Abstract
The aim of the present thesis was to investigate the pathophysiology of chronic tension-type headache with special reference to central mechanisms. Increased tenderness to palpation of pericranial myofascial tissues is the most apparent abnormality in patients with tension-type headache. A new piece of equipment, a so-called palpometer, that makes it possible to control the pressure intensity exerted during palpation, was developed. Thereafter, it was demonstrated that the measurement of tenderness could be compared between two observers if the palpation pressure was controlled, and that the Total Tenderness Scoring system was well suited for the scoring of tenderness during manual palpation. Subsequently, it was found that pressure pain detection and tolerance thresholds were significantly decreased in the finger and tended to be decreased in the temporal region in chronic tension-type headache patients compared with controls. In addition, the electrical pain threshold in the cephalic region was significantly decreased in patients. It was concluded that the central pain sensitivity was increased in the patients probably due to sensitization of supraspinal neurones. The stimulus-response function for palpation pressure vs. pain was found to be qualitatively altered in chronic tension-type headache patients compared with controls. The abnormality was related to the degree of tenderness and not to the diagnosis of tension-type headache. In support of this, the stimulus-response function was found to be qualitatively altered also in patients with fibromyalgia. It was concluded that the qualitatively altered nociception was probably due to central sensitization at the level of the spinal dorsal horn/trigeminal nucleus. Thereafter, the prophylactic effect of amitriptyline, a non-selective serotonin(5-HT) reuptake inhibitor, and of citalopram, a highly selective 5-HT reuptake inhibitor, was examined in patients with chronic tension-type headache. Amitriptyline reduced headache significantly more than placebo, while citalopram had only a slight and insignificant effect. It was concluded that the blockade of 5-HT reuptake could only partly explain the efficacy of amitriptyline in tension-type headache, and that also other actions of amitriptyline, e.g. reduction of central sensitization, were involved. Finally, the plasma 5-HT level, the platelet 5-HT level and the number of platelet 5-HT transporters were found to be normal in chronic tension-type headache. On the basis of the present and previous studies, a pathophysiological model for tension-type headache is presented. According to the model, the main problem in chronic tension-type headache is central sensitization at the level of the spinal dorsal horn/trigeminal nucleus due to prolonged nociceptive inputs from pericranial myofascial tissues. The increased nociceptive input to supraspinal structures may in turn result in supraspinal sensitization. The central neuroplastic changes may affect the regulation of peripheral mechanisms and thereby lead to, for example, increased pericranial muscle activity or release of neurotransmitters in the myofascial tissues. By such mechanisms the central sensitization may be maintained even after the initial eliciting factors have been normalized, resulting in the conversion of episodic into chronic tension-type headache. Future basic and clinical research should aim at identifying the source of peripheral nociception in order to prevent the development of central sensitization and at ways of reducing established sensitization. This may lead to a much needed improvement in the treatment of chronic tension-type headache and other chronic myofascial pain conditions.
Introduction
Tension-type headache is the most common (1) and, as far as socio-economic impact is concerned, the most important type of headache (2). In spite of this, the scientific interest in tension-type headache has previously been sparse, and knowledge about the pathophysiological mechanisms leading to this disorder is therefore limited. However, since the publication of the International Headache Classification (3) an increasing number of well-performed studies have been published. Myofascial tissues have been extensively studied, and it has been demonstrated that increased tenderness to palpation of pericranial myofascial tissues is the most apparent abnormality in patients with tension-type headache (4, 5). It is assumed that nociceptive impulses from the pericranial muscles may be referred to the head and perceived as headache, and that myofascial tissues therefore do play an important role in tension-type headache (6–8).
The pathophysiological mechanisms leading to the increased tenderness are poorly understood, which may partially be caused by methodological difficulties in the quantification of tenderness by manual palpation (9). Possible mechanisms leading to myofascial pain and tenderness include: (i) sensitization of peripheral myofascial nociceptors, (ii) sensitization of second order neurones at the level of the spinal dorsal horn/trigeminal nucleus, (iii) sensitization of supraspinal neurones, and (iv) decreased anti-nociceptive activity from supraspinal structures. Central mechanisms have only been sparsely investigated in tension-type headache, although it becomes increasingly evident that central factors are involved in the pathophysiology of this disorder (10–12). Thus, one of the most exciting developments in basic pain research over the past decades has been the recognition that the response generated by the somatosensory system to a defined input is not fixed or static. In particular, the increased knowledge of central sensitization, i.e. increased excitability of neurones in the central nervous system, has been a major breakthrough in the understanding of chronic pain (13). Central mechanisms are complex and often difficult to investigate in clinical studies. However, it is necessary to do so in order to examine whether the knowledge obtained from basic pain research is valid in patients with chronic pain, and in order to increase our understanding of the central mechanisms leading to tension-type headache and other chronic pain disorders.
The aims of the present thesis were to evaluate and improve the methodology used for manual palpation of myofascial tissues, to investigate the mechanisms leading to myofascial tenderness, and to investigate central pain mechanisms in patients with chronic tension-type headache, with a special focus on the role of serotonin. The thesis is based on a series of methodological and clinical studies in patients with chronic tension-type headache (14–20) and in patients with other types of chronic myofascial pain (21).
Peripheral mechanisms: myofascial tenderness
Myofascial pain probably plays an important role in tension-type headache. In this section, the methodology used for the evaluation of myofascial tenderness, the findings on pericranial tenderness in patients with tension-type headache and possible peripheral mechanisms leading to myofascial pain will be discussed.
Evaluation of tenderness by manual palpation
Tenderness, which may be defined as pressure-induced pain, is a very common sign in medical practice. Tenderness may be a normal physiological sign, e.g. following excessive muscle activity, or it may signal underlying pathology, e.g. inflammation in a joint. Manual palpation is the only clinically relevant method for evaluation of tenderness, and it is also an important tool in myofascial pain research including studies of tension-type headache. A precise quantification of tenderness by manual palpation is however, difficult. Thus, the reliability of manual palpation has been reported to be low (22, 23) or acceptable (24–27). Among the factors which may contribute to the variability in the evaluation of tenderness, the intensity of the palpation pressure and the scoring of tenderness are probably the most important (25, 28, 29).
In order to be able to measure the pressure intensity exerted during palpation a new piece of equipment, a so-called palpometer, was developed (14). The palpometer consists of a thin pressure-sensitive plastic film connected to a meter, a principle first described by Atkins et al. (30). The film is attached to the tip of the second finger, and the pressure exerted during palpation is measured in arbitrary units on the meter scale (Fig. 1). The relation between the forces applied to the plastic film and the palpometer output was found to be approximately linear, and the intra- and interobserver variation of exerted force at given palpometer values was found to be acceptably low (14). The authors concluded that the palpometer is a valuable tool for the measurement of the pressure intensity exerted during palpation. In the same study, it was demonstrated that the palpation pressure was stable within observers from week to week, while there was a considerable difference in pressure intensities between observers (14).
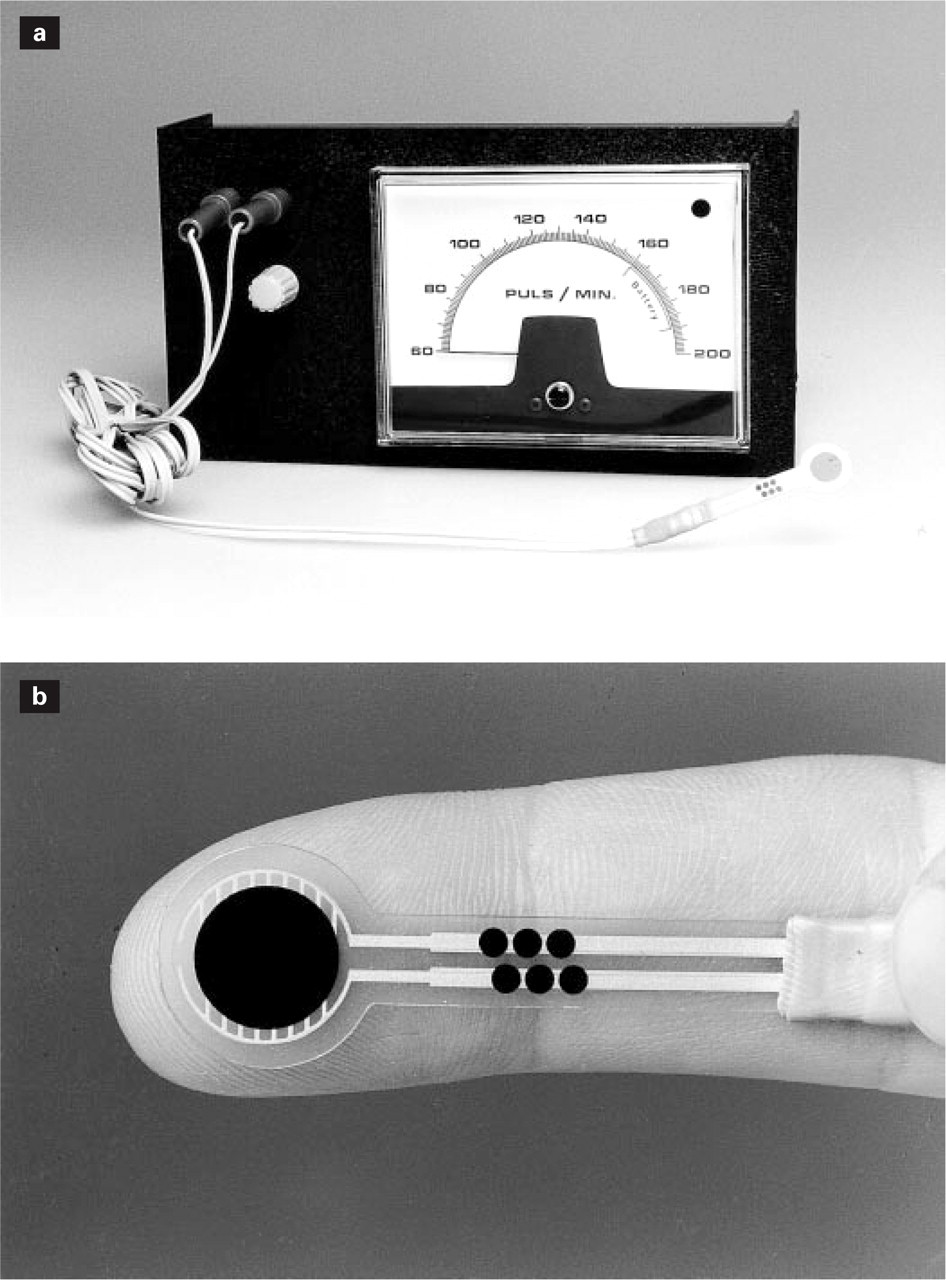
(a) The palpometer consists of a Force Sensing Resistor connected to a meter. The force is recorded by the meter scale, which is divided into arbitrary units (U) from 60 to 200 U. (b) The Force Sensing Resistor is a thin polymer film device, which is attached to the fingertip by means of thin adhesive tape (Micropore®, not shown). Reproduced from (14) with permission.
