuolated cells surrounded by a perinotochordal sheath rich in extracellular components such as laminin and coatomers. 1 The interaction of Notch and Hedgehog signaling is essential for appropriate formation of the sheath. 17 , 31 The sheath is thought to help support the mechanical displacement of the notochord from the vertebrae during formation of the spine. This process involves recruitment of somites alongside the notochord and condensation of the sclerotome region of the somite to form bands corresponding to regions of the future vertebrae and IVD, respectively (Fig. 2). 1 , 32 , 33 The notochord is removed from the regions of the developing vertebra through involution; however, it persists in the region of the developing IVD to form the NP. The sclerotome region goes on to form the AF, end plate, and vertebrae. All mammals possess a notochordal NP; however, in avian species the notochord persists as a rodlike structure throughout development and the IVD originates from the somites only. 17 , 31 , 34
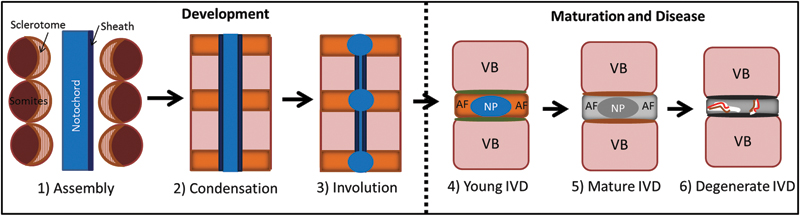
Stages of spine development through maturation and disease. (1) Assembly with the somites alongside the notochord with the sclerotome (orange/pink) situated toward the notochord. (2) Condensation of the sclerotome giving rise to regions of the future intervertebral disk (IVD; orange) and vertebral bodies (VB; pink). (3) Involution of the notochord that expands in the regions of the future IVDs to produce the notochordal cell (NC)-rich nucleus pulposus (NP) and the organized annulus fibrosus (AF) (4). The young NP is highly gelatinous and cellular with large vacuolated NCs. There is distinct demarcation between the NP and AF. (5) The mature IVD transitions with a more fibrous NP region with increased extracellular matrix and decreased cellularity; small nucleus pulposus cells are the predominant cell type in the NP. (6) The degenerate IVD is characterized by a loss of demarcation between the NP and AF, loss in cellularity, structural fissures (white), which provide a path for neurovascular ingrowth (red and brown lines). This figure and concepts are modified from Smith et al, 33 Stemple, 1 and Fleming et al. 32
Developmental Anabolic Ligands, Transcription Factors, and Pathways
Therapeutic candidates with the potential to be used to treat painful IDD include ligands and their associated transcription factors expressed by NCs during development. Ligands comprise of members of the Hedgehog, TGFβ (e.g., Nodal, bone morphogenetic proteins [BMPs], and Activin) and fibroblast growth factor (FGF) families as well as matricellular proteins (e.g., CTGF). Developmental studies in the mouse have highlighted a role for SHH in the maintenance and homeostasis of IVDs. SHH from the notochord is necessary and sufficient for formation of the perinotochordal sheath and patterning of the NP. 17 , 26 , 35 SHH signaling is also essential for postnatal differentiation and proliferation of the AF cells and SNPCs of the IVD. 26 , 36 The NP and end plate both express SHH and its receptor Patch but the AF does not. SHH interacts with many signaling ligands during disk development: both TGFβ and the BMP-antagonist Noggin function downstream of SHH. 36 , 37 TGFβ induces SHH-mediated effects in the NP and end plate, and Noggin upregulates transcription factor Pax-1 expression in the AF and maintains the IVD space through blockade of BMP4.
The TGFβ family plays an influential role during embryonic development of the IVD. Nodal is responsible for specification of cell fate and transition of the notochord from the mesoderm. 38 Similar to Nodal, Activin (another member of the TGFβ family) has been used to differentiate mouse embryonic stem cells into Foxa2- and T-expressing cells in vitro, and these cells subsequently differentiate into Noto-expressing NCs through antagonism of BMP, Wnt/β-catenin, and retinoic acid. 39 TGFβ is expressed throughout development in the mouse IVD and demonstrates a diffuse pattern in the mesenchyme surrounding the notochord. 26 , 40 The pattern of TGFβ expression is similar in the human IVD but increases between gestational weeks 4 and 5 in the notochordal NP. 41 Studies examining the role of TGFβ receptor 2 have shown that TGFβ prevents chondrocyte differentiation in the presumptive IVD and promotes differentiation of the AF from the sclerotome. 42
The FGF family also plays a role in maintenance of the notochord and NC fate. FGF2 together with Activin promotes Foxa2 and T expression in embryonic stem cells. FGF8 is essential for initiation of T expression, as loss of function of this growth factor disrupts the induction of the T gene. 39 , 43 CCN2/CTGF is part of the extracellular matrix heparin-binding family of proteins, and developmental knockdown studies in zebrafish as well as studies examining the effects of NC-conditioned media (NCCM) have demonstrated an essential role in IVD maintenance and homeostasis. Specifically, CCN2/CTGF is expressed in the early notochord of zebrafish, and when the gene is knocked down, the notochord fails to form correctly. 44 , 45
Transcription factors (e.g., Foxa2, T, Noto, SOX5/-6, and Jun) as well as Notch and Wnt/β-catenin signaling pathways support the formation, development, and differentiation of the notochord and NP. Foxa2 is the master transcription factor for notochord specification and differentiation and together with Foxa1 influences the formation of the IVD. 19 , 46 , 47 T and Noto function downstream of Foxa2 and play critical roles in differentiation of the rostral and caudal/posterior regions of the notochord, respectively. 48 , 49 , 50 SOX5/-6 regulates the formation of the perinotochordal sheath and NC survival. In animals lacking SOX5/-6, the IVD is devoid of an NP and the sheath is much thinner. 20 Transcription factor Jun is also critical in later stages of embryogenesis as selective deletion of the Jun allele increases NC apoptosis and disk hypocellularity. 51 Notch and Wnt/β-catenin signaling play influential roles during patterning and help to maintain the notochord during embryogenesis. 16 Notch is required for formation of the perinotochordal sheath and favors differentiation of small nonvacuolated cells over large vacuolated cells including antagonism of Hedgehog signaling. 31 Wnt/β-catenin plays a predominant role in posterior extension of the notochord and fate of the NCs in later stages of development. 18
