Abstract
Our understanding of the importance of lipid biology to neurodevelopment, neurodegeneration and behavioural neurobiology (all factors proposed as playing a pivotal role in schizophrenia) has increased rapidly in recent years [1]. Early reports of lipid alterations in the cerebrospinal fluid [2] or peripheral cell membranes of patients with schizophrenia [3–5], together with clinical reports suggesting a prostaglandin–deficit syndrome (metabolites of membrane lipids) [6], led to the formulation of the membrane hypothesis of schizophrenia [7–10]. In addition, the observation that inherited neuronal lipid storage diseases, such as adult-onset Tay-Sachs disease (a GM2 gangliosidosis), present with psychotic symptoms in 30% to 50% of the cases, indicates the dramatic consequences of a disturbed lipid metabolism [11]. This article attempts to summarize the current English literature investigating lipid biology in schizophrenia. Related areas of interest such as oxidative damage of the cell membrane [12], the relevance of bioactive lipids in movement disorders [13] or the effects of oestrogens on lipid metabolism [14] have been reviewed elsewhere.
Methods
Medline databases were searched for English language publications dating from 1966 to February 2001 for the term schizophrenia combined with each of the following medical subject headings: essential fatty acids (EFA); polyunsaturated fatty acids (PUFA); arachidonic acid (AA, 20 : 5, n-6); docosahexaenoic acid (DHA, 22 : 5, n-3); eicosapentaenoic acid (EPA, 20 : 5, n-3); linoleic acid (LA, 18 : 2, n-6); alpha-linolenic acid (α-LA, 18 : 3, n-3); eicosanoids; prostaglandins; platelet-activating factor (PAF); arthritis (rheumatoid arthritis, RA); phospholipids; phosphatidylethanolamine (PtdEtn); phosphatidylcholine (PtdCh, also called ‘lecithin’); phosphatidylserine (PtdSer); phospholipases (PLA); diet; fish; and Phosphorus-31 Magnetic Resonance Spectroscopy (31P MRS). Cross-checking of references led to the identification of additional relevant references.
Background
Lipids make up a very high proportion of the brain (50–60% of human dry brain weight). A major proportion of these lipids are essential fatty acids (EFA) mainly bound to glycerophospholipids (GPL). The term ‘essential’, in this context, describes substances that cannot be synthesized de novo in the mammalian body. Due to their unique chemical structure comprising a polar head group (e.g. choline) and two non-polar arms (consisting mostly of one saturated and one polyunsaturated fatty acid), GPL spontaneously form bi-layers and are the basic molecules of all types of cell membranes. Different types of GPL exist. In order of decreasing concentration in the adult human brain they are phosphatidylethanolamine (PtdEtn), phosphatidylcholine (PtdCh, also called ‘lecithin’), phosphatidylserine (PtdSer) and phosphatidylinositol (PI). PtdEtn and PtdSer are mainly located at the inner, cytosolic layer, whereas PtdCh and sphingomyeline (SM) are mainly part of the outer layer of the cell membrane.
Each type of GPL in a given tissue has a characteristic EFA composition. For example, white matter PtdEtn contains 3% DHA, whereas grey matter PtdEtn contains 24% DHA [15]. There are two series of fatty acids that are classified as essential, the n-3 and n-6 series (see Fig. 1). The terms n-6 and n-3 are synonymous with omega-6 and omega-3, and indicate the location of the last double bond, counted from the carbon tail. Linoleic acid (LA, 18 : 2, n-6) and alphalinolenic acid (α-LA, 18 : 3, n-3) are the parent compounds of these two EFA series, both with 18 carbon atoms. In this article we will apply the term ‘essential fatty acids’ when we refer to n-3 and n-6 fatty acids in general, although sometimes the term EFA is applied exclusively to LA and α-LA, with their metabolites being called ‘derived EFA’. Both series compete for the same enzymes (desaturases, elongases, phospholipases) and cannot be interconverted (i.e. n-3 EFA cannot be transformed into n-6 EFA and vice versa). ‘Derived EFA’ can be synthesized from their essential parent C-18 compounds or obtained directly from diet (e.g. fish [16], and breast milk [17]).