In headache research, manual palpation is usually performed according to the method described by Langemark & Olesen (4). Briefly, the palpation is performed bilaterally with small rotating movements of the second and third fingers, and the induced tenderness is scored according to the Total Tenderness Scoring system. The validity of this method was recently examined (15). By the use of the palpometer it was demonstrated that the measurement of tenderness differed significantly between two observers using non-instrumental palpation, while this difference was eliminated during pressure-controlled palpation. Tenderness scores did not differ significantly within observers from week to week even without control of the palpation pressure. This supports that palpation pressures are stable within observers over time as previously suggested (14). Thus, the intensity of the palpation pressure has to be controlled if tenderness scores have to be compared between research centres, or if more than one observer is used in a study, while palpation can be performed without control of the pressure intensity if the same observer is used throughout a study.
The Total Tenderness Scoring system scores tenderness on a four-point combined behavioural and verbal scale as follows: 0 = denial of tenderness, no visible reaction; 1 = verbal report of discomfort or mild pain, no visible reaction; 2 = verbal report of moderate pain, with or without visible reaction; 3 = verbal report of marked pain and visible expression of discomfort. The values from left and right sides are summed to a Total Tenderness Score (TTS). The Total Tenderness Scoring system is based on two important assumptions. First, the four-point scale must reflect the pain perceived by the subject. Second, it must be valid to summate the individual tenderness scores to a total score, which is not uncomplicated seen from a statistical point of view. This requires, as a minimum, that the four-point ordinal scale can be considered as a ratio scale, i.e. that increases in tenderness scores from 0 to 1, 1 to 2 and 2 to 3 reflect equal increases in pain perceived by the subjects. A recent study (15) indicated that each of the scores on the four-point tenderness scale corresponded well with the pain intensity recorded by the subjects on a visual analogue scale (Fig. 2). In addition, the pain recorded by the subjects increased approximately linearly with increasing tenderness score. Assuming the visual analogue scale to be a ratio scale (31), this indicates that also the four-point tenderness scale roughly can be considered as a ratio scale. This was confirmed by the finding of a remarkably high correlation between the sum of pain scores recorded by the subjects and the TTS recorded by the observer (15). This indicates that the Total Tenderness Scoring system is well suited to the recording of pain provoked by palpation. It should be emphasized that the scoring is subjective, and that blinding of the observer is important if tenderness has to be compared between groups.

Tenderness scored by a blinded observer according to a four-point (0–3) scale vs. pain intensity recorded by thesubjects on a 100-mm visual analogue scale during palpation of the trapezius muscle. Thirty subjects were palpated, each with seven different pressure intensities. Boxes indicate quartiles and median of pain intensities recorded at each tenderness score. The number of observations was 79, 40, 40 and 51 fortenderness scores 0, 1, 2 and 3, respectively. Vertical lines extend to the 2.5th and 97.5th centiles. Outliers are indicated separately. Reproduced from (15).
It can be concluded that the method for manual palpation described by Langemark & Olesen (4) makes it possible to compare pericranial tenderness between groups if the observer is blinded, and to compare pericranial tenderness within subjects if only one observer is used throughout a study. This method is therefore a valuable tool for the evaluation of pericranial tenderness.
Pericranial tenderness in patients with tension-type headache
The most prominent clinical finding in patients with tension-type headache is a considerably increased tenderness to palpation of pericranial myofascial tissues. This abnormality was documented for the first time in a blinded study in 1987 (4), and it has been confirmed in several blinded (5, 32, 33) and unblinded (17, 34–39) studies. The increased pericranial tenderness has been found both in patients with episodic and in patients with chronic tension-type headache (5, 40). The tenderness seems to be uniformly increased throughout the pericranial region and both muscles and tendon insertions have been found excessively tender (4, 5, 17) (Fig. 3). In addition, it has been demonstrated that the pericranial tenderness is positively associated with both the intensity and the frequency of tension-type headache (5), and that evaluation of tenderness by manual palpation is the most specific and sensitive test for the subdivision (3) of tension-type headache patients into those with and those without a muscular disorder (41).

Local tenderness scores ((right + left sides)/2) recorded by manual palpation at eight pericranial locations (mean ±
It is not known for certain whether the increased tenderness in tension-type headache is a primary or a secondary phenomenon to the headache. Migraineurs do have increased pericranial tenderness during migraine attacks (42), which could indicate that the tenderness is induced by the headache. The tenderness in the migraineurs did not however, correlate with the frequency of migraine attacks but with the frequency of interval headaches (42), and a large population study has demonstrated that there is no difference in pericranial tenderness between migraineurs studied outside of attack and non-migraineurs (5). Furthermore, the pericranial tenderness in patients with tension-type headache is increased not only on days with headache but also on days without headache (33, 37, 39, 40). This indicates that tenderness is not solely a consequence of an actual headache episode. A recent study (32) demonstrated that prolonged experimental tooth clenching induced headache in 68% of tension-type headache patients and in 16% of healthy controls. Shortly after clenching, pericranial tenderness was increased in those subjects who subsequently developed headache, whereas the tenderness score was unchanged in patients who remained headache-free. Experimental change of bite function in healthy controls (43) and experimental tooth clenching in migraine patients (44) also generated tension type-like headaches in a large proportion of the subjects. Together, these studies indicate that tenderness precedes headache and that muscular factors are of importance for the development of tension-type headache.
Possible peripheral mechanisms leading to myofascial tenderness
The term ‘myofascial pain’ can be defined as pain originating in striated muscle including its fascia and tendon insertions. Under normal conditions, myofascial pain is mediated by thin myelinated (Aδ) fibres and unmyelinated (C) fibres, while the thick myelinated (Aα and Aß) fibres normally mediate innocuous sensations (45). Various noxious and innocuous events such as ischaemia, mechanical stimuli and chemical mediators may excite and sensitize Aδ fibres and C fibres (46) and thereby play a role in the increased tenderness in tension-type headache.
It has been assumed for decades that contraction of head and neck muscles is of importance for the development of tension-type headache, as reflected in the former term for this disorder, ‘muscle-contraction headache’ (47). However, numerous laboratory-based electromyographic (EMG) studies have reported normal or, more often, only a slightly increased muscle activity in tension-type headache (37, 48–55). This probably cannot give rise to generalized muscle ischaemia. Alternatively, the increased muscle activity could be a normal protective adaptation to pain rather than its cause (56). The finding of a slightly increased muscle activity in patients with tension-type headache also on days without headache (40) however, makes this explanation less likely. The above-mentioned studies all used surface electrodes which record from relatively large areas of the muscle. In a recent study, Hubbard & Berkoff (57) used needle electrodes and reported EMG activity in so-called myofascial trigger points (7) to be significantly increased compared with EMG activity in adjacent non-tender muscle. Furthermore, EMG activity in the trigger points was significantly higher in patients with chronic tension-type headache than in healthy controls. The trigger point is apparently only a few millimetres in diameter, which explains why the increased EMG activity may only partially be detected with surface electrodes. Continuous activity in a few motor units over a long time may be sufficient for the development of myofascial pain and headache (58). Thus, it can be concluded that myofascial tenderness is not caused by generalized excessive muscle contraction leading to muscle ischaemia as was formerly believed (59). This is in agreement with a study demonstrating normal blood flow in the temporal muscle during rest as well as during dynamic exercise in chronic tension-type headache (60). However, it cannot be excluded that a slightly increased muscle activity may result in microtrauma of muscle fibres and tendon insertions and thereby to accumulation of metabolites, or that excessive activity in a few motor units may excite peripheral nociceptors.
