Abstract
Sumatriptan and butorphanol nasal sprays are commonly used agents for the management of migraine headaches. Under certain circumstances, these two agents may be administered closely in time. However, the possibility of a pharmacokinetic interaction and the safety of this regime have not been examined. In this crossover design study, 24 healthy subjects received the following four treatments, each separated by at least 7 days: 1 mg butorphanol (Stadol NS7®); 20 mg sumatriptan (Imitrex® Nasal Spray); or both formulations together with butorphanol administered either 1 or 30 min after sumatriptan. Serial plasma samples were collected for 24 h post-dose and analysed for butorphanol and/or sumatriptan by HPLC-MS/MS. Butorphanol plasma concentrations were reduced when it was administered 1 min (mean 28.6% decrease in AUC0-∞)
, but not 30 min, after sumatriptan. The pharmacokinetics of sumatriptan were not substantially altered by butorphanol. The combination of nasally administered sumatriptan and butorphanol appeared safe. However, if butorphanol nasal spray is administered < 30 min after sumatriptan nasal spray, the analgesic effect of butorphanol may be diminished due to reduced nasal absorption resulting from probable transient vasoconstriction of nasal blood vessels by sumatriptan.
Introduction
Sumatriptan and butorphanol are agents with differing mechanisms of action frequently used in the management of migraine headaches in adults (1, 2). Both compounds are commonly employed as nasal spray formulations, and sumatriptan is also available as a subcutaneous injection and as oral tablets for the treatment of migraine. Butorphanol nasal spray is recommended as a rescue medication to alleviate pain in patients not adequately responding to medications, such as sumatriptan, intended to relieve migraine headache. It is likely, therefore, that patients who still suffer from intractable moderate-to-severe migraine or headache pain after taking sumatriptan nasal spray may take butorphanol nasal spray shortly afterwards (3). However, the occurrence of a possible pharmacokinetic interaction and the safety of this regime have not been examined to date.
Butorphanol is a synthetic agonist-antagonist acting at µ-opioid receptors and is an effective analgesic (4). Sumatriptan is an agonist at vascular 5-hydroxytryptamine (serotonin) type 1B and 1D receptors (5-HT1B/1D) which play a role in the mediation of vasoconstriction and is therefore useful in managing migraine headaches (5). Sumatriptan has been reported to induce venoconstriction and to decrease the compliance of conduit arteries (6). In consequence, concurrent use of other vasoconstrictor agents used in migraine headache treatment, such as ergotamine or methysergide-containing preparations, are contraindicated for 24 h following administration of sumatriptan nasal spray (7, 8). There is no apparent pharmacokinetic interaction between butorphanol nasal spray and subcutaneously administered sumatriptan succinate (9). However, it is currently unclear whether there is any effect due to localized sumatriptan nasal spray-induced vasoconstriction on the subsequent absorption of other nasally administered drugs.
The aim of this study was to investigate the potential pharmacokinetic interaction when butorphanol and sumatriptan nasal sprays are administered concomitantly and 30 min apart. The secondary objective of this study was to evaluate safety of co-administered butorphanol and sumatriptan nasal sprays.
Subjects and methods
Subjects
The demographic characteristics of the study objects are given in Table 1. Subjects were between 19 and 40 years of age, and within 10% of the desirable weight for height and frame as determined from Metropolitan Life Insurance Company Tables. The health of the subjects was determined to be good by medical history, physical examination, electrocardiogram (ECG) and clinical laboratory tests performed within 2 weeks prior to the start of the study, and continued health confirmed by a physical examination conducted within 24 h prior to the first dosing. Female subjects were at least 1 year post-menopausal, surgically sterile, or used adequate barrier method of contraception. All female subjects had a negative serum or urine pregnancy test on the days of dosing. All subjects gave informed written consent prior to participation in the study. The Western Institutional Review Board (Olympia, WA, USA) approved the protocol and informed consent. The study was carried out according to the guidelines of good clinical practice.
Demographic characteristics of the study subjects

Data are mean±SD (range).
Study design
This was an open-label, single-dose, randomized, four-way crossover study. Twenty-eight healthy subjects participated and 24 completed the study. According to a randomization schedule, on four separate occasions each subject received a 1-mg dose of butorphanol tartrate nasal spray (Stadol NS®; Bristol-Myers Squibb Co., Princeton, NJ, USA), or a 20-mg dose of sumatriptan nasal spray (Imitrex®; Glaxo Wellcome Inc., Research Triangle Park, NC, USA), or a combination of 1 mg butorphanol and 20 mg sumatriptan nasal sprays. Butorphanol and sumatriptan nasal sprays were co-administered in two periods. When the two drugs were co-administered, butorphanol was administered after waiting for either 1 or 30 min, following administration of sumatriptan nasal spray. There was at least a 7-day washout period between treatments. Serial blood samples were collected before dosing and at 5, 10, 15, 20, 30, 35, 40, 45 min and 1, 1.25, 1.5, 2, 2.5, 4, 6, 8, 12 and 24 h after administration of the first drug in each period. Plasma was harvested from the blood samples and immediately frozen and kept frozen until analysed. Vital signs, ECGs and nasal status (colour, wetness, mucosal patency) were monitored throughout the study.
