Abstract
Acute headache is a very frequent symptom, responsible for significant demand at primary care units and emergency rooms. In such sets in Brazil, metamizol is easily found but, on the other hand, neither ergotics nor triptans are available. The aim of this study is to compare intravenous metamizol with placebo in the acute treatment of migraine with aura, migraine without aura and episodic tension-type headache. Fifty-four migraine with aura patients, 95 migraine without aura patients and 30 tension-type headache patients were treated with metamizol. Ninety patients (30 migraine with aura, 30 migraine without aura and 30 tension-type headache patients) received placebo. Pain intensity, nausea, aura, photo- and phonophobia were investigated at 30 min and 60 min after the administration of the drug. Significant improvement of pain after 30 min and 60 min post-dosage was achieved from metamizol groups compared with placebo groups. Significant improvement of all other symptoms was achieved after 60 min post-dosage. Side-effects were mild and with small incidence. Metamizol is an effective, safe and low price drug. It may be regarded as a good alternative drug for the treatment of common acute primary headaches.
Introduction
In the majority of countries, acute headache treatment is performed by general practitioners both in out-patient and in-patients sets (1). Several studies show that headache is responsible for a significant share of the health units' demand (2, 3). Acute headache is responsible for about 9% of all medical consultations in the primary care units of Brazil (4). However, in most of these units and even in public Hospitals in Brazil, as probably in other emerging countries, neither ergotics nor triptans are easily available (5–7). The therapeutic arsenal is restricted to painkillers, non-steroidal anti-inflammatory drugs and neuroleptics.
The acute medication treatment of primary headaches, in particular migraines, has been extensively studied. Special attention has been paid to the study of ergotics and triptan compounds. Other kinds of acute treatments are not so easily seen. This is the case with metamizol, which is commonly used in Brazil. As a matter of fact, two previous studies carried out in emergency units in Brazil (8, 9) have shown that i.v. metamizol is the most frequently used drug for the treatment of acute headaches. This fact suggests that metamizol is regarded as a very effective drug for physicians working in these units. However, we were not able to find in the literature a single protocol recommending the use of metamizol for the acute treatment of migraine and tension-type headache.
The aim of this study is to evaluate, in a controlled way, the effectiveness of i.v. metamizol in the treatment of migraine and episodic tension-type headache (ETTH).
Subjects and methods
Data were gathered from two primary health care units located in the inland of São Paulo State in Brazil. One of them is located downtown in the city of São Carlos serving the whole city population (about 160 000). The other unit is in the outskirts of the city of Ribeirão Preto and serves a population of 120 000 inhabitants.
The people who seek care in these units do not have prebooked appointments and usually suffer from a wide range of acute or chronic illnesses. About 9.3% of these patients come to the units due to headaches. All the individuals in this study were examined by the same physician (M.E.B.), who is a neurologist, trained in the use of the International Headache Society (IHS) criteria for headache and facial pain (10). Patients suffering from migraine without aura (MO), migraine with aura (MA) and ETTH aged ≥18 years, who had not taken any medicine for that headache attack, were enrolled into this study after giving informed consent. Patients on prophylactic treatment for migraine were excluded from the study.
The study was ‘single-blind’, prospective. The patient did not know if he was taking metamizol or placebo. First, 179 consecutive patients seen by the physician (M.E.B.) with the diagnoses of MO, MA and ETTH, were treated with metamizol (metamizol group, n = 179). Later, the following consecutive 30 patients of each one of the above diagnostic categories (namely MO, MA and ETTH) were treated with placebo (placebo group, n = 90).