During manual palpation it is often the clinical experience that tender muscles are harder, i.e. have a higher consistency than normal muscles. This was confirmed by Sakai et al. (61) who found a significantly higher degree of trapezius muscle hardness in patients with chronic tension-type headache than in healthy controls. This finding was made possible by the development of a new instrument for the measurement of muscle hardness, a so-called pressure/displacement transducer (62). A recent study (39) found a significant positive correlation between hardness and tenderness of the trapezius muscle in patients with chronic tension-type headache. Furthermore, there was no difference in hardness recorded on days with and on days without headache (39), indicating that increased hardness is not a direct consequence of actual pain. The mechanisms contributing to increased muscle hardness are not known, but may include slight tonic muscle contraction and tissue swelling due to the accumulation of chemical mediators (61).
Chemical mediators may also sensitize the nociceptive nerve endings. Particularly effective stimulants for skeletal muscle nociceptors are endogenous substances such as serotonin, bradykinin and potassium ions (63). These substances can be generated by numerous, not yet clarified, mechanisms. Serotonin can, for example, be released from platelets, bradykinin can be cleaved from its precursor plasma molecule kallin and potassium can be released from muscle cells, when pathologic conditions occur (e.g. lowering of pH during ischaemia, vascular damage, injury to muscle cells) (46). The release of neuropeptides, e.g. substance P and calcitonin gene-related peptide, from muscle afferents may also play a role in myofascial pain (64). The mode of action of the various mediators is complex and only poorly understood. Thus, the peripheral sensitization induced by a given mediator may be a rather specific process affecting only some aspects of receptor function, i.e. the sensitivity to local pressure (65), and the various mediators may interact and potentiate each other's effect. Thus, when serotonin and bradykinin are injected separately into the temporal muscle in healthy volunteers no pain is induced, while a mixture of the two substances induces both spontaneous muscle pain and tenderness (66). Much more research is needed to elucidate the role of chemical mediators in myofascial pain.
Search for other peripheral mechanisms responsible for sensitization of myofascial nociceptors has largely been negative (67–69), including temporal muscle biopsy studies (70) and studies on muscle metabolism (71). Thus, firm evidence for peripheral abnormalities as a cause of myofascial tenderness is still lacking.
Central mechanisms I: central sensitization
Central mechanisms are probably far more important for the pathophysiology of tension-type headache than previously anticipated. In this section, the importance of psychological stress and exteroceptive suppression will be briefly reviewed. Thereafter, central pain sensitivity in tension-type headache will be discussed with special reference to supraspinal mechanisms and mechanisms at the level of the spinal dorsal horn/trigeminal nucleus. Finally, the important interactions between peripheral and central mechanisms will be discussed.
Psychological stress
To the clinician, it is obvious that tension-type headache can be aggravated by psychological stress. In agreement with this, it has recently been found that stress and mental tension are the most conspicuous precipitating factors in tension-type headache (72–74), a series of experimental studies has demonstrated that tension type-like headaches can be induced by psychological stress (75, 76), and psychological and behavioural therapies appear to be as effective for the treatment of tension-type headache as pharmacotherapy (77). The mechanisms by which psychological stress plays a role in tension-type headache are not known, but central factors such as involuntary contractions of cephalic muscles, a decrease in supraspinal descending pain-inhibitory activity, and supraspinal hypersensitivity to nociceptive stimuli may be involved (78, 79). Thus, while it is evident that psychological stress is of importance in tension-type headache, the exact role of this factor in the generation, exacerbation, and maintenance of headache remains unclear (80).
Exteroceptive suppression
In 1987 Schoenen et al. (81) reported that the duration of a brainstem reflex, the so-called late exteroceptive suppression period (ES2) of temporal muscle activity, was reduced in patients with chronic tension-type headache. ES2 is probably mediated via inhibitory interneurones located in the brainstem (82, 83). These inhibitory interneurones are strongly modulated by cortical structures (84, 85), and it has therefore been suggested that ES2 may provide information about the central mechanisms related to tension-type headache (51). The original paper by Schoenen et al. (81) was received with great interest, and the finding of reduced ES2 periods in patients with chronic tension-type headache was later confirmed by two other research groups (86, 87). Unfortunately, the applied methodology, which is crucial for the reliability of ES2 measurements (88–91), was far from optimal in most of the earlier ES2 studies. A computerized method for recording and analysing ES2 was therefore developed (92). The computerized averaging method made it possible to analyse ES2 with low intra- and interobserver variations (92). By means of the computerized method and a blinded design, Bendtsen et al. then measured ES2 in 55 patients with chronic tension-type headache and in 55 healthy controls. Surprisingly, no difference in ES2 duration between patients and controls could be detected (93). The same result was found by Zwart & Sand (94) in a blinded study including 11 patients with chronic tension-type headache, and by Lipchik et al. (33) in a blinded study including 25 young women with chronic tension-type headache. In addition, ES2 has been found normal in an open study including 27 young women with chronic tension-type headache (38). Finally, it has not been possible to detect any significant correlations between ES2 duration and either clinical characteristics or various pain parameters in patients with tension-type headache (33, 48, 93, 95), and ES2 duration has been found to be reduced, rather than increased, during prophylactic treatment with amitriptyline in patients with chronic tension-type headache (95). Thus, while ES2 may be of great interest, e.g. in the study of how experimental stressors and drugs influence central mechanisms (92, 95–97), present knowledge indicates that ES2 is probably not closely related to the pathophysiology of tension-type headache (98).
Central pain sensitivity
Supraspinal pain sensitivity
The abnormal pericranial tenderness in patients with tension-type headache may be due to an increased sensitivity in the central nervous system to nociceptive stimuli from the periphery. Pain sensitivity has therefore been extensively studied in tension-type headache. The pressure pain detection threshold, i.e. the lowest pressure stimulus that is perceived as painful, has been found normal in patients with episodic tension-type headache (5, 50) and in groups of mixed episodic and chronic tension-type headache patients (55, 99). In contrast, pressure pain detection thresholds have been found decreased in patients with chronic tension-type headache in two studies (12, 17) (Fig. 4). The same result was indirectly found in an earlier study that compared patients with chronic tension-type headache with historical controls (10). One population study found normal pressure pain detection thresholds in 14 subjects with chronic tension-type headache (5). This study is however, difficult to compare with the above-mentioned studies because of the low number of subjects examined, and, more important, because the subjects in the population study probably were clinically less affected than the patients in the three other studies, who were recruited from specialized headache centres. The pressure pain tolerance threshold, i.e. the maximal pressure stimulus that is tolerated, has only been compared between chronic tension-type headache patients and healthy controls in one study (17), although pain tolerance is generally considered a better and more reproducible correlate to clinical pain than pain detection (12, 100–101). In this study (17), pressure pain tolerance thresholds in the finger were significantly lower in chronic tension-type headache patients than in healthy controls (Fig. 4). The lowered pressure pain detection and tolerance thresholds indicate the presence of both allodynia, i.e. pain elicited by stimuli which are normally not perceived as painful, and hyperalgesia, i.e. increased sensitivity to painful stimuli, in patients with chronic tension-type headache.
