Abstract
Migraine patients show abnormalities of cerebral electrophysiology that manifest themselves mainly during the attack interval. Cortical-evoked potentials of migraineurs fail to habituate to repetitive presentations of visual stimuli, and the amplitude of components of their auditory cortical-evoked potentials have a higher dependence on the stimulus intensities than in healthy subjects. A computer model of a neural network has been developed that is able to reproduce both these neurophysiological dysfunctions. It predicts a positive correlation between the magnitudes of both these dysfunctions. The model also offers an explanation of why mutations in the same ion channel gene with opposite consequences on channel function, e.g. P/Q Ca2+ channels in migraine, may lead to similar electrophysiological abnormalities.
Introduction
In healthy subjects, the amplitude of evoked cortical responses normally decreases with repetitive presentation of a stimulus. This phenomenon is commonly referred to as habituation [1], and one of its metabolic advantages could be protection against over-excitation and lactate accumulation in sensory cortices [2]. Migraine patients, unlike healthy controls, show a dissimilar electrophysiological behaviour, as shown for visual-evoked potentials, which fail to habituate to repeated stimuli and sometimes even potentiate [3, 4]. This also holds true for event-related potentials [5, 6, 7]. It has been hypothesized that the simultaneous occurrence of this deficit of habituation and a reduced mitochondrial energy reserve might lead to the activation of the trigeminovascular system, culminating in the migraine attack [8].
Migraine patients are also distinguished by a higher dependence of their auditory-evoked cortical responses on stimulus intensity than healthy volunteers [9, 10]. This increased intensity dependence of auditory potentials (IDAP) is, like sensitization, only detectable interictally [11]. The middle latency auditory potentials are complex neuronal phenomena that depend on thalamo-cortico-thalamic activities and various state-setting subcortico-cortical pathways; there is nonetheless indirect evidence that IDAP is inversely correlated with central serotonergic transmission [12]. If deficient, the latter may lead to a decreased cortical pre-activation level, and possibly be the basis for the observed lack of habituation in migraineurs [8]. It has been hypothesized that the strong IDAP found in migraineurs might be due to a deficit of habituation to high intensity stimuli [9], and thus these two distinct phenomena may in fact be expressions of the same dysfunction in habituation.
The use of computer models has been beneficial in the investigation of neurophysiological abnormalities underlying disorders such as epilepsy [13, 14, 15]. Much could therefore be learnt by applying the same tool to other neurological disorders also caused by abnormalities in cortical excitability. A neural network model of cortical habituation has been formerly proposed by Stanley [16, 17]. The author had found that his network model reproduced various aspects of habituation or sensitization to stimuli of varying intensities. We also chose this model because it is based on synaptic depression, which has been found to play a critical role in several classic models of habituation, such as the defensive reflexes of aplysia, the escape reflexes of crayfish and the startle reflexes of vertebrates [18]. The model could therefore be used for investigating the question of whether a lack of habituation and an abnormality in intensity dependence could arise for the same underlying dysfunction. Moreover, electrophysiological recordings involve methodological complications such as an order effect [19]. The availability of a model might thus favour a better understanding of the underlying neurobiological mechanisms and make it possible to predict test results and possibly correct methods, in order to improve retest reliability.
