Abstract
The effects of chronic, low-dose amitriptyline on serotonin (5-HT) synthesis rate were measured in rat brain using autoradiography and the trapping of α-[14C]-methyl-L-tryptophan (α-[14C]-MTrp). Rats received amitriptyline (2 mg/kg per day) or saline via intraperitoneal osmotic minipumps for 21 days. Amitriptyline had no effect on any physiological parameters measured, or on free or total plasma tryptophan levels. However, amitriptyline exerted selective decreases of 15% and 17% (P < 0.001) in serotonin synthesis rates in the dorsal and median raphe nuclei, respectively. There was no reduction in any of the projection areas studied, including the cerebral cortex, hippocampus, thalamus, hypothalamus or striatum. The data suggest that chronic low doses of amitriptyline can lead to sustained 5-HT re-uptake inhibition selectively in the raphe nuclei, an effect compatible with tonic activation of 5-HT1A autoreceptors and inhibition of 5-HT synthesis. The failure of chronic amitriptyline treatment to affect 5-HT synthesis rate in the projection areas may ensure an adequate regulation of pain pathways implicated in migraine headache, an effect possibly related to amitriptyline anti-migraine efficacy.
Introduction
Tricyclic antidepressants (TCAs) are one of the most commonly prescribed drugs for the treatment of chronic pain. They have proven efficacy in the treatment of a variety of pain disorders, including diabetic neuropathy (1), post herpetic neuralgia (2), fibromyalgia (3), atypical facial pain (4) and, more specifically for amitriptyline and its active metabolite, in migraine (5) and tension-type headache (6) (for review (7)). In migraine prophylaxis, the beneficial effects of the TCAs have been demonstrated within 4 weeks of treatment (8). Early studies on the use of amitriptyline in migraine and other chronic pain disorders indicated that the anti-nociceptive effect of these medications was independent of their antidepressant action (1, 5). Couch and colleagues (5) studied amitriptyline in 110 patients with migraine headache. Amitriptyline improved migraine by> 50% in 72% of patients, and> 80% in 57% of patients. Depression was absent in 40 patients, borderline in 53, and moderate to severe in 17. Depression improved only minimally with treatment, with only a weak relationship between improvement in depression and improvement in migraine demonstrated in this study. This and other recent studies confirming similar results further suggested that different mechanisms are responsible for the success of amitriptyline in the treatment of depression and disorders of chronic pain (for review (7)).
In experimental models of neuropathic pain (9, 10), acute and chronic treatment with TCAs, including amitriptyline, clomipramine and desipramine (at doses of 0.5–3 mg/kg) have proven efficacious. The acute onset of the pain-relieving properties of low-dose TCAs has led researchers to suspect that acute blockade of serotonin (5-HT) and norepinephrine uptake are involved in the anti-nociceptive response. The ability of the opioid antagonist naloxone to reduce the anti-nociceptive effect mediated by TCAs (11) further suggests that the mechanisms underlying TCA-induced analgesia relate to the interaction between the monoaminergic and opiate systems. As the recommended dose of TCAs in the treatment of migraine and chronic pain is a fraction (10–50 mg/kg per day) (7, 12, 13) of that used in depression (75–150 mg/kg per day) (14, 15), it has been hypothesized that such a dose regimen may have a specific effect on nociception. Considering that serotonergic neurotransmission is a pivotal system in the manifestation of migraine headache (16), the possibility exists that low doses of TCAs exert their prophylactic anti-migraine effect through regulation of brain serotonergic activity.
In order to assess this possibility, we evaluated the effect of chronic, low-dose amitriptyline, compatible with that used in the treatment of migraine and other chronic pain disorders (9, 10), on brain 5-HT synthesis rate measured in vivo using the α-[14C]-methyl-L-tryptophan (α-[14C]-MTrp) method. This method has been well characterized over recent years in animal models as well as in humans using positron emission tomography (17, 18). It is based on the observation that α-MTrp, an analogue of tryptophan not significantly incorporated into proteins (19), is trapped in brain proportionally to the in vivo activity of tryptophan hydroxylase (20, 21), the rate-limiting enzyme in the synthesis of 5-HT. Despite its limitations (22–24), which have been addressed in recent publications (18, 25, 26), the method has allowed clarification of the effects of various drug treatments on 5-HT synthesis rate in various brain areas. More specifically, the SSRIs fluoxetine (27) and paroxetine (28), the 5-HT1A receptor agonist buspirone (29) or 5-HT-releasing drugs MDMA (30), reserpine (31) and D-fenfluramine (32) were found, differently and region-specifically, to affect brain 5-HT synthesis. The results of the present investigation show that chronic low dose of amitriptyline reduces 5-HT synthesis rates selectively in raphe nuclei while having no significant effect in projection areas.