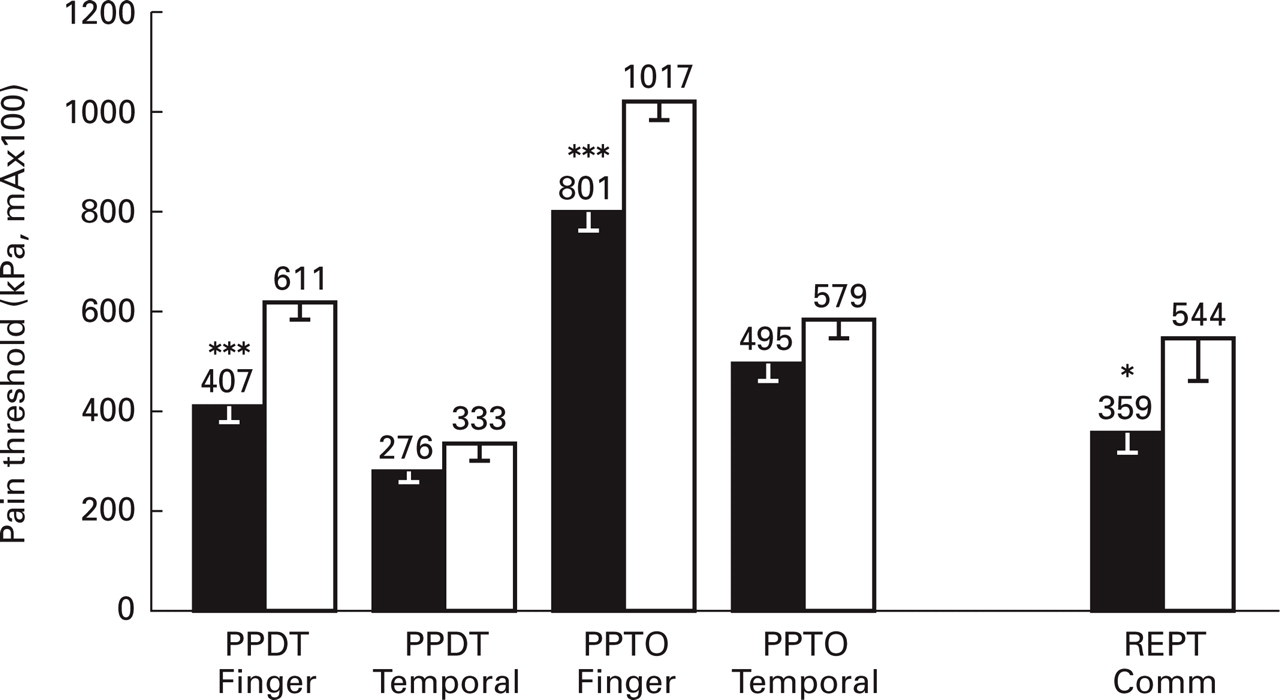
Pain thresholds to mechanical and electrical stimuli in 40 chronic tension-type headache patients (▪) and in 40 healthy controls (□) (mean ±
Patients with chronic tension-type headache have also been found hypersensitive to stimuli other than pressure. Langemark et al. (10) found significantly decreased pain detection thresholds to thermal stimuli in patients with chronic tension-type headache, and both the pain detection threshold (17) (Fig. 4) and the pain tolerance threshold (102) to electrical stimuli have been found decreased in these patients. The sensitivity to the various stimulus modalities (pressure, thermal and electrical) has been found increased both at cephalic and extra-cephalic locations (10, 12, 17, 102). The fact that pain hypersensitivity has been demonstrated for various types of stimuli applied both at cephalic and at extra-cephalic, non-symptomatic locations strongly indicates that the pain sensitivity in the central nervous system is increased in patients with chronic tension-type headache. The widespread and unspecific nature of the hypersensitivity indicates that the general pain sensitivity is affected at the supraspinal level rather than at the segmental level of the spinal dorsal horn/trigeminal nucleus. Thus, it can be concluded that the general pain sensitivity in the central nervous system is increased in patients with chronic tension-type headache, while the central pain processing seems to be normal in patients with episodic tension-type headache.
In the study by Bendtsen et al. (17) pressure pain detection and pressure pain tolerance thresholds were decreased in parallel in chronic tension-type headache patients compared with controls, and the two thresholds were significantly correlated both in the finger and in the temporal region in the headache patients. In addition, pressure pain thresholds recorded at cephalic and extra-cephalic sites were highly correlated. These data indicate that pain detection and pain tolerance, as well as pain perception in different parts of the body, are modulated by a common, probably supraspinal, factor in patients with chronic tension-type headache. Moreover, pressure pain detection and tolerance thresholds recorded both at cephalic and extra-cephalic sites, as well as the pain threshold to electrical stimuli, were significantly and negatively correlated to the TTS in patients with chronic tension-type headache (17). In agreement with this, Langemark et al. (10) found a significant negative correlation between pressure pain detection thresholds in the temporal region and TTS in patients with chronic tension-type headache, while Jensen et al. (40) found a significant negative correlation between TTS and both pressure pain detection and pressure pain tolerance thresholds in patients with chronic tension-type headache, but, importantly, not in patients with episodic tension-type headache. These data demonstrate that there is a relationship between the central hypersensitivity and the increased pericranial myofascial tenderness in patients with chronic, but not in patients with episodic, tension-type headache. However, the increase in myofascial tenderness has been found much more pronounced than the increase in general pain sensitivity, as can be seen by comparing Figs 3 and 4, and the correlation between pain thresholds and TTS was only moderate in the above-mentioned studies (10, 17, 40). Thus, the general hypersensitivity can explain only a minor part of the increased pericranial tenderness in patients with chronic tension-type headache. It is therefore likely that other factors contribute to the increased tenderness.
Pain sensitivity at the level of the spinal dorsal horn/trigeminal nucleus
In 1990 it was suggested by Jensen (9) that myofascial tenderness may be the result of a lowered pressure pain threshold, a stronger response to pressures in the noxious range (as illustrated by a steeper stimulus-response function) or a combination of both. However, it was not possible to study the relation between palpation pressure and pain until the development of the palpometer (14). By means of this instrument, the stimulus-response function for pressure vs. pain was investigated in 40 patients with chronic tension-type headache and in 40 healthy controls (16). The stimulus-response function recorded from normal muscle was well described by a power function. From highly tender muscle, the stimulus-response function was displaced towards lower pressures and, more important, it was approximately linear, i.e. qualitatively different from that of normal muscle (Fig. 5). When the patients were subgrouped on the basis of their degree of tenderness, it was found that the stimulus-response function was almost perfectly linear in the most tender patients and almost perfectly described by a power function in the least tender patients. Thus, the abnormal stimulus-response function was related to the degree of tenderness and not to the diagnosis of tension-type headache. This was confirmed in a subsequent study (21) in patients with fibromyalgia, where the relation between pressure and pain in the trapezius muscle was almost perfectly linear and not slightly curved as in the patients with chronic tension-type headache. This was in accordance with the finding that the trapezius muscle in patients with fibromyalgia was more tender than in the headache patients. The finding of a qualitatively altered response to nociceptor stimulation in tender muscle indicates that myofascial pain, at least in part, is caused by qualitative changes in the processing of sensory information. The question is whether these changes are located in peripheral nerve endings, in the spinal cord or in higher order neurones.

Stimulus–response functions for pressure vs. pain in the trapezius muscle in 40 patients with chronic tension-type headache (•) and in 40 healthy controls (▴) (mean ±
Spinal dorsal horn neurones that receive inputs from deep myofascial tissues can be classified as high-threshold mechanosensitive (HTM) neurones requiring noxious intensities of stimulation for activation, and as low-threshold mechanosensitive (LTM) neurones, which are activated by innocuous stimuli (63). Yu & Mense (103) have shown that HTM dorsal horn neurones have a positively accelerating stimulus-response function, whereas the stimulus-response function of LTM neurones is approximately linear. This suggests that the linear stimulus-response function in tender human muscle may be caused by activity in LTM afferents. At first this seems unlikely, because LTM afferents normally mediate innocuous sensations. However, Woolf (13) has demonstrated that a prolonged noxious input from the periphery is capable of sensitizing spinal dorsal horn neurones, so that LTM afferents can mediate pain. The original finding by Woolf on spinal dorsal horn sensitization has later been confirmed by numerous independent laboratories (63, 104–107), and a similar sensitization of trigeminal brainstem nociceptive neurones following stimulation of craniofacial muscle afferents has been reported by Hu et al. (108). While the above-mentioned studies have been performed on animal models, Torebjörk et al. (109) demonstrated similar changes in the central processing of inputs from LTM afferents in humans following intradermal injection of capsaicin. It therefore seems likely that the abnormal stimulus-response function in tender muscle can be explained by changes in neuronal behaviour at the spinal/trigeminal level. A decrease of the supraspinal descending inhibition probably does not explain the finding, because it has been reported that the descending inhibition acts via a parallel shift or via a decreased slope of the stimulus-response curve (103, 110), while it does not change the shape of the stimulus-response curve. Sensitization or increased stimulation of normally active peripheral nociceptors would probably induce a quantitative rather than a qualitative change of the stimulus-response curve (111). However, activation of a new class of peripheral nociceptors, e.g. silent nociceptors, might result in a qualitatively changed stimulus-response function. Although silent nociceptors have not yet been identified in skeletal muscle (63, 64), this might be an alternative explanation for the abnormal stimulus-response function. In conclusion, the finding of qualitatively altered nociception from tender human muscles indicates that the central nervous system is sensitized at the level of the spinal dorsal horn/trigeminal nucleus in patients with chronic myofascial pain.
It has long been a puzzle why patients with chronic tension-type headache are much more tender to manual palpation than controls, while there is only a slight difference in pain thresholds between the two groups. The abnormal stimulus-response function (16) showing that the largest difference between tender and normal muscle is found at moderate pressure intensities, together with the finding that most observers use a moderate pressure intensity during manual palpation (14), explains this (Fig. 6). Thus, the central sensitization at the level of the spinal dorsal horn/trigeminal nucleus probably accounts for a large part of the increased tenderness in patients with chronic myofascial pain. This does not necessarily imply that spinal mechanisms are more important than supraspinal mechanisms in chronic myofascial pain. A supraspinal dysfunction may, for example, be a prerequisite for the development of alterations at the level of the spinal dorsal horn/trigeminal nucleus. Furthermore, the relative importance of spinal and supraspinal pain mechanisms and the interaction between these is only poorly understood. The proposed separation between supraspinal and spinal pain sensitivity in tension-type headache should therefore be regarded only as a working hypothesis.