Neurovascular Guidance Proteins
The notochord performs an essential role in axonal guidance and neoangiogenesis in the developing embryo through secretion of neurovascular guidance molecules, which results in a largely aneural and avascular IVD. Studies by Keynes et al have demonstrated that notochords from chick embryos are able to repel dorsal root ganglion (DRG) in vitro. 23 More recent work has implicated the axonal guidance molecule Semaphorin3A (Sema3A) as a potential candidate for repulsive effects induced by the notochord. When the notochord was cocultured with embryonic DRGs in the presence of neuropilin-1, a soluble receptor for Sema3A, the axonal repulsion effects of the notochord were negated. 2
CS proteoglycans are present within the notochord and are biochemically and immunologically distinct from proteoglycans found in cartilage. 52 A role for CS has also been suggested with respect to patterning of the developing nervous system. 53 Work by Masuda et al has shown a direct interaction between CS from the notochordal core and DRG repulsion. 54 During later stages of development, repulsion is transferred to the perinotochordal sheath. The mechanism by which CS interacts with and inhibits nerve ingrowth is not completely understood, although a role for the receptor contactin interacting with CS directly has been proposed. 55 CS can interact with antineural inhibiting factors by either facilitating inhibition directly or by acting as a reservoir for these proteins. 56
The notochord is also the primary signaling center for inhibition of blood vessel formation along the embryonic midline. Noggin and Chordin are possible candidates for mediating NC-derived repulsive effects. 4 Mechanistic studies have shown that notochordal BMP-antagonists Noggin and Chordin inhibit angiogenesis in the mesoderm in cell culture studies and that these effects are mediated through inhibition of endothelial cell differentiation and assembly of tubules in vivo. 57 Studies by Meadows et al have confirmed the neurovascular inhibiting potential of the notochord and suggest Semaphorin 3E has the ability to inhibit both innervation and angiogenesis simultaneously. 58
Growth and Maturation of the IVD
During growth, the NP is composed of a gelatinous core populated predominantly by large vacuolated NCs with a small percentage of SNPCs. 5 , 6 , 59 The NP is contained circumferentially by the AF and cranially and caudally by cartilage end plates. During maturation the notochordal NP transitions to a more fibrous structure populated predominantly by SNPCs. Studies have characterized the molecular profile of both NCs and SNPCs and identified several differences in matrix protein, enzyme, and integrin expression (e.g., collagen 1, tissue inhibitor of metalloproteinase 1, biglycan, and matrix metalloproteinase [MMP]2/3) between the two cell types. 60 Immature NP cells (or NCs) expressed increased levels of cell surface proteins CD44 and CD24 and transcription factor T compared with mature NP cells. 61 , 62 These differences were also highlighted by Cappello et al, who showed that NCs produce a matrix that is distinct from SNPCs and lack an abundant and protective pericellular matrix. 63
The transition of the NP from NCs to predominantly SNPCs is also thought to be associated with the onset of disk degeneration as species such as rabbit and pig that retain NCs into adulthood do not experience IDD. 5 , 6 The young IVD with a notochordal NP matures with an increase in fibrous matrix and decrease in cellularity (Fig. 2). In humans, this is often followed by progression to IDD with an increase in structural disruption, neurovascular ingrowth, and pain.
Mechanisms of Maturation, Loss of Notochordal Cells, and Origin of Small Nucleus Pulposus Cells
Maturation of the IVD and transition of cellularity from NCs to SNPCs is associated with changes in the microenvironment of the IVD that occur simultaneously with growth and aging. Nutrition plays an important role in maintenance of the NC population. 64 , 65 , 66 NCs have higher metabolic rates when compared with SNPCs, and hypoxia can increase rates of glycolysis, suggesting that NCs require more nutrients than SNPCs. 65 Guehring et al showed that mechanical compression of the IVD in an in vivo rabbit model can decrease the number of NCs over time from 70 to 10% at 56 days. 64 Thus mechanical stress and reduced nutrition are both considered essential factors in maturation of the IVD. This is supported by computational studies by Louman-Gardiner et al suggesting an insufficient nutrient supply and altered physical microenvironment are responsible for loss of NCs over time. 66 Injury can also induce loss of NCs in species that normally retain NCs into adulthood. Needle puncture injury in a rat tail model transitions the IVD first to a “chondrogenic” phenotype with an increase in SNPCs and proteoglycan, then to a more degenerate fibrocartilaginous phenotype with loss of SNPCs and proteoglycans at 6 to 12 weeks. 67
In species that experience IDD, NCs are believed to disappear by early adolescence 59 ; however, studies in both the adult bovine and human IVD have recently demonstrated the presence of cells expressing NC markers such as vimentin, cytokeratin-8, and galectin-1 that are phenotypically similar to NCs in cytomorphology. 68 , 69 This is in conflict with previous studies that suggest NCs disappear from the adult IVD altogether in species such as bovine and human. 5 , 59 In the mouse IVD, T is expressed in the adult disk although at reduced levels. 70 The function of this small population of NCs in the adult IVD is not clear, nor is it evident how they have adapted to survive in the mature IVD where microenvironmental conditions are much harsher and nutrition more limited compared with the developing and young IVD.
