Abstract
The aim of this systematic review was to determine the incidence of sentinel headache reported by patients with aneurysmal subarachnoid haemorrhage, and whether they are likely to be due to recall bias or to misdiagnosis of a previous haemorrhage. Nine studies of good quality, which reported the number of patients with aneurysmal subarachnoid haemorrhage with a history of sentinel headache, gave rates of 10% to 43%. Two case-control studies, in which the frequency of a history of sentinel headache in patients with aneurysmal subarachnoid haemorrhage was compared with that in controls with non-aneurysmal subarachnoid haemorrhage or with stroke, gave an incidence of 5% (95% confidence interval 0.5, 16) in controls, suggesting that only a small number of apparent sentinel headaches are due to recall bias. Sentinel headaches appear to be a real entity. Their true incidence may vary from near zero to about 40% according to the rate of misdiagnosis in the community under consideration.
Introduction
For over 80 years the observation has been made that patients admitted to hospital with a subarachnoid haemorrhage (SAH) frequently describe a similar previous episode of headache which was ignored, either by the patient or by the physician of first contact (1). The importance of this sentinel headache is that early treatment of an aneurysm, if found, may be successful in preventing the disability or mortality associated with a subsequent bleed. Reviews of the literature report that up to 50% (2) or 60% (3, 4) of patients with a confirmed diagnosis of SAH describe a previous episode of headache. It is important to know how reliable these figures are. Educational interventions to reduce the misdiagnosis of SAH are recommended and indeed are underway (5, 6). Their potential effectiveness depends on the baseline incidence of misdiagnosis.
Gillingham (7) is credited with first using the term ‘warning leak’ in the belief that these premonitory headaches represent the first, albeit small, haemorrhage from an aneurysm. By its very nature, representing as it does an undiagnosed SAH, information about it is bound to be indirect, although Ball (8) has reported a case in which histological evidence of a healed tear in the aneurysm wall was found at autopsy. The degree of organization of the tear was consistent with a leak at the time when the patient experienced a sudden severe headache 6 weeks before her death.
Researchers from Utrecht have cast doubt on the idea that the warning leak is a frequent occurrence. Their prospective study (9) found only two patients out of 21 with aneurysmal SAH who gave a history of a previous characteristic headache. They suggested that the higher rates usually quoted may be largely due to recall bias. Headache, unrelated to an aneurysm, is such a frequent symptom that many patients who rupture an aneurysm will be able to recall a recent headache which they may be tempted to link to their current illness. In a more recent study workers from the same department, using a different approach, concluded again that, at least in their study, warning leaks were uncommon (10). The hypothesis presented was that, if the sentinel headache is due to a first, undiagnosed bleed, when those patients present they are presenting with their second bleed. They should therefore resemble other patients who are presenting with a second documented bleed. However, Linn and colleagues found no difference in clinical and computed tomography (CT) features between those admitted for the first time, whether or not they had a history of sentinel headache. Conversely, those sustaining a second documented bleed were in a worse condition clinically and on CT scan than those being admitted for the first time with or without a history of a sentinel headache. However, there are three problems with this approach. First, the groups being compared are not similar. The fact that those with a sentinel headache were not admitted to hospital at that stage suggests that they were different in some way from those who were. This difference might also be responsible for their better condition when they bleed (again) and are admitted. Second, the Utrecht comparative study (10) found that the intervals between sentinel headache and SAH, and between SAH and rebleed, were strikingly similar. Most occurred within 2 weeks with a peak within 1 day and a smaller peak at 7–14 days. This argues in favour of the sentinel headache being a bleed. Third, other studies have found that patients who give a history of a sentinel headache are in significantly worse condition on admission (11) and have a worse outcome (5) than those with no such history.
A final point, which the Utrecht authors raise (10), is that sentinel headaches can occur even if they are not warning leaks. They may be due to aneurysmal wall stretching or dissection. Whatever their mechanism, if they exist they matter.
