Abstract
The acoustically evoked cortical potentials of 20 migraine patients were investigated using a combined conditioning-testing and oddball paradigm. The short and long-term habituation results of the P50 and P300 waves were compared with 16 healthy subjects. Migraineurs were characterized by a sensory gating deficit of the P50 wave (reduced short-term habituation) in the non-target condition and a reduced long-term habituation of the P300 wave in the target condition. The study describes disturbances of information processing on the automatic and cognitive levels in migraine patients and emphasizes the role of sensory gating and orienting response in migraine pathogenesis.
Introduction
When a stimulus is repeatedly applied, the amplitudes of the scalp evoked (EP) and event-related potentials (ERP) tend to decline. Most of these EP amplitude decrements can be defined as habituation (1, 2). Extensive research on EP habituation described two main kinds of this physiological phenomenon – short-term habituation (STH) and long-term habituation (LTH) (3).
The STH occurs when individual stimuli are repeated at short inter-stimulus intervals and is often attributed to a refractory period (2) or sensory gating (4). It has been proposed that, if the stimuli are presented in pairs, the first stimulus activates excitatory inputs that cause a neuronal response, as well as inhibitory pathways that induce a suppression of neuronal activity on the following stimulus. These processes result in STH (5). This form of habituation is independent of stimulus intensity, psychological characteristics of the stimulus and attentional demands, and is possibly under strong genetic control (4–6). Especially the STH of the P50 (P1), a positive peak occurring about 40–90 ms after stimulus onset, has been intensively investigated because of its relevance in a number of psychiatric and neurological disorders (5, 6). The LTH occurs across blocks of stimuli lasting minutes and is often explained with the ‘dual-process’ theory of Thompson and Spencer (7) or ‘comparator’ theory of Sokolov (8). It has been suggested that the habituation of a neuronal response occurs when the external events of the environment and internal neural model are matched and no longer produce an orienting response of attentional focus (1, 2, 7, 8). This habituation depends on the intensity, psychological significance of the stimulus and attentional resources (1–3). The LTH was investigated most often for the P300 wave in the ‘oddball’ paradigm, during which the subject is exposed to two different stimuli, one of which occurs relatively infrequently (target stimulus) and requires allocation of attentional resources (9). There are very few studies which have established the relevance of the LTH to any neuropsychiatric disorders. In summary, the STH is more probably related to the preattentive screening out the irrelevant stimuli (4–6), while the LTH is attributed to the attentive processes of the orienting response (7–9).
Migraine patients are characterized by reduced habituation of EPs and ERPs (10, 11). These findings were replicated in a number of studies analysing various EP components using different modalities and kinds of stimulation (12–16). Reduced habituation in migraine may be discussed as a basic pathophysiological mechanism of this headache, since it is more pronounced a few days before a migraine attack, representing attack anticipation and increased susceptibility of the brain to provoking agents (17, 18). It shows similarities between relatives of migraine families and may be described as a genetically determined abnormality closely associated with the individual risk of migraine (19, 20).
However, all of the known studies on habituation in migraine used the LTH paradigm. There was no comparison between a target (rare stimuli which are subjectively more significant and related to a larger informational content) and non-target (frequent stimuli which have to be neglected) condition if the ‘oddball’ design was employed. To investigate the type of habituation impaired in migraine, the LTH and STH paradigms were assessed in a common study design as described by Guterman et al. (21) and Schall and Ward (22). Using a classical ‘conditioning-testing’ experiment (4–6), we performed long-term stimulation using target pairs of tones combined with non-target pairs, similar to the ‘oddball’ paradigm (9). The experimental design answered the following questions: are migraine patients characterized by sensory gating deficit, and how are the different kinds of EP habituation related to cognitive information processing in migraineurs? Because the habituation phenomenon in migraineurs was often discussed as a result of either mitochondrial energy metabolism abnormalities or serotoninergic and noradrenergic dysfunction (10, 11), it could be expected that migraineurs will show impairment in habituation mechanisms which are independent of attentional and contextual characteristics of the stimulus, that is, in both kinds of habituation without any effect of ‘target’–‘non-target’ (attentional) condition. The power calculations demonstrate that in order to achieve the test power e = 0.8 (β= 0.20) and the significance level α= 0.05, at least 20–25 subjects in each group have to be enrolled (23).
