Abstract
Sleep recordings were performed in eight patients to analyse sleep alterations preceding migraine attacks. Polysomnographic recordings from nights before an attack were compared with nights without following migraine. We analysed standard sleep parameters and electroencephalogram (EEG) power spectra. The main findings preceding migraine attacks were a significant decrease in the number of arousals, a decrease in rapid eye movement (REM) density, a significant decrease of beta power in the slow wave sleep, and a decrease of alpha power during the first REM period. The results suggest a decrease in cortical activation during sleep preceding migraine attacks. According to the models of sleep regulation, alterations in the function of aminergic or cholinergic brainstem nuclei have to be discussed.
Introduction
Although not widely recognized, many findings connect migraine and sleep. Noradrenergic and serotonergic neurotransmission together with the neuroanatomical sources, the locus coeruleus and the dorsal raphe, seem to be crucial in migraine pathophysiology (1, 2). Weiller et al. (3) studied patients suffering from migraine without aura using position emission tomography (PET) and found an increase in brainstem regional cerebral blood flow (rCBF) coinciding with the anatomical location of the dorsal raphe nucleus and the locus coeruleus.
According to the reciprocal interaction model of rapid eye movement (REM) sleep regulation, both structures are also involved in basic sleep regulation (4, 5). Suppression of REM sleep through serotonergic and noradrenergic activity has been reported (6, 7). Sleep abnormalities, especially in REM sleep, can be expected therefore in connection with a migraine attack. In fact, early morning headache has been associated with increased REM sleep and slow wave sleep as well (8). On the other hand, a decrease in REM latency was found in migraine patients during the interval between attacks (9). Self-reports showed lower subjective sleep quality and shorter sleep duration before migraine attacks (10). Objective parameters of sleep continuity in nights preceding migraine are therefore of interest.
The purpose of the present study was to compare polysomnographic recordings within the same subjects from the nights before migraine attacks with nights without following migraine. We studied alterations of sleep duration, sleep continuity, REM sleep and slow wave sleep.
Subjects and methods
Subjects
Patients were chosen for the study if their migraine attacks occurred during sleep or on awakening rather than at random times during the day. Eight patients (median age 48 years; six women and two men) spent up to four nights in our sleep laboratory and reported a migraine attack during or following one night (see Table 1). Seven patients diagnosed with migraine without aura according to the diagnostic criteria of the International Headache Society (IHS) (11) took part in the study. These patients showed a mean frequency of 3.9 migraine attacks per month. The patients slept in the laboratory when the onset of a migraine attack was most probable. One patient sleeping in our laboratory for other reasons reported a typical migraine attack with intense unilateral pulsating headache and vomiting which lasted for more than 4 h. Three other participants spent nights in the laboratory without developing a migraine attack.
Characteristics of patients

Six out of the eight probands reported frequent sleeping problems. The anamnesis was recorded and a physical examination performed by each patient. Four patients had accompanying disorders (see Table 1). Patient 1 had a mild hypertonus, but showed normal blood pressure values under treatment with metoprolol 47.5 mg/day. The study was approved by the local ethics committee and all patients gave their written consent prior to the study.
Sleep recordings and experimental design
Sleep was recorded employing standard procedures: horizontal electro-oculogram (EOG), submental electromyogram (EMG) and electroencephalogram (EEG) (C3-A2; C4-A1). Recordings were visually scored according to standard criteria (12) by a rater unaware of the experimental conditions. The following parameters were computed:
Total sleep time (in minutes)
Sleep efficiency (ratio of total sleep time to time in bed in percent)
Stages 1, 2, slow wave sleep (stages 3 and 4 combined) and REM, all expressed in percent of total sleep time
Latencies, i.e. time from beginning of recording to the first epoch of stage 2 (sleep onset latency) and from sleep onset to the first epoch of stage REM (REM latency) in minutes
Number of awakenings
Number of arousals (abrupt changes in EEG frequency > 3 s, according to the ASDA definition (13))
Total REM density (ratio of 3-s mini-epochs including rapid eye movements to the total number of 3-s mini-epochs per REM sleep of the whole night)
The C4-electrode was selected for calculation of the power spectra. Spectral analysis was performed using the fast Fourier transform (FFT) algorithm and the truncating error was reduced by applying a Hanning window. The EEG signals were A/D converted at 100 samples per second with a 12-bit resolution. Only artefact-free epochs of 30 s duration were analysed. Distinct frequency ranges were defined for the assessment of EEG power spectra: δ (1–3.5 Hz), θ (3.5–7.5 Hz), α (7.5–12.5 Hz), β1 (12.5–15 Hz), β2 (15–26 Hz) and β3 (26–30 Hz). The mean values were calculated for each sleep stage.