After clinical examination and before the drug administration, patients were asked to answer a questionnaire about: (i) pain intensity (the patient should score on a 0–10 scale, where 0 means no pain and 10 the maximal pain intensity); (ii) intensity of nausea, phonophobia, photophobia and aura that were quantified as following, according to the patient's own opinion: none (0), mild (1), moderate (2) or severe (3). Later on, the patient went to the observation room. Saline at 1 ml/min was administrated through a peripheral vein. Out of the patient's sight, 1000 mg of metamizol (2 ml diluted in 8 ml of saline) was administrated (metamizol group), or alternatively 10 ml of saline were given to the patients from the placebo group. After receiving the active drug or placebo, patients were allowed to remain lying on a bed, in a collective room (observation room), which is very bright, without acoustic isolation and sometimes busy. Thirty and 60 min post-dosage, patients were asked to respond to the same above-mentioned questions. Sixty minutes after dosage, if the headache were deemed to be severe enough to receive rescue treatment, i.v. chlorpromazine, 0.1 mg/kg, was given. They were also asked about any adverse events. Any event was considered adverse which was a symptom that the patient did not present before drug administration (any new symptom), even those which could be deemed part of a migraine attack and regardless of relationship to the use of the study medication. This means that, if a given migraine patient had no nausea prior to or during drug administration, and such a symptom was present 30 and/or 60 min later on, nausea was to be considered an adverse event.
Statistic procedures used were the following: χ2 test in contingency tables for gender comparison; for other comparisons, non-parametric Mann–Whitney test was used. For the comparison of data from the same group of individuals at 30 min and at 60 min post-dosage, the Wilcoxon non-parametric test for paired data was used.
This study received the approval by the Committee on Ethics in Medical Research from the Institution.
Results
Migraine without aura
From 95 MO patients undergoing metamizol treatment, 69 (72.6%) were females and 26 (27.4%) were males. The average age was 32.8 years (
Comparison of pain intensity between the metamizol group and the placebo group is presented in Fig. 1. Significant decrease in pain intensity is seen as soon as t = 30 min (P < 0.001), which persisted at t = 60 min. Comparisons between the metamizol group and the placebo group with regard to nausea, photo- and phonophobia are shown in Fig. 2. All of these accompanying symptoms showed significant improvement in the metamizol group when compared with the placebo group at t = 30 min and t = 60 min. The exception was photophobia, in which significant improvement was observed only at t = 60 min.

Migraine without aura: time vs. pain intensity. Comparison between metamizol and placebo. ∗∗P < 0.01. ▪, Metamizol; •, placebo.

Migraine without aura: time vs. associated symptoms. Comparison between metamizol and placebo. ∗P < 0.05; ∗∗P < 0.01. •, Metamizol; ▪, placebo.
Migraine with aura
Of 54 MA patients undergoing metamizol treatment, 37 (68.5%) were females and 17 (31.5%) were males. The average age was 36.4 years (
Comparison of pain intensity between the metamizol group and the placebo group is presented in Fig. 3. Similar to MO patients, a significant decrease in pain intensity was observed as soon as t = 30 min (P < 0.001) and such a significant decrease persisted at t = 60 min. Comparisons between metamizol and placebo groups for nausea, photo-, phonophobia and aura are shown in Fig. 4. The IHS criteria were considered to define aura. All of these symptoms showed significant improvement (P < 0.001) in the metamizol group when compared with the placebo group at t = 30 min and t = 60 min. The exception was aura intensity, which had significant improvement only after 60 min.