Essential fatty acid pathways.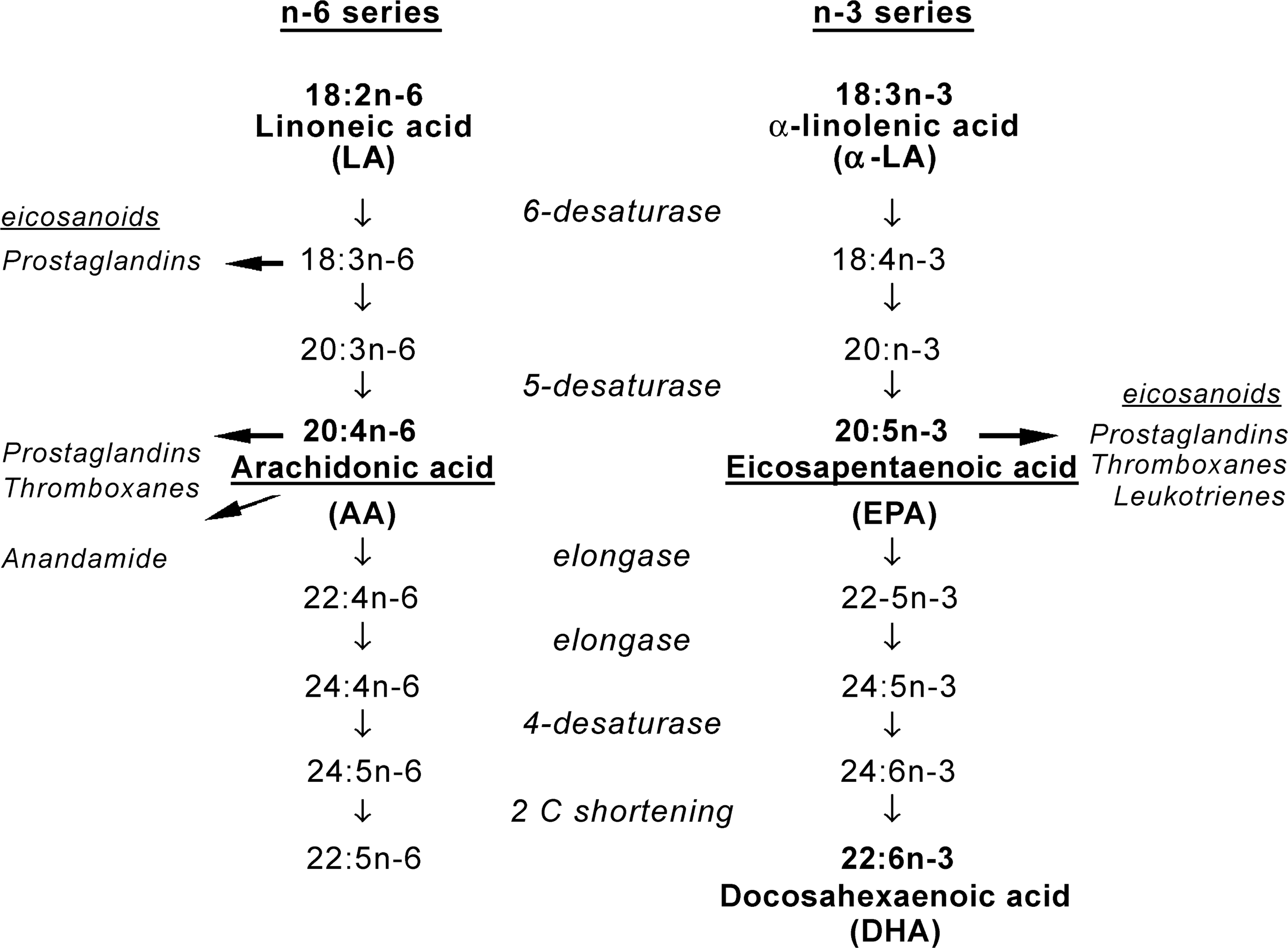
The structure and fluidity of cell membranes are determined by their EFA and cholesterol composition, which in turn modulates ion channels, receptor activity and neurotransmitter release (see Fig. 2) [18]. Excitable membranes such as synapses have a particularly high concentration of EFA. It is now becoming apparent that ‘derived EFA’ such as arachidonic acid (AA, 20 : 5, n-6), docosahexaenoic acid (DHA, 22 : 5, n-3) or eicosapentaenoic acid (EPA, 20 : 5, n-3) and their products (eicosanoids) such as prostaglandins, thromboxanes, prostacyclins and leukotrienes, termed as ‘bioactive lipids’, regulate processes such as neuronal migration, and determine synaptic plasticity [19], factors proposed to be disrupted in schizophrenia [20, 21]. In addition, AA linked with ethanolamine as anandamide, or esterified with glycerol, is an endogenous ligand for the brain cannabinoid receptor [22], a receptor system proposed to be of relevance for the pathogenesis of neurodegenerative disease [23].
