Abstract
The interval between indomethacin administration and clinical response may be extremely relevant in the assessment of chronic paroxysmal hemicrania (CPH) and other unilateral headache disorders like cluster headache (CH), with which CPH can be confounded. Indomethacin is inactive in CH; however, in some anecdotal reports in recent years, doubt has been cast on the ineffectiveness of indomethacin in CH. In this study, we have re-assessed the effect of indomethacin treatment in a group of 18 patients with episodic CH (three females and 15 males). From the day 8 of the active period, indomethacin 100 mg i.m. was administered every 12 h, for 2 consecutive days, in an open fashion. The mean daily attack frequency before the test (1.6 ± 0.6) was not statistically different from that on day 1 (2.1 ± 0.9) and day 2 (1.9 ± 0.8) after indomethacin administration. The mean interval between indomethacin injection and the following attack (day 1 and day 2) was 4.6 + 1.1 h. We did not observe any refractory period in any patient after indomethacin. Since the ‘expected’ attack occurred when there theoretically could have been a protective effect after indomethacin administration, it can be reasonably assumed that there is no such protective effect. The use of a test dose of 100 mg i.m. indomethacin (INDOTEST) appears to provide a clear-cut answer in this situation. It may be a useful tool for a proper clinical assessment of unilateral headache with relatively short-lasting attacks when problems of classification arise. A correct diagnosis of CPH or CH is important, since a CPH diagnosis may imply a lifelong treatment with a potentially noxious drug.
Keywords
Introduction
Chronic paroxysmal hemicrania (CPH) is a strictly unilateral headache disorder, characterized by an absolute responsiveness to indomethacin (1, 2). Since the indomethacin response is a fundamental element in establishing the differential diagnosis vs. cluster headache (CH), it is extremely important that this pharmacological test be conducted in an optimal way. In recent literature, there are several reports in which a dubious diagnosis of CPH has been made on the basis of hazily or even erroneously executed indomethacin tests.
It was demonstrated many years ago that indomethacin is ineffective in CH: 10 CH patients suffering from either the episodic or chronic variety of CH were treated with oral indomethacin, in an open fashion; the treatment was of no benefit (1). Similarly, Bogucki (3) treated a CH patient with the chronic form and an unusually high frequency of attack with oral indomethacin and with no clinical response.
Moreover, in two cases with coexisting CH and CPH (4), oral indomethacin provided an absolute effect as regards the CPH part of the picture, but not the CH part, while verapamil was effective only against the latter.
However, some slightly disturbing reports have also appeared: in one report with an alleged coexistence of CH and CPH, the authors claimed that the results were less clear-cut, with a putative effect of indomethacin on the alleged CH part of the complex picture (5). It goes without saying that nosographic and interpretation problems have been immense in a case like this. A case of this nature is not particularly suitable for drawing sweeping conclusions.
The indomethacin-responsive episodic CH case reported by Geaney (6) was, however, probably not a true CH, but a case of the remitting form of CPH, masquerading as CH. Likewise, the clinical data regarding the patient described by Klimek (7) appear to be so indistinct that any firm conclusion can hardly be drawn from it. These anecdotal case reports are obscure and bewildering. The fluctuating course of CH makes drug effect difficult to assess in many cases. These cases should accordingly be reconsidered in this light. They may, nevertheless, to some extent have cast doubt on the original, clear-cut message: that of non-effectiveness of indomethacin in CH.
The aim of the present study was therefore to search for further confirmatory evidence for the ineffectiveness of indomethacin in CH. In doing so, we intended to optimize the conditions for indomethacin bioavailability by administering the drug parenterally.
Patients and methods
Patients
The present series consisted of 18 episodic CH patients, all in the active period of disease (three females and 15 males, aged 44 ± 16 years, mean ± SD). The diagnosis was established according to the International Headache Society (IHS) criteria (8). Non-hospitalized patients followed in the out-patient clinic were asked to fill in a headache diary from the first day of the cluster period for 7 days onwards; after this run-in period, the indomethacin test (‘INDOTEST’) (9) was carried out. Subcutaneous sumatriptan injection was allowed for treatment of attacks in the run-in period and during breakthrough attacks during the test. The study design is shown in Fig. 1.

Cluster headache and INDOTEST: schematic illustration of the temporal relationship between the onset of the cluster period and indomethacin administration. Hatched bar denotes the expected refractory period under indomethacin regimen (in case of clinical efficacy of indomethacin).