Recent in vivo lineage studies support the concept that all cells from the NP are derived from the embryonic notochord. 3 , 21 , 71 These studies are supported by work that has shown NCs can differentiate into multiple cell types (polygonal, giant, and vacuolated) with similar features to the SNPCs cells. 72 Although it has also been suggested in rodents and rabbits that NCs die via apoptosis and are replaced by cells originating from the cartilaginous end plate. 73 , 74 , 75 Studies have also demonstrated the presence of a small population of multipotent “progenitor cells” with the ability to differentiate into the trilineages of cartilage, bone, and fat. 76 However, more recent work by Sakai et al has shown that such progenitor cells decrease with aging and degeneration in the human condition. 43 Whether these progenitor cells are of notochordal or mesenchymal/somite origin remains to be elucidated.
Intervertebral Disk Degeneration
IDD is a complex multifactorial disorder associated with genetic, mechanical, and environmental components. Due to its complexity, the mechanisms associated with disease onset and what makes a degenerate IVD painful versus nonpainful are poorly understood. We consider painful IDD to be a pathologic cell-mediated remodeling of the IVD characterized by structural disruption, loss of cellular homeostasis, and increased catabolism as well as inflammation and neurovascular ingrowth.
Structural Degradation and Disease
Aging and degeneration of the IVD leads to changes in biochemistry and structure. Homeostasis of anabolic and catabolic processes shifts to catabolism and increased matrix degradation. Increased cell death coupled with an upregulation of inflammatory cytokines, such as interleukin (IL)-1β and tumor necrosis factor-α (TNFα), also perpetuate matrix breakdown through increased expression of MMPs and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTs). 77 , 78 , 79 , 80 Loss of glycosaminoglycans causes a decrease in osmotic and hydrostatic pressure impairing mechanical function with uneven force distribution and stress in the disk. 81 Continued loss of mechanical function can lead to AF tears and herniation of the NP. Furthermore, calcification and thinning of the end plates result in decreased permeability and nutrient diffusion. 82 Overall, disk degeneration is characterized by structural changes with loss of matrix and calcification of the end plates, which leads to decreased mechanical function and a hostile cellular microenvironment with increased inflammatory cytokines, catabolic enzymes, and decreased nutrition.
Symptomatic Changes: Neurovascular Ingrowth, Inflammation, and Pain
The healthy IVD is largely avascular and aneural; however, in painful IDD an increase in neurovascular ingrowth has been observed. Ingrowth of nociceptive nerve fibers that express the neuropeptide Substance P and axonal elongation marker GAP43 have been identified within the inner AF and NP of painful human IVDs. 13 , 14 This is corroborated by studies that have demonstrated ingrowth of nerves expressing the high-affinity receptor for nerve growth factor (NGF) TrkA alongside microvascular blood vessels, which also express NGF. 83 Seventy-five percent of the nerves that innervate the rat lumbar IVDs express TrkA including the low-affinity receptor for NGF p75NTR. 84 We have identified nerve fibers and blood vessels in the AF, NP, and end plates of degenerate human IVDs including their presence in Schmorl nodes using antibodies for PGP9.5 and von Willebrand (Fig. 3). Neurovascularization in our histologic sections was located in areas characterized by limited proteoglycan (Alcian blue) and substantial collagen (picrosirius red) as demonstrated by FAST staining. 85
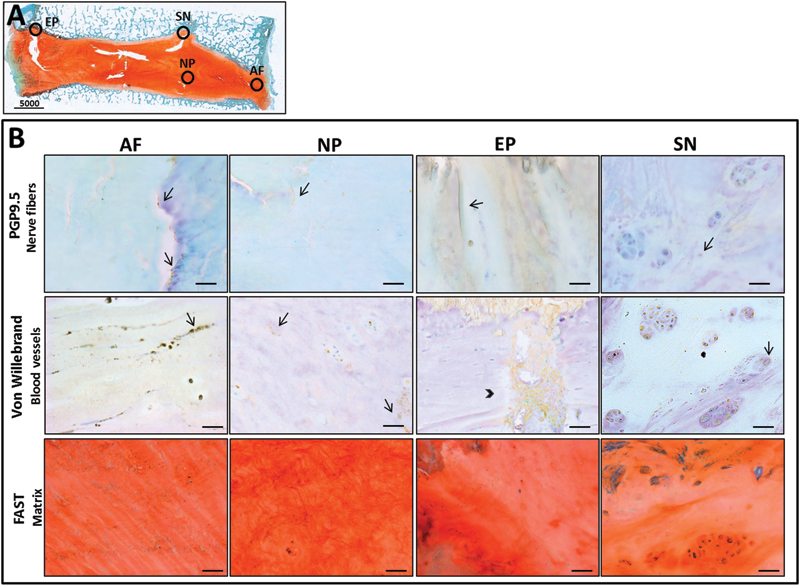
(A) Stitched image of the sagittal section of degenerated human intervertebral disk (IVD) tissue (Thompson grade IV) stained with FAST demonstrating the regions from which annulus fibrosus (AF), nucleus pulposus (NP), end plate (EP), and Schmorl node (SN) were sampled (circles). (B) Histology of human IVD tissue showing pathologic targets of nerves (top panel), blood vessels (middle panel), and decreased proteoglycan content (bottom panel) colocalized in similar areas of AF, NP, EP, and SN stained with PGP9.5 (Abcam no. ab109261, Cambridge, MA, USA), von Willebrand (Abcam no. ab6994), and FAST (red = collagen, blue = proteoglycan), respectively. Arrows indicate nerves and blood vessels and arrowhead indicates a large capillary network.