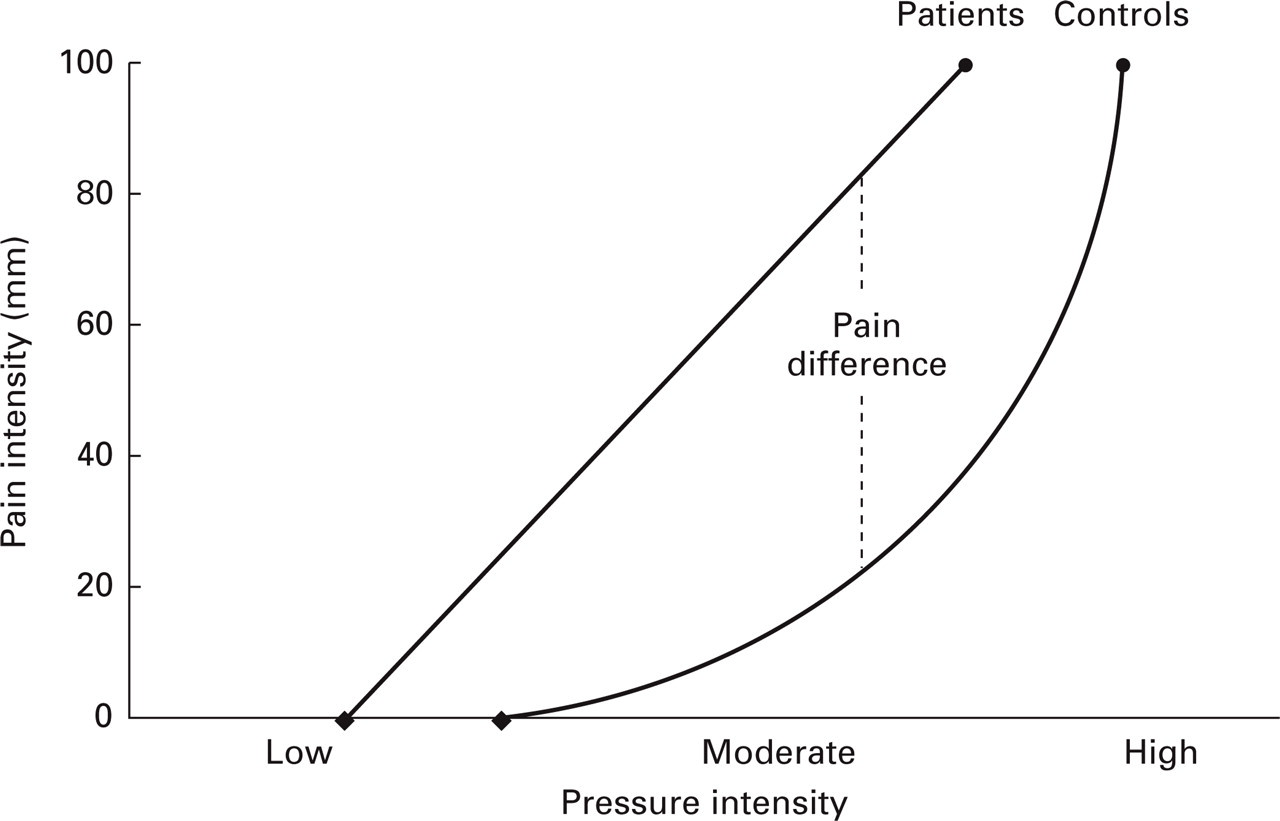
Theoretical explanation for the markedly increased tenderness in patients with chronic tension-type headache. Thestimulus–response functions for pressure vs. pain in patients and in healthy controls have revealed that there is only a smalldifference between tender and normal muscles at low and high pressure intensities, while there is a considerable difference intenderness at moderate pressure intensities (16). Together with the finding (14) that most observers use a moderate pressure intensity during manual palpation, this may explain the markedly increased tenderness but only slightly decreased pain detection andtolerance thresholds in patients with chronic tension-type headache (17).
Possible peripheral mechanisms leading to central sensitization
As described above, the central nervous system is probably sensitized both at the supraspinal level (10, 12, 17) and at the level of the spinal dorsal horn/trigeminal nucleus (16) in patients with chronic tension-type headache. Which factors are responsible for the central sensitization? Basic pain research has, as previously mentioned, demonstrated that central sensitization can be generated by prolonged nociceptive inputs from the periphery. This mechanism is particularly liable to be of importance in patients with chronic myofascial pain, because inputs from muscle nociceptors are more effective in inducing prolonged changes in the behaviour of dorsal horn neurones than are inputs from cutaneous nociceptors (112). The previously described finding of a relation between the increased tenderness and the general pain hypersensitivity in patients with chronic tension-type headache (17) was recently investigated further by Jensen et al. (40) by comparing pain thresholds in tension-type headache patients with and in tension-type headache patients without abnormal pericranial tenderness. This study demonstrated that chronic tension-type headache patients with abnormal tenderness had significantly lower pressure pain detection and tolerance thresholds than chronic tension-type headache patients without abnormal tenderness. No such difference was detected in patients with episodic tension-type headache. In addition, it was found that chronic tension-type headache patients with abnormal tenderness tended to have lower mechanical pain thresholds than healthy controls, while patients without abnormal tenderness had significantly higher pain thresholds than controls (40). Thus, in agreement with previous studies (16, 17), this study indicates that central pain sensitivity is increased only in chronic tension-type headache patients with increased pericranial tenderness.
The demonstration of a relation between pericranial tenderness and central sensitization does not reveal the cause–effect relationship between these factors. However, since patients with episodic tension-type headache have increased pericranial tenderness but normal central pain sensitivity, and since chronic tension type-headache usually evolves from the episodic form (113), it is most likely that the central sensitization in patients with chronic tension-type headache is induced by prolonged nociceptive inputs from myofascial tissues, as previously suggested (16, 17). The central sensitization could theoretically also be secondary to the chronic pain condition itself, but this is most unlikely, because the central nervous system is not sensitized in patients with chronic tension-type headache who are not tender to palpation (40). However, until prospective studies have investigated peripheral and central pain mechanisms in episodic tension type-headache patients who later develop the chronic form, this question cannot be definitively answered. In conclusion, the central sensitization in patients with chronic tension-type headache is probably induced by prolonged nociceptive inputs from pericranial myofascial tissues.
Central mechanisms II: biochemical mechanisms
A disturbance in pain-modulating transmitters as well as cellular changes in the central nervous system are likely to be involved in the altered pain perception in patients with chronic tension-type headache. Serotonergic mechanisms in tension-type headache and the possible synaptic and cellular events in the spinal dorsal horn/trigeminal nucleus involved in central sensitization will be discussed in this section.
Serotonergic mechanisms
Serotonin (5-hydroxytryptamine, 5-HT) is a widely distributed neurotransmitter that has an important but complex role in pain modulation. Thus, while 5-HT has mainly algogenic actions on peripheral nerves as previously described, it seems to have predominantly anti-nociceptive effects in the central nervous system (114). 5-HT is an important transmitter in the anti-nociceptive pathways descending from the brainstem to the spinal dorsal horn, and it is probably also involved in ascending anti-nociceptive pathways (115, 116). The anti-nociceptive effects of 5-HT are mediated via many different 5-HT receptor subtypes, e.g. 5-HT1, 5-HT2 and 5-HT3 receptors (117). The complexity of the 5-HT pain modulatory system is underscored by the fact that the effect of 5-HT may vary even within the same 5-HT receptor subtype (118). 5-HT may, for example, have both a facilitatory and an inhibitory action on spinal nociceptive processing when acting via the 5-HT1A receptor (78). In addition, 5-HT has effects on other modalities than pain, i.e. vascular effects, that indirectly may influence pain mechanisms. This further complicates the investigation of the role of 5-HT in pain. Thus, while it is certain that 5-HT has important anti-nociceptive effects, the exact role of 5-HT in pain modulation is far from understood.
Efficacy of serotonergic drugs in tension-type headache
The prominent role of 5-HT in anti-nociception together with the findings of increased pain sensitivity in patients with chronic tension-type headache render it probable that 5-HT is of relevance for the pathophysiology of tension-type headache. One way to test this assumption is to examine the effect of various serotonergic drugs in patients with tension-type headache. Lance & Curran (119) and Diamond & Baltes (120) reported more than two decades ago that the non-selective 5-HT reuptake inhibitor amitriptyline had a prophylactic effect in chronic tension-type headache. However, recent studies have questioned these findings. Göbel et al. (121) found a significant effect of amitriptyline over placebo only in the last week of a 6-week study, and Pfaffenrath et al. (122) found no significant effect of amitriptyline compared with placebo in a multicentre study. In the latter study however, the frequencies of side-effects were similar in amitriptyline and placebo. Usually, amitriptyline has marked side-effects and the inability to detect these suggests insensitivity of the trial for reasons which remain obscure. In an attempt to clarify whether amitriptyline has an effect in chronic tension-type headache, and in order to investigate whether such an effect could be attributed to blockade of 5-HT reuptake, Bendtsen et al. (18) conducted a double-blind, placebo-controlled, three-way crossover study of the effect of amitriptyline and of the effect of citalopram, a highly selective 5-HT reuptake inhibitor, in patients with chronic tension-type headache. The patients had been resistant to numerous previous treatments and were not suffering from depression. Amitriptyline reduced the area under the headache curve (calculated as headache duration times headache intensity) by 30% compared with placebo, which was highly significant, while citalopram had only a slight (12%) and insignificant effect (Fig. 7). Amitriptyline also significantly reduced the secondary efficacy parameters headache duration, headache frequency and intake of analgesics.