Sleep was recorded between light out and light on according to the wish of the patient. The first night in the sleep laboratory served the adaptation. The patients were questioned, whether migraine had occurred or not the following morning.
The aim was to record one night preceding migraine and one control night without a following migraine attack for each proband. The adaptation night was repeated if the interval between the sleep recordings was greater than 2 weeks.
Additionally, the patients completed questionnaires on habits and internal and environmental factors. Mood and sleep quality were estimated by a visual analogue scale (range: 1=very bad to 9=very good).
Statistical analysis
For descriptive statistics of sleep variables, mean and standard deviation (SD) were calculated. A non-parametric Wilcoxon test (two-tailed) was used to compare nights preceding migraine and control nights. The level of significance was set at 5%.
Results
We obtained polysomnographic recordings from eight patients, who reported typical migraine headache during or following a night in the sleep laboratory. Two patients reported nocturnal migraine headache at 3.10 and 4.30 a.m., respectively, and fell asleep again. Six patients noticed a beginning or present migraine attack while awaking in the morning. In two patients migraine occurred after the first, adaptation night in the sleep laboratory. Four patients reported a migraine attack following the second night and two patients after the third night of polysomnography.
Questionnaires and self-rating scales
Only one patient had the feeling of an oncoming attack in the evening before the migraine attack. Self-estimation of the mood using an analogue scale in the evening showed no differences between nights with following migraine and control nights (median and range: 6 (2–8) to 6 (4–7); scale between 1 (=very bad) and 9 (=very good)). The mood in the morning differed significantly: 4 (3–5) during the migraine attack and 6 (5–7) after the control night. The quality of sleep showed marginal differences (median and range: 5 (2–9) for the night with following migraine and 6 (4–7) for the control night).
Sleep recordings of the two patients with nocturnal migraine
Two patients suffered from a migraine attack during the night and fell asleep again (Figs 1 and 2). Patient 2 awoke out of non-rapid eye movement (NREM) sleep with migraine at 3.10 a.m. Slow wave sleep was seen less than 10 min before awakening. She took sumatriptan orally and fell asleep after 60 min, with a normal sleep profile including two REM periods (Fig. 1a). In comparison with the control night (Fig. 1b), the first sleep cycle showed a longer REM latency, a shorter REM duration and a lower total REM density. Patient 5 awoke out of REM sleep with migraine at 4.30 a.m. (Fig. 2a). The following sleep lasted 2 h and was interrupted by 10 short awakenings. Only five short awakenings occurred during the last 2 h of sleep in the control night (Fig. 2b). The sleep profile before migraine onset showed a longer REM latency and a lower total REM density.

Figures 1 and 2 show the sleep profiles of nights with migraine and control nights of two patients. The times of sleep are presented in the abscissa and the different sleep–wake stages, arousals and rapid eye movements (EM) in the ordinate. (a) This demonstrates that patient 2 awoke at 3.10 a.m. out of non-REM sleep (sleep stage 3 less than 10 min before awaking). The patient suffered from a migraine attack (indicated by the arrow) and took sumatriptan orally. She fell asleep after 60 min and showed a normal sleep profile including two REM periods. The control night is shown in (b).

(a) Patient 5 awoke out of REM sleep with migraine at 4.30 a.m. (indicated by the arrow). The patient took no medication and fell asleep again 20 min later. The control night is shown in (b).
Statistical analysis of sleep recordings
Table 2 shows the means and standard deviations of different sleep parameters of all eight patients for eight nights with following migraine and eight control nights. The number of arousals was significantly lower (P = 0.05; Z =−1.96) and there was a tendency towards a decrease in total REM density (P < 0.1; Z =−1.69) in the nights preceding a migraine attack. Furthermore, there was a marginal increase of slow wave sleep (in percentage) and of REM latency in the nights before migraine. The number of awakenings within the first 2 h of sleep was marginally decreased in the nights preceding migraine. There were no differences in the total sleep time, sleep efficiency, total number of awakenings or in the duration of the different sleep stages.