The cell membrane GPL as ‘depot’ for lipid second messengers.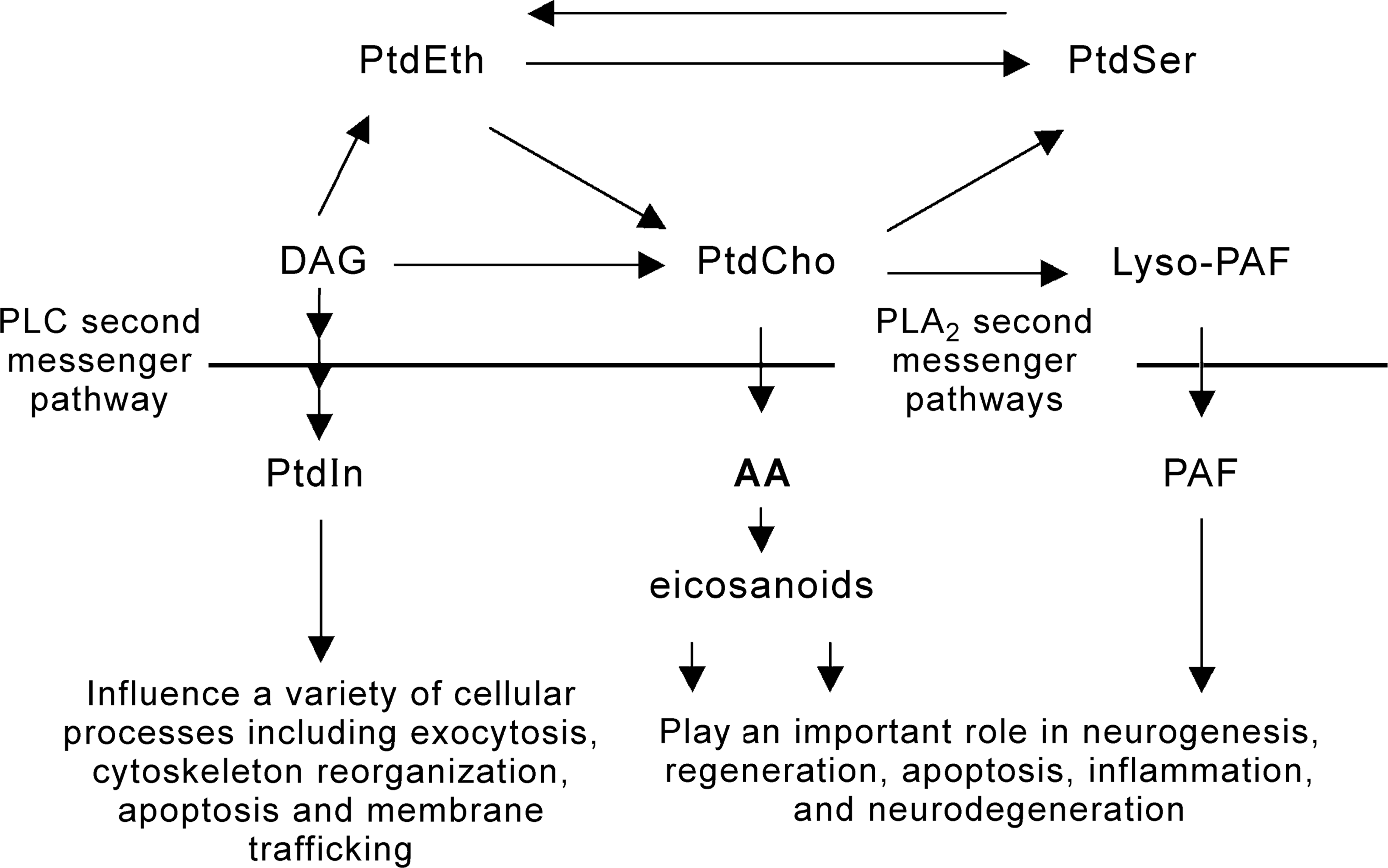
Bioactive lipids in schizophrenia
As EFA have such an important role in brain development and nerve functioning, the question emerges whether nutritional deficits in pregnancy and early childhood might contribute to an increased risk of schizophrenia, in line with the neurodevelopmental concept of the disorder [24]. The importance of EFA for normal brain development during pregnancy and early infancy is well documented [25]. Despite some evidence for a protective role for breastfeeding [26, 27], there have also been negative findings [28, 29]. However, the results of the case-control studies [26, 27] may indicate a protective effect of breastfeeding in ‘high-risk babies’ with other predisposing factors, such as a family history of schizophrenia, very premature birth, birth complications, viral infections, or extreme forms of malnutrition, whereas population-based studies [28, 29] do not provide evidence of a protective effect of breastfeeding in the general population. Further studies using carefully designed randomized case-control designs to investigate the protective role of breastfeeding or EFA supplementation in ‘high-risk baby populations’ (selective prevention [30]) seem warranted.
While it is well known that patients with schizophrenia live an unhealthy lifestyle (increased intake of saturated fats, lower intake of EFA, increased cigarette consumption and alcohol use, less exercise and obesity), it remains unclear if the reduced intake of EFA associated with this has a direct negative impact on the course of the illness [31]. A retrospective analysis [32] correlating the course and outcome figures from the International Pilot Study of Schizophrenia (IPSS) [33] with the national dietary characteristics of the eight participant countries suggested that the course of illness was better in those countries where the diet contained less saturated (animal) and more polyunsaturated fats (e.g. vegetables and seafood). These results need to be interpreted with caution, as the methodology was poorly described, the study was retrospective, and the results may be better explained as due to common underlying variables such as more urban living, migration or stress factors [34]. Nonetheless, future epidemiological studies should consider dietary factors as a potential cofactor especially in subgroups with an increased risk of schizophrenia such as a family history of mental disorders, or birth complications. In this context a recent report of lower incidence of depression in populations with high intake of marine or sea fish (rich in n-3 fatty acids) is noteworthy [35, 36].