Materials and methods
Subjects
Twenty females suffering from migraine without aura (IHS criteria (24), code 1.1, mean age 33.2 ± 6.5 years, duration of the disease 14.2 ± 9.4 years, attack frequency 4.77 ± 4.8 days with migraine/month, attack duration 26.7 ± 8.3 h) took part in the study. The patients were recruited from the Out-patient Department of the Institute of Medical Psychology (University of Kiel). None of the patients had used oral contraceptives or prophylactic medication within the 8 weeks prior to the investigation. Attack-related conventional analgesic medication, as well as the use of ergotamine and triptans, was not considered. None of the subjects had a hearing impairment or drug or alcohol abuse. Care was taken that the time interval between the attacks and the recordings was at least 5 days. The interval was registered at the follow-up visit or by phone call. The recording was repeated if an attack occurred within 5 days – this was the case in six subjects. The recordings were not performed in the premenstrual phase of the ovarian cycle. Twenty healthy females (mean age 33.3 ± 6.9 years) with no history of migraine in at least two generations served as controls. The subjects were informed about the course of the experiment and gave written informed consent according to the Helsinki convention.
Physiological recordings
The participants were seated in an armchair with open eyes in an electrically shielded sound-attenuated room with dimmed light. The subjects were told not to fall asleep and to count the high tones distributed among the low tones. The auditory evoked potentials (AEP) recordings were performed in the conditioning–testing paradigm described by Guterman et al. (22). Auditory stimuli (70 dB SPL, 10 ms duration) were presented in pairs with an inter-stimulus interval of 500 ms (conditioning and test stimuli, respectively), with a background noise of 40 dB (SPL), through headphones (Sony 2812, bandwidth 300 Hz to 18 kHz). If the subjects showed startle reactions or electro-oculogram (EOG) activity to the stimulus, the intensity was reduced by 5 dB (25). Two types of stimuli pairs were generated. The target pair of tones (1 kHz) was presented with one of three non-target pairs of tones (2 kHz) in a pseudo-randomized order (target:non-target = 1:3). One hundred and twenty pairs of stimuli were stored. The inter-trial interval varied between 8 and 12 s (10 s being the average). According to the procedure outlined by Nagamoto et al. (25), data acquisition was performed 100 ms before to 500 ms after each tone.
The EEG was recorded using non-polarizable Ag/AgCl electrodes over Cz (10–20 system) with linked mastoids as reference. The ground was placed at the middle of the forehead. The electrode site on the scalp was prepared with alcohol and scraped with rough paper, resulting in an electrode impedance of <5 kΩ. The EEG signals were amplified 20 000 times using a Nihon Kohden amplifier with the low frequency filter of 1 Hz and high frequency filter of 250 Hz (12 dB octave/slope), digitized at a rate of 1000 Hz.
Vertical eye movement artefacts were excluded by parallel recording of the EOG using electrodes (Ag/AgCl) positioned 1–1.5 cm above and below the right eye. Each trial was observed during recording. Trials with EOG activity> 50 µV, α waves, or myogenic artifacts> 35 µV were rejected. The operator was blinded to the subject's diagnosis and suppression of the test response. A protocol listed the number of rejected trials (approximately 12%) for each recording. There were no significant differences between the groups according to the number of rejected trials. Although the rejection procedure may influence the course of habituation in an individual case, by group comparisons this effect may be neglected if the rejected trials are randomly distributed among subjects in each group and if the number of these trials is minimal. In our study three migraine patients and two healthy controls were excluded from the sample because of a large number of eye movement artefacts (>30 trials), so that the evaluation of the course of habituation in these subjects was impossible.
