Abstract
Clinical studies indicate anti-migraneous efficacy of the probably GABAergic anti-convulsants valproate and gabapentin. For the GABAergic anticonvulsants vigabatrin and tiagabine, studies about antimigraneous efficacy are missing. The aim of this study was to test the GABAergic potency of these drugs in vitro before further clinical studies. Intracellular recordings were obtained from hippocampal pyramidal cells. Spontaneous GABAergic hyperpolarizations (SGH) elicited by 75 μ
Introduction
Enhancing the cerebral GABA system is one of many proposed mechanisms to limit convulsive seizures. Furthermore, enhanced GABA potentials are also discussed as one possible mechanism of valproate's action in migraine prophylaxis (1). While the anticonvulsant potency of the probably GABAergic substances valproate and gabapentin (2) are proven, first clinical trials indicate that migraine patients benefit from the prophylactic treatment with either drug, too (3–8). The GABAergic anticonvulsants vigabatrin and tiagabine have not yet been studied with respect to their anti-migraneous effectiveness (9). Table 1 summarizes commonly postulated mechanisms of how GABAergic anti-convulsants can act in epileptic tissue. Taken together, in addition to the GABAergic mechanisms, valproate as well as gabapentin act on various ion channels and transmitter systems, while for vigabatrin and tiagabine only GABAergic mechanisms are described. Both drugs increase GABA levels in brain but interact with different cellular components: vigabatrin by irreversible inhibition of the GABA-degrading enzyme GABA aminotransferase (26), and tiagabine by reversible inhibition of a GABA re-uptake via high-affinity binding to the GABA carrier, GAT-1 (27).
Postulated mechanisms of tested anti-convulsants

In the present study we used GABAergic hyperpolarizations experimentally induced by 4-aminopyridine(4-AP) which is commonly used to elicit hyperexcitation in cortical neurones (28). 4-AP evokes spontaneous GABAergic events in hippocampal pyramidal neurones, as previously shown by several investigators (29–32). This was interpreted to result from a 4-AP-induced inhibition of special potassium channels of GABAergic interneurones whose pronounced firing rates are likely to induce spontaneous GABAergic events in post-synaptic pyramidal neurones.
Compared with a local application of GABA (33), the 4-AP-treated hippocampal slice is a very convenient model to study the potency of GABAergic drugs whose different modes of action on the GABA system are of special interest concerning the pathophysiology and the treatment of migraine.
Methods
Transverse hippocampal slices (200–400 μm thick) were prepared from brains of ether-anaesthetized adult guinea pigs (300–400 g). Slices were preincubated for 2 h in 28°C warm saline equilibrated with 5% CO2 in O2. This saline contained (in m
The following drugs were added to CTR: 4-AP (75 μ
Intracellular recordings were obtained from the somata of CA3-neurones with sharp glass microelectrodes filled with 2
Results
In this study we investigated the effect of the GABAergic anticonvulsants on spontaneous GABAergic hyperpolarizations (SGH). Therefore, intracellular recordings were obtained from 47 CA3-neurones of 45 slices. Only CA3-neurones were selected which had AP-amplitudes > 50 mV and a resting membrane potential <−50 mV. In all tested 47 CA3-neurones epileptiform activity consisted of periodically appearing bursts whichdeveloped during application of 75 μ
SGH induced by 4-AP
SGH occurred in 28 out of 47 CA3-neurones. These SGH had a duration of 2–4 s. The hyperpolarizing changes in membrane potential were strictly monophasic and reversal potentials of −58 to −67 mV were measured. The SGH persisted during the blockade of NMDA- and AMPA-receptors with 50 μ
Effects of valproate, gabapentin, vigabatrin on SGH
In a first step those drugs acting on GABA-degrading or-synthesizing enzymes were tested. The testing period was limited to 60 min, which is a reasonable duration for intracellular recordings of CA3-neurones.
In seven neurones SGH remained unchanged in amplitude and duration when slices were exposed to 1–2 m
Effects of tiagabine on SGH
In contrast to the above mentioned drugs the GABA re-uptake inhibitor tiagabine clearly changed SGH: within 10–15 min after washin, the duration of SGH increased by 50–70%. Thereafter, the monophasic SGH converted into triphasic membrane potential fluctuations with an early hyperpolarization (component a), a following depolarization (component b), and a late hyperpolarization (component c) in all neurones (n = 10, Fig. 1a). This triphasic event was completely reversible within 30 min after the onset of washout.