Methods
Since in CPH patients the time span between indomethacin administration and attack recurrence was previously found to be longer after 100 mg than after 50 mg indomethacin (i.e. 10.4 ± 2.5 h vs. 8.2 ± 4.8 h, respectively) (9), the former dose was chosen. The test was carried out by invariably administering indomethacin from day 8: 100 mg intramuscularly at 8.00 a.m. and 8.00 p.m. for 2 consecutive days. The effect of indomethacin on any ongoing actual CH attack was not the aim of the present study. Attacks did not occur during or right after injection. The ‘protective’ effect of 100 mg indomethacin i.m. is known to start between 22 and 73 min after drug administration (9). Sumatriptan 6 mg s.c. was allowed in the event of attacks materializing during the test, in order to ameliorate the suffering. This would naturally prevent attacks for 3–4 h.
In order to assess the response during the test, the following parameters were evaluated: (i) the mean daily attack frequency over 7 consecutive days prior to the first injection and during the 2 days of the test; (ii) the mean duration of attacks prior to indomethacin injection and during the 2 days of the test; (iii) the mean interval between indomethacin injection and the first subsequent attack.
The beginning of the cluster period is likely to be the most pertinent one for the evaluation of drug efficacy. The chances of a spontaneous stop in the flow of attacks increase with the advance of the bout. By adhering to the model described, a fast and reliable answer to a possible protective effect of parenteral indomethacin during the 48-h treatment period could be obtained.
Statistical analysis
The statistical evaluation of the attack frequency before and after indomethacin administration was carried out using the Friedman's test, while the Wilcoxon test for paired data was applied when comparing attack duration before and during the test period (day 1–day 2).
Results
In our patient series, the previous mean cluster period duration had been 35.7 + 12.4 days, while the mean duration of the penultimate period had been 31.2 ± 10.9 days. As shown in Table 1, there was no significant difference between the mean daily attack frequency during the 7 days preceding the test (1.6 ± 0.6) and that of day 1 and day 2 of the indomethacin test (2.1 ± 0.9 and 1.9 ± 0.8, respectively; P = 0.073 and P = 0.074, Wilcoxon test). Attack duration (with sumatriptan s.c. being given immediately upon attack onset) was similar before and during the test (11.5 ± 2.2 min and 10.9 ± 2.5 min, respectively) (Table 1). Not too much emphasis can be put on these figures, due to the interference of sumatriptan. From our data, the influence of sumatriptan injection on the forthcoming attacks may partly affect the test: the peak plasma concentration after injection being 14 min (10) and the effect duration 3–4 h. Emphasis was put on the occurrence of attacks within the ‘window’ of indomethacin protection. The mean interval between indomethacin administration and the first subsequent attack was 4.6 + 1.1 h, with attacks at times appearing even after 3 h. These data definitively confirm that within the expected refractory period of 7.5–13 h following 100 mg parenteral indomethacin, there is no protection from the drug.