Confusion can arise from the different terms used to describe different aspects of the same phenomenon. In this review the term ‘warning leak’ will be avoided since it assumes that the pathology underlying the premonitory symptom is a bleed. The term ‘sentinel headache’ will be used for sudden severe headache which is followed by recovery, only for the patient to suffer a subsequent SAH. It therefore represents a ‘missed diagnosis’ whether it is the patient or a doctor who misses the significance of the headache. Where that same headache eventually brings the patient to medical attention, and to the correct diagnosis, without a second attack the term ‘delayed diagnosis’ is preferable. The underlying pathology may be the same but it cannot be called ‘sentinel’ since it is the presenting symptom, not a previous symptom. Delay in diagnosis is an important issue, but it describes a different sequence of events.
The incidence of sentinel headache is dependent on the behaviour of the patients and clinicians concerned. The ‘true’ incidence is therefore only true for the time and place of the study under consideration. It is not surprising, therefore, that studies show a wide variation in results. The purpose of this review is to examine the degree to which these variations reflect genuine differences in rates of misdiagnosis and, unlike previous reviews of this topic (12, 13), to do so in a systematic fashion.
Methods
MEDLINE, EMBASE and the Expanded Science Citation Index were searched from inception to September 2002 using ‘headache’ or ‘diagnosis’ or ‘warning’ or ‘sentinel’ and ‘subarachnoid hemorrhage’. Separate searches were made using ‘subarachnoid hemorrhage’ plus the diagnostic filter of the Institute of Health Sciences, Oxford, and ‘subarachnoid hemorrhage – diagnosis’. The ‘Related Articles’ feature of PubMed and the Cited Reference Search in the Web of Science were searched. The grey literature was searched via SIGLE. All languages were included. The reference lists of retrieved articles were scanned. Contact was made with experts in the field.
Studies were included if they described the number of unselected patients, regardless of age, with aneurysmal SAH, proven radiologically, at surgery or at autopsy, who were able, or whose relatives were able, to give a history of the presence or absence of previous headaches; and the number who gave such a history in which the headaches were characteristic of SAH (sudden and unusually severe) and occurred in the previous 3 months. If no time scale was given studies were included if, in the judgement of the clinician, the preceding headache was deemed to be relevant, provided it met the other criteria above. Lack of blinding of patient, relative and assessor was not an exclusion criterion because of the practical difficulties involved.
All relevant studies were included which met the above criteria, whether they were cross-sectional, case–control or cohort studies. They were restricted to aneurysmal SAH, since that is the condition where early diagnosis is important, and to headaches that were sudden and severe, since that is the premonitory symptom most likely to alert the physician of first contact to the fact that SAH is a possibility.
The search and initial selection of studies was performed by the author. The author and an independent reviewer extracted data, and made decisions on the final inclusion of studies, with good agreement (κ= 0.88). Disagreements were resolved by discussion.
Exact (Clopper–Pearson) confidence intervals were calculated. Heterogeneity was assessed by the χ2 test.
Results
Thirty-three relevant studies were retrieved, of which 24 were excluded for the reasons given in Table 1. In eight of the nine included studies (see Table 2) the existence of a sentinel headache was assessed by interview of the patient and/or a relative or by questionnaire. In one it was established by a review of the medical records. Excluding poor-quality studies has removed those with the highest estimates but those that remain still give a range of 10% to 43% with marked heterogeneity. It is therefore not possible to summarize the results into a single figure that would be meaningful. Separating studies in which information about sentinel headache was gathered by direct interview from those in which it was gathered from the medical records does not improve the homogeneity, nor does limiting the estimate to the numbers who reported their sentinel headache to a doctor.
Excluded studies

∗These were patients not referred by their general practitioners: a different group from the 21 patients in the same study in Table 2 who were referred.