Methods
Model
The constructed circuit was based on a previously existing network model of habituation [16, 17]. A diagram of the network is shown below (Fig. 1). Each node in the model can be interpreted as a population of neurones. The activity of the node O is taken to represent the magnitude of the evoked potential to a given stimulus. While the units for O in the model are arbitrary, they are presented here in microvolts (µV) in order to simulate electrophysiological experiments. Output from the node O is determined by input from the nodes H and S. Afferent auditory input I, varying between 40 and 70 dB, is received at the node H. Output from the node H reaches node O through a direct path mediated by the synapse w 2 , and indirectly via the node S, mediated by the synapse w 3 . The following equations describe how each node integrates input from the other nodes of the circuit:
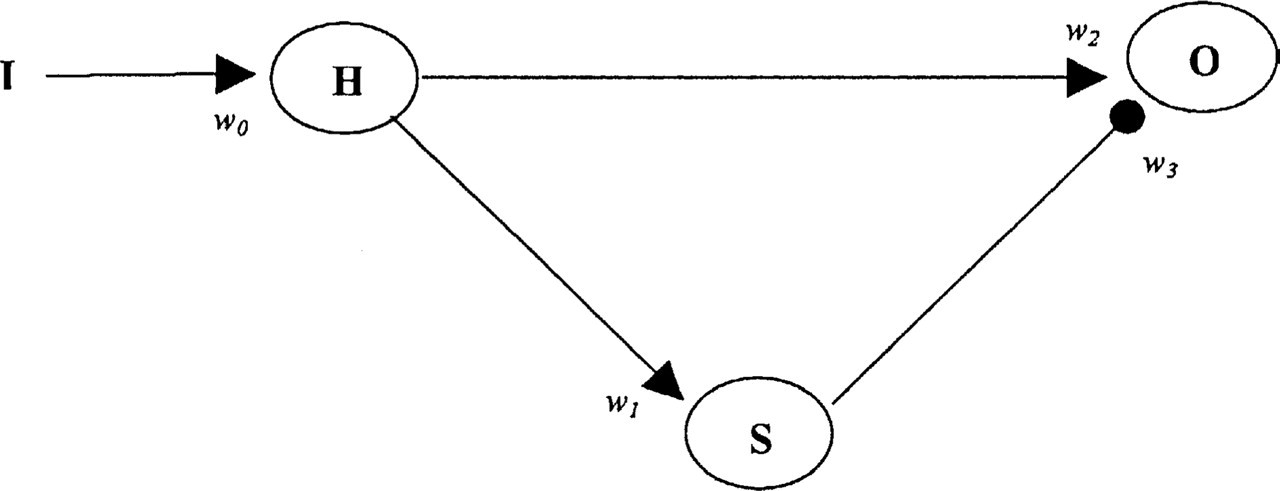
Circuit diagram of network model (modified from Stanley 1976).
While the synapse w 2 depresses with repetitive presentations of I, the synapse w 3 is potentiated in this condition. For each synapse w i , the change in strength is determined by the initial strength w i0 , the time constant of change τ i and the value of a i . If a i is negative, the synaptic strength decreases and the synapse is a depressing synapse. A positive value of a i on the other hand, would lead to a potentiating synapse. The following differential equation describes synaptic evolution:
The synapse w 0 and w 1 were set to remain constant. As in the case of the model by Stanley [16, 17], the value of τ i was not a constant but varied based on the value of I. In our model, the value of τ i was linearly related to the value of I.
All simulations were run using a fourth-order Runge-Kutta integrator. Each simulation was repeated several times using slightly varying parameters in order to ensure that the results were stable to slight variations.
Adjustment of synaptic strength
The strength of a synapse, w i , was adjusted by varying the parameter a i of equation 4. Thus the strength of the potentiating synapse w 3 was increased by increasing a 3 , while the strength of the depressing synapse w 2 was decreased by decreasing the absolute value of a 2 .
Stimulus presentation and response measurements
The presentations of I and the measurement of the responses of O were carried out in blocks of trials. A block usually consisted of 40 trials, in which the duration of each square pulse stimulus within the block was 20 ms. A block therefore usually lasted 800 ms. The response of O, or the simulated evoked potential, in each block was measured as the mean value of O in that block. The value of O is presented in units of microvolts (µV). In some cases, the value of an output is expressed as a percentage of its value in the first block. This mode of presentation allows us to highlight any habituation or sensitization that may have taken place.
The conditions that would give rise to habituation or sensitization upon the repetitive presentation of a stimulus were first investigated. In other words, the ‘normal’ and ‘migraine’ synapse models (NSM and MSM) were developed by modifying the strengths of the potentiating synapse w 3 and the depressing synapse w 2 . As in the case of the experimental studies carried out up to now, the stimulus intensities used were 40 and 70 dB.
Subsequently, the dependence of the amplitudes of responses on the stimulus intensity was tested by using the normal synapse model and the migraine synapse model. Stimulus amplitudes of 40, 50, 60 and 70 dB were utilized, and stimuli were presented in blocks of 100 trials for each stimulus intensity. The recorded response was the mean response in each block. The measure of the intensity dependence of responses of O on the presentations of I was obtained by plotting the value of O obtained for each intensity of I. The value of the intensity dependence was given by the slope of the I–O curve.