Data reduction and analysis
The 120 trials were divided into blocks of stimuli: four blocks of non-target pairs and two blocks of target pairs. Twenty artefact-free trials in each block were averaged to produce an evoked potential. The averages were digitally filtered with a seven-point low-pass smoothing routine (25). P50 and P300 waves were identified for each stimulus (conditioning and test stimuli). The computer algorithm for P50 peak identification according to Nagamoto et al. (25) was applied. The algorithm identified the conditioning P50 wave as the most positive peak between 40 ms and 90 ms after the first stimulus. If more than one peak was identified, the later one was selected. The amplitude was measured relative to the preceding negativity. The test P50 was identified as the most positive peak with a latency within ±10 ms of the latency of the conditioning P50 response. The test amplitude was also measured relative to the preceding negativity. If no peak amplitude was detected within the latency range established from the conditioning stimulus ±10 ms, the amplitude of the test response was set to zero. The P300 amplitude was identified for both conditioning and test stimuli using the computer algorithm according to Polich and McIsaac (9). The maximal positive peak in the latency range 250–500 ms was selected and the amplitude of P300 calculated relative to the prestimulus baseline average.
The STH of P50 and P300 was described in terms of the testing/conditioning ratio (T/C ratio – the amplitude of the test P50 or P300 wave divided by the amplitude of the conditioning P50 or P300 wave expressed in percent) (4, 25). Lower values indicated increased STH or auditory sensory gating. The T/C ratios were compared using ANOVA statistics. Since the data were normally distributed (non-significant results in the Kolmogorov–Smirnov test) and characterized by homogeneous variances (F-test), analysis of variance was performed with a within-subject factor Habituation (conditioning vs. test tone for the STH or trial blocks for the LTH), a between-subject factor Group (migraineurs vs. healthy controls), and P50 or P300 amplitudes as dependent variables, separately for the target and non-target conditions to characterize LTH and STH. The comparisons between migraineurs and healthy controls within each block of recording were carried out using two-tailed t-tests . P < 0.05 was considered to be significant, the Bonferroni correction was applied for multiple comparisons.
Results
