Abstract
The involvement of Gi proteins in the modulation of pain perception has been widely established, and mutations in G-proteins have already been identified as the aetiopathological cause of human diseases. The aim of the present study was to determine whether a deficiency or a hypofunctionality of the Gi proteins occurred in primary headache. The functionality and the level of expression of Gi proteins were investigated in lymphocytes from migraine without aura, migraine with aura and cluster headache sufferers. A reduced capability to inhibit forskolin-stimulated adenylyl cyclase activity in headache patients was observed. Migraine patients also showed basal adenosine cAMP levels about four times higher than controls. The reduced activity of Gi proteins seems not to be related to a reduction of protein levels since no significant reduction of the Giα subunits was observed. These results indicate Gi protein hypofunctionality as an aetiopathogenic mechanism in migraine and cluster headache.
Introduction
Headache is the most common pain syndrome. It is also the most frequent symptom in neurology, where it may be a disease in itself (primary headache) or indicate an underlying local or systemic disease (secondary headache). In the complete absence of physical and laboratory alterations, diagnosis of primary headache remains purely clinical, based on the detailed description of symptoms by the patients. The criteria for classification of headache disorders was proposed in 1988 by the International Headache Society (IHS) (1) that classified primary headache into three major clinical subtypes: migraine, cluster headache and tension-type headache. Migraine is a multifaceted disorder of which the head pain is only one component. It is an extremely frequent and sometimes incapacitating condition and female preponderance is a characteristic feature (2). The two most frequent varieties of migraine are migraine without aura and migraine with aura in which neurological symptoms precede or accompany the headache (2).
Numerous factors can trigger or predispose to migraine attack. Some are identifiable, such as stress, hormonal and dietary factors, while the majority remain unknown. The exact pathogenesis of primary headache is, indeed, still unknown. Many theories have been elaborated, but none can account for all the clinical features and pathophysiological aspects of this syndrome. The ‘vascular’ hypothesis, which states that the headache phase of migrainous attacks is caused by vasodilatation and that the neurologic symptoms, occurring during the migraine prodrome, are produced by intracranial vasoconstriction (3), was widely accepted for many years. The ‘serotoninergic’ hypothesis implicated a primary neuronal origin in migraine which may represent a hereditary perturbation of serotoninergic neurotransmission. It was found that platelet levels of serotonin fall consistently at the onset of headache and migrainous episodes may be triggered by drugs that release this biogenic amine from tissue store (4). The ‘trigeminovascular’ hypothesis is one of the most accepted theories of migraine origin. A depolarization of the trigeminal ganglions or its perivascular nerve terminals activates the trigeminovascular system, giving rise to central transmission of nociceptive information and retrograde perivascular release of powerful vasoactive neuropeptides, such as CGRP, substance P, neurokinin A (5, 6). In more recent years, however, it was supposed that in migraine both vascular and neural components are relevant and most probably interrelated (7). Recently, ‘neuronal hyperexcitability’ has been proposed as the biological basis for susceptibility to migraine. Enhanced excitability of cell membrane, perhaps in part genetically determined, renders the brain susceptible to attacks. Factors that increase or decrease neuronal excitability determine the threshold for triggering attacks (8). However, from the numerous studies conducted so far, no single receptor abnormality has emerged that can completely explain why some people are more prone to suffer from migraine than others. These considerations led us to look beyond the receptor at the more universally distributed components of cell signalling that are G-proteins.
Nearly all inhibitory neurotransmitters able to enhance the pain threshold utilize Gi proteins as signal transduction system. G-proteins are heterotrimeric molecules with α, β and γ subunits. The α subunits can be classified into families, depending on whether they are targets for cholera toxin (Gs), pertussis toxin (Gi and Go) or neither (Gq and G12) (9). Gi proteins represent the most widespread modulatory signalling pathway in neurones (10) and are responsible for inhibition of adenylate cyclase activity and modulation of several K+ and Ca2+ channels in order to reduce cell excitability (11, 12). Involvement of Gi proteins in the modulation of pain perception has been well established. The administration of pertussis toxin, which selectively inactivates Gi proteins (13), produced hyperalgesia and allodynia in laboratory animals (14, 15), clearly indicating that a lack of functionality of Gi proteins enhances the sensitivity to pain. Hypofunctionality of Gi proteins also produced insensitivity to analgesic treatments. It has been observed that pertussis toxin prevents the enhancement of the pain threshold induced by widely used analgesic drugs such as opioids, antihistamines, and tricyclic antidepressants (14, 16, 17).