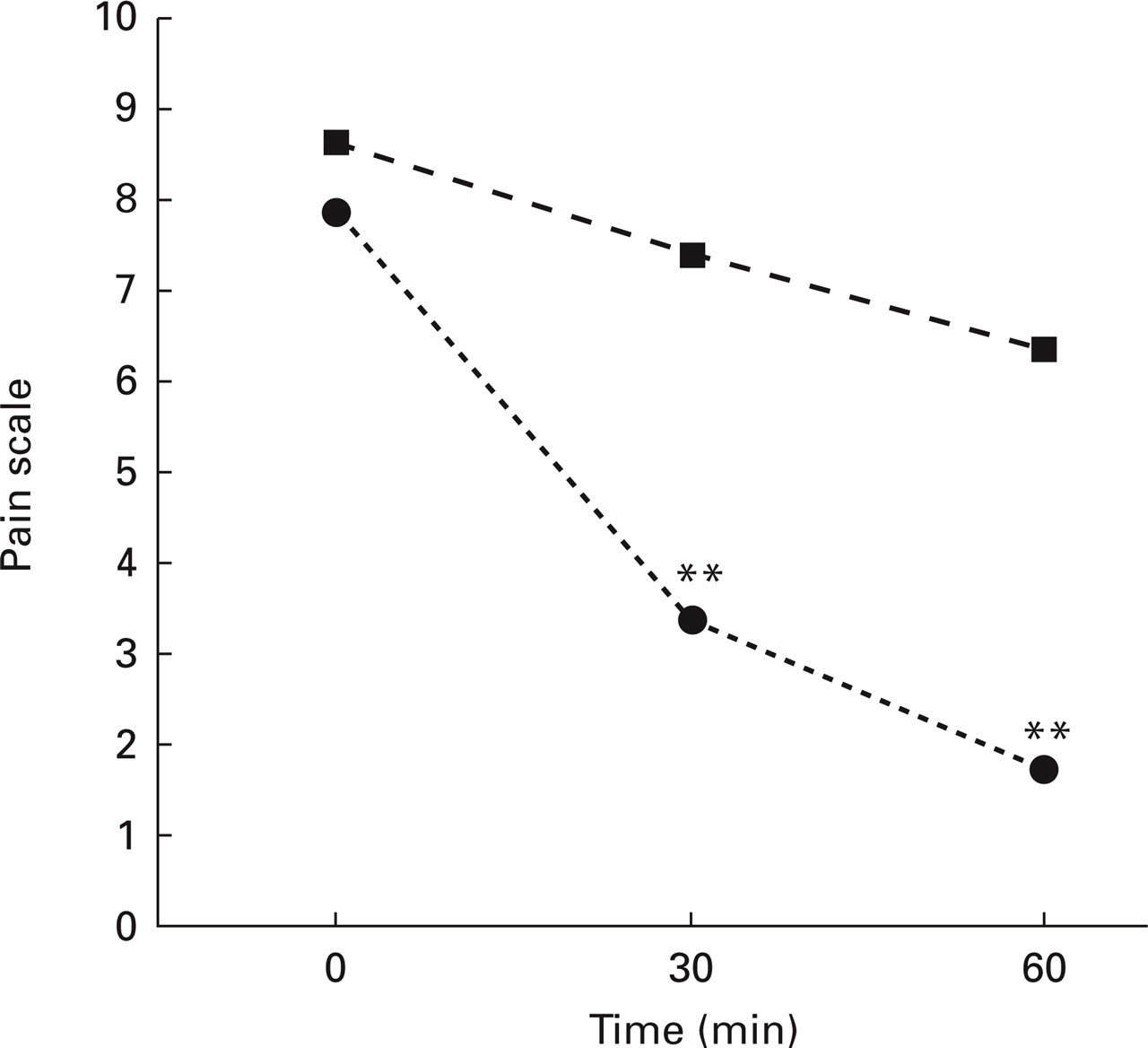
Migraine with aura: time vs. pain intensity. Comparison between metamizol and placebo. ∗∗P < 0.01. •, Metamizol; ▪, placebo.
