Abstract
Transient cortical depolarization is implicated in the pathology of migraine. SB-220453 is a potent anti-convulsant which inhibits neurogenic inflammation and cortical spreading depression (SD)-evoked nitric oxide release via a novel but unknown mechanism. This study further investigates the effects of SB-220453 on generation and propagation of repetitive SD in the anaesthetized cat. Vehicle or SB-220453 1, 3 or 10 mg/kg was administered intraperitoneally 90 min prior to induction of SD in the suprasylvian gyrus (SG). Changes in d.c. potential were recorded in the SG and the adjacent marginal gyrus (MG). In vehicle-treated animals (n = 7), a brief exposure (6 min) to KCl induced a median (25–75% range) number of five (four to six) and three (two to four) depolarizations over a duration of 55 min (32–59 min) and 51 min (34–58 min) in the SG and MG, respectively. SB-220453 produced dose-related inhibition of the number of events and period of repetitive SD activity. SB-220453 also reduced SD-induced repetitive pial vasodilatation but had no effect on resting haemodynamics. However, when SD events were observed in the presence of SB-220453, it had no effect on metabolic coupling. These results show that SB-220453 produces marked inhibition of repetitive SD in the anaesthetized cat. SB-220453 may therefore have therapeutic potential in treatment of SD-like activity in migraine.
Introduction
Cortical spreading depression (SD) is a slowly propagating (2–5 mm/min) transient disturbance in electroencephalographic activity which has been detected in a variety of anaesthetized animals [1–4] and in human tissue in vitro[5].
More recently, SD-like activity has been implicated in migraine pathophysiology and particularly with the neurophysiological disturbances associated with migraine which include aura [6, 7]. Slowly spreading oligaemia has been observed following both experimental SD [8, 9] and migraine with aura [10–14]. Furthermore, in animal models SD also induces release of CGRP [15, 16] and nitric oxide [16–19] and produces a reflex activation of perivascular nerves [20]. SD may therefore also link cortical excitability with activation of the sensory afferent pathways [21].
We have recently identified a novel mechanistic class of compounds acting at a novel CNS site which possess potent anti-convulsant activity [22–25]. In a recent publication [26] we have demonstrated that the novel benzopyran, SB-220453, decreases cortical nitric oxide concentrations during SD activity. As nitric oxide may be a key mediator of C-fibre activation during migraine [27], this activity of SB-220453 may be an important potential anti-migraine action. Indeed, SB-220453 blocks electrically evoked C-fibre neuro-inflammation [22] and inhibits trigemino-vascular reflexes [28]. Thus our aim in the current study was to evaluate whether SB-220453 may directly influence the metabolism of SD generation and propagation in a gyrate brain. Clearly, by studying both generation and propagation of SD following SB-220453 administration, an understanding may emerge of whether the compound may temporally and spatially constrain cortical hyperexcitability during SD. In this manner, SB-220453 may limit receptive field involvement as proposed by Woolf [29]. We also explored the relationship between repetitive SD-induced cortical electrical activity and pial vasomotor responses in the presence or absence of SB-220453.
Methods
Surgical procedures
All procedures were approved by an internal ethics committee and conducted in accordance with the UK Animals (Scientific Procedures) Act 1986. Male cats (2.6–5.6 kg) were fasted overnight, anaesthetized using 5% halothane in oxygen, maintained with α-chloralose (100 mg/kg i.v.) and mechanically ventilated with room air. Animals were monitored for mean arterial blood pressure, arterial blood gas composition, rectal and cranial temperatures by procedures previously described [19]. Heart rate was derived from the arterial blood pressure signal. Surgical exposure of the marginal and suprasylvian gyri of the left parietal cortex was undertaken by craniotomy and durectomy and the brain covered by a prewarmed (37°C) layer (4 cm) of mineral oil [19].