Materials and methods
Animals and chronic drug treatment
Male Sprague-Dawley rats (Charles River, 160–180 g) were housed in the animal facility (room temperature 22°C, 12 h day–night cycle) for at least 48 h before use in an experiment. To insure a stable and continuous exposure of re-uptake sites and receptors to the drug, osmotic minipumps (Alzet osmotic pump model 2ML2; ALZA, Palo Alto, CA, USA) containing amitriptyline or normal saline were implanted intraperitoneally under ketamine anaesthesia using sterile conditions, as described by the manufacturer. Amitriptyline (Sigma Biochemical, St Louis, MO, USA) was dissolved in saline and given at a constant delivery rate of 2 mg/kg per day for 21 days, which corresponds to a fraction of the dose used (10–30 mg/kg per day) to assess its antidepressant effects (33, 34). The same volume of saline was given to control rats for 21 days (approx. 2 ml).
After day 21 of drug or saline treatment, rats were fasted overnight with water supply ad libitum in preparation for the autoradiographic procedure. This was done in an attempt to obtain reasonably stable plasma concentrations of the amino acids. To avoid the influence of circadian rhythm, the radioactive tracer was injected between 12.00 h and 14.00 h, and all rats were sacrificed between 14.00 h and 16.00 h. All animal procedures were approved by the Institution Animal Care Committee and conformed to the guidelines of the Canadian Council of Animal Care.
Experimental procedure
The femoral artery and vein were cannulated with plastic catheters under halothane (0.5–1%) anaesthesia. The posterior limbs of the rats were fixed using a loose fitting plaster cast, and the rats were allowed to wake prior to tracer injection. Rats were injected with 30 µCi of α-[14C]-MTrp (specific activity approx. 55 mCi/mmol), synthesized in house according to Mzengeza and colleagues (35), over a 2-min period using a constant infusion injection pump. Arterial blood samples were taken at progressively increasing intervals, starting from the beginning of tracer injection to decapitation of the rats. The blood samples were centrifuged for 5 min at 9300
The plasma concentrations of total and free tryptophan were measured during the experiment. At three points of blood sampling during the experiments, an additional 50 µl of plasma were taken and deproteinized with 25 µl of 20% trichloroacetic acid for the measurement of the plasma tryptophan concentration. The sample was mixed with a vortex and centrifuged. Supernatant (50 µl) was stored at −84°C until measurement of the total tryptophan concentration using HPLC, as previously described in detail (29, 30). A further 50 µl of plasma were filtered through an Ultrafree-MC filter spinning at 9300
Rats were killed using a guillotine 60 and 150 min after tracer injection. The brains were removed, frozen in isopentane (−45°C), and sliced into 30 µm thickness in a cryostat at about −20°C. The brain slices were mounted on glass slides and exposed to X-ray films along with 14C-polymer standards (American Radiolabel) for 3 weeks to obtain radioautograms. The films were developed, and radioactivity concentrations in different structures identified with reference to a rat brain atlas using the Microcomputer Imaging Device consisting of a video camera, a frame grabber, and appropriate software (MCID, Imaging Research Inc., Ste-Catherine, ON, Canada). Optical density was converted into tissue tracer concentration (nCi/g) based on appropriate standard calibration curves obtained from the calibration 14C-standards.