Table 1 demonstrates amplitudes and latencies of P50 and P300 waves and the T/C ratios for the target and non-target pairs of tones. Figure 1 represents original traces of event-related potentials recorded in a patient suffering from migraine and in a healthy subject. Figure 2 shows the course of LTH of both P50 and P300 waves.

Examples of scalp evoked potential (EP) curves obtained in a healthy subject (a) and a migraine patient (b).
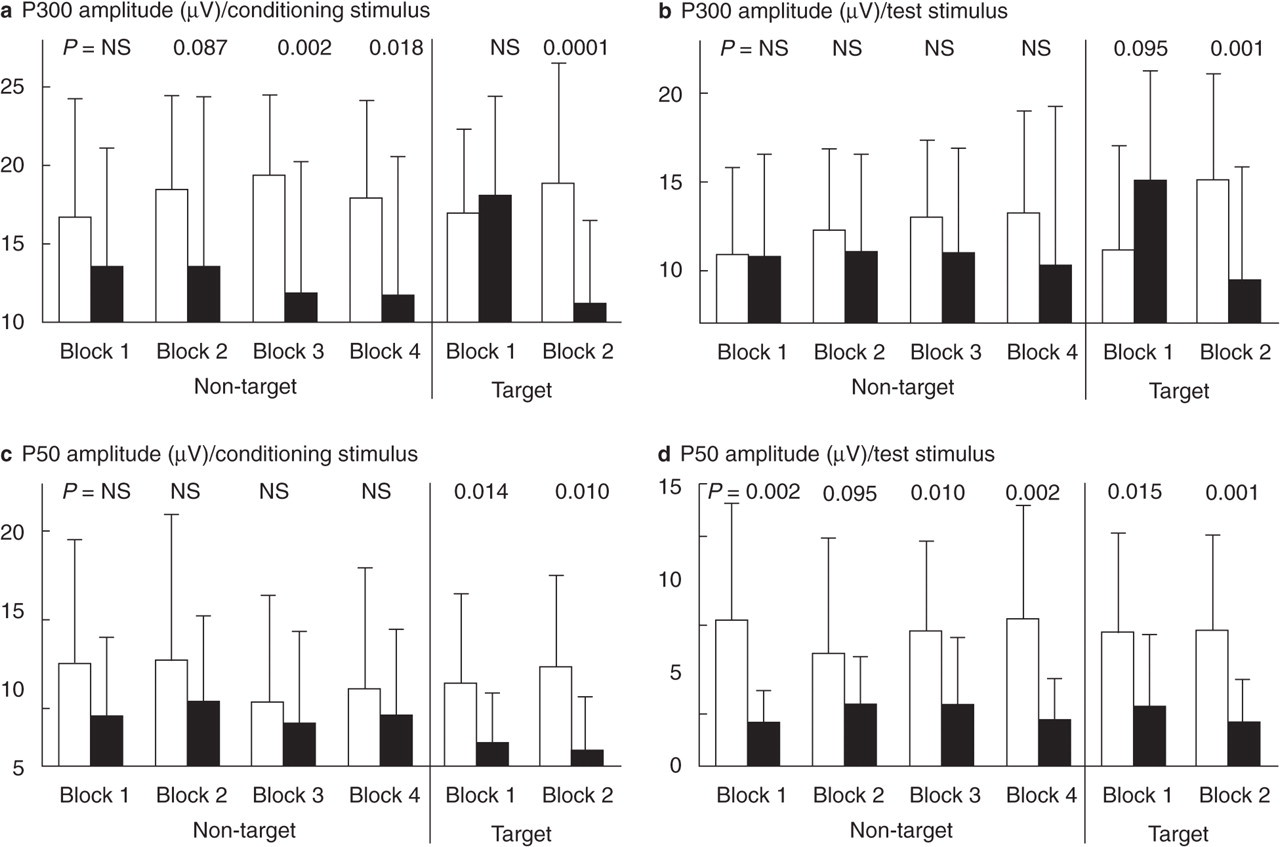
Long-term habituation over four trial blocks in the non-target condition and two trial blocks in the target condition shown for the P300 (a,b) and P50 (c,d) waves of auditory evoked cortical potential recorded in migraine patients and healthy controls. The comparisons between migraineurs and healthy subjects according to scalp evoked potential (EP) amplitudes in each trial block were performed using two-tailed t-tests with the following Bonferroni adjustment. The differences between EP parameters were assumed being significant if the P-value was < 0.002. In order to demonstrate nearly significant results all P-values < 0.05 are shown. Significant differences were found in the second block of recordings of the target condition for the P300 potential and in the last block of the non-target condition and second block of the target condition for the P50 wave elicited on the test stimulus. □, Migraine patients; ▪, healthy controls.
Amplitudes and latencies of P50 and P300 waves (mean (SD)) on the conditioning and the test clicks and the T/C ratio (%) for the target and non-target pairs of clicks

Short-term habituation
As shown in Table 1, the amplitudes of the P50 and P300 waves were larger in migraineurs compared with healthy subjects, although they became significant only for the P50 potential elicited on the test stimulus (P < 0.01 for both target and non-target conditions). The T/C ratio of the P50 wave was larger in migraine patients then of the P300 peak in healthy controls. The latencies of both P50 and P300 potentials did not differ between the groups in all conditions.
The effect Habituation was significant for the non-target condition (F (1, 34) = 27.404, P < 0.001) for the STH of the P50 wave. The Group demonstrated a significant interaction with the Habituation (F (1, 34) = 4.098, P = 0.05). This means that the migraine patients investigated were characterized by a significantly less pronounced STH of P50, or sensory gating, compared with the age-matched healthy controls. The T/C ratio was also significantly larger in migraineurs than in healthy subjects (one-way ANOVA: F (1, 34) = 8.337, P = 0.007).