Means and standard deviations of sleep variables—a comparison of nights preceding migraine with control nights of eight probands
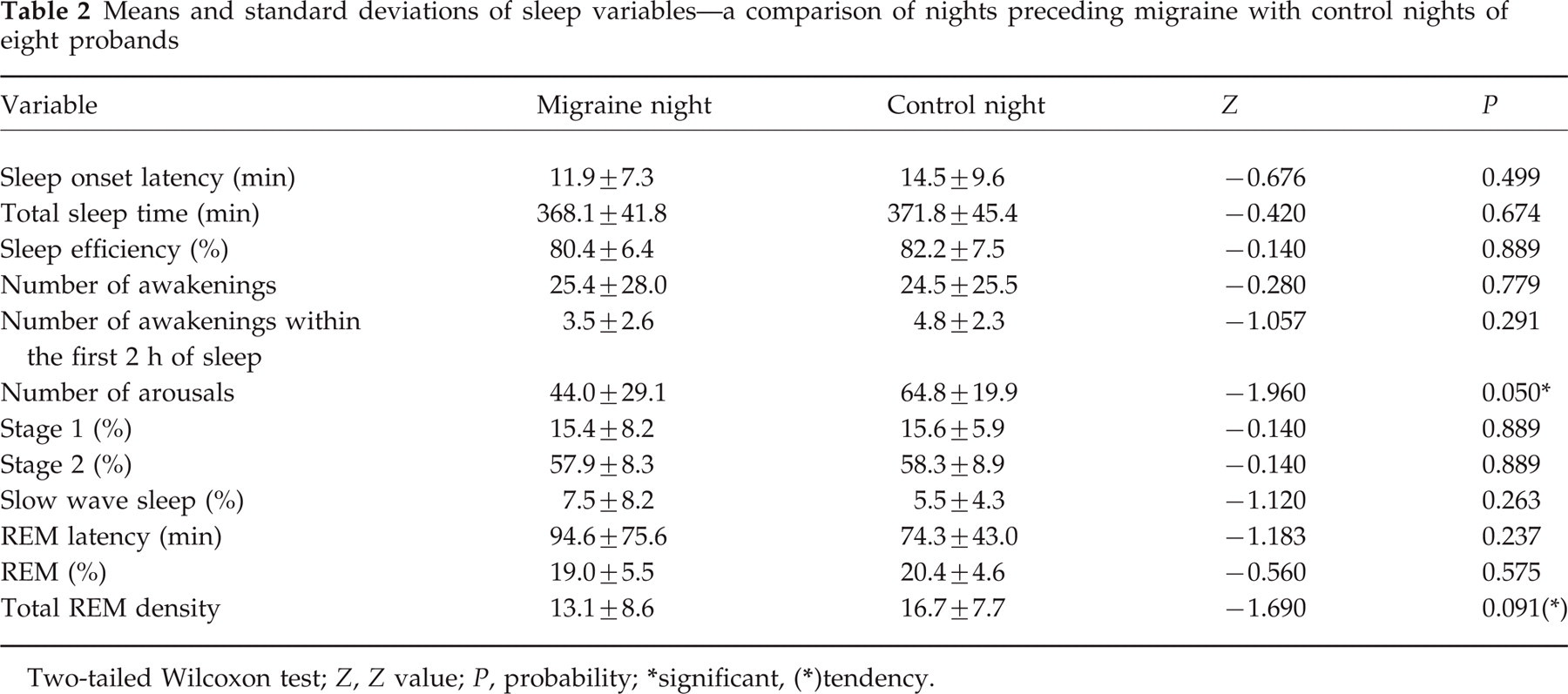
Two-tailed Wilcoxon test; Z, Z value; P, probability;
significant,
tendency.
Power spectrum
Table 3 shows the mean power in different sleep stages during the whole night, Table 4 for the first sleep cycle. In comparison with the control nights, there was a significant decrease of the total power and the power of the β2 band (15–26 Hz) in slow wave sleep in the nights preceding migraine. Additionally, there was a tendency to a decreased power in the β3 band (26–30 Hz). The power of the β2 band was also significantly decreased in the slow wave sleep of the first sleep cycle in nights before migraine. There was a tendency towards a decrease in α power in the first REM period.
Mean power in different sleep stages of the first sleep cycle—a comparison of nights preceding migraine with control nights of eight patients

Unit = μV
; two-tailed Wilcoxon test; Z, Z value; P, probability;
significant;
tendency; migraine, migraine night; control, control night; SWS, slow wave sleep; REM, REM sleep.
Mean power in different sleep stages of the whole night—a comparison of nights preceding migraine with control nights of eight patients

Unit = μV
; two-tailed Wilcoxon test; Z, Z value; P, probability;
significant;
)tendency; migraine, migraine night; control, control night; SWS, slow wave sleep; REM, REM sleep.
Discussion
The main alterations in sleep preceding a migraine attack were a significant decrease in the number of arousals and a trend to a decrease in REM density (Table 2), furthermore a significant decrease of β power in the slow wave sleep and a tendency towards a decrease of α power in the first REM period (Tables 3 and 4).