Extracellular field potential recordings
Extracellular direct current (d.c.) field potential recordings were made using Teflon-insulated 0.125 mm diameter silver/silver chloride microelectrodes (Advent Research Materials, Halesworth, UK). Two, channel-separated recording electrodes were utilized, one situated on the suprasylvian gyrus (SG) parenchyma, the other on marginal gyrus (MG) parenchyma of the left parietal cortex. Both electrodes were equidistant from the site of SD induction and referenced to the left limb semitendinous muscle. All electrodes were connected to a direct current-coupled field effect input stage and amplified using a Neurolog system NL834 4-channel d.c. preamplifier. D.c. signals were low pass filtered and passed to a data acquisition system for offline analysis (Biopac MP100 data acquisition system; Linton Instruments, UK). Changes in extracellular field potential in both gyri were analysed for amplitude (mV), event number and the period of repetitive SD activity. Figure 1 shows a schematic representation of the experimental set up.

Schematic representation of (a) the orientation of the crainiotomy over the cat cortex and (b) localization of stimulation and recording sites within the cranial window. (c) KCl pellet used in the experiment. The pellet was formed in an Eppendorf tube with volume < 100 μl. High magnification shows a uniform structure when the pellet is removed from the Eppendorf tube. (d) Schematic representation of the changes in d.c. potential (mV) following application of KCl pellet for 6 min to the supersylvian gyrus. Measurements of time to repolarization (min) (1), amplitude of d.c. depolarization (mV) (2), total period of spreading depression activity (min) (3) and number of depolarizations were made.
Pial artery diameter measurements
Suprasylvian pial artery diameter measurements were made using a Wild M400 photomicroscope (× 64 magnification) (Leitz, Milton Keynes, UK) and recorded on video tape for off-line analysis using a Hamamatsu C2400 high intensity camera and video microscaler (For-a IV 550; Cameron Communications, Glasgow, UK).
Induction of spreading depression
Spreading depression was induced using a standardized KCl pellet. Crystals of KCl were prepared uniformly in the base of an open 1.5 ml Eppendorf tube. KCl (30 mg) was placed into the tube with 10 μl of sterile water and allowed to dry in air overnight (Fig. 1). This technique therefore produces a standard mass and surface area (approx. 2.5 mm diameter across top surface) of chemical stimuli for induction of SD. KCl pellets were then placed on the brain using forceps with the top (largest) surface to cortex and removed after 6 min using a saline-soaked swab. As the brain is covered in a layer of prewarmed mineral oil, KCl is easily removed with an aqueous environment, leaving no residual crystal.
Experimental protocol
Two separate studies assessing SB-220453 were performed. In the first, SD activity was induced using a 6-min application of KCl solid placed directly onto the suprasylvian parenchyma in a region approx. 1 cm distant from both the marginal (MG) and suprasylvian gyrus (SG) recording sites. Four separate treatment groups were utilized: vehicle (labrasol, 1 ml/kg; Alfa Chemicals Ltd., Binfield, UK) (n = 7), SB-220453 (cis-(–)-6-acetyl-4S-(3-chloro-4-fluorobenzoylamino)-3,4-di-hydro-2,2-dimethyl-2H-1-benzopyran-3S-ol; Medicinal Chemistry, SmithKline Beecham Pharmaceuticals, Harlow, UK) 1 mg/kg (n = 4), 3 mg/kg (n = 5) and 10 mg/kg (n = 6). All agents were administered intraperitoneally, 90 min prior to induction of SD. Recordings of extracellular field potential were made in both SG and MG and maintained for a further 54 min after removal of KCl.
A separate study was conducted to assess changes in blood concentrations of high-dose SB-220453 over the period of repetitive activity. SD activity was induced as above in vehicle (n = 4) and SB-220453 (10 mg/kg i.p.; n = 4)-treated animals dosed 90 min prior to SD induction. Blood samples were removed from a venous line at 10-min intervals throughout the experiment to assess concentrations of SB-220453.