Mutations in G-proteins have already been identified as the aetiopathological cause of human diseases. Alterations at Gsα subunit level have been observed in the Albright hereditary osteodystrophy, an autosomic dominant genetic pathology, as well as in the acromegaly, hyperfunctional tyroid nodules and McCune–Albright syndrome (18).
The aim of the present study was to determine whether a deficiency or a hypofunctionality of the Gi proteins occurs in headache. The functionality and expression levels of Gi proteins were therefore investigated in lymphocytes from patients with clinically well-defined primary headache.
Patients and methods
Participants
Eighteen healthy volunteers, 21 primary-headache and 12 painful-disease (neuropathic pain, arthrosis, rheumatoid arthritis) sufferers participated in the study after informed consent was obtained from all. Patients were drug-free for a period of at least 1 week and had undergone laboratory and physical examination to exclude the presence of other concomitant infections. Furthermore, healthy subjects had no headache familial history. Patients suffering from primary headache were clinically classified following the criteria established by the IHS (1) and divided into three subgroups: migraine without aura, migraine with aura and cluster headache. Patients belonging to the painful diseases group were classified as suffering from arthrosis, rheumatoid arthritis and neuropathic pain mainly deriving from entrapment syndromes. For all patients the visual analogue scale (VAS) value, which represents an arbitrary indication of the pain experienced during the attack, was evaluated. Blood samples were collected during the quiescent state (period between migraine attacks or cluster periods) and experiments were performed in blind.
Adenylyl cyclase activity assay
Lymphocytes were isolated as described by Böyum (19). Mononuclear cell counts were made. After permeabilization with the detergent digitonin (10 μg/ml) (20), lymphocytes from healthy subjects were preincubated with or without pertussis toxin (PTX) at the concentration of 100 ng/ml at 37°C for 90 min, whereas lymphocytes from headache and painful disease sufferers were all preincubated without PTX. After preincubation, lymphocytes (1–2×106 intact cells/assay) were incubated either with the vehicle dimethyl sulphoxide (DMSO) or forskolin 10−4
Western blot analysis
Aliquot of lymphocyte lysates (100 ml), or recombinant Gi2α and Gi3α, used as reference proteins, were boiled for 5 min with 30 ml of sample buffer (4% SDS, 20% glycerol, 10% 2-mercaptoethanol, 125 m
Specific bands were detected using the ECL system (Amersham) according to the manufacturer's instructions. (Densitometric analysis of the bands obtained was carried out using the Scion image program.)
Statistical analysis
Results are given as the means±
Results
Clinical characteristics of patients
As reported in Table 1, all patient groups showed a comparable VAS value indicating homogeneity of intensity of pain experienced. Laboratory values in patients affected by headache were similar to healthy controls (data not shown).
Clinical parameters of patients suffering from headache and painful diseases



VAS, Visual analogue scale.
Evaluation of Gi protein functionality
The functionality of Gi proteins was assessed in peripheral blood intact lymphocytes by investigating their ability to inhibit adenylyl cyclase activity. We first stimulated adenylyl cyclase by using forskolin 10−4

Evaluation of adenylyl cyclase activity in lymphocytes from healthy and migraine subjects. (A) Lymphocytes from healthy subjects were preincubated with or without pertussis toxin (PTX). ∗P < 0.05; ∗∗P < 0.01 vs. forskolin-stimulated lymphocytes. ○, Healthy; •, headache; □, PTX. (b) Lack of reduction of cAMP levels in patients suffering from migraine without aura (MO; ▪), migraine with aura (MA; □) and cluster headache (CH; •). ∗P < 0.05 vs. corresponding value of headache group. ○, Healthy.