Matrix breakdown is one of the hallmarks of IDD. Intact human aggrecan, isolated from the NP and AF of healthy IVDs, inhibits neurite outgrowth from neuroblastoma cells and endothelial cell migration in vitro. 86 , 87 Such inhibition is blocked by enzymatic degradation of CS, suggesting that aggrecan in the healthy IVD forms a barrier inhibiting neurovascular ingrowth—a barrier that decreases with severity of degeneration. The proteoglycan decorin also inhibits neurite outgrowth, and repulsion is thought to be mediated by integrin α subunits. However, not all glycosaminoglycans show nerve-inhibiting potential as oversulfated dermatan sulfate side chains can induce axonal elongation. 88 Annular defects (rim lesions/transdiscal tears) and needle puncture injury in combination with an inflammatory stimulus provide permissive environments for neurovascularization with increased ingrowth and expression of IL-1β, TNFα, and neuropeptides calcitonin gene-related peptide and substance P, providing a pathway for pain sensitization. 89 , 90 Increases in the neurovascular and pain-promoting factors NGF and brain-derived neurotrophic factor have been reported in the degenerate human IVD and such neurotrophins are upregulated by IL-1β. 91 , 92 TNFα also upregulated NGF to a more limited extent and induced expression of substance P in degenerate human NP cells. The role of NGF as a mechanism for nerve ingrowth has been further investigated in coculture and conditioned media studies of human disk cells and human SH-SY5Y cells/rat DRGs. 93 , 94 NP cells from the degenerate disk enhanced neurite outgrowth. Stimulation of neurites was blocked by addition of an anti-NGF but not brain-derived neurotrophic factor. 93 Downregulation of the axonal inhibitor Sema3A has also been observed in the degenerate human IVD, in particular for the AF, which may also contribute to increased neurovascular ingrowth in painful IDD. 95
Can We Harness the Therapeutic Potential of Notochordal Cell–Derived Candidates?
The notochord has the ability to synthesize and express anabolic ligands, transcription factors, and neurovascular guidance molecules, which are essential during IVD development, and NCs in the immature IVD maintain homeostasis through the secretion of soluble factors. Such factors could be extrapolated from both the embryonic and immature NCs, identified and recombinant biologics generated to be utilized for therapeutic effect. We propose a novel hypothetical model by which both structure (anabolic) and symptom-modifying (anti-inflammatory/antineurovascular) candidates derived from the notochord can be isolated and used to treat targets identified in the painful IVD (Fig. 4). The concept that NCs are a source of symptom-modifying factors is indeed recent, and there are few publications with respect to such candidates in the IVD field of research. Most of these studies are limited to in vitro investigations, and whether NCs or the factors they secrete can inhibit neurovascular ingrowth in vivo remains a hypothesis requiring further testing. Larsson et al have demonstrated that both NCs and SNPCs from rat IVDs are able to repel neurite outgrowth from rat DRGs in vitro, although the mechanisms underlying repulsion have not been investigated. 96 Developmental studies by Keynes et al and Anderson et al have demonstrated repulsion of DRGs by the notochord with CS and Sema3A as candidate factors. 2 , 23 There is also literature to suggest that notochord-derived factors Noggin, Semaphorins, and CSs have antineurovascular inhibiting roles in adulthood as well as during development, which we speculate could be extrapolated to IDD. 97 , 98 , 99 , 100 Sema3A has been shown to reduce pain behaviors in an in vivo adult rat sciatic nerve injury model and suppression of Noggin in adult human adipose-derived stromal cells enhances angiogenesis both in vitro and in vivo. 98 , 99 We propose a theory in which such factors could be combined to generate a cocktail therapy to address both structural regeneration of the disk as well as inflammation and neurovascular ingrowth (Table 1).
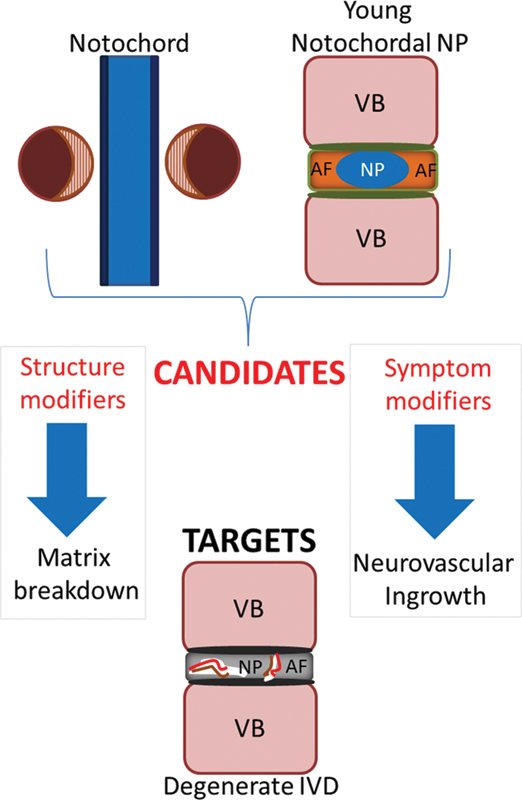
Hypothetical model describing therapeutic candidates and targets for painful disk degeneration. Candidates are identified from the notochord due to their importance in patterning the developing intervertebral disk (IVD) and can be isolated from either the notochord or young notochordal nucleus pulposus (NP). Candidates have structure and symptom-modifying potential to treat targets to limit matrix breakdown and neurovascular ingrowth present in the degenerate IVD. Abbreviations: AF, annulus fibrosus; IVD, intervertebral disk; NP, nucleus pulposus; VB, vertebral bodies.