Detection of SB-220453
Blood samples (0.25 ml) were removed and placed in heparinized tubes and transferred to glass extraction tubes containing sodium hydroxide (1 ml of 0.01
Statistical analysis
Data were expressed mean ±
The amplitude and time to repolarization was calculated as shown in Fig. 1. As the number of SD events was observed over a predefined period, the SD event number and period over which activity was recorded are expressed as median (25–75% range). Since these parameters were not normally distributed, differences in outcome were determined by Kruskal–Wallis test (non-parametric
Results
Effects of SB-220453 on resting cardiovascular parameters
Administration of vehicle or SB-220453 (1, 3 or 10 mg/kg i.p.) had no significant effects on mean arterial blood pressure or heart rate. For example, at the time of SD induction, mean arterial blood pressure (MABP) was 75± 11 mmHg, 94 ± 17 mmHg, 77 ± 9 mmHg, 75 ± 8 mmHg and heart rate was 150 ± 11 beat/min, 136 ± 7 beat/min, 141 ± 8 beat/min and 150 ± 8 beat/min in vehicle- and SB-220453 1, 3, 10 mg/kg-treated animals, respectively. No acute, transient effects on mean arterial blood pressure or heart rate were observed throughout the protocol in any experimental groups.
Effect of SB-220453 on generation of SD and propagation into contiguous gyri
Suprasylvian gyrus SD was found to elicit similar extracellular field potential waveform changes in the contiguous MG. There was an attenuation in the number of d.c. deflections observed in the MG relative to that observed in the SG. Following vehicle treatment, a median 5 (25–75% range 4–6) events were recorded (n = 7) in the SG, whereas in the MG of the same animals a significant reduction in the number of SD events was detected with only a median 3 (25–75% range 2–4) d.c. deflections being observed (n = 7) (Fig. 2). Pre-dosing with SB-220453 (1–10 mg/kg i.p.) induced a dose-dependent decrease in number of negative shifts in extracellular d.c. field potential observed in the SG, reaching significance at 3 mg/kg (median 2 (25–75% range 2–3), P < 0.05, n = 5) and 10 mg/kg i.p. (median 1 (25–75% range 1–1), P < 0.01, n = 6) when compared with vehicle (Mann–Whitney U-test) (Fig. 2). SB-220453 produced a similar inhibition of SD in the MG, with animals exhibiting a median of only 0.5 (25–75% range 0–1, n = 6, P < 0.05, Mann–Whitney U-test) events at 10 mg/kg (Fig. 2).

Histogram showing median and range (25–75%) of the number of spreading depression (SD) extracellular d.c. field depolarizations in the suprasylvian gyri (SG) and marginal gyri (MG). Animals were pretreated with either vehicle (labrasol 1 ml/kg i.p.) or SB-220453 1, 3 or 10 mg/kg i.p. SD was elicited 90 min later in the SG and propagation of the resultant repetitive d.c. depolarizations followed in both the suprasylvian and contiguous marginal gyri using Ag/AgCl electrodes (n = 5–7). Data represented as median and range. ∗P < 0.01 vs. corresponding gyri in vehicle group (Mann–Whitney U-test). ∗∗P < 0.05 between gyri within group (Mann–Whitney U-test).
In all experimental groups there was a failure of SD waves to propagate into neighbouring gyri, with the vehicle-treated animals demonstrating a statistical difference in the number of events recorded. However, the ratio of the number of SD events observed in the MG was between 40% and 60% of the events recorded in the SG in all experimental groups (Fig. 2); this indicates a lack of effect of SB-220453 on the likelihood of propagation of SD into adjacent gyri.
Although significantly more SD events were observed in the SG compared with the MG, the duration of the activity was not different between gyri in vehicle-treated animals (n = 7). SD activity occurred for a median 55 min (25–75% range 32–59 min) in the SG and 51 min (25–75% range 34–58 min) in the MG (Fig. 3). SB-220453 produced a dose-related inhibition of SD duration (Fig. 3) and following administration of the highest dose (10 mg/kg, n = 6), SD activity was only observed for a period of 4 min (25–75% range 4–5 min) in the SG and 3 min (25–75% range 0–7 min) in the MG (Fig. 3).