Results
Habituation or sensitization to repeated stimuli
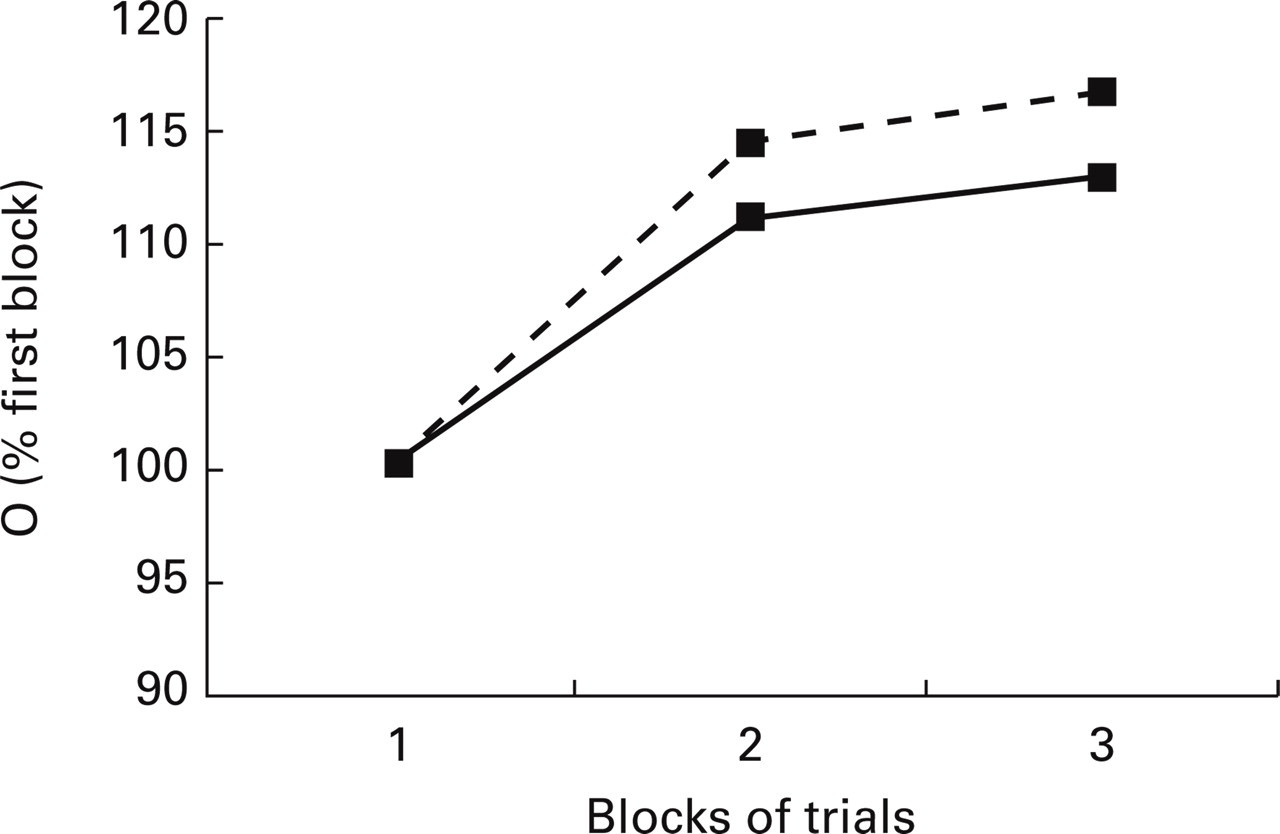
The normal synapse model NSM was found to habituate both at stimulus intensity 40 and 70 dB. In the case of the migraine synapse model MSM, however, sensitization took place instead (Fig. 2). The difference in the migraine and normal models lay partly in the higher strength of the potentiating synapse w 3 for the MSM (for the MSM, the factor a 3 had a value 2.1 as opposed to the value 1.5 for the NSM). This therefore leads to a greater value of O, the response to the stimulus.

Responses of normal synaptic (—▪—) and migraine synaptic (–––▪–––) network models upon repetitive presentations of stimuli. (a) Responses with 40 dB input. (b) Responses with 70 dB input.