Excess of bioactive lipids in rheumatoid arthritis – a neuroprotective factor?
The negative association between rheumatoid arthritis (RA) and schizophrenia give further evidence for the involvement of bioactive lipids in the pathophysiology of schizophrenia [37, 38]. Rheumatoid Arthiritis is associated with an overproduction of bioactive lipids, in particular platelet-activating factor (PAF), AA and its eicosanoids [6]. A recent meta-analysis of 16 published data sets [39] encompassing over 70 000 patients with schizophrenia and over 350 000 patients with other psychiatric conditions estimated that the rate of RA in schizophrenia is less than a third of the rate of RA in other psychiatric diagnoses. The relative risk of RA in schizophrenia versus the general population was estimated to be even lower at around 10%. These findings would be in keeping with the hypothesis that schizophrenia is associated with reduced EFA levels.
The niacin flush test, a marker for a prostaglandin deficit in schizophrenia?
The oral intake of niacin (nicotinic acid, vitamin B3) in normal volunteers induces a dose-dependant flush reaction of the face and upper body that is proposed to be mediated via prostaglandin D2, a metabolite of AA [40]. Early studies reported a reduced flush response in some patients with schizophrenia [38, 41] and its restoration after remission of psychosis [42]. Two studies trying to quantify the flush reaction using methodologies such as measuring malar temperature changes [43] or plethysmography [44, 45] to measure blood flow had mixed results. Hudson and colleagues [46] used thermo-coupled sensors to measure the skin temperature relative to the core body and ambient room temperature after oral intake of 200 mg niacin and demonstrated that 42.9% of the patients with schizophrenia had no change in temperature, whereas 94% of patients with bipolar disorder and 100% of normal controls did. In addition, the lack of flushing was associated with low levels of EFA in red cell membranes. The conversion from non-flusher to flusher was associated with an increase in EFA levels in red cell membranes and treatment response [47].
In contrast, in a topical variant of the test, where different concentrations of niacin patches (0.1, 0.01, 0.001 and 0.0001 M) are put on the skin, over 70% of patients with schizophrenia had either no or a markedly reduced flush reaction compared with normal controls [48], which was not explained by neuroleptic medication [49]. Shah and colleagues [49] replicated the latter study using a score system that integrated the oedema and could separate patients from normal controls at all but the lowest niacin concentration (0.0001 M). Our own group has confirmed these findings in a group of first-episode psychosis patients using a new semiquantitative, descriptive assessment scale that integrates erythema, oedema and time course to assess the overall flush reaction. Using this scale we were able to separate normal controls from first episode psychosis patients with a sensitivity of 81% and a specificity of 81.5% (unpublished data). Normal controls had a normal distribution, whereas first episode patients showed a bimodal distribution supporting a previous report suggesting a bimodal distribution of AA levels in patients with schizophrenia [50]. As membrane fluidity correlates with the amount of AA in the skin and is proposed to be predictive of relapse in schizophrenia, the topical niacin flush test might have the potential to detect people at risk for transition or relapse to psychosis [51] or to detect a subgroup of patients with lower AA levels, proposed to be a subgroup of patients with negative symptoms and poor prognosis [52]. There is also preliminary evidence that about 40–50% of first degree relatives of schizophrenia patients show an impaired niacin flush response [53].
Reduced membrane EFA in schizophrenia
A recent review of 15 published studies (Fenton et al. [54]) found a depletion of LA, AA and DHA in red cell membranes, thrombocytes, and fibroblasts of patients with schizophrenia, which is thought to be independent of drug treatment [55]. Primate studies have demonstrated that peripheral EFA composition of blood cell membranes correlates with EFA composition of nerve cell membranes [56]. Unsurprisingly therefore reductions in EFA have also been found in post-mortem brains of patients with schizophrenia, relative to normal control brains [57, 58]. Yao and colleagues [55] suggested a defective uptake of EFA into membrane GPL as a possible aetiopathological mechanism in schizophrenia, whereas Peet and colleagues [59], who also found an increase in EFA peroxidation products, suggested an increased breakdown of membrane GPL. Glen and colleagues [47, 50] demonstrated that high levels of saturated fatty acids and low levels of red cell membrane EFA (AA, 20 : 4, n-6 and DHA, 22 : 6, n-3) were found in a subgroup of patients with predominantly negative symptoms of apathy and withdrawal. Patients with positive symptoms showed a normal EFA distribution, suggesting that EFA deficiency (in particular AA) might be more relevant to negative symptom schizophrenia, and may be relevant to a poorer prognosis or be relevant for a poorer treatment response to antipsychotics.