Studies that describe patients with aneurysmal SAH and the number with a history of sentinel headaches that were sudden and severe
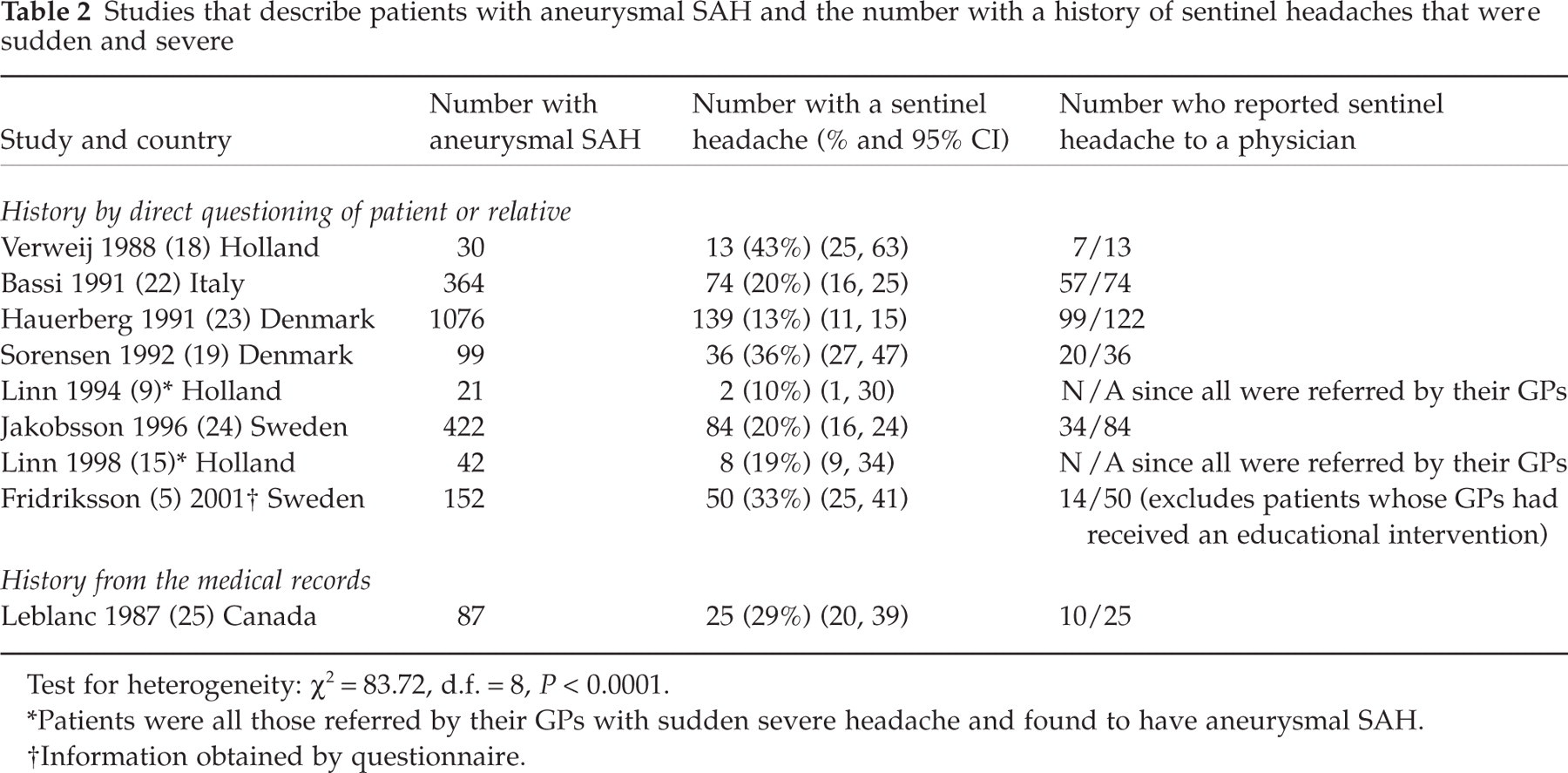
Test for heterogeneity: χ2 = 83.72, d.f. = 8, P < 0.0001.
∗Patients were all those referred by their GPs with sudden severe headache and found to have aneurysmal SAH.
†Information obtained by questionnaire.
Five studies were found in which an attempt can be made to estimate the contribution made by recall bias by comparing sentinel headaches in patients with aneurysmal SAH with sentinel headaches in controls (see Table 3). Two studies (14, 15) allow a comparison of the frequency of sentinel headache in patients with aneurysmal SAH with that in non-aneurysmal SAH, which is thought not to rebleed (16). Two (17, 18) compare the frequency of sentinel headache in patients with aneurysmal SAH with that in patients with strokes. Of these one (18), plus one other (19), compare patients with aneurysmal SAH with non-neurological in-patients. The quality of the studies are good; controls were admitted consecutively to the same hospital as cases, data were collected in the same way and, in the cases of non-aneurysmal SAH controls especially, symptoms were similar to those experienced by cases.