Drug analysis
Plasma samples were assayed for butorphanol and/or sumatriptan concentrations by validated high performance liquid chromatography–tandem mass spectroscopy methods (10, 11). The lower limit of quantification (LLQ) of these methods were 13.7 pg/mL and 0.71 ng/mL for butorphanol and sumatriptan, respectively. The standard curves were linear (r 2>0.98) over the ranges from 13.7 to 1372 pg/mL and 0.71–71.4 ng/mL for butorphanol and sumatriptan, respectively. Quality control samples prepared at the time of sample analysis were analysed along with the study samples to assess assay accuracy and precision. Within and between-run precision values for quality control samples were <7% for butorphanol and <11% for sumatriptan at low, intermediate and high concentrations over the ranges of the standard curves.
Statistical analysis
Pharmacokinetic parameters (maximum plasma concentration (Cmax), time to Cmax (tmax), area under the plasma concentration vs. time profile extrapolated to infinity (AUC0–∞); and apparent half-life (t1/2)) for both butorphanol and sumatriptan were determined by standard non-compartmental methods (12). For treatment comparisons, an analysis of variance (
Results
Safety
Twenty-four subjects completed the study. No subjects were dropped from the study due to adverse events. The most frequently reported treatment-emergent adverse event in this study was lightheadedness, reported by a total of 13 subjects (46%). Four subjects (14%) each receiving butorphanol alone or a combination of sumatriptan and butorphanol, and one subject (4%) receiving only sumatriptan reported lightheadedness. The second most common adverse event was asthenia, which was reported by a total of four subjects (14%), receiving either butorphanol alone or a combination of sumatriptan and butorphanol. All of the adverse events reported were judged by the investigator to be mild to moderate in intensity. Other adverse events were judged to be mild in intensity and unlikely to be related to study drug(s) and were not resolved at discharge.
Pharmacokinetics
Five subjects were excluded from the pharmacokinetic/statistical analyses because of unusually low plasma concentrations of sumatriptan and/or butorphanol in one of the treatment periods. These unexpectedly low plasma concentrations for both were likely to be due to errors in drug administration with the nasal spray devices by the subjects themselves.
Mean plasma concentration vs. time profiles for butorphanol following administration of butorphanol nasal spray alone and both 1 and 30 min post-sumatriptan nasal spray are depicted in Fig. 1. Mean plasma concentration-time profiles of sumatriptan following administration of sumatriptan nasal spray alone along with butorphanol nasal spray are depicted in Fig. 2. Mean pharmacokinetic parameters for butorphanol and sumatriptan in all treatments are provided in Table 2. The 90% CIs for Cmax and AUC0–∞ for butorphanol and sumatriptan are also included in Table 2.
Mean (± SD) pharmacokinetic parameters for butorphanol (B) and sumatriptan (S) nasal sprays administered alone (1 and 20 mg, respectively) and together with butorphanol administered 1 or 30 min after sumatriptan to 19 healthy subjects. Point estimates (co-administered : alone) and their 90% confidence intervals are given for the primary pharmacokinetic parameters of Cmax and AUC0–∞

∗Median (range).

Mean plasma concentration vs. time profiles of butorphanol following a single 1-mg intranasal dose, • administered alone, and ▵ 1 or □ 30 min after 20 mg intranasal sumatriptan to 19 healthy subjects. The exploded view shows early time points.