An attractive feature of using NC-derived candidates is that they are naturally occurring and diverse with a multitude of functions. However, one of the downsides of using NCs is that the factors secreted are complex and can serve as morphogens with distinct functions based on the time of treatment, dosage, and even concentration gradients. This is certainly the case with SHH, TGFβ, and other factors that serve multiple functions, such as Hedgehog proteins demonstrating the potential to promote angiogenic processes. 101 As a consequence, screening studies to examine efficacy and safety in appropriate in vivo animal models are necessary to validate the effects of the potential candidates.
Notochordal Cell–Derived Structure-Modifying Factors
Bioactive factors secreted by the notochord and NCs can target structural restoration of the degenerate IVD. Yet few candidate ligands (only CTGF and TGFβ3) from the notochord have been identified to date as demonstrating therapeutic potential. 7 , 45 Signaling ligands and pathways such as Hedgehog, TGFβ, FGF, Wnt/β-catenin, and Notch can be added or modified as recombinant proteins to target degenerate SNPCs or adult human stem cells and promote anabolic processes and matrix synthesis as well as enhance a young NC-like phenotype in the cells. We also suggest that transcription factors regulated by these pathways such as Foxa2, T, and Noto can be considered candidates and modified to induce similar effects. Recent studies have used transfection of chondrogenic transcription factor genes SOX5, -6, and-9 to induce differentiation of bone marrow– and adipose-derived MSCs down the chondrocyte lineage. 102 , 103 It maybe then possible to transfect human bone marrow– or adipose-derived MSCs with candidate transcription factors that can differentiate cells toward an immature NC/SNPC phenotype and then transplant these cells into animal models of IDD. By differentiating MSCs toward an immature NC/SNPC phenotype, transcription factors may also indirectly reduce symptoms associated with IDD.
Early studies by Vasan et al first demonstrated the potential of the notochord to enhance chondrogenic differentiation including increased synthesis of cartilage-specific proteoglycans. 104 More recent studies examining the therapeutic potential of NCs have looked at the effects of NCs or NCCM interacting with IVD cells and MSCs. They demonstrate proanabolic structure-modifying capabilities, with several possible mechanisms of action. Cocultures of NCs derived from nonchondrodystrophoid dogs and mature bovine SNPCs showed that NCs increased proteoglycan synthesis of SNPCs compared with cultures of SNPCs alone. 9 This effect was not dependent on cell contact but rather soluble factors secreted from the NCs themselves. This has been corroborated by studies examining the effects of canine NCCM on bovine SNPCs. 45 Porcine-derived NCCM promotes a healthy NP phenotype in human degenerate SNPCs and MSCs where increases in NP phenotypic marker genes as well as proteoglycan accumulation were observed. 7 , 8 , 105 NCs cannot be used for direct treatment of IDD because they are likely to die in the harsh human degenerate IVD microenvironment. Furthermore, xenogeneic safety concerns exist with regard to injecting nonhuman NCCM into human IVDs. Therefore, development of successful treatments strategies will require identification and characterization of candidates from the notochord or NCCM. Characterization of candidates in NCCM using proteomic analysis has so far identified matricellular proteins CTGF, tenascin, and clusterin. 8 , 45 However, a more systematic characterization is warranted examining efficacy and safety of the previously identified soluble candidates and those newly identified candidates proposed in the current review in animal models.
Notochordal Cell–Derived Symptom-Modifying Factors
During embryonic development of the spine, the notochord secretes neurovascular inhibiting candidates such as the Semaphorins, Noggin, Chordin, and CS, and these factors also function in adulthood to maintain homeostasis. We suggest that such candidates could be isolated and used as recombinant biologics to target neurovascular ingrowth in painful IDD. Similarly, the receptors on the ingrowing nerves to which some of these ligands bind, such as neurophilins, contactin, and axonin-1, can also be modulated with the potential to inhibit neural ingrowth.
Studies have demonstrated that NCs isolated from species that retain these cells through to adulthood (rabbit and canine) have anti-inflammatory, antiapoptotic, and neural inhibiting effects. NCCM from rabbit NCs decreases the expression of inflammatory markers such as inducible nitric oxide synthase and Cyclooxygenase-2 in AF cells treated with IL-1β. 10 Similar effects were observed when rabbit NCs were cocultured with both human AF cells and macrophages with additional decreases in IL-6 and IL-8 gene and protein expression. 106 NCCM from canine NCs reduces cell death and has an antiapoptotic effect on bovine NP cells treated with IL-1β in vitro as well as upregulating aggrecan and collagen type II and downregulating MMP3. 107 These studies suggest that NCs can produce soluble factors that inhibit inflammation and catabolism; however, the specific soluble factors remain to be elucidated. Coculture studies examining the effect of NCs and mature NP cells from rat IVDs on rat DRGS showed that NCs at intermediate and low concentrations with high concentrations of NP cells induced the greatest inhibition of neurite outgrowth. 96 , 107 , 108 These studies support the proposed model that the notochord and NC-derived candidates can be utilized to target neurovascular ingrowth and inflammation associated with painful IDD; however, their validation for therapeutic effect warrants further investigation.
Limitations and Challenges of Notochordal Cell–Derived Therapies
The IVD is faced with several challenges with respect to biological therapies that have the potential to restore structure/function and limit pain. Due to its low cellularity, limited nutrition, and lack of blood supply, the inability of the IVD to repair itself has been described by some as “frustrated healing.” 82 , 109 Sclerosis of end plates contributes to a hostile microenvironment that is not favorable for the injection of stem cells or growth factors. Therefore the time at which to intervene or the grade of degeneration to target becomes critical, and this is largely dependent on diagnosis of disease or identification of biomarkers characteristic of IDD. For NC-derived therapies to be successful, when to intervene is important and we suggest that such therapies should target early stages of degeneration: Thompson grades III to IV. However, later stages of disease are often associated with symptomatic changes, and therefore in such cases it may be plausible to still intervene if the pain can be reduced or limited directly by neurovascular inhibiting factors. Alternatively notochordal-based therapies could be combined with treatments that may address calcification of the end plate and improve nutrition.