Case–control studies that assess the frequency of sentinel headache
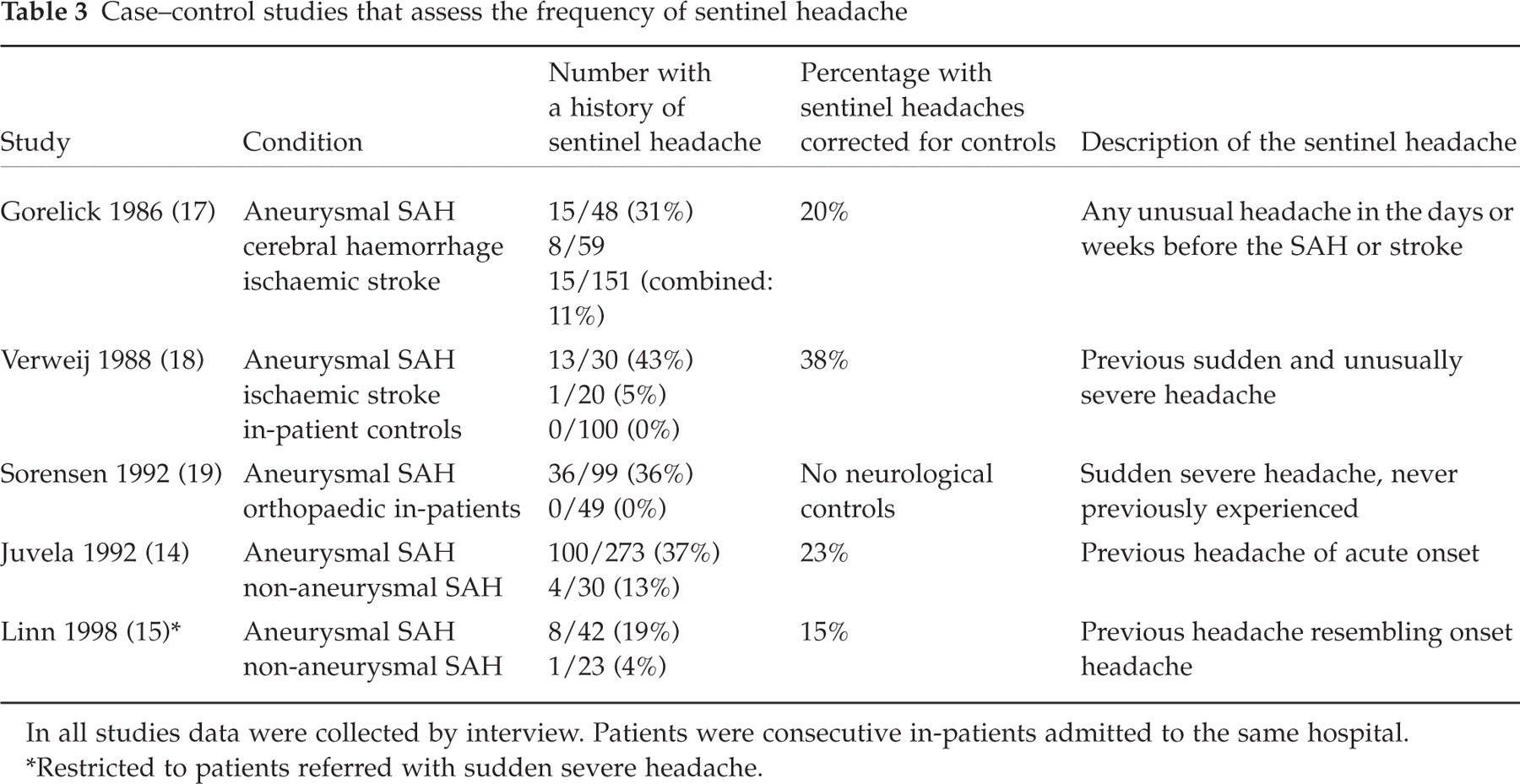
In all studies data were collected by interview. Patients were consecutive in-patients admitted to the same hospital.
∗Restricted to patients referred with sudden severe headache.