Although the main effect Habituation remained significant (F (1, 34) = 35.959, P < 0.001) for the target condition, the groups did not differ according to the STH of P50 (non-significant interaction Habituation × Group: F (1, 34) = 0.747, P = 0.756). This was due to a nearly significant decrease of the conditioning P50 amplitude and increase of the T/C ratio in the healthy control group during the target condition compared with the non-target condition (two-tailed paired-sample t-test: t (15) = 1.87, P = 0.08 and t (15) =−1.91, P = 0.07, respectively). In the migraine group, the amplitudes of both P50 waves, and the T/C ratio, remain unchanged compared with the non-target pairs of tones.
Analysis of STH of the P300 wave revealed a significant effect Habituation and a non-significant interaction Habituation × Group for both non-target (F (1, 34) = 26.184, P < 0.001 and F (1, 34) = 0.409, P = 0.527, respectively) and target (F (1, 34) = 28.194, P < 0.001 and F (1, 34) = 0.455, P = 0.889, respectively) conditions. The differences in T/C ratios of P300 between migraine patients and control subjects were also non-significant. Therefore, migraineurs and healthy controls did not differ according to the STH of P300.
Long-term habituation
Figure 2a–d shows LTH of the P50 and P300 waves in the target and non-target conditions. It is obvious that the P300 wave tended to habituate in the healthy subjects and particularly in the target condition. In migraine patients no clear habituation was evident but more probably a potentiation of both P50 and P300 potentials could be observed. The potentiation of evoked potentials in migraineurs is more pronounced in target than in non-target conditions and also for the P300 wave than for the P50 potential.
Analysis of the LTH for the non-target condition showed no significant main effect or interaction for either of the ERP waves or conditioning and test stimuli. For the target condition and the P300 amplitude, however, the main effect Habituation was significant, especially for the conditioning stimulus (F (1, 34) = 11.66, P = 0.002 for the conditioning and F (1, 34) = 3.128, P = 0.08 for the test tones). The Group enhances the significance, emphasizing the observation that migraineurs demonstrate a loss of P300 habituation on the target stimuli compared with healthy controls (interaction Habituation × Group: F (1, 34) = 32.65, P < 0.001 for the conditioning and F (1, 34) = 22.824, P < 0.001 for the test tone). No significant effects or interactions in any of the conditions evaluated were found for the LTH of the P50 amplitude.
Discussion
The study demonstrates that migraineurs are characterized by a reduced STH or sensory gating deficit. On the one hand, the loss of STH in migraine may result from the abnormal refractory period (2) of nervous cells in the brain following their, possibly genetically determined, impaired excitability (26, 27). This explanation is less likely, since we did not observe any differences in the P300 STH between migraineurs and controls, although the interstimulus interval (ISI) used was in the range of the P300 refractory period (1, 2). On the other hand, ‘gating’ has been defined as the ability to screen out and filter excess and trivial stimuli (4, 5). Therefore, inability to filter the non-significant stimuli may cause an overload of the brain and, under disturbance of the brain energy metabolism (28) or abnormal regulation of cortical excitability (27), lead to a migraine attack. Relation of a deficient sensory gating to the functioning of noradrenergic, acetylcholinergic and dopaminergic systems, as well as a large genetic component in the variance of this phenomenon (4, 5), enables only speculation on the role of this abnormality in migraine pathogenesis. Abnormal functioning of all these systems, as well as calcium channel disregulation, have been described in migraine (26, 29) and may be responsible for the reduced STH. Therefore, the mechanisms of sensory gating deficit in migraine remain unknown and need to be investigated in more detail.