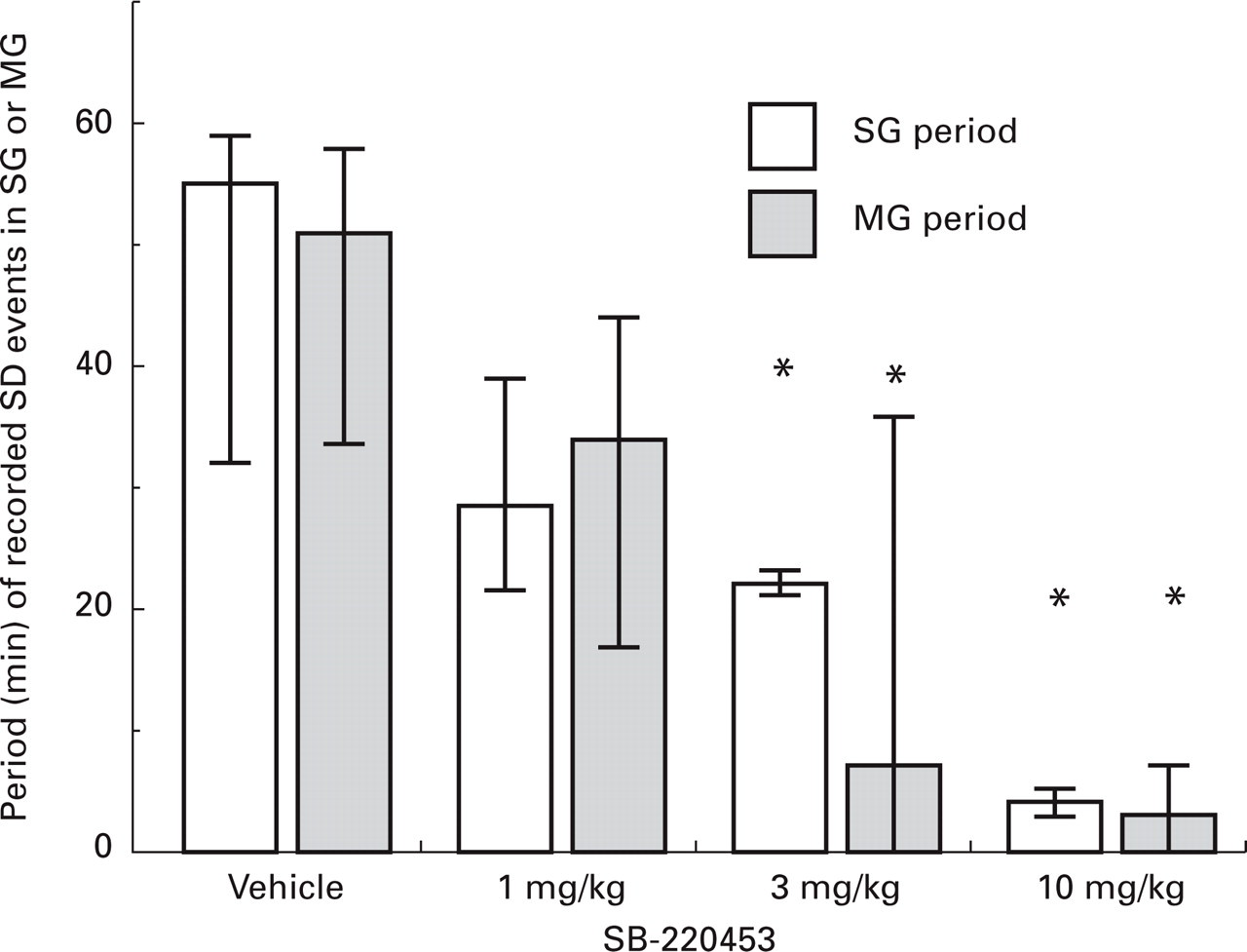
Histogram showing median (25–75% range) of the total period of spreading depression (SD) activity in which SD extracellular d.c. field potential changes were detected in the suprasylvian gyri (SG) and marginal gyri (MG). Animals were pretreated with either vehicle (labrasol 1 ml/kg i.p.) or SB-220453 1, 3 or 10 mg/kg i.p. SD was elicited 90 min later in the SG and propagation of the resultant repetitive d.c. depolarizations followed in both the suprasylvian and contiguous marginal gyri using Ag/AgCl electrodes (n = 5–7). Data represented as median (25–75% range) ∗P < 0.05 vs. corresponding gyri in vehicle group (Mann–Whitney U-test).