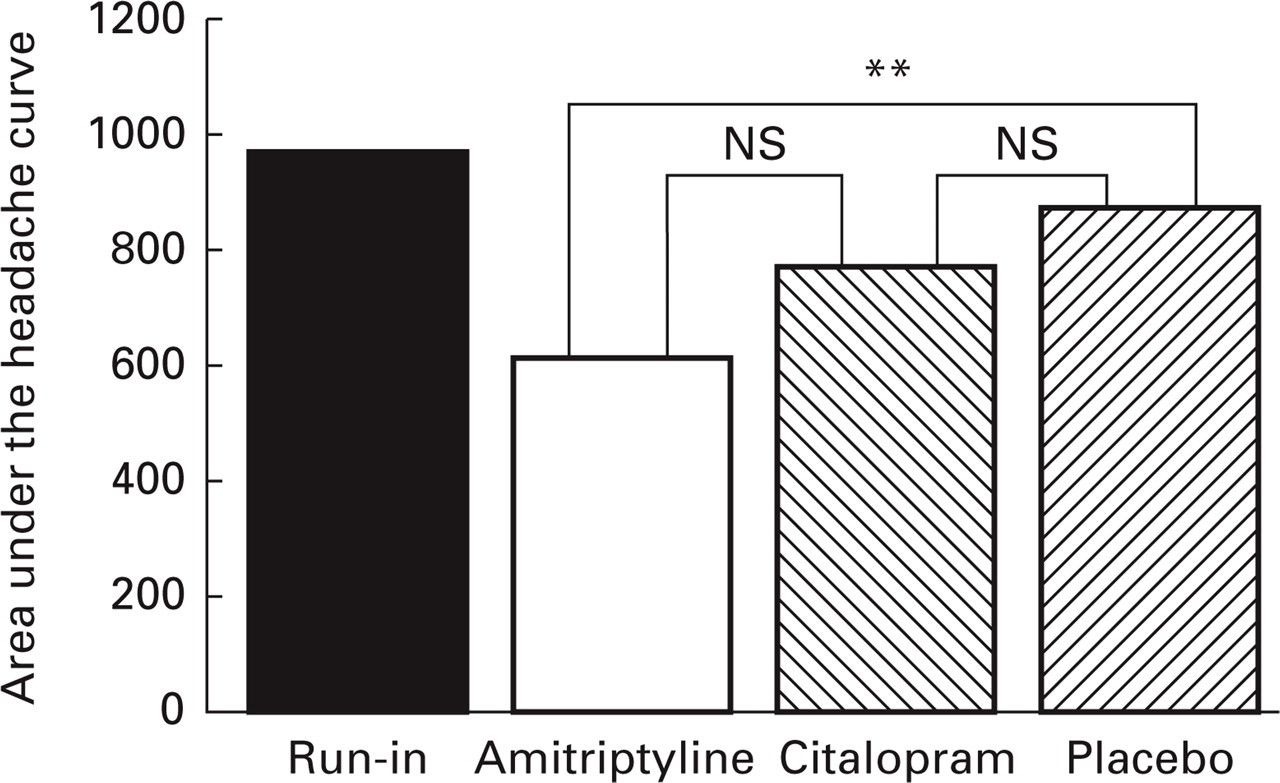
The prophylactic effect of amitriptyline, a non-selective 5-HT reuptake inhibitor, and of citalopram, a highly selective 5-HT reuptake inhibitor, was compared in a double-blind, placebo-controlled, three-way crossover study including 40 patients with chronic tension-type headache. The barsrepresent the area under the headache curve (duration × intensity) during a 4-week run-in period and during the last 4 weeks of treatment with amitriptyline, citalopram andplacebo. ∗∗P = 0.002; NS, not significant. Data from (18).
Previously it was assumed that the analgesic properties of the tricyclic antidepressants could be ascribed to the blockade of serotonin reuptake in the central nervous system (123–126), but this has recently been questioned. In an animal model, Ardid et al. (127) found that both noradrenaline reuptake inhibitors and selective serotonin reuptake inhibitors had analgesic effects, but that amitriptyline was more effective than both these drugs. Watson & Evans (128) found amitriptyline more effective than the selective serotonin reuptake inhibitor zimeldine in postherpetic neuralgia, and Sindrup et al. (129) and Max et al. (130) have reported tricyclic antidepressants to be more effective than selective serotonin reuptake inhibitors in painful diabetic neuropathy. These results are in line with the finding of a clear effect of amitriptyline but only a trend towards an effect of citalopram in tension-type headache (18). While citalopram is an extremely specific serotonin reuptake inhibitor (131), amitriptyline also has effects on reuptake of noradrenaline (132) as well as effects on serotonergic (133), adrenergic (134), cholinergic (133), and histaminergic (135) receptors. Of these effects, especially inhibition of noradrenaline reuptake (132, 136) and the effects on various serotonin receptor subtypes (117, 137) have been considered important. The efficacy of the 5-HT1D-like receptor agonist sumatriptan has recently been examined in patients with tension-type headache, but sumatriptan had only a minimal effect (138, 139). In addition to the above-mentioned properties of amitriptyline, it is known that the drug potentiates the effect of endogenous opioids (123, 140). This may be of importance, because cerebrospinal fluid Met-enkephalin levels have been found increased in chronic tension-type headache patients (141). However, the cerebrospinal fluid β-endorphin levels were found to be normal in the same group of patients (142). Most interestingly, it has been demonstrated that amitriptyline may act as a N-methyl-
In conclusion, our present knowledge on the effects of serotonergic drugs in tension-type headache does not allow definite conclusions on the role of 5-HT in the pathophysiology of this disorder. It is likely that the blockade of 5-HT reuptake plays some role in the efficacy of amitriptyline in tension-type headache, but that also other of the actions of amitriptyline, e.g. blockade of noradrenaline reuptake or modulation of 5-HT or NMDA receptors, are required in order to achieve a clinically significant reduction in headache.
Plasma and platelet 5-HT levels in tension-type headache
The role of 5-HT in tension-type headache may also be investigated by studying 5-HT in the peripheral blood. Platelets are considered a model for serotonergic neurones, because there are several morphological, biochemical and pharmacological similarities between platelets and serotonergic nerve endings (146). Platelets themselves do not synthesize 5-HT but accumulate it from the low concentrations which circulate free in the plasma and which mainly originate from the enterochromaffin tissue of the gastrointestinal tract (147). Platelet 5-HT is stored in dense granules and represents a slow turnover reserve pool, which is pharmacologically inactive. In contrast, extracellular plasma 5-HT has a rapid turnover and is pharmacologically active (148). The platelet and plasma levels of 5-HT may reflect the 5-HT content in the serotonergic nerve endings and in the synapses, respectively. The term plasma 5-HT has previously been inconsistently defined (for a review see (149)). In the following, the term plasma 5-HT refers to the extracellular platelet-poor plasma portion of 5-HT in the blood.
The numerous studies on peripheral 5-HT metabolism in tension-type headache have given very inconsistent results. Platelet 5-HT has been reported to be reduced (150–153), normal (19, 154, 155) or increased (156, 157), while plasma 5-HT has been found increased during headache-free periods (156, 158), normal during headache (19, 155), or normal during headache-free periods and increased during headache (159). These studies seem conflicting but are in fact incomparable because of large clinical and methodological differences. In addition, the characterization of the patients has been inadequate in many studies. If only studies including patients with episodic or chronic tension-type headache (3) and excluding patients with concomitant serious disorders, e.g. depression, are considered, a more simple picture emerges. These studies indicate that patients with episodic tension-type headache have increased platelet 5-HT and plasma 5-HT levels (156), while patients with chronic tension-type headache have normal or decreased platelet 5-HT and plasma 5-HT levels (19, 152, 155).
Simultaneous with the measurement of plasma and platelet 5-HT levels, the plasma level of beta-thromboglobulin (β-TG) which is regarded as a marker of in vivo platelet activation (160), and the urinary excretion of the main 5-HT metabolite 5-hydroxyindoleacetic acid (5-HIAA) were measured in patients with chronic tension-type headache (19). Both of these parameters were found to be normal. The normal levels of platelet 5-HT and plasma β-TG suggest that the platelets are not continuously activated (161). This is in line with the finding of a normal urinary excretion of 5-HIAA, which indicates that the systemic 5-HT turnover is not increased (147) in patients with chronic tension-type headache. It can be concluded that present knowledge, which is rather limited, indicates that plasma 5-HT and platelet 5-HT levels are increased in patients with episodic tension-type headache, while the peripheral 5-HT metabolism seems to be largely normal in patients with chronic tension-type headache.