In the MSM described above, sensitization was partly obtained because of a potentiating synapse w 3 that had a higher strength than that of the NSM. In the case where the strengths of the potentiating synapses were the same for both the MSM and NSM, we found that sensitization could still be achieved when the strength of the depressing synapse w 2 was low (a 2 =−1.8 for the NSM, a 2 =−1.2 for the MSM). As shown in Fig. 3, this MSM gave rise to a sensitization that was similar to that obtained with a strong potentiating synapse. The stimulus used in all cases was 70 dB. Both the cases of sensitization in Fig. 3 were achieved by processes that appear diametrically opposed. One calls for the increase in the strength of a synapse (stronger potentiating synapse for MSM), while the other calls for its decrease (weaker depressing synapse for MSM).
Sensitizing responses of migraine synaptic model in two different cases. One in which the strength of the potentiating synapse w 3 is higher than in the normal (–––▪–––) and the second in which the strength of the depressing synapse w 2 is lower than in the normal synaptic model (—▪—).
Dependence of the response amplitude on the stimulus intensity
By using the migraine synapse model (MSM) characterized by a strong potentiating synapse (high w 3 ), we found that the slope of the I–O curve, and hence the dependence of responses on stimulus intensity, is greater in MSM than in NSM (Fig. 4).

Higher dependence of amplitude of responses on stimulus intensity in the migraine synaptic model (high w 3 –––-; low w 2 –––) than in the normal model (—) as shown by the higher I–O slopes for the migraine synaptic model.
The same tests conducted for the MSM with a weak depressing synapse (low w 2 ) led to similar results & (Fig. 4).
Intensity dependence and sensitization of the model
The dependence of the amplitude of responses on stimulus intensity in MSM was found to be positively correlated with the sensitization of the same model (Fig. 5). The latter was altered by varying a 3 from 2.1 to 3.1. In Fig. 5 intensity dependencies expressed as the values of amplitude-stimulus function (ASF) slopes (in µV/10 dB) are plotted against values of a 3 . The higher the sensitization of the model, the higher the ASF slopes (or slope of I–O curve) and thus the intensity dependence.

Correlation between intensity dependence and strength of the potentiating synapse (a 3 ) in the migraine synaptic model.
A similar correlation between intensity dependence and sensitization was observed in the MSM with a weak depressing synapse. Once again the intensity dependence was found to increase with the increase of a 3 or sensitization (figure not included).
Discussion
A computer model of competing potentiating and depressing synaptic processes, based on a previously published model [16, 17], was used to reproduce the amplitudes of auditory-evoked responses of normal volunteers and migraine patients to repeated stimuli of different intensities [9]. The normal synapse model was found to habituate upon repetition of the stimulus. In the case of the migraine synapse model, however, the responses did not only fail to habituate, but in fact, increased in amplitude. This is in keeping with experimental observations in migraine patients [3].
This study on the relationship between synaptic alterations, habituation and IDAP has been carried out using a simple neuronal network model. The nervous system is certainly infinitely more complex than any of the models currently available, or the model that has been presented in this paper. The purpose of computer modelling, however, is to find the simplest mechanism that can reproduce the phenomenon being studied, i.e. to abstract certain essential characteristics of the phenomenon. This simple model should then be used to gain insights into the phenomenon being studied and used to make predictions. A more complex model should only be constructed once the simple model is no longer able to ‘fit’ the experimental data and the questions being addressed. This approach has been very successfully applied in the field of neuroscience, e.g. much insight and successful experimental predictions have been made using thalamocortical models where a neurone is represented as a unicompartmental R-C circuit with no dendritic trees [13, 14, 15].
The sensitization in the migraine synapse model, as opposed to the habituation in the normal synapse model, could be achieved using two different types of neural network frameworks. In the first one, the potentiating synapse was stronger than in the normal synapse model; in the second, the depressing synapse was weaker. This observation may help to address an existing puzzle in the genetic studies of the pathogenesis of migraine. There is ample evidence for a genetic involvement in migraine [20]. In familial hemiplegic migraine (FHM), a rare form of migraine with aura, mutations have been found in the alpha 1 A subunit gene of the neuronal P/Q-type Ca2+ channel (CACNA1A on chromosome 19p13) [21]. Sibpair and linkage analyses [22, 23, 24] point to the involvement of this locus also in the common forms of migraine, where some indirect signs of possibly dysfunctioning P/Q Ca2+ channels have already been provided [25, 26]. P/Q Ca2+ channels control neurotransmitter release and neuronal excitability.