The amplitude of SD events was similar between SG and MG in vehicle-treated animals. The initial SD event produced a mean amplitude of 13.0 ± 2.4 mV in SG and 10.9 ± 6.8 mV in MG with a respective duration of time to repolarization of 3.5 ± 3.9 and 3.3 ± 1.7 min (n = 7). In SB-220453-treated animals, the amplitude of the d.c. potential change and the time taken for repolarization of the initial SD depolarization were similar to vehicle-treated animals (see Table 1 for SG).
Mean±

Data are pooled from two separate experiments with n animals per group.
Effect of SB-220453 on SD metabolism–pial artery diameter coupling
Pooling data from both studies shows that a median 5 (25–75% range 4–6) events were observed in the SG lasting over a median of 55 min (25–75% range 35–59 min, n = 11) in vehicle-treated animals. This activity was significantly inhibited by SB-220453 (10 mg/kg) where only 1 (25–75% range 1–1) event (n = 10, Mann–Whitney U-test, P < 0.001) was observed over a period of 5 min (25–75% range 4–14 min, n = 10, Mann–Whitney U-test, P < 0.0001).
There were no significant differences in resting pial artery diameter in vehicle- or SB-220453-treated animals between groups. Immediately prior to SD induction, pial artery diameter was 188 ± 41 μm in vehicle-treated animals (n = 11) and 204 ± 32 μm (n = 4), 211 ± 12 μm (n = 5) and 183 ± 32 μm (n = 10) in SB-220453 1, 3 and 10 mg/kg-treated animals, respectively (P > 0.05).
The application of KCl to SG parenchyma in vehicle-treated animals induced a repetitive negative deflection in extracellular d.c. field potential and associated repetitive episodes of pial artery vasodilatation. An example trace is shown in Fig. 4 for vehicle- and SB-220453-treated animals. Interestingly, in the vehicle-treated animal, despite a close coupling of vasodilatation to cortical d.c. potential changes, seven episodes of vasodilatation were recorded with six SD events in a nearby region of interest.

Superimposed original trace recordings for individual animals of artery diameter (μm) and extracellular d.c. field potential (mV) over time following KCl induction of spreading depression (SD) at time = 0 min. Animals were pretreated with vehicle (labrasol 1 ml/kg i.p.) or SB-220453 10 mg/kg i.p. 90 min prior to SD induction.
In all treatment groups, at least one SD event was recorded in the SG which was associated with a marked pial vasodilatation in most animals. No significant differences were observed between vehicle- or SB-220453-treated animals (Table 1) on the first SD event. However, as SB-220453 produced marked inhibition of repetitive SD there was an associated inhibition of repetitive episodes of pial vasodilatation following KCl application (Fig. 4).
Blood concentrations of SB-220453
In animals in which serial blood samples were removed, the number and the duration of SD activity were comparable with the previous study. Thus, in vehicle-treated animals, SD activity was sustained for at least 50 min, during which period a median 5.5 (25–75% range, 4.5–7.5) events was recorded (n = 4). Pretreatment with SB-220453 10 mg/kg i.p. inhibited SD generation to a median 2 (25–75% range, 1.5–2.5) events (n = 4). In these experiments, mean blood concentrations (μM) of SB-220453 ranged from 2.2 ± 0.2 at initiation of SD to 2.5 ± 0.25, 3.1 ± 0.5, 3.3 ± 0.3, 3.4 ± 0.3, 3.7 ± 0.4 and 3.7 ± 0.4 μ
Discussion
The main finding of the present study was that the novel benzopyran, SB-220453, produced marked inhibition of regenerative SD in the cat. SB-220453 [22] is a potent and selective ligand for a novel CNS-specific binding site identified in rat, human [23] and cat [28] brain, and is part of a novel family of anticonvulsant agents [22–25]. Affinity at this site has been correlated with marked anticonvulsant properties for a number of structurally related benzopyran analogues [23]. To date, over 80 standard pharmacological agents have no affinity for this site [22–25]. High affinity for the SB-220453 CNS binding site is correlated with in vivo potency in producing elevation of seizure threshold [24, 25]. Furthermore, SB-220453 has been shown to block neurogenic inflammation in rat meninges following electrical stimulation of the trigeminal ganglion [22].