Arousals are characterized by abrupt changes in EEG frequency, which may include θ, α and/or frequencies greater than 16 Hz (13). They are longer than 3 s, but do not alter the sleep stage. Spontaneous arousals in sleep are normal, and increases with age (14). EEG arousals may represent a general physiological phenomenon of cortical activation during sleep (15), which is possibly decreased in the night preceding a migraine attack.
Additionally, we found a significant decrease in β power (15–26 Hz) in slow wave sleep and a trend to a decrease of α power (8–12 Hz) in the first REM period of sleep before a migraine attack. More research is needed to determine the physiological meaning of changes in frequency bands. A decrease of β power possibly reflects a hypoactivity of the central nervous system (16). It is remarkable that alterations have already been observed in the first sleep cycle in the first 2 h of sleep preceding a migraine attack. Corresponding to our findings, a decrease of activity in the α and β ranges were observed in a frequency analysis of nitroglycerin-induced migraine attacks (17).
The trend to a decrease of the total REM density preceding migraine may be explained by increased aminergic influences (18). Interestingly, it is assumed that the noradrenergic nucleus locus coeruleus and the serotonergic dorsal raphe nucleus are involved in migraine pathophysiology (1, 2), as a result of blood flow (19) and PET studies (3). The decrease in REM density may indicate a dysfunction of these nuclei prior to the headache. According to the reciprocal interaction model of REM sleep regulation (4, 5), an impairment of cerebral cholinergic input may also occur in sleep preceding migraine, also explaining the decreased number of arousals. It was recently shown that the antimigraine drug sumatriptan was able to induce antinociception by increasing cholinergic activation in the CNS (20).
We found no alterations in the occurrence of specific sleep stages in the nights preceding a migraine attack. This is in contrast to the findings of Dexter (8). He studied four migraine patients for a total of 29 nights. Headache in the morning was reported 10 times. Except for one night, all nights showed long periods of slow wave sleep and REM sleep in comparison with nights without headache the next morning. This discrepancy is probably due to differences in the patients studied. Dexter's patients had a mean headache frequency of 3–4 days per week, in our study less than 1 day per week. It is possible that the patients in Dexter's study also suffered from tension-type headaches. Dexter's patients discontinued all medications 1 week prior to admission, so rebound effects may have occurred. On the other hand, we could confirm the finding that migraine occurs in association with REM sleep or slow wave sleep (8, 21). Two patients in the present study reported a migraine attack beginning in the night, waking up out of REM sleep or less than 10 min after slow wave sleep.
Typical premonitory signs during the 24 h preceding a migraine attack are: irritability, withdrawal, depression and drowsiness (2, 22). In a time-series analysis of self-ratings of five patients suffering from migraine it seems that a typical subject was alert 2 days before the attack and drowsy during the migraine (23). In a similar study of 20 migraine patients Spierings et al. (24) showed that the stress of daily hassles as well as tension, irritability and tiredness, were significantly increased in the 2 days before a migraine headache. Kropp & Gerber found a corresponding periodicity in cortical activation in migraine patients. The increases in the contingent negative variation (CNV) amplitudes, indicating cortical hyperactivity in the days before migraine, normalized during migraine attacks (25, 26).
The decrease in the number of arousals and β power may reflect a decrease in cortical activity some hours before the migraine headache occurs. In addition, the proportion of slow wave sleep was marginally higher in the nights preceding a migraine attack. The decrease of the REM density may be interpreted as an increase in the need for sleep in the nights preceding migraine: Systematic sleep recordings have shown that the REM density linearly decreases with the reduction of prior sleep duration (27).
A limitation of our study was the small number of patients examined. Two patients suffered from a migraine attack following the adaptation night, which may have had an effect on the results. However, the sleep onset latency did not differ in the nights preceding migraine from the control nights. In two patients a migraine attack occurred during the night which may have affected the following sleep. Nevertheless, we believe that sleep recordings offer an interesting insight into the pathophysiology of alterations preceding migraine headache. The decreased number of arousals, the decreased β power in slow wave sleep and the trend to a decreased REM density indicate a decreased cortical activation. This may be caused by changes in the function of the aminergic nuclei or an impairment of cerebral cholinergic input. The predictive value of these findings should be investigated in future studies.
Footnotes
Acknowledgements
We thank Susanne Dardemann and Lars Friege for technical assistance. This study was supported by a grant from the Faculty of Medicine of the Christian-Albrechts-University of Kiel.