Based on our exclusion criteria, we eliminated publications on “chordoma” from this literature review as it was considered unlikely that such articles would identify any potential candidates. Yet at the same time we excluded publications that may yield important information on factors or signaling pathways to avoid based on their association with cancer, which could be considered a limitation of the review. However, the mechanism of how chordomas arise is largely unknown, and such concerns can be addressed in future studies validating efficacy and safety of candidates.
The concept that NC-derived factors could be used for therapeutic effect is recent, and as a consequence, there are very few studies to date that have examined their symptom-modifying potential or effects in vivo, although several in vitro studies have examined the effect of NCs and the soluble factors they secrete on IVD cells and MSCs. This review highlights a need to validate the efficacy and safety of possible candidates described here; a series of in vivo studies are required in suitable animal models representing IDD and painful IDD.
Conclusions
Understanding concepts from development can help to inform minimally invasive strategies for discogenic back pain. The notochord and NCs pattern the IVD during development and help to maintain biosynthesis/homeostasis in the avascular and aneural IVD through the secretion of a multitude of factors that have the potential to be used as therapeutic candidates. This systematic review supports the theory that proteins, developmental ligands, and transcription factors important in development could be used as candidates with potential to target the structural changes and pain often associated with IDD. NC-derived candidates could be used in combination as a therapeutic cocktail treatment with the potential to promote anabolism and structural repair while limiting neurovascular ingrowth. This review synthesized the literature to identify several possible factors that may be able to be translated directly or with modification to therapeutic strategies for IDD. Remaining research priorities include identification and isolation of factors produced by NCs, mechanistic evaluation of those factors on identified targets, and validation of their therapeutic potential under physiologically relevant conditions in in vivo animal models of disk disease that can then be translated to the human condition.
Disclosures
D. Purmessur, None
M. C. Cornejo, None
S. K. Cho, None
A. C. Hecht, None
J. C. Iatridis, None
Footnotes
Acknowledgments
Thanks to Benjamin Walter and Damien Laudier for technical support with human IVD histology staining and imaging. This work was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases/National Institutes of Health (Grants R01 AR051146 and R01 AR064157) and the AO Foundation (Grant S-13-50P of the AO Research Fund).
Data extraction table from primary search
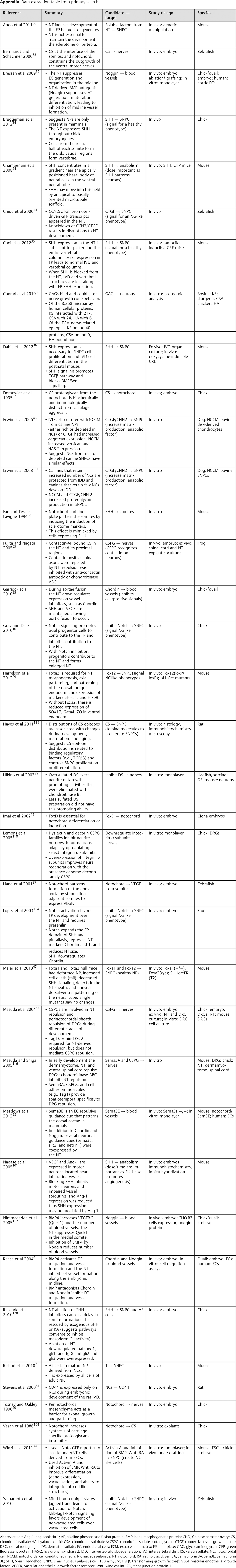
| Reference | Summary | Candidate → target | Study design | Species |
|---|---|---|---|---|
| Ando et al 2011 30 |
NT induces development of the FP before it degenerates. NT is not essential to maintain the development the sclerotome or vertebra. |
Soluble factors from NT → SNPC | In vivo: genetic manipulation | Mouse |
| Bernhardt and Schachner 2000 53 | • CS at the interface of the somites and notochord. constrains the outgrowth of the ventral motor nerves. | CS → nerves | In vivo: embryo | Zebrafish |
| Bressan et al 2009 57 |
The NT suppresses EC generation and organization in the midline. NT-derived-BMP antagonist (Noggin) suppresses EC generation, maturation, differentiation, leading to inhibition of midline vessel formation. |
Noggin → blood vessels | In vivo: embryo ablation/ grafting; in vitro: monolayer | Chick/quail: embryo; human: aortic ECs |
| Bruggeman et al 2012 34 |
Suggests NPs are only present in mammals. • The NT expresses SHH throughout chick embryogenesis. Cells from the rostral half of each somite form the disk; caudal regions form vertebrae. |
SHH → SNPC (signal for a healthy phenotype) | In vivo | Chick |
| Chamberlain et al 2008 24 |
SHH concentrates in a gradient near the apically positioned basal body of neural cells in the ventral neural tube. SHH may move into this field by an apical to basally oriented microtubule scaffold. |
SHH → anabolism (dose important as SHH patterns neurons) | In vivo: SHH::GFP mice | Mouse |
| Chiou et al 2006 44 |
CCN2/CTGF promoter-driven GFP transcripts appeared in the NT. Knockdown of CCN2/CTGF results in disruptions to NT development. |
CTGF → SNPC (signal for an NC-like phenotype) | In vivo | Zebrafish |
| Choi et al 2012 35 |
SHH expression in the NT is sufficient for patterning the entire vertebral column; loss of expression in FP leads to normal IVD and vertebral columns. When SHH is blocked from the NT, IVD and vertebral structures are lost along with FP SHH expression. |