Calculation of 5-HT synthesis rate from trapping constant of α-[14C]-MTrp
Tissue concentrations of α-[14C]-MTrp were measured at least in triplicates in different brain areas using the rat brain atlas of Paxinos and Watson (36). Regions including the 5-HT cell body and primary projection areas were considered in the analysis. Despite a high tryptophan hydroxylase activity in the pineal gland (PG), 5-HT is only a precursor in melatonin synthesis in this region and, thus, it was not included in the analysis. The brain tissue regional concentration of tracer was converted into the tissue volume of distribution (DV) by division with the plasma tracer concentration at the end of the experiment. The net plasma to brain clearance (K∗; ml/g per min) values of α-MTrp were calculated from the linear relationship between the DV (ml/g) and the exposure time (Θ; min), as described in detail previously (18). An input function for each rat was smoothed using plasma radioactivities obtained from arterial sampling, and the plasma radioactivity at the time the rat was killed was estimated by a least squares fit using the plasma tracer concentration data between 2.5 min and the end of the experiment. Using the mathematically fitted curve (sum of two exponential) of the input function, the exposure time was calculated as a ratio between the integral under the plasma curve and the plasma tracer concentration at the end of the experiment. The biological model on which calculation is based consists of three compartments and contains one irreversible compartment into which α-[14C]-MTrp is transported and in part trapped (18). The regional rates of trapping can then be converted into the regional 5-HT synthesis rates (21). The brain 5-HT synthesis rates are calculated from the brain trapping constant of α-MTrp and the concentration of non-protein-bound tryptophan in the plasma (37) using an in vivo measured conversion constant (lumped constant, LC) evaluated in rat brain at 0.42 (38). The LC converts the α-[14C]-MTrp trapping constant into the trapping constant of tryptophan via the 5-HT metabolic pathway. The synthesis rate (R; pmol/g/min) values were calculated by the equation R = K∗Cp Free-Trp/LC, where Cp Free-Trp (pmol/ml) stands for the concentration of plasma-free tryptophan.
Statistical analysis
The statistical evaluation of group data was achieved by comparing the ratios of 5-HT synthesis rates between control and treated groups in each brain region using one-group two-tailed t-tests. The null hypothesis was that the mean of the ratios should not be significantly different from unity, which would indicate that there was no drug effect on the synthesis. After finding that this was the case, the synthesis in the individual structures was compared by two-tailed t-test for two groups (control and treatment). Results are expressed as mean ± SD and statistical significance was defined as P < 0.05.
Results
Average values for physiological measures and free and total serum tryptophan concentrations of the control and amitriptyline-treated rats are presented in Table 1. There was no significant difference between control and amitriptyline-treated rats in final body weight or weight gain, nor in arterial pH, PO2, PCO2, or free and total plasma tryptophan levels. Free plasma tryptophan levels were 8.2 ± 2.5 nmol/ml and 8.6 ± 1.9 nmol/ml, and total plasma tryptophan levels were 86 ± 18 nmol/ml and 77 ± 15 nmol/ml, in the control and amitriptyline groups, respectively.
Physiological measures and free and total serum tryptophan concentrations of control and amitriptyline-treated rats

Values are expressed as mean ± SD. There was no significant difference in any of the parameters between controls and amitriptyline-treated rats.
A set of autoradiograms illustrating the tracer uptake in brains of control and amitriptyline-treated rats is shown in Fig. 1. Images a, c, e and g are from saline-treated rats sacrificed 150 min after injection of the tracer, while images b, d, f and h are from amitriptyline-treated rats sacrificed at the same time point. Levels have been selected arbitrarily to illustrate the overall distribution of the tracer in the brain. It can readily be observed that areas such as the dorsal and median raphe nuclei (DR and MR on Fig. 1g,h) and the pineal gland (PG on Fig. 1g), which are known to contain high levels of tryptophan hydroxylase, exhibit greater tracer concentrations. In most projecting areas, the distribution of the tracer is rather homogeneous. However, heterogeneity was apparent in the ventral hypothalamus, auditory and occipital cortex, and between the caudate putamen and globus pallidus (Fig. 1).