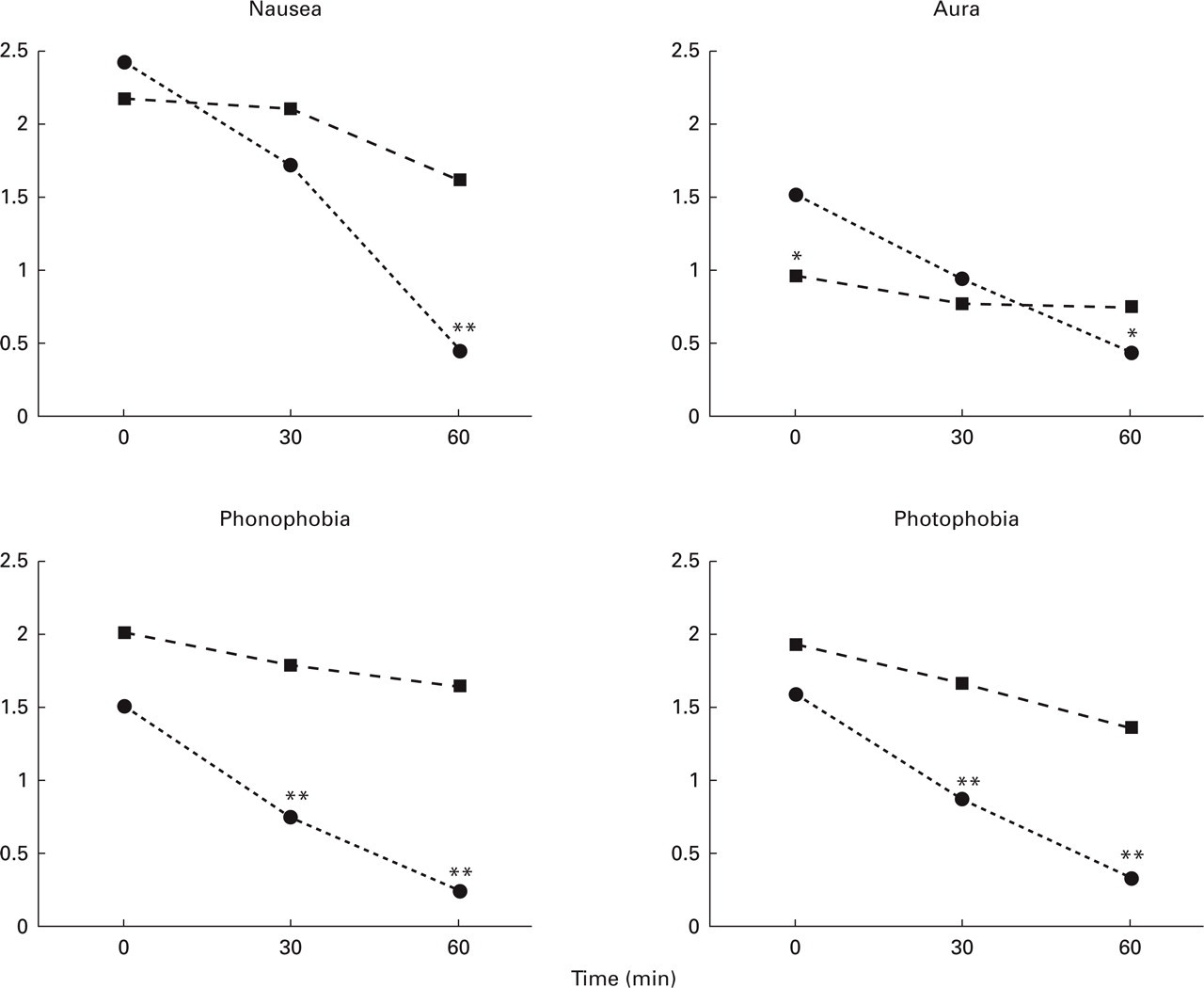
Migraine with aura: time vs. associated symptoms. Comparison between metamizol and placebo. ∗P < 0.05; ∗∗P < 0.01. •, Metamizol; ▪, placebo.
ETTH
Of 30 ETTH patients undergoing metamizol treatment, 15 (50%) were females and 15 (50%) were males. The average age was 43.1 years (
Comparison of pain intensity between metamizol and placebo groups is shown in Fig. 5. Similar to MO and to MA groups, a significant decrease in pain intensity was seen as soon as t = 30 min (P < 0.05) and this difference was maintained at t = 60 min (P < 0.001).

Episodic tension-type headache: time vs. pain intensity. Comparison between metamizol and placebo. ∗P < 0.05; ∗∗P < 0.01. •, Metamizol; ▪, placebo.