Clinical features of cluster headache patients before and during INDOTEST
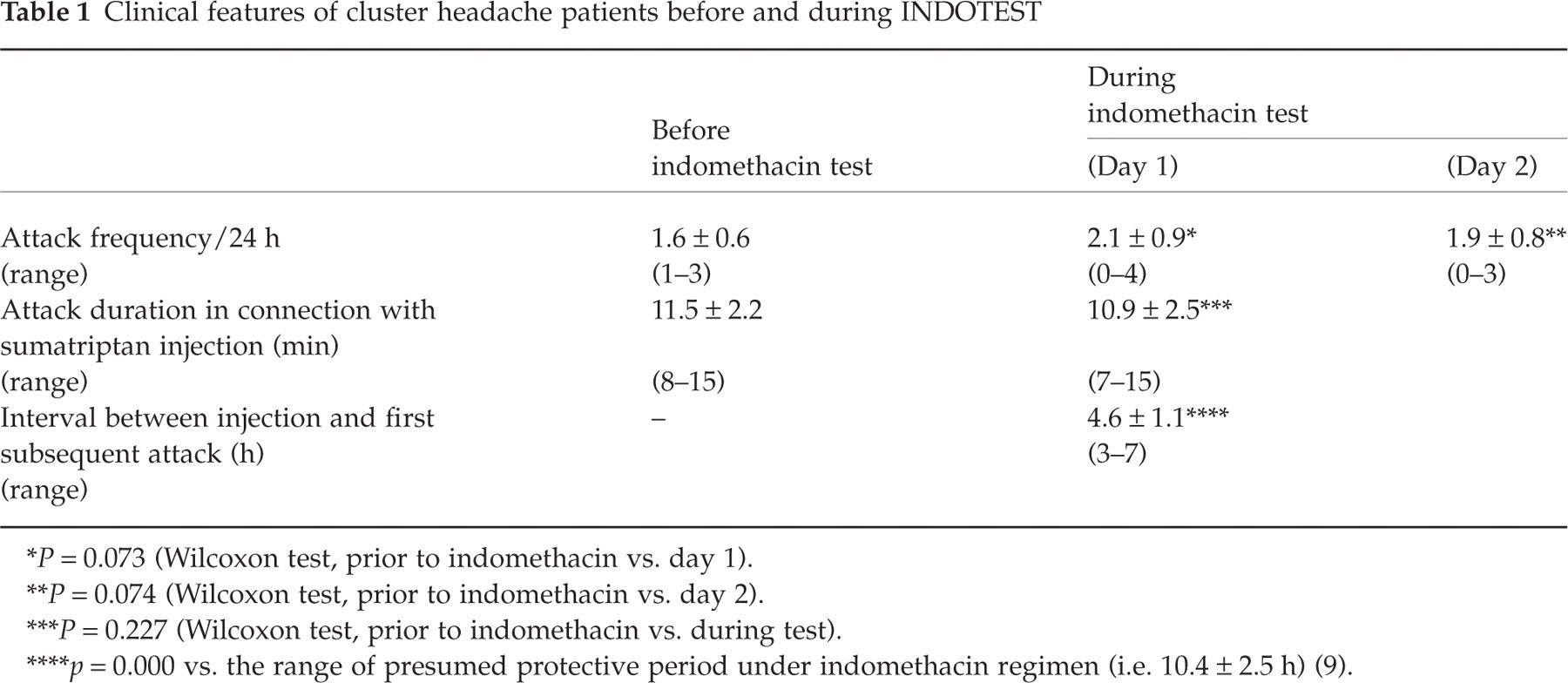
∗ P = 0.073 (Wilcoxon test, prior to indomethacin vs. day 1).
∗∗ P = 0.074 (Wilcoxon test, prior to indomethacin vs. day 2).
∗∗∗ P = 0.227 (Wilcoxon test, prior to indomethacin vs. during test).
∗∗∗∗ p = 0.000 vs. the range of presumed protective period under indomethacin regimen (i.e. 10.4 ± 2.5 h) (9).
Discussion
In our opinion, controlled experiments in drug studies should actually not be necessary in CPH and CH with daily recurring short-lasting attacks, unlike in other headache forms, such as migraine. The main reason for this is that, to the best of our knowledge, a placebo effect is apparently mild in CH (11, 12), while it has never been properly demonstrated in CPH (1, 12).
Our study was carried out according to an uncontrolled design, although in CH patients the use of placebo may be as important as in other cases (11, 12).
The purpose of the present study was to reconfirm the lack of protective effect of an indomethacin regimen in CH patients, employing the recently developed parenteral indomethacin test (INDOTEST). A clear-cut answer was obtained, and the non-efficacy findings of the original study were confirmed (1).
For indomethacin to have been effective a complete disappearance of attacks would have been essential during the test. Similarly, it would have been extremely relevant that any attacks recurring during the test would have displayed a pain pattern close to that of the pretest phases.
The less suitable period for investigating the effect of pharmacological treatments in CH is the medium–late phase of a cluster period, when the chances of a spontaneous recovery are high. Accordingly, we chose in our study the onset of a cluster period, with a well-established headache pattern.
The rationale for carrying out the INDOTEST in CH patients is debatable. In principle, it may not be advisable to perform this test when the patient has less than one attack/24 h. In patients with three to four or more attacks per 24 h, the differential diagnosis vs. CPH may be more involved. In such a case, a fast and reliable differential diagnostic answer can be obtained by the INDOTEST. In this situation, repeated parenteral administration of 100 mg indomethacin will allow for a protective period of 10.4 ± 2.5 h in case of CPH. In countries where parenteral indomethacin is not available, one should use the oral build-up dose strategy for the differential diagnosis in unclear cases (13).
Footnotes
Acknowledgements
This study was supported by a grant from the Ministry of Public Health No ICS 57.2/RC95.60.