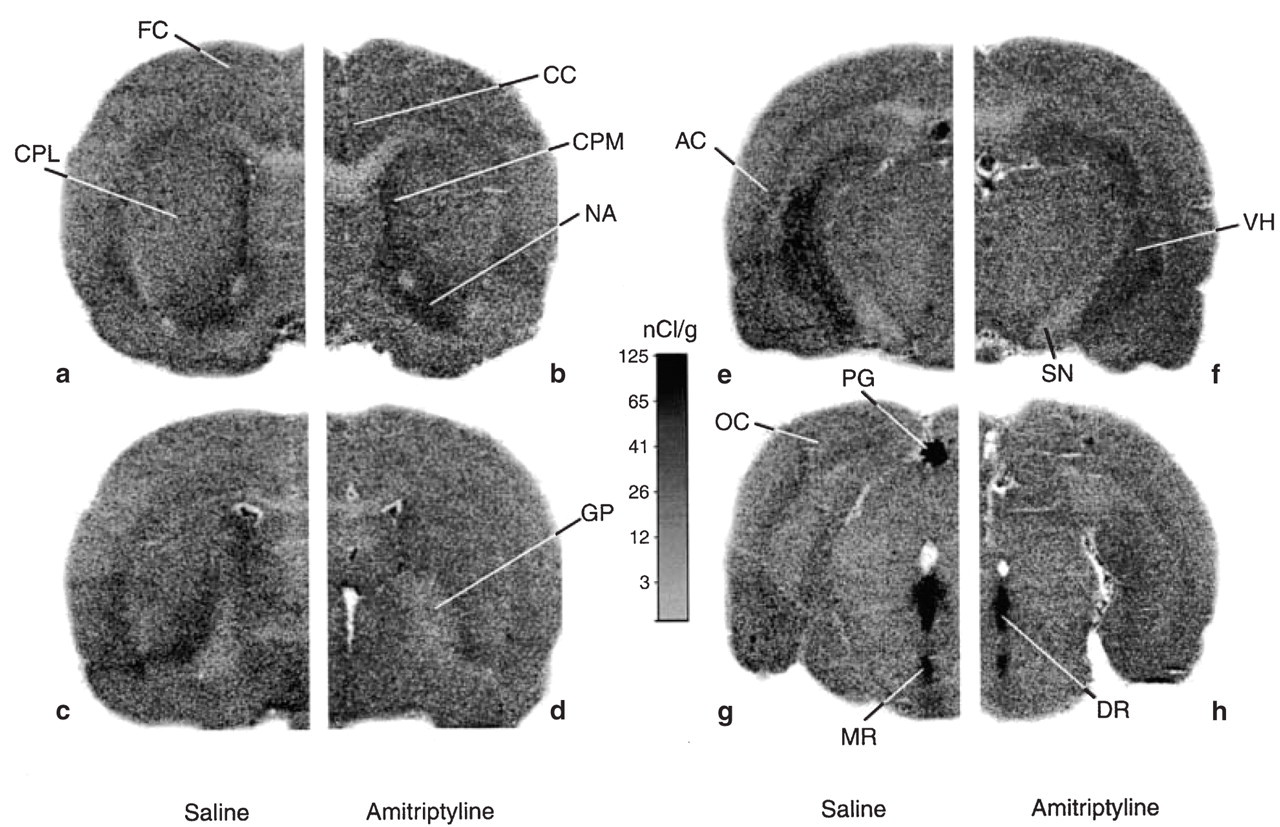
Representative autoradiograms obtained from rats treated with saline (a,c,e,g) or amitriptyline (2 mg/kg per day, b,d,f,h) for 21 days with osmotic minipumps implanted intraperitoneally. The autoradiograms represent the tissue radioactivities in rat brain 150 min after the injection of α-[14C]-methyl-L-tryptophan and were generated from 30 µm thick slices exposed to X-ray films for 3 weeks. AC, Auditory cortex; CC, cingulated cortex; CPL, caudate putamen lateral; CPM, caudate putamen medial; DR, dorsal raphe; FC, frontal cortex; GP, globus pallidus; MR, median raphe; NA, nucleus accumbens; OC, occipital cortex; PG, pineal gland; VH, ventral hippocampus.