Phospholipase A2 (PLA2) in schizophrenia and other neurological conditions
Phospholipases are key enzymes in determining the composition of the cell membrane, many regulatory processes and second messenger pathways. The phospholipases A2 (PLA2) consist of a broad range of enzymes (the PLA2 superfamily) defined by an ability to catalyze the hydrolysis of the middle (sn-2) ester bond of substrate GPL in order to release bioactive lipids, usually a free fatty acid (e.g. AA) and a lysophospholipid (e.g. lyso-platelet-activating factor (lyso-PAF)) [60]. Historically PLA2 was classified as either calcium-dependant or calcium-independent. Since the cloning of a cytosolic 85 kDa group IV PLA2 (often referred to as cPLA2) the categorization is genetically determined (currently XI subgroups) [60]. Most of the studies measuring PLA2 activity in peripheral tissue of patients with schizophrenia were carried out before acquisition of the current knowledge of the different types of PLA2. Gattaz and colleagues [61–63] were the first to demonstrate an over-active PLA2 in plasma, serum and platelets of patients with schizophrenia, using a fluorometric methodology that primarily measures a calcium-independent PLA2 (most likely the cytosolic 85 kDa group IVA PLA2). Ross et al. [64] demonstrated that contradictory findings in previous reports [62–69] could be explained by different PLA2 test kits used. In accordance with a reduction of EFA in peripheral and central cell membrane tissue in schizophrenia, an increased calcium-independent PLA2 activity has also been demonstrated in post-mortem studies of the temporal lobes of patients with schizophrenia [70, 71]. In contrast, calcium-dependent PLA2 activity was decreased by 27–29% in the temporal cortex and prefrontal cortex and by 44% in the putamen, relative to controls. Similar alterations of changed PLA2 activity have been found in other psychiatric and neurologic conditions [65], although it is known that PLA2 is inhibited by neuroleptic medication [68]. Allelic association studies in families with a history of schizophrenia suggest that the cPLA2 gene locus (Ban I) might be a candidate gene predisposing an individual to schizophrenia [72–74], yet two studies not restricted to patients with a family history of schizophrenia have failed to replicate such findings [75, 76]. These studies did not stratify patients according to niacin flush response [69, 77].
Altered glycerolphospholipid (GPL) membrane composition in schizophrenia
Whereas the evidence for reduced EFA and increased activity of the calcium-independent PLA2 (probably the cytosolic 85 kDa group IVA PLA2) seems to be quite robust, attempts to measure GPL composition of the cell membrane were less conclusive [3–5]. Initial reports found a marked increase in PtdSer (up to 50%) and a decrease in PtdEtn and Phosphatidylcholine (PtdCho) [78, 79] leading to the proposal that the ratio of Phosphatidylethanolamine (PtdEtn) to Phosphatidylserine (PtdSer) might even be a biochemical marker for schizophrenia. The most consistent finding throughout most studies [3–5] was a decrease in PtdEtn (9–47%), although two groups [80, 81] failed to replicate these findings. Significantly lower amounts of PtdEtn and PtdCho were also found in post-mortem brain tissue from patients with schizophrenia [58], even after accounting for potential confounds. Serial phospholipid analysis of peripheral cell membranes in first episode drug-naïve patients and nonaffected first degree relatives in combination with measurement of enzyme activities (e.g. phospholipases) and EFA analysis would be required to elucidate the exact mechanism behind these membrane abnormalities.
In conclusion, the available evidence in peripheral tissue and postmortem tissue of patients with schizophrenia suggests that EFA, especially AA and its precursors (n-6), and DHA (n-3) are reduced, and the enzyme for the cleavage of these EFA is more active. Whether these alterations are related to a lack of EFA incorporation [55], an increased breakdown of GPL by an over-active PLA2 activity or from an underlying disturbance (e.g. an intracellular dysfunctional EFA transport [82]) remains to be clarified. Tests, such as the topical niacin flush test [69], sensory gating [77], breath analysis [83] or cognitive tests might clarify subgroups out of the schizophrenia spectrum where bioactive lipids are of relevance.
In vivo membrane chemistry assessed by 31Phosphorus Spectroscopy (31P-MRS)
Advances in brain imaging have allowed measurement of GPL metabolites in vivo using 31P-MRS. For the purposes of this review, there are two important resonances from this spectrum – the phosphomonoester (PME) and phosphodiester (PDE) peaks [83]. The phosphomonoester peak (PME) includes phosphatidylethanolamine (PtdEth) and phosphatidylcholine (PtdCh) peaks (associated with cell membrane precursors) that can be separated using proton-decoupling or high magnetic field strengths. These two peaks account for approximately 45% of the PME peak [84]. The PDE peak can be separated into glycerophosphatidylcholine (GPCh), glycerophosphatidyl-ethanolamine (GPEth) and the mobile phospholipids (MP). The PDE subfractions GPCh and GPEth are associated with the breakdown products of the cell membrane, but represent only about 15% of the total peak area. The remaining signal most likely arises from underlying intracellular mobile phospholipids (probably lysosomal and peroxisomal). The role of 31P-MRS in the investigation of high-energy metabolites [83, 85, 86] and other methodological aspects [87] are reviewed elsewhere.