Increasing concentrations of Gpp(NH)p produced a dose-dependent reduction of cAMP levels in lymphocytes from neuropathic pain sufferers, patients affected by rheumatoid arthritis or arthrosis similar to that observed in healthy subjects. The values indicating the adenylyl cyclase activity were reported as difference (Δ) between forskolin and forskolin+Gpp(NH)p 10−5
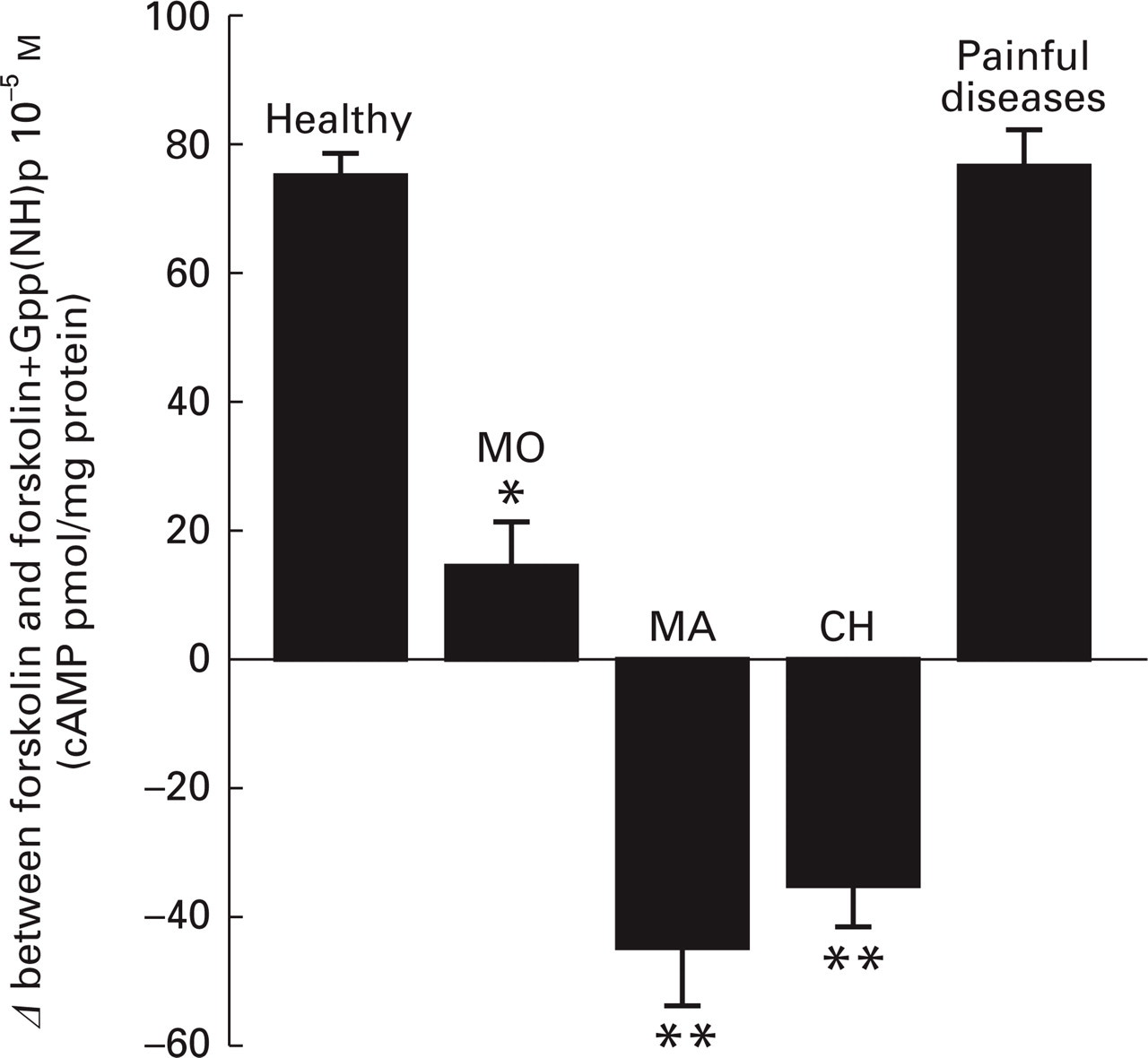
Evaluation of adenylyl cyclase activity in lymphocytes from headache patients in comparison with patients suffering from neuropathic pain, rheumatoid arthritis and arthrosis. ∗P < 0.05; ∗∗P < 0.01 vs. healthy group. MO, Migraine without aura; MA, migraine with aura; CH, cluster headache.
Determination of cAMP basal levels
Under standard conditions of assay, in healthy subjects basal levels of cAMP were 21.2 ± 2.6 pmol/mg protein. Even if there was no statistically significant correlation of cAMP levels with age or sex, older subjects showed tendentially slightly higher levels of cAMP (data not shown). Primary headache patients showed a highly significant enhancement of basal cAMP levels when compared with healthy subjects. This effect was particularly evident in patients suffering from migraine with or without aura rather than in cluster headache sufferers, since the cAMP basal levels where, respectively, about four times and twice higher than controls. By contrast, the basal cAMP levels of patients suffering from neuropathic pain, arthrosis and rheumatoid arthritis were not statistically different from healthy subjects. Patients with arthrosis differed from other groups because, even if they maintain Gi protein functionality, they showed higher levels of cAMP (Fig. 3).