The tissue DV of each brain structure as a function of exposure time had a significant linear relationship (P < 0.05) with a positive slope (K∗; ml/g per min). Slopes were converted into 5-HT synthesis rates by the equation described in Materials and Methods. Rates of serotonin synthesis (R) were calculated using the mean values of plasma-free tryptophan concentration (Cp Free-Trp) in each group. In the group analysis of R-values, there was a significant difference in serotonin synthesis rate between the control and amitriptyline-treated group for the dorsal and median raphe nuclei only, with no significant difference in serotonin synthesis in all other brain structures measured (Table 2). Specifically, amitriptyline induced decreases in serotonin synthesis rates of 15% (P < 0.001) and 17% (P < 0.001) in the dorsal and median raphe nuclei, respectively, compared with control rats.
Effect of saline (control) and amitriptyline on 5-HT synthesis rate (R) in selected brain regions of rats treated chronically for 21 days

Values are expressed as mean ± SD.
The n in parentheses represents the number of animals in each group.
There was no statistical difference between the control and the treated rats except in the dorsal and median raphe nuclei (P < 0.001), where there was a 15% and 17% decrease in 5-HT synthesis rate, respectively.
Discussion
The present study shows that chronic treatment with low doses of the tricyclic antidepressant amitriptyline induces a selective decrease in 5-HT synthesis rate in the brainstem raphe neurones cell body area, while synthetic activity remains unaltered in the nerve terminal projection areas. Although the exact site(s) of action of amitriptyline have not been identified in the present study, the findings are best explained by a sustained inhibition of 5-HT re-uptake having more pronounced effects on 5-HT levels in the raphe nuclei than in the projection areas. The decreased synthesis rate in the raphe areas would be consistent with a tonic activation by endogenous 5-HT of somatodendritic 5-HT1A autoreceptors, which have been implicated in the regulation of 5-HT synthesis (29, 39). In contrast, changes in the terminal areas, if present, are negligible as they could not be evidenced with the trapping α-[14C]-MTrp, a method that has allowed clarification of the effects of various clinically relevant compounds on 5-HT synthesis rate in many brain regions, including terminal areas (28, 40, 41).
Previous studies have shown that mRNA expression (34) and sensitivity (42) of brainstem raphe 5-HT1A somatodendritic autoreceptors does not change after chronic amitriptyline or TCA treatment, allowing sustained feedback regulation of serotonergic activity at the cell body level despite prolonged use of the medication. There was no significant difference in 5-HT neurones firing rate to LSD or 5-HT application between controls and rats treated (14 days) with imipramine (5 and 10 mg/kg per day) or desipramine (5 mg/kg per day), and therefore, unaltered responsiveness of the 5-HT autoreceptor was observed (42). A possible explanation for these findings is that the tricyclics are not potent enough re-uptake inhibitors of 5-HT to raise extracellular 5-HT concentrations high enough to alter the sensitivity of autoreceptors.
Our experiments show that amitriptyline, at a dose shown to have anti-nociceptive effect in a rat model of acute and chronic neuropathic pain (9, 10), selectively alters 5-HT synthesis rate in the dorsal and median raphe nuclei, two areas involved in nociceptive control and processing. The selective decrease in 5-HT synthesis observed here in the raphe nuclei probably results from a greater concentration of extracellular 5-HT in this area following TCA treatment, thus allowing interaction with somatodendritic 5-HT1A autoreceptors. Indeed, marked increases (up to five-fold) in extracellular 5-HT levels have been reported in the raphe region after acute systemic injection of amitriptyline, clomipramine or the SSRI fluvoxamine, with 5-HT levels remaining unaltered or being minimally increased in terminal areas such as the frontal cortex (43–45). The greater rise in extracellular 5-HT in the raphe nuclei following 5-HT re-uptake blockade may explain the selective change seen in the present study in serotonin synthesis rates using low-dose amitriptyline. Thus, our results suggest that greater extracellular levels of 5-HT in the raphe nuclei may cause a decrease in the activity of tryptophan hydroxylase in serotonergic cell bodies, thereby decreasing 5-HT synthesis. Such an effect is consistent with a tonic effect of endogenous 5-HT on somatodendritic 5-HT1A autoreceptors, the activation of which decreases firing of 5-HT neurones in the raphe nuclei (39).