31P-MRS in different stages of schizophrenia
In a landmark study, Pettegrew et al. [88] demonstrated decreased PME and increased PDE peaks in the prefrontal cortex of drug-naïve first-episode patients, indicative of a higher cell membrane turnover. This pattern may also be present before the onset of psychosis [89]. Reduced PME and increased PDE peaks have also been identified in the temporal lobes of drug-naïve first-episode patients [90]. Thus, the previously described alterations of the cell membrane in peripheral cells and post-mortem brain tissue have also been demonstrated in vivo, in the early stages of schizophrenia. Indeed, first-episode drug-naïve psychosis patients showed a strong correlation between an elevated PDE peak in prefrontal cortex and low red cell membrane AA levels, suggesting that alterations in peripheral tissue were closely related to in vivo brain alterations of cell membrane metabolism [91].
Stanley et al. [92] examined whether this pattern was present at different illness stages by examining three groups: drug-naïve; newly diagnosed and medicated patients; and chronic medicated patients with schizophrenia. PME were lower in all three groups when compared to age and sex-matched control groups. However, PDE were only increased in drug naïve and newly diagnosed medicated patients, implying a higher membrane phospholipid turnover at the onset of illness. Recently, the same group explored these findings using protondecoupling to distinguish subfractions of the phospholipid peaks [94]. Using this technique in chronic medicated patients with schizophrenia, Potwarka et al. [94] presented results that were consistent with previous observations of decreased PME, but did not support the hypothesis of increased membrane breakdown. Unexpectedly the glycerol-3-phosphoethanolamine (GPEth) peak, and glycerol-3-phosphocholine (GPCh) peak were both normal, but the mobile phospholipid (MP) peak was higher suggesting an altered intracellular lipid metabolism. In another study Blüml et al. [95] did not replicate these findings in an examination of the parietal cortex in 11 chronic and 2 neurolepticnaïve patients. The single breakdown peaks (GPEth, GPCh) and G-phosphocreatine were increased, but the total PDE peak was not different. Further, while the total PME peak area was elevated, the individual PtdEtn and PtdCh peak areas did not differ. While differences in subject age between studies may explain the observed differences [94], a more likely explanation is that these abnormalities may be apparent in particular brain regions, such as prefrontal cortex and temporal lobes. The latter would be consistent with the available evidence implicating these areas in schizophrenia [96, 97]. Preclinical data also suggest that EFA distribution in the brain is specific for different brain regions [98].
In conclusion, the majority of in vivo 31P-MRS studies have confirmed alterations in GPL metabolism, and most studies have shown a reduced PME peak independent of treatment and stage of illness. Furthermore, four studies have found an increased PDE peak in early psychosis, while in chronic schizophrenia the pattern has been inconsistent. The changes observed are reported most often in frontal and temporal lobes suggesting that there is increased membrane turnover in these regions, particularly in drug-naïve first-episode patients. While the evidence suggests that neuroleptic treatment is associated with a reduction of the PDE peak, this reduction was associated with improvements in symptoms [93]. Future serial 31P-MRS investigations using proton-decoupling, high-field imaging (3 Tesla or higher), or novel analysis techniques need to address the question of which phospholipids are most relevant for the phosphoester alterations observed in previous studies [99]. Longitudinal phosphorus MRS investigations of drug-naïve first-episode psychosis patients, using a volume of interest that maximizes the amount of grey matter (rich in EFA), with concurrent examination of EFA composition and PLA2 activity in peripheral cells, should clarify the implications of these findings for the understanding and treatment of schizophrenia.
EFA-supplementation studies in animals and humans
The importance of bioactive lipids and EFA supplementation has been studied extensively in animals (reviewed by Wainwright [116, 117]). Animals (mice, rats, cats, primates) fed an EFA deficient or depleted diet showed significantly aberrant sleeping patterns, adipsia, polydipsia, anorexia, hyperalgesia, reduced self-grooming activity, a decrease in exploration and social interaction, as well as a disruption of established food-motivated lever-pressing responses. EFA supplementation had a positive impact on 18 out of 24 altered behavioural variables in EFA deficient animals (reviewed by Wainwright [117]). Improvements in behavioural tasks after EFA supplementation were correlated with alterations in brain lipid composition and were specific for different brain areas such as the temporal lobe [114, 118]. The n-3/n-6 ratio in the diet was of particular importance in determining the availability of EFA for the incorporation of EFA into animal brains with a preference for n-3 EFA, especially EPA and DHA [117, 119]. There is now growing evidence that EPA competes with AA by inhibiting AA metabolism via inhibition of 5-lipoxygenase activity at low concentrations and PLA2 activity at high concentrations [120]. The structural similarity between AA and EPA could explain the enhanced effectiveness of EPA if it behaves as an analogue of AA [121]. In addition, preclinical studies demonstrating an inhibitory effect of EPA on protein kinase C [122, 123] suggests it may be neuroprotective [124].