This investigation demonstrates that SB-220453 produces a dose-related inhibition of the number of SD-induced field potential changes in the SG over a similar dose range to previous studies [22]. Associated with the decrease of SD event number, the period of repetitive SD activity was also significantly reduced by SB-220453 without any effect on the initial individual event. Interestingly, one SD event was observed in the SG during the period of KCl administration which was not inhibited by SB-220453 (10 mg/kg) in any of the animals investigated. SB-220453 therefore appears to block selectively the generation of SD activity once the KCl stimulus has been removed (Fig. 2).
In a separate group of animals we confirmed the inhibitory actions of SB-220453 on repetitive SD activity and demonstrated similar blood concentrations throughout the experimental period. SB-220453 has a high affinity (pKi 7.3) for its binding site in cat brain homgenates [28], and blood concentrations in excess of 2 μ
The pharmacological profile of activity of SB-220453 is quite distinct from that of NMDA antagonists [30, 31] and inhalational anaesthetics which almost abolish SD [9]. Indeed, in our model of repetitive SD activity, i.v. administration of MK801 following initiation of SD activity produces a rapid and complete inhibition of subsequent events but it has no effect on trigeminal neurovascular reflexes [32], demonstrating a different pharmacological profile from SB-220453 [28].
Repetitive spreading depression in suprasylvian and marginal gyri
In this study we investigated both the generation of SD activity and the propagation in neighbouring gyri. Reduced SD propagation has been noted at architectonic boundaries [3, 4, 33]. Following a single application of KCl to a non-ischaemic cortex, we have also found that the characteristics of the initial and subsequent SD wavefronts show marked differences in rate and spread of the propagated wavefront [33, 34]. Detailed analysis of the primary and secondary events clearly shows marked reductions in the velocity of propagation and the total area covered by the SD wavefront [34]. These observations are not dependent on the integrity of the experimental preparation, as in these models we can induce further SD events with repeated KCl stimulation (Smith & Parsons, unpublished observations).
This study demonstrates less SD events in the MG compared with the neighbouring SG. However, the period of SD activity was similar in both the SG and MG, as was the amplitude of individual SD events. The mechanism(s) underlying this observation is not clear, but only 40–60% of recorded SD events penetrated into adjacent gyri. Collectively these results show that SB-220453 produced marked inhibition of the generation of SD in both the SG and MG, but had little effect on the likelihood of propagation. Similar observations have been reported with SB-220453 using serial diffusion weighted images as a marker of the SD wavefront [35].
SB-220453 and SD-induced vascular changes
SB-220453 had no effect on the resting cardiovascular parameters of arterial blood pressure, heart rate or pial artery diameter. Concomitant with the reductions in the number of SD events, SB-220453 also reduced the total number of repetitive SD-induced dilatations (Fig. 4). However, SB-220453 had no effect on SD-induced vasodilatation when it occurred to the initial event (Fig. 4, Table 1). These observations indicate that SB-220453 has no effect on local coupling of metabolism and pial artery responses.
We conclude that SB-220453 represents a novel class of benzopyran, which has the ability to inhibit repetitive SD in the cat. SB-220453 was well tolerated, with no effects on arterial blood pressure, heart rate or pial artery diameter. As cortical excitability or depolarization has been implicated in conditions such as migraine, this study provides rationale for the evaluation of this agent in this disorder.
Footnotes
Acknowledgements
The authors would like to thank Dr Stephanie Yeulet and Ms Beverley Smith for help with detection of SB-220453.