Adverse effects
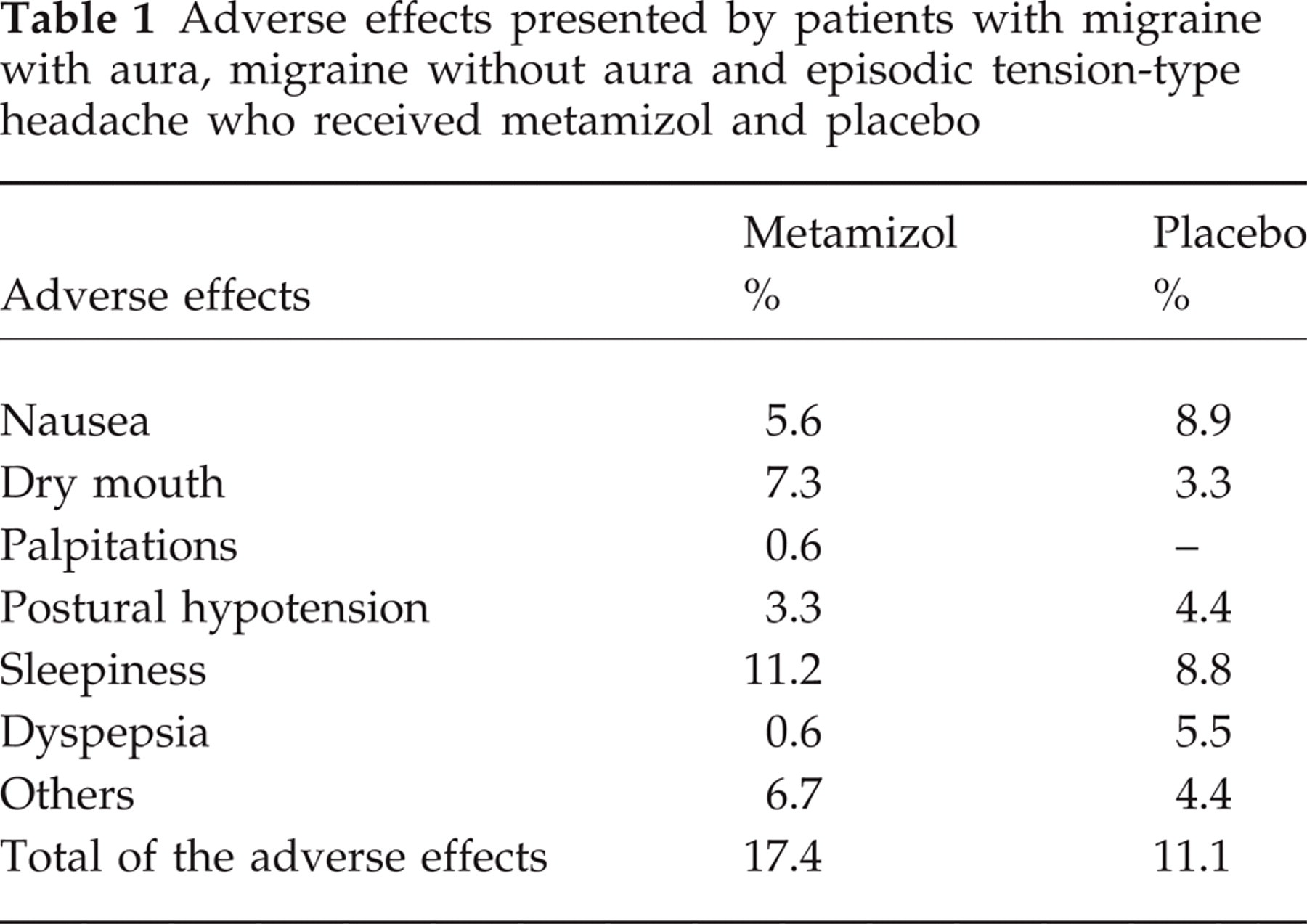
The adverse effects reported by the patients are presented in Table 1. They were seldom reported, usually mild or moderate. Only one patient who took the metamizol reported dyspepsia. Another one, an ETTH patient, reported palpitations 40 min after taking metamizol. However, her heart rate was 72 beats per minute, and an electrocardiogram recorded at the time the patient was symptomatic showed no relevant alterations.