In vitro, the CACNA1A mutations detected in migraine may have opposite effects on Ca2+ channel function, some producing a gain, others a loss of function [27, 28]. If this is true also in migraineurs, it may seem difficult to understand how opposite dysfunctions may lead to the same disorder and the same electrophysiological pattern. As these ionic channels play a role in determining the efficacy of a synapse, their dysfunctions may lead to both increase and decrease of synaptic strengths. The results from the model may help to explain how two opposing changes might give rise to the same disorders. This paragraph should not be taken to indicate that we consider only P/Q Ca2+ channel dysfunctions as responsible for abnormalities in habituation and sensitization.
As mentioned before, habituation involves complex neurobiological mechanisms, among them neuronal excitability and transmitter release (e.g. in the Aplysia model [18]). The P/Q Ca2+ channels are known to play a pivotal role in neurotransmitter release [29]. However, a myriad of factors at the synapse, such as the dissociation constant of a ligand for a receptor, the number of post-synaptic receptors, the rate of re-uptake of a neurotransmitter, alterations in the N-type Ca2+ channels in the pre-synaptic terminal, etc., could modify the synaptic strength. Our model only sheds light on how the same abnormalities in electrophysiological responses may be obtained even with opposing signs and location (potentiating or depressing synapse) of the synaptic dysfunction.
Output from the synaptic model also offers an explanation for the stronger stimulus intensity dependence of cortical responses observed in migraine patients compared with healthy volunteers. This phenomenon was indeed found to be displayed by the same neural network model that showed deficiencies in habituation. The migraine synapse model with a higher sensitization also displayed a higher dependence on stimulus intensity. Assuming that a model with a higher sensitization should necessarily show a higher dependence on stimulus intensity is not correct. If the inputs from the different supra-threshold intensities produced indeed the same level of sensitization, there would be no significant increase in response amplitude with increasing intensity. Our observation may thus suggest that the two electrophysiological patterns in migraineurs, namely the lack of habituation and the strong intensity dependence, may be explained as a unified phenomenon.
Results from the model predict that migraineurs who demonstrate a higher stimulus intensity–response slope should also show a greater sensitization to repeated stimuli. A preliminary study of the degree of sensitization and IDAP has been performed for auditory-evoked potentials. It shows indeed a positive correlation between the sensitization at high stimulus intensities and IDAP [30].
Further studies using the model would have to take the following points into account. The first is that the conclusions of this paper were based on results that were obtained for auditory stimuli and on parameters required to reproduce patterns observed for this modality. A repetition of these results using the parameters from different sensory modalities will ensure the generality of the conclusions drawn here. Secondly, the stimulus that was used in the model was a square pulse. The stimulus used in the experimental studies, however, was a pulse of 50 ms with 10 ms rise and fall times. Future improvements of the model should take this aspect into account. Finally, even though synaptic depression is known to play the primary role in several classic models of habituation, a more complex network including inhibitory circuits could be constructed to study the role of inhibition in shaping the network output.
Conclusions
A classical synaptic model of competing potentiating and depressing synapses [16, 17] was used to model cortical information processing of healthy volunteers and migraine patients. The classical model was adapted to reproduce the results of a study of auditory-evoked potentials [9] and the resulting models were used to investigate possible underlying mechanisms of habituation and intensity dependence abnormalities such as those found in migraineurs. Our main results can be summarized as follows. (i) This classical synaptic model of habituation was adapted to result in habituation (normal synapse model, NSM) or sensitization (migraine synapse model, MSM). (ii) Sensitization of the MSM could be achieved in two ways: by increasing the strength of the potentiating synapse or diminishing the strength of the depressing synapse. (iii) The sensitizing model (MSM) produced a higher stimulus intensity dependence than the habituating model (NSM), suggesting a common underlying neuronal mechanism for these two abnormalities observed in migraine patients. (iv) The amount of sensitization correlated with the magnitude of the dependence on stimulus intensity predicting a similar correlation in vivo.
Footnotes
Acknowledgements
This study was supported by grant no. 3.4523.00 from the Belgian Fund for Medical Research (Brussels, B), grant no. 125 from the Migraine Trust (London, UK), Belgium and Concerted Research Action 99/044–241 from the French Community of Belgium.