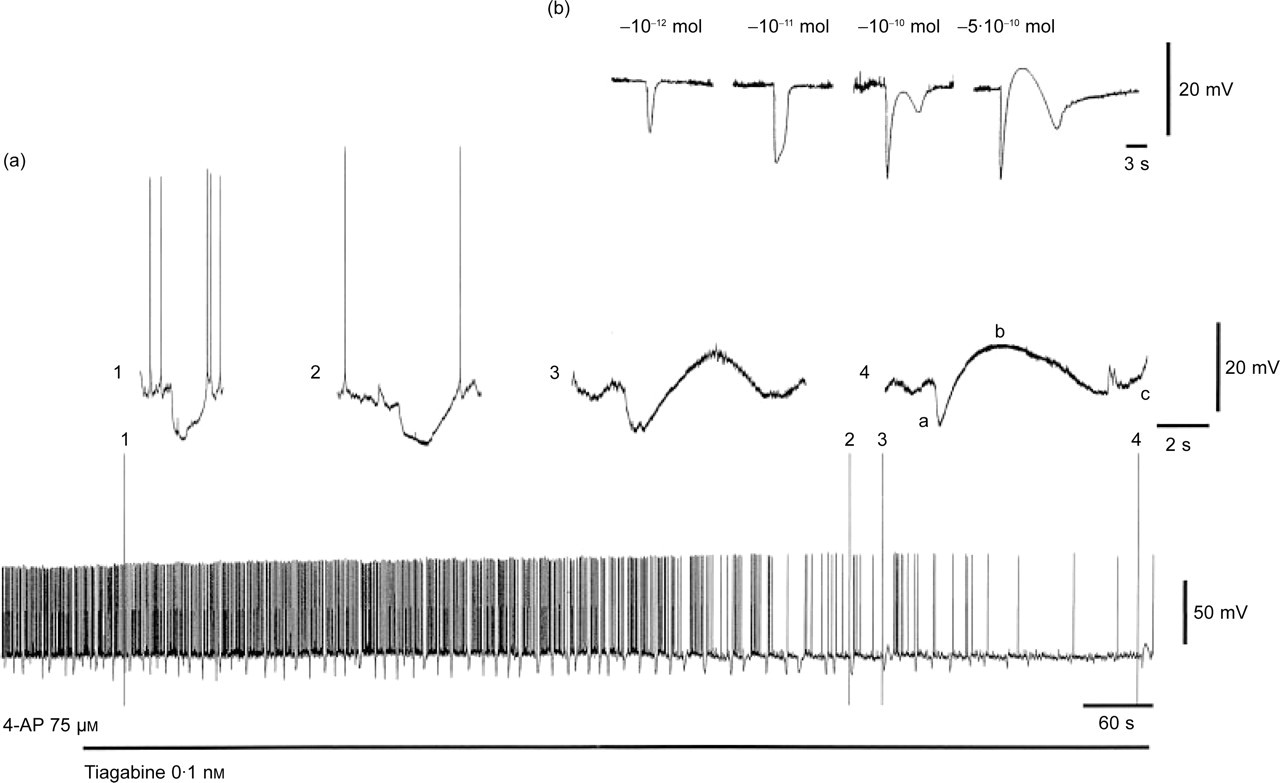
(a) Conversion of monophasic spontaneous GABAergic hyperpolarizations (SGH) induced by 4-aminopyridine (4-AP) into triphasic membrane potential fluctuations (a,b,c) under tiagabine 0.1 m
Discussion
The objective of the study was to compare the GABAergic potency of the four anticonvulsants valproate, gabapentin, vigabatrin, and tiagabine. As a parameter for assessment of GABAergic potency, SGH induced by 4-AP were selected. In contrast to valproate, gabapentin and vigabatrin, only tiagabine prolonged the duration of the SGH after 10–15 min of application.
This observation was supported by the study of Thompson & Gähwiler (35), who also described a clearly prolonged duration of evoked mono- and polysynaptic inhibitory post-synaptic potentials (IPSP) of CA3-neurones exposed to tiagabine (10–25 μ
When 0.1 m
There are several lines of evidence that GABergic drugs modulate biochemical and physiological events involved in the pathophysiology of migraine. Spreading depression, which is a cortical event that underlies the migraine aura, may be suppressed by an increase of inhibitory GABAergic neurotransmission induced, for example, by valproate (1). The release of vasoactive neuropeptides (substance P, neurokinin A, calcitonin-gene-related peptide) from activated sensory nerve terminals (40) can be decreased by valproate (41). Furthermore, the activation of the trigeminal nucleus caudalis, responsible for modulation of the nociceptive signals, can be suppressed by GABAergic drugs. Expressing of immediate early gene c-fos as a marker of neuronal activity within trigeminal nucleus caudalis was reduced by valproate (42) as well as allopregnanolone, a progesterone metabolite which modulates GABAA receptor activity (43). The effect of valproate could be reversed by bicuculline (42). Together, these studies suggest that an augmentation of the inhibitory central GABA system plays a pivotal role in suppressing migraine-related events.
We conclude that the re-uptake inhibitor tiagabine augments the level of synaptic GABA faster compared with the other drugs which work on enzymatic pathways. Keeping in mind tiagabine, administered intraperitoneally, increases extracellular GABA overflow in the globus pallidus of Sprague-Dawley rats with peak values at about 40 min after application (44), this drug appears well suited as a fast-acting anti-migraine drug. The clinical efficacy of tiagabine in migraine therapy, especially in acute migraine attacks, should be investigated.