Platelet 5-HT uptake in tension-type headache
The study of 5-HT uptake may be a more direct method of investigating the role of 5-HT in tension-type headache than the study of platelet 5-HT and plasma 5-HT levels, because the presynaptic 5-HT uptake mechanism is crucial for the regulation of 5-HT levels in the neuronal synapses. 5-HT uptake can be measured indirectly by measuring the number of 5-HT transporters in the platelet membrane, because there is a high correlation between the number of 5-HT transporters and 5-HT uptake (162). As mentioned above, human platelets have for many years been considered a useful presynaptic neurone model for the 5-HT nerve terminal in the central nervous system (163), and recent studies have confirmed that the amino acid sequence of the 5-HT transporter protein expressed in platelets and in 5-HT neurones is identical (164). Thus, it is possible that some pathophysiological abnormalities linked to the 5-HT transporter protein would occur in parallel in platelets and in the brain (165). As the 5-HT transporter contains specific binding sites for several antidepressant drugs, receptor binding analysis can be used to determine the number of transporters. This subject has been investigated in only a few tension-type headache studies. Marazziti et al. (166) reported decreased imipramine binding in patients with episodic tension-type headache, while Jarman et al. (167) found no difference in this parameter between patients with tension-type headache and healthy controls. Headache frequency was not reported in the latter study. Using tritiated paroxetine as the binding ligand, Bendtsen & Mellerup (20) found the number of platelet 5-HT transporters to be normal in patients with chronic tension-type headache. From a methodological point of view, tritiated paroxetine is a better ligand than imipramine because the number of 5-HT transporters can be determined with greater accuracy (162, 165, 168). In addition, the number of platelet 5-HT transporters in the chronic tension-type headache patients did not predict the outcome of subsequent treatment with amitriptyline (20). This is in agreement with a study on patients with idiopathic pain (169) and supports the previous assumption that mechanisms other than inhibition of serotonin reuptake are involved in the analgesic effect of the tricyclic antidepressants (18). Using radioactively labelled 5-HT, platelet 5-HT uptake has been reported increased (154) in patients with tension-type headache (headache frequency not reported) and normal (170) in patients with chronic tension-type headache. Thus, the above-mentioned studies indicate that platelet 5-HT uptake is decreased in patients with episodic tension-type headache and normal in patients with chronic tension-type headache. This is in line with the findings of increased, respectively, normal plasma 5-HT levels in the two groups. It is therefore possible that the increased plasma 5-HT level in patients with episodic tension-type headache is related to a decreased number of platelet 5-HT transporters. However, further studies are needed to confirm these findings.
In summary, present knowledge indicates that patients with episodic tension-type headache have a decreased platelet 5-HT uptake and increased plasma 5-HT levels, while patients with chronic tension-type headache have a normal platelet 5-HT uptake and normal plasma 5-HT levels. Furthermore, it has been found that plasma 5-HT increases during a headache attack in a mixed population of episodic and chronic tension-type headache patients (159), while there is a significant negative correlation between plasma 5-HT and headache frequency in patients with chronic tension-type headache (19). Thus, patients with chronic tension-type headache may have an impaired ability to increase plasma 5-HT, and therefore possibly also synaptic 5-HT levels, in response to increased nociceptive inputs from the periphery. Such a serotonergic dysfunction could contribute to central sensitization at the level of the spinal dorsal horn/trigeminal nucleus and thereby to the conversion from episodic to chronic tension-type headache. However, this must be regarded as only a preliminary hypothesis. First, the above-mentioned results should be confirmed in future studies. Second, we do not know whether the peripheral changes in 5-HT actually do reflect similar mechanisms in central neurones.
Possible synaptic and cellular mechanisms leading to central sensitization
In the previous section, it was suggested that the central sensitization in patients with chronic tension-type headache may be induced by prolonged nociceptive inputs from pericranial myofascial tissues. The recent progress in basic pain research has increased our knowledge about the mechanisms that may underlie the central neuroplasticity. Thus, there is ample experimental evidence showing that persistent activity in peripheral nociceptors may lead to increased responsiveness of second order dorsal horn neurones, and that neurotransmitters released from C-fibres may be responsible for the altered neuronal function (171). Neurotransmitters known to be involved in the development of central sensitization include the tachykinins substance P and neurokinin A and the excitatory amino acid glutamate (172). Prolonged release of these neurotransmitters may activate post-synaptic receptors that are normally blocked, e.g. NMDA receptors (173). Activation of NMDA receptors leads to increased influx of calcium, which initiates a cascade of biochemical events including increased production of nitric oxide, prostaglandins, protein kinases and protein products of immediate early genes (174, 175). This may lead to long-term metabolic changes and increased excitability of the affected cell (176).
The increased excitability of the dorsal horn neurones significantly alters the pain perception in the individual patient. In the sensitized state, previously ineffective low-threshold Aß-fibre inputs to nociceptive dorsal horn neurones become effective (177). This means that pain can be generated by low-threshold Aß-fibres, which clinically will manifest itself as allodynia. Woolf & Doubell (171) have suggested that the major cause of increased pain sensitivity in the chronic pain condition is an abnormal response to inputs from low-threshold Aß-fibres. In addition, the response to activation of high-threshold afferents is exaggerated, which clinically will manifest itself as hyperalgesia.
Mechanisms other than increased excitability of second order neurones that have been proposed to be involved in central sensitization include: (i) structural reorganization such that novel synapses between Aß-fibres and nociceptive dorsal horn neurones are formed (178); (ii) decreased supraspinal inhibition of Aß-fibre inputs to nociceptive dorsal horn neurones (171); and (iii) the formation of a presynaptic link between Aß-fibres and nociceptive dorsal horn neurones as a consequence of C-fibre-induced activation of spinal interneurones (179). Thus, in spite of the complexity of the nociceptive system, our understanding of the mechanisms involved in sensitization at the level of the spinal dorsal horn has dramatically improved in recent years. It is likely that similar mechanisms are of importance at the supraspinal level, but present knowledge on supraspinal neuroplasticity is very limited.
A pathophysiological model for chronic tension-type headache
On the basis of the previously described findings in basic pain and headache research, a simplified pathophysiological model for chronic tension-type headache can be created. In healthy subjects, the processing of pain is finely regulated by multiple pathways, so that the degree of perceived pain is appropriate for the actual situation (Fig. 8a). The model shows two of the pain modulatory structures: the periaquaductal grey (PAG) in the midbrain that gives rise to inhibitory pathways descending to the spinal dorsal horn, and the rostral ventromedial medulla (RVM) in the brain stem that, in addition to so-called off-cells that inhibit nociceptive transmission, also contains on-cells that facilitate nociceptive transmission in the spinal dorsal horn (78). The nociceptive system allows the detection of potential harmful events and enables the individual to react appropriately to these, e.g. to avoid unphysiological working positions that cause painful pericranial muscles and headache. The painful stimulus from the periphery is usually eliminated by actions from the individual and, if necessary, by local reparative mechanisms in the myofascial tissues, and the properties of the nociceptive system will normally not be altered after a short-lasting painful episode. This is termed ‘normal pain processing’ in the model and will probably be representative of the nociceptive system in subjects with rare episodes of tension-type headache.