31P-MRS in schizophrenia (focus on phospholipid metabolites)
In conclusion, preclinical data support the role of bioactive lipids, especially EFA, in brain development and behavioural neurobiology, which can be influenced by dietary intake of such lipids. These findings raise the question about whether different types of EFA supplementation can influence EFA deficiencies and GPL alterations described in schizophrenia and whether normalization in EFA levels has an impact on the pathogenesis or course of schizophrenia.
The first supplementation studies were in patients with tardive dyskinesia (TD) (predominantly patients with chronic schizophrenia) and used formulae with a majority of omega-6 EFA (e.g. one capsule Efamol Evening Primrose Oil® contains 360 mg LA, and 45 mg γ-linolenic acid). Two initial small studies of Efamol Evening Primrose Oil® supplementation in chronic patients with tardive dyskinesia (TD) did not identify any beneficial effect on TD or symptomatology [125, 126]. However, in a double-blind randomized, crossover study [127] there was marginal improvement in TD but an unexpected moderate improvement in psychopathology (20–30%) and overall cognitive functioning. During the entire trial period the effects of Efamol Evening Primrose Oil® could not be explained by practice effects, since there was clear deterioration when patients switched from Efamol Evening Primrose Oil® to placebo.
Research investigating the effect of omega-3 EFA has been more successful [128, 129]. The latter study, using 10 g MaxEPA® per day (enriched n-3 EFA formula; 171 mg EPA/g, 114 mg DHA/g), demonstrated a significant improvement in psychopathology and TD [129]. In addition, there was an inverse correlation between EPA and AA levels in red cell membranes suggesting that mainly EPA were responsible for its effect on the AA levels. Similar findings were also observed in another small open-label study using 1 g of an EPA-enriched oil (Kirunal®; EPA:DHA = 3 : 1) per day in 10 chronic, symptomatic patients with schizophrenia [130]. On the basis of these results Peet et al. [131] randomly allocated symptomatic patients with chronic schizophrenia to an EPA enriched oil (Kirunal) and a DHA enriched oil (Doconal) or placebo. Only the group with the EPA enriched oil improved in psychopathology, while patients taking the DHA-enriched oil or placebo oil showed no significant changes. The greatest difference was between the EPA and DHA groups [131]. Consequently, Peet and colleagues [131] undertook a randomized study to compare 2 g of EPA enriched oil/day with placebo supplementation in 63 unmedicated first and multi-episode patients with schizophrenia as an initial sole treatment. After the study period of 12 weeks, only 66% of patients in the EPA arm required neuroleptic medication (haloperidol), compared with 100% of those in the placebo arm. Four out of 15 patients in the EPA arm did not need antipsychotics at any time during the study period. Total days on haloperidol were significantly lower (46.3 days vs 71.7 days), and those in the EPA group showed a greater improvement in positive and negative symptoms, despite a lower cumulative antipsychotic dose. When the effect of different doses of EPA (1, 2, and 4 g EPA per day) was studied in 117 symptomatic patients with chronic schizophrenia, a significant impact on psychopathology was only found for 2 g EPA, and normalization of RBC membrane AA levels was only observed in the 2 g EPA group [132].
Recently, Fenton and colleagues [133] presented a randomized study supported by the Stanley Foundation. The study was placebocontrolled and included 87 patients with chronic schizophrenia and residual symptoms. These patients had a mean age of 40 [range 18–65], an average duration of illness of 20 years and were supplemented with 3 g purified EPA/placebo daily for a period of 16 weeks. The randomized trial showed no between-group differences for either psychopathological parameters, or for cognitive functioning in patients with chronic schizophrenia and residual symptoms. The selection of chronic patients with residual symptoms on a stable neuroleptic medication and the use of a higher dose of EPA in comparison to Peet's study of younger and more acutely psychotic patients may explain the difference in outcome between the two studies. Future placebocontrolled studies should address issues such as illness duration, acuity of symptomatology, background treatment (flexible or fixed dose), and dose of EPA.