None out of 149 non-neurological controls reported a sentinel headache. In the two studies in which only headaches that were sudden and severe were accepted as sentinel headaches the frequency of apparent sentinel headaches in neurological controls was 4% and 5%{summary figure 5%[95% confidence interval (CI) 0.5, 16]}. The two studies in which the headaches were more loosely defined had higher rates but are excluded from this study because of that loose definition. Sudden severe headache is sufficiently uncommon, other than in patients with SAH, for its occurrence in controls to be attributed to recall bias. Such bias therefore contributes approximately 5% of the rates of sentinel headache recorded in the studies that are included in this review.
Discussion
The potential problems to which these observational studies are prone are referral bias, selection bias, recall bias, assessor bias and bias due to inaccurate diagnosis.
Referral bias could arise because most of the studies come from tertiary referral centres, which tend to receive those patients who are in better condition (20). Such a bias would only be important if such patients were more, or less, likely to have experienced a sentinel headache and there is no agreement that patients with sentinel headache are in a worse condition when they finally present (10).
Selection bias and bias due to inaccurate diagnosis have been minimized by the inclusion only of studies with an unselected group of patients with proven aneurysmal SAH.
The possibilities of recall bias and assessor bias remain, because of the impracticality of blinding patients or assessors. The headache that patients describe is so striking that it seems unlikely that the figures will be falsely low because patients or their relatives have forgotten it. The possibility that the figures might be falsely high has prompted the inclusion of the case–control studies. The comparison of cases with controls suggests that approximately 5% of apparent sentinel headaches are due to recall bias.
It is not possible to make a similar assessment of the contribution of assessor bias. The fact that the assessors were not blind to the diagnosis nor, presumably, to the hypothesis under investigation makes them more ready to describe headache as sentinel when they know the patient has a SAH.
A final possible source of error comes from publication bias. Studies whose main point is to report on the incidence of sentinel headache are more likely to be published if their finding is that it is high than low. However, studies in which the reporting of the incidence of sentinel headache is incidental (15, 17) have less reason for publication bias, and those studies comprise one that has one of the highest incidences and one that has one of the lowest. Publication bias in observational studies cannot be explored by means of a funnel plot (21), since increasing study size does not necessarily correlate with increasing reliability.
In summary, bias is likely to be inflating the incidence of sentinel headache, but not enough to explain it away.
More important, in the author's opinion, than bias in explaining the variation in results, is the fact that rates of misdiagnosis of SAH are likely to vary according to setting. Patients in one place may consult more readily with sudden severe headache than those in another, and physicians may vary in their referral rates. Confirmation of the importance of the competence of the referring physician in explaining the variation in rates of sentinel headache is offered by the fact that the studies from Utrecht (9, 10, 15) had the lowest rates of sentinel headaches. Local general practitioners (GPs) had been sensitized to the importance of sudden severe headache by being enrolled in a prospective study that began in 1988. Fewer missed diagnoses (i.e. sentinel headaches) would be expected. Similarly, Fridriksson's study from south-east Sweden (5) compared sentinel headaches in patients from the city of Linköping, whose GPs had been specifically educated about the importance of sudden severe headaches, with patients from the surrounding area. GP education was associated with a fall in the incidence of sentinel headache from 33% to 17% (odds ratio = 0.42, 95% CI 0.16, 1.08). Furthermore, the remaining 17% was due entirely to the failure of patients to report their headache. Variations in rates of sentinel headache do not mean that they are due to bias, but that misdiagnosis is more common in some areas than others.
In conclusion, a review of the literature of the frequency of sentinel headaches in aneurysmal subarachnoid haemorrhage reveals that the higher rates quoted, of 50–60%, are only found in studies which do not meet the criteria of this review. Good-quality studies, however, in which patients gave a clear history of a sudden and unusually severe headache, still give a range of 10–43%. Recall bias may be inflating those figures, but not by more than about 5%. Since the event under consideration is the misdiagnosis of SAH in cases where the symptoms are relatively mild, genuine variations will exist between countries and between hospital catchment areas because of real differences between the inclinations of different groups of patients to report symptoms and of different clinicians to interpret them correctly.
Footnotes
Acknowledgements
The author wishes to acknowledge the help of Tricia Maxwell in the independent extraction of data, the advice of Dr Betty Kirkwood on the statistical aspects, and the assistance of the librarians of the Sussex Postgraduate Medical Centre, Brighton.