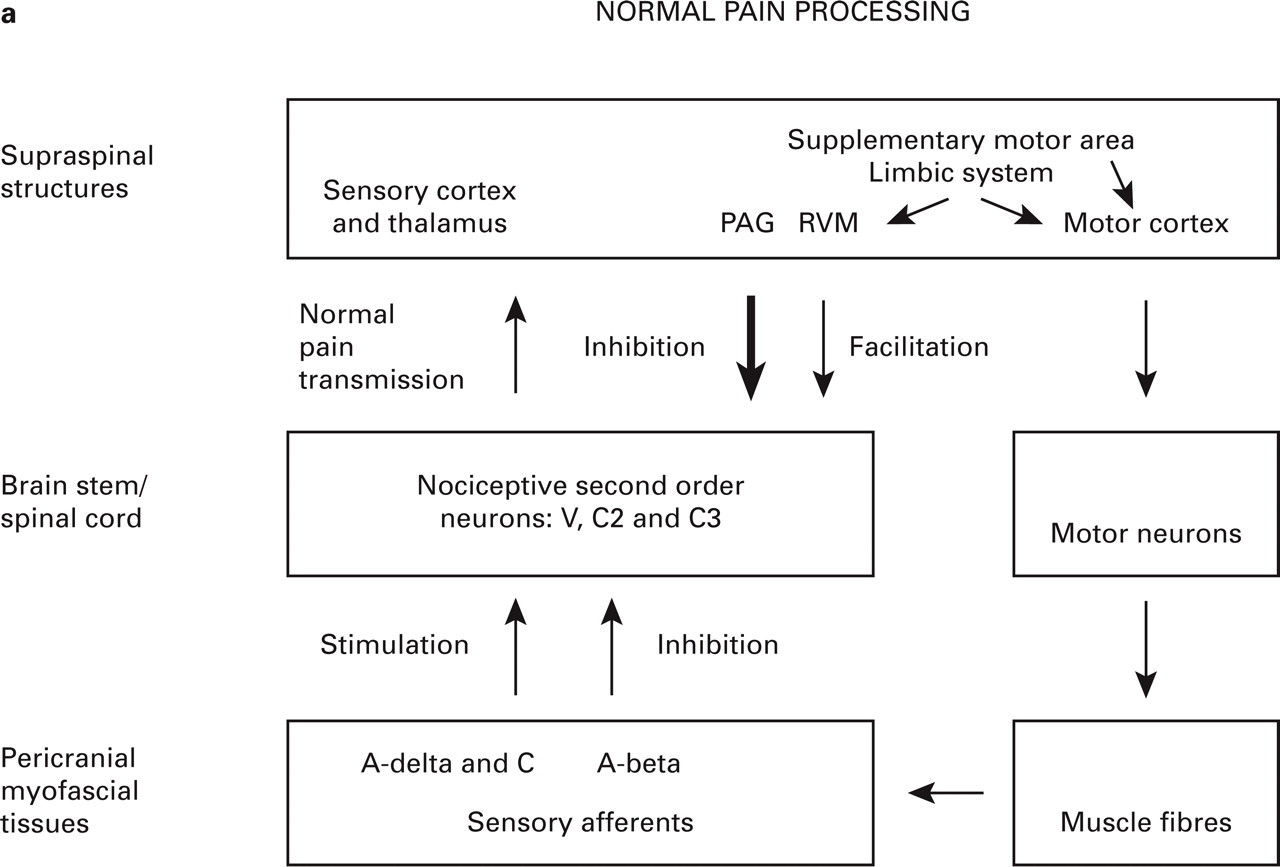
(See previous page.) A simplified theoretical model of chronic tension-type headache. The model states that the main problem in chronic tension-type headache is sensitization of dorsal horn neurones due to increased nociceptive inputs from pericranial myofascial tissues. (a) Normal pain processing. (b) Aberrant pain processing. Important alterations from the normal pain state are presented in bold: the nociceptive input from myofascial A-delta- and C-fibres is increased for unknown reasons, resulting in plastic changes in the spinal dorsal horn/trigeminal nucleus. As a consequence, the normally inhibitory effect of low-threshold A-beta-fibres on pain transmission in the spinal dorsal horn is altered to a pain stimulatory effect, and the response to nociceptive A-delta- and C-fibres is potentiated. The increased nociceptive stimulation of supraspinal structures may result in increased facilitation and decreased inhibition of pain transmission at the level of the spinal dorsal horn/trigeminal nucleus and in increased pericranial muscle activity. Together these mechanisms may induce and maintain the chronic pain condition. V, Trigeminal nerve; C2 and C3, second and third cervical segment of the spinal cord; PAG, periaquaductal grey; RVM, rostral ventromedial medulla.
Under some conditions, the painful stimulus from the pericranial myofascial tissues may be more prolonged or more intense than normal. As previously described, the mechanisms behind this are not known but may include increased muscle activity or the release of various chemical mediators secondary to local pathological conditions. Increased muscle activity secondary to psychogenic stress is likely to be of relevance in this respect, because the psychogenic stress condition may cause a prolonged increase of muscle tone via the limbic system and at the same time potentiate pain facilitation from the brain stem to the spinal dorsal horn (79). In most subjects these conditions will be self-limiting due to central pain modulatory mechanisms and local reparative processes, and will be experienced as frequent headache episodes for a limited period of time. However, in predisposed individuals the prolonged nociceptive input from the pericranial myofascial tissues may lead to sensitization of nociceptive second order neurones at the level of the spinal dorsal/trigeminal nucleus (Fig. 8b). The pathophysiological basis for the increased susceptibility to central sensitization is unknown. Possible mechanisms include an impaired supraspinal inhibition of nociceptive transmission in the spinal dorsal horn due to a serotonergic dysfunction, as proposed in the previous section. In the sensitized state, the afferent Aß-fibres that normally inhibit Aδ- and C-fibres by presynaptic mechanisms in the dorsal horn will on the contrary stimulate the nociceptive second order neurones. In addition, the effect of Aδ- and C-fibre stimulation of the nociceptive dorsal horn neurones will be potentiated, and the receptive fields of the dorsal horn neurones will be expanded (173). The nociceptive input to supraspinal structures will therefore be considerably increased, which may result in increased excitability of supraspinal neurones (180) as well as decreased inhibition or increased facilitation of nociceptive transmission in the spinal dorsal horn (181), i.e. in generalized pain hypersensitivity. The central neuroplastic changes may also increase the drive to motor neurones both at the supraspinal and at the segmental level (13), resulting in slightly increased muscle activity and in increased muscle hardness. It is possible that low-grade tension that normally does not result in pain does so in the presence of central sensitization (182). Moreover, it is possible that the biochemical changes in the dorsal horn may alter the properties of the sensory afferents, so that these release inflammatory mediators, i.e. substance P and CGRP, from the receptive endings in the myofascial tissues (64), thereby creating a vicious cycle. For the sake of simplicity this is not indicated in Fig. 8b. By these mechanisms the central sensitization may be maintained even after the initial eliciting factors have been normalized, and the individual will experience daily headaches.
The proposed model explains the sensitization at the level of the spinal dorsal horn/trigeminal nucleus, the slightly increased supraspinal hypersensitivity, the slightly increased muscle activity, the increased muscle hardness, the chronic pain and the absence of objective signs of peripheral pathology in patients with chronic tension-type headache with increased pericranial tenderness. The model is also compatible with the increased pericranial tenderness but normal central pain processing in patients with episodic tension-type headache. The model does not account for the mechanisms that initiate the central sensitization or for the pain in the minority (41) of chronic tension-type headache patients without abnormal pericranial tenderness (40). The proposed model is based mainly on experimental animal studies and observational clinical studies. Headache is however, a subjective feeling that cannot be directly studied in animals, and in observational studies it is difficult to clarify if an abnormal finding is a consequence or a cause of the disorder. Experimental human studies are therefore needed in order to bridge the gap between basic animal and clinical research and in order to study the cause–effect relationship between myofascial nociception and central sensitization. Thus, the proposed pathophysiological model of chronic tension-type headache needs to be tested and probably modified, but it may be helpful in providing appropriate directions for future research. To summarize, the model states that the main problem in chronic tension-type headache is central sensitization due to prolonged nociceptive inputs from pericranial myofascial tissues, and that this mechanism is of major importance for the conversion of episodic into chronic tension-type headache.
Future perspectives
An important implication of the proposed model is that the future search for a satisfactory treatment for chronic tension-type headache should have two major aims: (i) to identify the source of peripheral nociception in order to prevent the development of central sensitization in patients with episodic tension-type headache, and (ii) to reduce established central sensitization in patients with chronic tension-type headache (Fig. 9). The possibilities for studying peripheral nociception have been greatly improved with the recent development of experimental human models of localized myofascial pain that allow infusion of chemical mediators into muscles for prolonged periods of time (183, 184). Similarly, experimental human models of tension-type headache could be extremely valuable, but so far only a few such models have been developed (for a review see Bendtsen & Jensen (185)). Recently, a genetic predisposition to chronic tension-type headache was reported (186). Future genetic studies may help to identify the mechanisms that make some individuals susceptible to the development of central sensitization. Basic pain research has demonstrated that NMDA receptor antagonists are effective in preventing the development of central sensitization as well as in reducing established central sensitization (187). In line with this, the NMDA receptor antagonist ketamine inhibits central sensitization in experimental human studies (188) and reduces pain in patients with fibromyalgia (189). Furthermore, an effect of the nitric oxide synthase inhibitor NG-mono-methyl-

The proposed pathophysiological model of chronic tension-type headache delineates two major aims for future research: (i) to identify the source of peripheral nociception in order to prevent the development of central sensitization in patients with episodic tension-type headache, and (ii) to reduce established central sensitization in patients with chronic tension-type headache.
Footnotes
Acknowledgements
This thesis is based on work carried out at the Department of Neurology, Glostrup Hospital from 1993 to 1996 during my appointment as a research fellow at the University of Copenhagen. I express my profound gratitude to Dr Rigmor Jensen for continuous encouragement, innumerable fruitful discussions and a valuable friendship, and to Professor Jes Olesen for having provided me in every way with optimal working conditions and continuous support and inspiration. The present thesis would not have been possible without them. I am indebted to laboratory technician Hanne Andresen for her enthusiasm and skilful help during the studies. I also owe thanks to Dr Messoud Ashina for constructive discussions and a valuable friendship. Furthermore, I wish to thank my co-authors Inge Hindberg, Jesper Nørregaard, Niels Kristian Jensen, Steen Gammeltoft and Erling Mellerup for their contributions in the specific studies. My sincere gratitude is due to the headache patients and healthy volunteers who participated in the studies. Without their participation it would have been impossible to do this work. Last, but not least, I would like to thank my family for their patience throughout this study. The work was supported by grants from the Lundbeck Foundation (145/91), the Pharmaceutical Foundation of 1991, the Foundation of Jacob Madsen and Olga Madsen, the Foundation of A.P. Møller and Chastine Mc-Kinney Møller, the Oak Foundation, Danish Health Insurance Foundation and the Foundation of Dr Eilif Trier-Hansen and Ane Trier-Hansen.