SHH → SNPC (signal for a healthy phenotype) | In vivo: tamoxifen-inducible CRE mice | Mouse |
| Conrad et al 2010 56 |
GAGs bind and could alter nerve growth cone behavior. Of the 8,268 microarray human cellular proteins, KS interacted with 217, CSA with 24, HA with 6. Of the ECM nerve-related epitopes, KS bound 40 proteins, CSA bound 9, HA bound none. |
GAG → neurons | In vitro: proteomic analysis | Bovine: KS; sturgeon: CSA; chicken: HA |
| Dahia et al 2012 36 |
SHH expression is necessary for SNPC cell proliferation and IVD cell differentiation in the postnatal mouse. SHH signaling promotes TGFβ pathway and blocks BMP/Wnt signaling. |
SHH → SNPC | Ex vivo: IVD organ culture; in vivo: doxycycline-inducible CRE | Mouse |
| Domowicz et al 1995 52 | • CS proteoglycan from the notochord is biochemically and immunologically distinct from cartilage aggrecan. | CS → notochord | In vivo; embryo | Chick |
| Erwin et al 2006 45 |
IVD cells cultured with NCCM from canine NPs (either rich or depleted in NCs) or CTGF had increased aggrecan expression. NCCM increased versican and HAS-2 expression. Suggests NCs from rich or depleted canine SNPCs have similar effects. |
CTGF/CNN2 → SNPC (increase matrix production; anabolic factor) | In vitro | Dog: NCCM; bovine: disk-derived chondrocytes |
| Erwin et al 2008 111 |
Canines that retain increased number of NCs are protected from IDD and canines that retain few NCs develop IDD. NCCM and CTGF/CNN-2 increased proteoglycan production in SNPCs. |
CTGF/CNN2 → SNPC (increase matrix production; anabolic factor) | In vitro | Dog: NCCM; bovine: SNPCs |
| Fan and Tessier-Lavigne 1994 28 |
Notochord and floor plate pattern the somites by inducing the induction of sclerotome markers. This effect is mimicked by cells expressing SHH. |
SHH → somites | In vitro | Mouse |
| Fujita and Nagata 2005 55 |
Contactin-AP bound CS in the NT and its proximal regions. Contactin-positive spinal axons were repelled by NT; repulsion was inhibited with anti-contactin antibody or chondroitinase ABC. |
CSPG → nerves (CSPG recognizes contactin on neurons) | In vivo: embryo; ex vivo: spinal cord and NT explant coculture | Frog |
| Garriock et al 2010 25 |
During aortae fusion, the NT down regulates expression vessel inhibitors, such as Chordin. SHH and VEGF are maintained allowing aortic fusion to occur. |
Chordin → blood vessels (inhibits overpositive signals) | In vivo: embryo | Chick/quail |
| Gray and Dale 2010 16 |
Notch signaling promotes axial progenitor cells to contribute to the FP and inhibits contribution to the NT. With Notch inhibition, progenitors contribute to the NT and forms enlarged NT. |
Inhibit Notch → SNPC (signal NC-like phenotype) | In vivo | Chick |
| Harrelson et al 2012 46 |
Foxa2 is required for NT morphogenesis, axial patterning, and patterning of the dorsal foregut endoderm and expression of markers SHH, T, and Hlxb9. Without Foxa2, there is reduced expression of SOX17, Gata4, ZO in ventral endoderm. |
Foxa2 → SNPC (signal NC-like phenotype) | In vivo: Foxa2(loxP/loxP); Isl1-Cre mutants | Mouse |
| Hayes et al 2011 119 |
Distributions of CS epitopes are associated with changes during development, maturation, and aging. Suggests CS epitope distribution is related to binding regulatory factors (e.g., TGFβ3) and controls SNPC proliferation or differentiation. |
CS → SNPC (to bind molecules to proliferate SNPCs) | In vivo: histology, immunohistochemistry microscopy | Rat |
| Hikino et al 2003 88 |
Oversulfated DS exert neurite outgrowth, promoting activities that were eliminated with chondroitinase B. Less sulfated DS preparation did not have this promoting ability. |
Inhibit DS → nerves | In vitro: monolayer | Hagfish/porcine: DS; mouse: neurons |
| Imai et al 2002 15 | • FoxD is essential for notochord differentiation or induction. | FoxD → notochord | In vivo: embryo | Ciona embryos |
| Lemons et al 2005 118 |
Hyalectin and decorin CSPG families inhibit neurite outgrowth but neurons adapt by upregulating select integrin α subunits. Overexpression of integrin α subunits improves neural regeneration with the presence of some decorin family CSPGs. |
Downregulate integrin α subunits → nerves | In vitro: monolayer | Chick: DRGs |
| Liang et al 2001 27 | • Notochord patterns formation of the dorsal aorta by stimulating adjacent somites to express VEGF. | Notochord → VEGF from somites | In vivo: embryo | Zebrafish |
| Lopez et al 2003 114 |
Notch activation favors FP development over the NT and requires presenilin. Notch expands the FP domain of SHH and pintallavis, represses NT markers Chordin and T, and reduces NT size. SHH downregulates Chordin. |
Inhibit Notch → SNPC (signal NC-like phenotype) | In vivo: embryo | Frog |