In addition to its ability to inhibit the re-uptake of serotonin, amitriptyline inhibits the re-uptake of noradrenaline, and has α1-adrenoreceptor antagonist properties. Re-uptake inhibition of noradrenaline indirectly reduces excitatory effect of α1-adrenergic tone on 5-HT neurones, while α1-adrenoreceptor antagonists inhibit 5-HT cell firing. This leads one to question whether the inhibitory effect of amitriptyline on 5-HT cell firing is mediated solely by the 5-HT1A receptor. This has been addressed and it appears to be the case. Indeed, Gartside and colleagues (46) have shown that the 75% inhibition of 5-HT cell firing induced by an acute i.v. 2 mg/kg dose of amitriptyline was reversed by the selective 5-HT1A receptor antagonist WAY100635, the latter having no effect on the firing inhibition induced by the selective α1-adrenoreceptor antagonist prazosin. An additional study has shown that 5-HT neurone firing rates remain unaltered following short and long-term treatments with reboxetine, a selective noradrenergic re-uptake inhibitor (47). These results suggest that the α1-adrenoreceptor antagonist and noradrenaline re-uptake blocker properties of amitriptyline do not contribute to the inhibitory effects on 5-HT cell firing and, therein, 5-HT synthesis. Also, amitriptyline is a potent 5-HT2 receptor antagonist, a property originally associated with efficacy in migraine prophylaxis, although this view has been revisited (for review (48)). This characteristic, however, is unlikely to explain the present findings, as there is no evidence that 5-HT2 receptors regulate 5-HT synthesis in brainstem raphe nuclei. However, it cannot be excluded that the success of amitriptyline in the treatment of headache and chronic pain is due to its combined activity at monoamine re-uptake sites and specific populations of 5-HT2 receptors, based on the failure of selective SSRIs in the treatment of these pain conditions. Other factors such as variance in brain penetration and individual variability in transport proteins (49) may also account for their differences in clinical efficacy.
The dorsal raphe nucleus exerts significant anti-nociceptive function. Stimulation of the dorsal raphe nucleus can cause powerful anti-nociception, and increases the analgesic effect of morphine, whereas a lesion of the dorsal raphe can partly abolish morphine-induced analgesia (50). Microinjection of 5-hydroxytryptophan into the dorsal raphe nucleus raises the threshold for pain sensation (50). Indeed, the dorsal raphe nucleus is intimately involved in both pain modulation and pain perception, with ascending and descending projections to many structures recognized to be involved in pain control, including the spinal trigeminal nucleus. The latter, which could not be measured in the present study as it was beyond adequate resolution of the method, is an important central relay nucleus in the transmission of vascular head pain associated with migraine headache (51). In this regard, 5-HT and 5-HT1B/D receptor agonists such as the triptans which are effective anti-migraine drugs, have been shown to inhibit trigeminal neurone activity evoked by craniovascular stimulation, a mechanism possibly involved in their therapeutic effect (51, 52). Recently, a similar central effect has been described for opioid receptors and suggested to account for their anti-migraine efficacy (53). As a reduction in central serotonergic activity has been implicated in the pathophysiology of migraine, it may lead to a reduced anti-nociceptive activity in the brainstem and, therein, central sensitization on spinal trigeminal neurones (54). Our data suggest that chronic amitriptyline treatment probably results in preserved 5-HT synthetic activity in all terminal areas measured. It is thus likely that 5-HT synthetic activity is also preserved in the spinal trigeminal nucleus, an effect which would be compatible with an adequate regulation of pain pathways in migraine sufferers kept under prophylactic treatment with low-dose amitriptyline. This may explain the efficacy of amitriptyline in the treatment of chronic pain or migraine prophylaxis, in doses much lower than those traditionally used for depression, and in the absence of depressive symptoms.
Footnotes
Acknowledgements
We thank Ms L. Michel for preparing the final manuscript. This work was supported by a CIHR/GlaxoSmithKline 2000 Canadian Headache Society Fellowship (T.P.), a grant from the Heart and Stroke Foundation of Québec (E.H.), and grant MOP-42438 from Canadian Institutes of Health Research (M.D.).