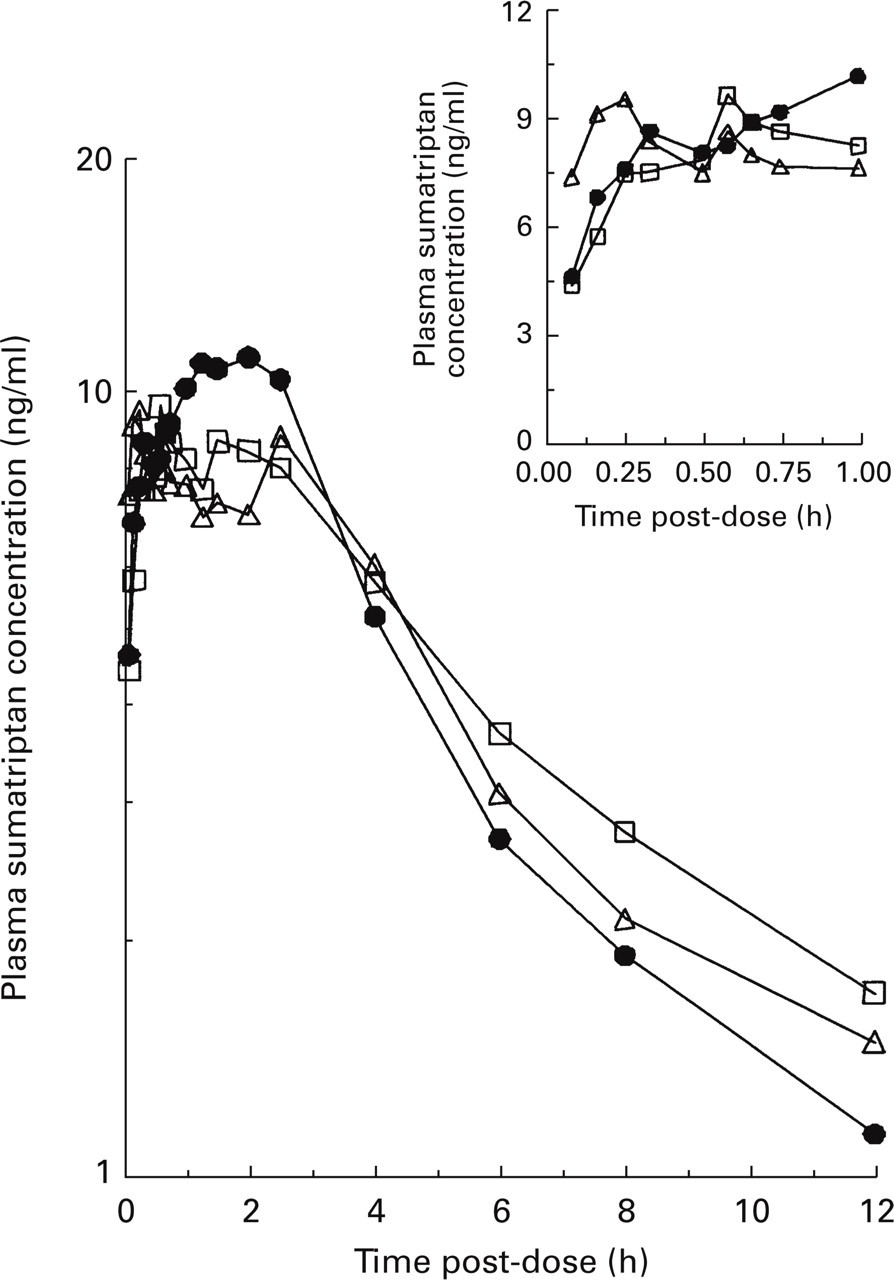
Mean plasma concentration vs. time profiles of sumatriptan following a single 20-mg intranasal dose, • administered alone, and 1 mg intranasal butorphanol given ▵ 1 or □ 30 min later to 19 healthy subjects. The exploded view shows early time points.
Administration of butorphanol 1 min post-sumatriptan significantly affected the absorption of butorphanol. The geometric means for AUC0–∞ and Cmax were 28.6% and 38.3% lower, respectively, when butorphanol was given 1 min after sumatriptan compared with butorphanol given alone. However, these parameters were not substantially altered when butorphanol was administered 30 min after sumatriptan administration. The geometric means for butorphanol AUC0–∞ and Cmax were 10.6% greater and 17.5% lower, respectively, when butorphanol was given 30 min after sumatriptan compared with butorphanol given alone (see Table 2). While the 90% confidence limits for AUC0–∞ met the prespecified limits for concluding lack of interaction, the lower limit of 90% CI for Cmax was marginally outside of the 0.67–1.50 boundary. The differences in tmax and t1/2 for butorphanol between treatments were not statistically significantly different. There were no statistically significant sequence or carryover effects for any of the variables analysed.
Administration of butorphanol 1 or 30 min post-sumatriptan had no significant effect on the pharmacokinetics of sumatriptan. The geometric means for sumatriptan AUC0–∞ and Cmax were 9.4% and 6.7% greater, respectively, when butorphanol was given 1 min after sumatriptan compared with sumatriptan given alone. Similarly, the geometric means for sumatriptan AUC0–∞ and Cmax were 9.8% greater and 6.6% lower, respectively, when butorphanol was given 30 min after sumatriptan compared with sumatriptan given alone. The differences in tmax and t1/2-values for sumatriptan between treatments were not statistically different. There were no statistically significant sequence or carryover effects for any of the variables analysed.