| Maier et al 2013 47 | • Foxa1 and Foxa2 null mice had deformed NP, increased cell death (tail), decreased SHH signaling, defects in the NT sheath, and unusual dorsal-ventral patterning of the neural tube. Single mutants saw no changes. | Foxa1 and Foxa2 → SNPC (healthy NP) | In vivo: Foxa1(−/−); Foxa2(c/c); SHHcreER(T2) | Mouse |
| Masuda et al 2004 54 |
CSPGs are involved in NT repulsion and perinotochordal sheath repulsion of DRGs during different stages of development. Tag1/axonin-1/SC2 is required for NT-derived repulsion, but does not mediate CSPG repulsion. |
CSPG → nerves | In vivo: embryo; ex vivo: NT and DRG culture; in vitro: DRG cell culture | Chick: embryo, DRGs, NT; mouse: DRGs |
| Masuda and Shiga 2005 116 |
In early development the dermamyotome, NT, and ventral spinal cord repulse DRGs; chondroitinase ABC inhibits NT repulsion. Sema3A, CSPGs, and cell adhesion molecules (e.g., Tag1) provide spatiotemporal specificity to DRG repulsion. |
Sema3A and CSPG → nerves | In vitro | Mouse: DRG; chick: NT, dermamyotome, spinal cord |
| Meadows et al 2012 58 |
Sema3E is an EC repulsive guidance cue that patterns the dorsal aortae in mammals. In addition to Chordin and Noggin, several neuronal guidance cues (sema3E, slit2, and netrin1) were coexpressed by the NT. |
Sema3E → blood vessels | In vivo: Sema3a −/−; in vitro: monolayer | Mouse: notochord/ Sem3E; human: ECs |
| Nagase et al 2005 101 |
VEGF and Ang-1 are expressed in motor neurons located near infiltrating vessels. Blocking SHH inhibits motor neurons and impaired vessel sprouting, and Ang-1 expression was reduced, thus SHH expression may be mediated by Ang-1. |
SHH → anabolism (dose/time are important as SHH also promotes angiogenesis) | In vivo: embryos immunohistochemistry, in situ hybridization | Mouse |
| Nimmagadda et al 2005 117 |
BMP4 increases VEGFR-2(Quek1) and the number of blood vessels. The NT suppresses Quek1 in the medial somite. Inhibition of BMP4 by Noggin reduces number of blood vessels. |
Noggin → blood vessels | In vivo: embryo; CHO B3 cells expressing noggin protein | Chick/quail: embryo |
| Reese et al 2004 4 |
BMP4 activates EC migration and vessel formation and the NT inhibits of vessel formation along the embryonic midline. BMP antagonists Chordin and Noggin inhibit EC migration and vessel formation. |
Chordin and Noggin → blood vessels | In vivo: embryo; in vitro: cell migration assays | Quail: embryo, ECs; human: ECs |
| Resende et al 2010 120 |
NT ablation or SHH inhibitors causes a delay in somite formation. This is rescued by exogenous SHH or RA (suggests pathways converge to inhibit mesoderm Gli activity). Ablation of NT downregulated patched1, gli1, and fgf8 and gli2 and gli3 were overexpressed. |
SHH → SNPC and AF cells | In vivo: embryo | Chick |
| Risbud et al 2010 71 |
All cells in mature NP derived from NCs. T is expressed by all cells of adult NP. |
T → SNPC | In vivo | Mouse |
| Stevens et al 2000 61 | • CD44 is expressed only on NCs during embryonic development of the rat IVD. | NCs → CD44 | In vivo: embryo | Rat |
| Tosney and Oakley 1990 29 | • Perinotochordal mesenchyme acts as a barrier for axonal growth and patterning. | Notochord → nerves | In vivo: embryo | Chick |
| Vasan et al 1986 104 | • Notochord increases synthesis of cartilage-specific proteoglycans in somites. | Notochord → CS | In vitro: explants | Chick |
| Winzi et al 2011 39 |
Used a Noto-GFP reporter to isolate node/NT cells derived from ESCs. Used Activin A and inhibition of BMP, Wnt, RA to improve differentiation (gene expression, vacuolization, and ability to integrate into midline structures). |
Activin A and inhibition of BMP, Wnt, RA → SNPC (create NC-like cells) | In vitro: monolayer; in vivo: node grafting | Mouse: ESCs; chick: embryo |
| Yamamoto et al 2010 31 | • Mind bomb ubiquitylates Jagged1 and leads to activation of Notch. Mib-Jag1-Notch signaling favors development of nonvacuolated cells over vacuolated cells. | Inhibit Notch → SNPC (signal NC-like phenotype) | in vitro; in vivo | Zebrafish |
Abbreviations: Ang-1, angiopoietin-1; AP, alkaline phosphatase fusion protein; BMP, bone morphogenetic protein; CHO, Chinese hamster ovary; CS, chondroitin sulfate; HA, hyaluronic acid; CSA, chondroitin sulphate A; CSPG, chondroitin sulfate proteoglycans; CTGF, connective tissue growth factor; DRG, dorsal root ganglia; DS, dermatan sulfate; EC, endothelial cells; ECM, extracellular matrix; FP, floor plate; GAG, glycosaminoglycan; GFP, green fluorescent protein; HAS-2, hyaluronan synthase 2; IDD, intervertebral disk degeneration; IVD, intervertebral disk; KS, keratin sulfate; NC, notochordal cell; NCCM, notochordal cell conditioned media; NP, nucleus pulposus; NT, notochord; RA, retinoic acid; Sem3A, Semaphorin 3A; Sem3E, Semaphorin 3E; SHH, Sonic Hedgehog; SNPC, small nucleus pulposus cell; T, Brachyury; TGFβ, transforming growth factor-β; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor; Wnt, wingless-int; ZO, tight junction protein-1.