Surprisingly, we observed significant differences in the P50 STH between migraineurs and controls only for the non-target tones. In the target condition, the P50 amplitude on the test stimulus increased, causing a significant increase in the T/C ratio in healthy subjects. This supports the previously published observations that attentional demands, which are greater during the processing of target tones, may lead to the enhancement of the test P50 amplitude and T/C ratio (21, 30). As in the present study, however, this effect was not observed in patients characterized by sensory gating deficit (30). The differences in P50 suppression under conditions of increased processing demands between migraineurs and controls emphasize that attentional processes play a less important role in the influencing of sensory gating in migraine, reflecting poorer adjustment possibilities of the early selective processing to changing environmental conditions.
In the analysis of the LTH, the only differences between migraineurs and healthy controls were observed for the P300 wave in the target condition. If the P50 wave characterizes an automatic information processing, the P300 component is related to the context updating and evaluation of cognitive characteristics of the stimulus (31). It was demonstrated that the P300 amplitude is resistant to habituation when the task situation requires active processing of each stimulus (9). This suggests that the requirement of stimulus discrimination involves allocation of attentional resources maintaining the signal value of the stimulus (31). The fact that migraineurs show abnormal habituation in the target condition, which is closely associated with an increased variability of and sensitivity to the changes in the subjective significance of the stimulus, suggests that migraine patients have difficulty in stimulus discrimination and abnormalities in cognitive information processing. It has been previously shown that abnormalities in the early component of the contingent negative variation, discussed as a cortical representation of the orienting response (32), as well as in the habituation of the classical orienting reaction, are associated with migraine (33). Because of the relevance of the P300 amplitude and its habituation to the orienting response (1), we suggest that the results of this study provide further support for the disturbed orienting activity in migraineurs.
Could the STH and the LTH in migraine be related to each other? This question has not been systematically investigated, so that one can only speculate about a common neurobiological explanation for both STH and LTH and their role in the pathogenesis of the headache. Traditionally, the phenomenon of habituation, especially of evoked and event-related potentials, has been attributed to the functioning of prefrontal cortex and thalamus, activities of the hippocampal neurones, and influences from the locus coeruleus, raphe nuclei and mesocorticolimbo-striatal system (1, 2, 4, 5, 7, 8). The overview of abnormalities during the attack-free interval in migraine patients provides evidences of low serotoninergic and dopaminergic and high noradrenergic activities as well as abnormal cortical excitability in migraineurs between attacks (26–29). All these abnormalities may influence both LTH of P300 and STH of P50. For example, the STH and LTH may be affected by modulation of the noradrenergic activity. P50 studies strongly support noradrenergic involvement, with greater noradrenergic function predicting poorer P50 suppression (34, 35). Investigations of P300 emphasize that increase in noradrenergic activity is accompanied by larger P300 amplitudes and reduced habituation, especially in the ‘target’ condition when attentional demands are required (36, 37). Abnormal function of the noradrenergic system in migraine is well described (for review see (38)). Even abnormalities in evoked and event-related potentials in migraine (increased amplitudes and lack of habituation) have been attributed to enhanced noradrenergic activity (11, 14, 39, 40). Therefore, the changes in arousal and preactivation of the migrainous brain influenced by the noradrenergic system may result in both abnormal STH and LTH. However, habituation is a complex phenomenon, and other pathophysiological mechanisms may be significant. Further investigations are needed to clarify these mechanisms and to specify the place of both LTH and STH in the puzzle of migraine pathogenesis.
In conclusion, this study demonstrates that migraine patients are characterized by two kinds of habituation deficits compared with healthy subjects – the sensory gating deficit, or reduced STH, of the P50 wave, independent of attentional demands to the stimulation, and the LTH of the P300 wave under circumstances of increased cortical processing and enhanced mobilization of attentional resources. These habituation deficits represent abnormalities of different levels of information processing in migraine. The reduced P50 STH possibly represents the impairment of an early stage of the automatic screening out of stimuli, whereas the P300 LTH deficit results from the disturbed cognitive processing. How the STH and LTH are related, and which mechanisms underlie these abnormalities, needs to be investigated in further studies.