Adverse effects presented by patients with migraine with aura, migraine without aura and episodic tension-type headache who received metamizol and placebo
Discussion
This trial, comparing metamizol with placebo, shows that metamizol is significantly superior to placebo in terms of pain relief at 30 min (P < 0.01) and at 60 min (P < 0.001). For all other migraine symptoms there was also significant improvement in the metamizol group when compared with placebo.
Metamizol is a pyrazolonic-derived, non-opiate analgesic drug, with antipyretics and spasmolytic properties. It was introduced for therapeutic use in 1922. It is a highly hydrophilic compound, and when in solution it is quickly hydrolysed in metil-amino-antipirine (MAA). Its analgesic effect ranges in peak from 20 to 45 min after the i.v. administration. Its active metabolites are MAA and amino-antipyrine (AA). The half-life of MAA/AA complex is about 2.7 h. The excretion is predominantly renal (11, 12).
Metamizol is an effective analgesic known world-wide. Its effectiveness has been shown in several painful situations, such as post-surgery pain, odontalgias and oncologic surgeries (13–15).
Our study shows that metamizol is also an effective drug in the acute treatment of common primary headaches.
It is known that there is no relationship between the analgesic effect of metamizol and its ability to inhibit prostaglandins (16). It is also known that metamizol acts in two main ways: in the periphery, turning nociceptor insensitive to pain activation (17), and centrally, acting on periaquedutal grey substance, activating the pain inhibitory pathways (18). An interesting finding arising from the present study is that metamizol action was more than analgesic: it produced significant relief of phono-, photophobia, nausea and aura.
The action of metamizol on the aura makes possible some speculation. There is evidence suggesting that migraine might be due to a cannalopathy (18, 19). Neuronal P-type calcium channels regulate 5-hydroxytryptophan (5-HT) liberation. Malfunctioning calcium channels might decrease 5-HT liberation from neurones, leading to migraine attack and making it difficult for these attacks to end. Besides, it is known that migraineurs have a decreased brain magnesium (20). Decreased brain magnesium and a cannalopathy may be relevant in starting cortical spreading depression, a phenomenon that is accepted as related to migraine aura. Further, aura is said to be associated with a state of neuronal hyperexcitability (21), and with increased CSF excitatory amino acids, glutamate and aspartate (21), and with low levels of magnesium in CSF. Metamizol might have an action on membrane permeability and ionic channels or might act on the excitatory amino acids or even on NMDA receivers. The fact is that somehow, this drug may have an action on the mechanisms underlying migraine aura.
The safety of metamizol has been called into question for many years due to the possible risk of agranulocytosis. This risk was accepted to be > 0.1% in the 1970s. However, in many countries, among them Brazil, the drug is widely used and considered safe. More recent studies (22) have shown that the risk of agranulocytosis is lower than 1.1 per million users and that the risk of aplasic (verifica) anaemia is close to zero. Unlike aspirin and others non-steroidal anti-inflammatory drugs, metamizol is devoid of gastrointestinal tract side-effects. Long-term adverse event evaluations were not performed in this study due to the fact that large multicentre studies had already shown the metamizol safety profile (22–24).
The small amount of adverse effects seen in the present study shows that metamizol has a good tolerability profile. In conclusion, i.v. metamizol is an inexpensive and safe drug, effective not only for pain relief, but also for the relief of migraine-associated symptoms. It should be regarded as an excellent alternative for the acute treatment of migraines and ETTH. It is our opinion that double-blind, randomized, placebo-controlled multicentre studies are needed in order to determine the proper role metamizol may play in the treatment of acute headaches.