Evaluation of basal (▪) and forskolin-stimulated (□) cAMP levels in patients suffering from migraine without aura (MO), migraine with aura (MA), cluster headache (CH), rheumatoid arthritis, neuropathic pain, and arthrosis. ∗P < 0.05 vs. healthy subjects.
By comparing forskolin-stimulated levels of cAMP, no statistically significant difference was seen among values observed in healthy subjects and in all the investigated groups of patients (Fig. 3).
Quantitative determination of Giα levels
The expression of Giα and of the subtypes Gi2α and Gi3α proteins were investigated. As illustrated in Fig. 4, levels of Giα, Gi2α and Gi3α, quantified by optical densitometry, were similar in lymphocytes from both healthy subjects and headache sufferers. Some representative examples of Western blot analysis bands are reported as inset.

Evaluation of Giα protein levels in lymphocytes of patients suffering from primary headache (M; ▪) in comparison with healthy (H; □) subjects. Data are reported as percentage of corresponding Giα subtype standard. Inset: bands obtained by Western blot analysis.
Discussion
Present results showed that lymphocytes from headache patients showed a reduced capability to inhibit forskolin-stimulated adenylyl cyclase activity, clearly showing a hypofunctionality of Gi proteins. The observed impaired capability to reduce cAMP levels in headache sufferers was similar to that produced by preincubation of lymphocytes from healthy subjects with pertussis toxin at a concentration consistent with a complete ADP-ribosylation of Gi proteins (25), further indicating a selective hypofunctionality of Gi proteins. All headache subtypes investigated, even if they had different clinical characteristics, showed a similar reduction of inhibitory functionality, suggesting a fundamental role of Gi proteins in the aetiopathogenesis of both migraine and cluster headache.
Lymphocytes from headache suffering subjects showed higher basal levels of cAMP. These results indicate that, at resting conditions, there is a disequilibrium between the inhibitory and stimulatory system of this enzyme, mediated, respectively, by Gi and Gs proteins, confirming the hypothesis of a hypofunctionality of Gi proteins. An alteration at the level of the adenylyl cyclase can also be excluded, since the direct stimulation of the enzyme by forskolin did not reveal any difference in cAMP levels produced between patients and healthy subjects.
Among the several classes of compounds that have been investigated as antimigraine agents, dihydroergotamine or triptans represent the reference drugs for acute migraine treatment (26). These compounds exert their activity by inducing vasoconstriction of cranial vessels, even if it is not completely elucidated whether these drugs exert their therapeutic action through an effect on 5-HT1B/D receptors or via a direct effect on vessels (27). It has long been known that cAMP is a potent vasodilator of cerebral vessels. The relaxation of vascular smooth muscle produced by compounds that activate adenylate cyclase, such as β-adrenergic agents and forskolin, is thought to depend on their capability to increase the concentration of cAMP (28). Rosenblum (29) demonstrated the existence in mice of an adenylyl cyclase–cAMP system for dilating cerebral arterioles. Furthermore, it has been reported that a suffusion with cAMP produced a concentration-dependent sustained vasodilatation of the resting pial arteries in rats (30). The clinical efficacy of vasoconstrictor agents is in agreement with our experimental findings. We observed higher basal levels of cAMP specifically in migraine patients, and this biochemical alteration could represent the origin of the vasodilatation which, in turn, represents the cause triggering the migraine attack. Further confirmation of this hypothesis is provided by the observation that among the drugs employed to prevent effectively migraine attacks are the β-blockers (3) which are responsible for lowering the intracellular levels of cAMP.