Discussion
Migraine is a chronic intermittent and debilitating disorder, characterized by episodes of moderate to severe head pain, which is usually unilateral and throbbing. Results from two controlled dose-ranging clinical studies examining the effectiveness of intranasal sumatriptan have shown headache relief rates (at 2 h post-dose) that ranged from 43% to 54% following a 10-mg dose and from 55% to 63% following a 20-mg dose (13, 14). Similarly, butorphanol nasal spray has been shown to be effective in relieving pain associated with migraine headache (15). It is likely that sumatriptan and butorphanol nasal sprays may be co-administered to patients who do not respond well to a single agent (3).
When butorphanol nasal spray was administered 1 min post-sumatriptan nasal spray, a statistically significant decrease in mean values for both Cmax (38.3%) and AUC0–∞ (28.6%) of butorphanol was observed. However, the mean Cmax of butorphanol decreased by only 17.5%, and the mean AUC0–∞ increased by 10.6% when butorphanol nasal spray was administered 30 min post-sumatriptan dosing. The observed interaction at 1 min post-sumatriptan dosing is probably due to nasal vasoconstriction caused by sumatriptan. It has been shown that sumatriptan causes vasoconstriction of several blood vessels including cerebral, carotid and temporal arteries (6, 16). Therefore, it is likely that intranasal administration of 20 mg of sumatriptan produced vasoconstriction of the blood vessels in the nasal cavity, which, in turn, decreased the nasal absorption of butorphanol.
One study has administered sumatriptan by the subcutaneous route and found there was no effect on the rate (Cmax) or extent (AUC0–∞) of butorphanol absorption given intranasally (9). This observation supports the hypothesis that vasoconstriction of the blood vessels in the nasal cavity after local administration of sumatriptan was most likely to be responsible for the decreased absorption of intranasal butorphanol administered 1 min post-sumatriptan. Furthermore, the vasoconstriction produced by sumatriptan appears to be transient, with the maximum vasoconstriction occurring within 5 min of subcutaneous injection and arterial pressures returning to preinjection (baseline) levels within 25 min (15). This is consistent, therefore, with the observation in our study that when butorphanol was administered 30 min post-sumatriptan, the absorption of intranasal butorphanol was not substantially altered.
The 90% CI limits for the ratio of means for Cmax and AUC0–∞ exceeded the pre-stated boundary of 0.67–1.50 and did not include 1.0 when butorphanol was administered 1 min post-sumatriptan. When butorphanol was administered 30 min post-sumatriptan the 90% CI for AUC0–∞ was within the specified boundary, and that for Cmax marginally missed the a priori limit. However, the 90% CI for Cmax contained 1.0, indicating that it is likely that with a larger number of subjects (i.e. with an appropriate power) the interval could become narrower to declare a ‘true’ lack of interaction. Whatever the case, the interaction was certainly smaller and unlikely to be of clinical significance when butorphanol was administered 30 min post-sumatriptan.
The pharmacokinetics of sumatriptan were not significantly influenced by the subsequent administration of butorphanol under the dosage regime examined in this study. The pharmacokinetic parameters of sumatriptan when administered alone were similar to those obtained following dosing of intranasal butorphanol. In many subjects a secondary peak was observed following intranasal administration of sumatriptan. This is probably due to oral absorption of sumatriptan, and has been reported in previous studies (14).
There was no evidence of any clinically important change in the nasal status or ECGs when sumatriptan nasal spray and butorphanol were co-administered compared with the baseline values. In addition, there were no clinically significant differences in the frequency of adverse events when butorphanol was administered alone or with sumatriptan nasal spray. Therefore, sumatriptan nasal spray and butorphanol may be co-administered without any safety concerns.
In conclusion, due to the probable transient vasoconstrictor effects of sumatriptan on nasal blood vessels, the absorption of butorphanol was significantly reduced when butorphanol was administered 1 min after sumatriptan nasal spray. When butorphanol was administered 30 min after sumatriptan, butorphanol pharmacokinetics were regarded as being not significantly altered. The pharmacokinetics of sumatriptan were not influenced by the subsequent administration of butorphanol at 1 or 30 min. The combination of nasally administered sumatriptan and butorphanol appears safe. However, the additive analgesic effect of butorphanol may be diminished if given soon after intranasal sumatriptan compared with when given 30 min or longer afterwards.
Footnotes
Acknowledgements
The assistance of Leonard Lachman MD (Phoenix International Life Sciences, Inc., Neptune, NJ, USA) and Anthony F. Grigor PhD (Savanet Inc., Savage, MD, USA) in assisting with the clinical and analytical aspects, respectively, of this study is gratefully acknowledged.