There are several limitations evident in the treatment studies reported to date. Different combinations and doses of oils, some with higher quantities of parent EFA compounds and others with different n-3/n-6 ratios, make interpretation difficult. Some studies used a sample of chronic treatment-resistant patients with TD, while other studies used drug-naïve first episode patients. Neuroleptic treatment has also been heterogeneous. In addition, the effects of sex steroids on EFA metabolism, susceptibility of EFA to oxidation (from smoking, starvation, and stress), developmental factors (e.g. birth complications, breast-feeding), the dependency of EFA metabolism on age (e.g. loss of desaturase activity) and environmental factors (e.g. cannabis use) should also be considered. While diet might also have a confounding effect, this would be unlikely, as the amount of EFA supplementation used in clinical trials (usually 1–10 g EFA/day) is much higher than the dietary intake, even if fish were consumed on a daily basis (a fish meal of 100 g of Atlantic salmon contains 71 mg AA, 171 mg EPA and 378 mg DHA [134] which is around 10% of the amount of EPA used in clinical trials).
In conclusion, while Peet et al. [132] and Horrobin et al. [53] have presented some preliminary positive results in the acute and early phase of psychotic illness, Fenton and colleagues [133] did not replicate these findings in a chronic patient population. Independent research groups should attempt to investigate the potential use of EPA at different illness stages (e.g. its main effect may be in the onset phase of illness) and elucidate the postulated dose-related mechanism(s) of action, considering the low cost and high acceptance of such treatments and the minimal side-effects.
Conclusions and limitations
The main findings from studies investigating the role of bioactive lipids in patients with schizophrenia are consistent with the notion of a membrane pathology throughout the body. This is evidenced by a general reduction of cell membrane EFA in peripheral tissue (e.g. red blood cells, fibroblasts) and post-mortem brain tissue, the most significant being in AA, its precursors and DHA. Increased activity of the EFA metabolizing enzymes such as an over-active calcium-independent PLA2 (most likely the cytosolic, group IVA PLA2) suggests an increased membrane breakdown of EFA. While differences might be expected at different illness stages, studies are predominantly of patients with established schizophrenia, with no studies examining phospholipase activity in drug-naïve first-episode psychosis.
In vivo 31P-MRS studies of patients with established schizophrenia have generally revealed that PME are reduced, while the results for PDE are inconsistent. Such changes may be related to changes in intracellular phospholipid transport [82, 94]. In contrast, the few studies examining the early course of the illness provide a more consistent pattern, showing lower PME and higher PDE peaks, indicating that membrane lipid turnover is high. While the PDE peak could be influenced by antipsychotic treatment [92], the first-episode studies in neuroleptic-naïve patients indicate that effects of medication do not explain the changes.
Related aspects, such as the impaired niacin flush reaction and the reduced incidence of schizophrenia in RA implying alterations of bioactive lipids originating from AA, further support the notion of altered membrane lipid chemistry in schizophrenia. Tools, such as the topical niacin flush test, and blood and breath peroxidation product analyses [83] may be of relevance in the understanding of biochemical subgroups within the schizophrenia spectrum. Such an understanding might expose potential new treatment targets such as the development and use of selective enzyme inhibitors (selective PLA2 inhibitors) or pharmacogenetic targets relevant for lipids (e.g. expression of apolipoprotein D [135]) in the schizophrenia spectrum.
The available preliminary data from intervention studies provide some support for the notion that purified EPA is effective in treating positive and negative psychotic symptoms when used in the acute illness stage at doses of 1–2 g/day. One multicentre study in patients with established schizophrenia and residual symptoms using 3 gram EPA/day failed to demonstrate an effect. Most studies have used EPA as an adjunct to neuroleptic medication with only one study using EPA alone. Further studies examining the efficacy of EPA at different doses, and at different illness stages would seem warranted. As well as clinical outcome measures, future studies should incorporate additional outcome measures related to lipid metabolism, such as the measurement of enzyme activities (e.g. phospholipases), in vivo 31P-MRS (preferably 1 H-decoupled), EFA analysis, the topical niacin flush test and breath peroxidation product analysis.
Finally, the question of specificity of the findings and any proposed interventions needs to be addressed. For example, in a study of patients with bipolar disorder, Stoll et al. [136] demonstrated that n-3 EFA supplementation resulted in fewer relapses, an improvement in global clinical outcomes and depressive symptoms. It is likely that the changes in lipid biology identified in schizophrenia may be relevant to other psychiatric conditions and more generally to other neurodevelopmental and neurodegenerative disorders.
Footnotes
Acknowledgements
Gregor Berger was supported by a Fellowship from the Swiss National Science Foundation and M. and W. Lichtenstein Foundation (University of Basel), the University of Melbourne and the North-Western Health Care Network. Stephen Wood was supported by NH & MRC grants (IDs: 145627, 981112). This work is supported by an NHMRC grant (IDs: 209062) and a Stanley Foundation grant (Berger et al. 2001). We thank David Horrobin (Laxdale, UK), Ulrich Honegger (University of Bern, Switzerland), Malcolm Peet (University of Sheffield, UK), Peter Williamson (University of Western Ontario, Canada), Martin Lambert (EPPIC, Australia) and Warrick Brewer, University of Melbourne, for their comments on various drafts of this review article. We thank the Mental Health Library of the Royal Melbourne Hospital.