The most recent theory of headache pathogenesis considers brain hyperexcitability as the biological basis for susceptibility to migraine (8). This theory is based on some studies which showed that occipital cortex neurones may be hyperexcitable in some 90% of patients suffering from migraine with aura who experience visual disturbances as part of their aura (31). Furthermore, psychological studies of visual discrimination showed that migraine sufferers had a greater sensitivity for low-level visual processing between attacks, showing functional impairment of inhibitory interneurones (32). It has also been reported that cortical and precortical visual and auditory processing in migraine patients, investigated by means of pattern-reversal visual evoked potentials (VEP) and auditory cortical evoked potentials (IDAP), is impaired between attacks (33, 34). Our results obtained with migraine sufferers are not only in agreement with the above-mentioned hypothesis, but the observed hypofunctionality of Gi proteins can represent the biological mechanism responsible for the cell hyperexcitability. It is well known, indeed, that Gi protein activation reduces cell excitability (12). A deficiency of the Gi protein-mediated inhibitory system increases cell excitability, leading to a hypersensitivity to stimuli that can also represent the main cause of the symptoms experienced by headache sufferers, such as hypersensitivity to pain, photophobia, phonophobia, etc.
Gi proteins are coupled to many receptor types and subtypes. It is also well known that Gi protein levels can be down-regulated at several levels, such as turnover, transcription, etc., by hormones and neurotransmitters. Considering that all subjects included in this study were drug-free for at least 1 week, we can assume that the observed Gi protein hypofunctionality represents an individual's predisposition to headache rather than a down-regulation produced by the pharmacological treatment.
It can also be excluded that the observed hypofunctionality of Gi proteins could be an adaptive response to a condition of chronic pain. Patients suffering from painful diseases such as neuropathic pain, arthrosis or rheumatoid arthritis, who experienced pain sensations comparable to headache sufferers, as indicated by the VAS values, showed an unaltered Gi protein functionality.
As the observed hypofunctionality of Gi proteins could imply a reduction of protein expression, levels of Gi proteins were determined in cell membrane preparations from headache sufferers using Western blot analysis. To this purpose, antibodies against Giα subunits, which represent the functional subunit of Gi proteins (9), were employed. Furthermore, specific antibodies against Gi2α and Gi3α subtypes were used, since Gi2 represents the Gi protein subtype mainly involved in the inhibitory regulation of adenylyl cyclase activity in vivo (35) and it plays an important role in the modulation of pain perception. The administration of selective antibodies against Gi2α as well as the inhibition of its expression by the use of specific antisense oligonucleotides prevented the analgesia induced by agonists of μ-opioid receptors (36, 37). Similarly to Gi2, Gi3 has also been reported to be involved in pain processing, since antibodies against Gi3α prevented δ-opioid analgesia (38). No statistically significant reduction of Gi protein levels was observed in headache patients, leading us unable to establish from these data that Gi protein hypofunctionality is subsequent to a reduction of protein levels.
The lack of Gi protein reduction seems to be in contrast to recent data in which a reduction of about 50% of Gi2α mRNA was observed in lymphocytes from migraine patients (39). These results were obtained using Northern blot analysis and it is therefore plausible that this discrepancy could be attributed to the different recognition sites of the probes used for the determination of mRNA levels in comparison with those of the antibodies employed for Western blot analysis. The hypofunctionality of Gi proteins could be, in fact, due to the presence of a mutation in the protein sequence. In this condition, the use of a cDNA probe to quantify mRNA contents, wherever the mutation is located, may show a level reduction. Conversely, using Western blot analysis a reduction of Giα protein levels might not be shown, since the short segment of the protein with which the employed antibodies interact might not contain the mutation.
A structural alteration of the Gi proteins in headache sufferers can be excluded since there was no difference in the molecular weight of the α subunits of any Gi protein subtype investigated in comparison with healthy subjects.
The investigation into Gi protein functionality was conducted in lymphocytes from peripheral blood taking into account that Gi proteins have an ubiquitous distribution (40) and that blood samples can easily be obtained from patients in a painless manner. Lymphocytes represent an appropriate tool to detect Gi protein functionality since they have cell surface recognition sites for pertussis toxin, the selective inactivator of Gi, which are lacking in other blood cell types such as erythrocytes (41). Lymphocytes have also been widely used to investigate the aetiopathogenesis of human diseases at a biochemical and molecular level (42–46).
In conclusion, our results indicate that lymphocytes from migraine and cluster headache patients are distinguishable from lymphocytes not only from healthy subjects but also from patients suffering from other painful diseases.
Footnotes
Acknowledgements
The authors thank Dr Silvia Quattrone for her methodological and technical advice, Dr David Beccani for linguistic revision and all subjects and patients for their availability and confidence. The study was partially supported by grants from Boehringer Ingelheim and Florence University (ex 60%). A diagnostic kit obtained from these results is under patent of the University of Florence.
