Abstract
The measurement of in vivo cerebral biochemistry, particularly the concentration of tissue metabolites, has been enabled by magnetic resonance spectroscopy (MRS) [1], which has been used to investigate patients with schizophrenia over the last 8 years. This review paper provides a critical reflection on the literature that has arisen from these investigations. It begins with a historical overview of MRS. This is followed by an account of the formative influences, limitations and future directions of the three MRS techniques used in schizophrenia research as they have been applied to the three primary regions of interest that have been explored.
Historical overview of the principles of magnetic resonance spectroscopy
A classical description of nuclear magnetic resonance (NMR) is outlined below. More recent models, informed by constructs of quantum mechanics, have been proposed, but are beyond the scope of this paper to explain adequately. The existence of NMR was first shown by Rabi et al. in 1939 [2]. Subsequently in 1946, Bloch et al. [3] and Purcell et al. [4] demonstrated that the magnetic dipoles of molecules alter or resonate when placed within a magnetic field [5–7]. An atom can be conceptualised as having a central region of positively charged proton(s), with or without neutrons, surrounded by a ‘shell’ of negatively charged electrons [6]. A proton has a charge and a spin, which generate a magnetic field that allows it to align either with or against the direction of an externally applied magnetic field [8]. However, only nuclei with an odd number of protons or neutrons have a net spin that makes them suitable for NMR [5–8]. The Lamor frequency is the unique amount of energy, in a given magnetic field, which resonates a particular nucleus (for example 31P, 1H, 19F) [5–11]. The electromagnetic energy associated with this resonant frequency is associated with the alignment of a proportion of protons at 180° to the magnetic field [8]. When this particular resonant frequency (RF) magnetic field is removed, the protons in a particular nucleus return to their original axes [5–8]. This process is called relaxation [5–11]. The voltage signal generated from the relaxation can be translated, by Fourier transformation, from the voltage signal decay with time domain to the frequency domain resulting in a particular magnetic resonance spectrum [5,6]. The relaxation process varies with time because the protons do not all return simultaneously to their former spin axes [5–8]. T1 and T2 relaxation times are orthogonal measures of these specific processes that relate to specific parameters of particular tissues [5–8]. T1 refers to the ‘longitudinal’ or ‘spin lattice’ relaxation time while T2 indicates the ‘transverse’ or ‘spin spin’ relaxation time [5–8]. In addition, the resonant frequency of a given nucleus is affected by the magnetic fields generated by the surrounding atoms [5,6]. This effect is termed the ‘chemical shift’ and it varies according to the bond configuration of a given molecule [5,6,8]. Thus, the same element in different biochemical compounds resonates at slightly different frequencies, which results in distinct magnetic resonance spectrum peaks [5,6]. The position of a peak on the horizontal frequency axis indicates the particular biochemical present. The area under a given peak represents a quantitative measure of a given biochemical. To detect such minute changes in the ‘chemical shift’, an ‘extremely homogenous’ magnetic field is required [5]. The process of achieving this homogenous magnetic field is called ‘shimming’ [5,6].
Technique of magnetic resonance spectroscopy in schizophrenia studies
As reviewed by Keshavan and colleagues [5,6], MRS requires a horizontal-bore magnet (1.5–4.0 tesla), a radiofrequency transmitter and receiver coil focused on a particular nucleus of interest (for example, 31P, 1H, 19F), and a computer. Magnetic resonance spectroscopy is of relatively high sensitivity, compared with magnetic resonance imaging (MRI), while the peaks of interest contained within the spectra are of low sensitivity. A range of radio-frequencies needs to be generated to focus on a given nucleus of interest [5,6]. Importantly, a volume of interest (VOI) needs to be determined from which the signal is generated. Initially, surface VOIs were studied by placing the surface coil near the area of interest [5,6]. However, the obtained spectra were often ‘contaminated’ by signals from more superficial structures, such as skin and muscle tissue [12]. Subsequently, gradient-localisation techniques have been developed that use magnetic gradients in which the radiofrequency varies with distance to define VOIs. These techniques are particularly well suited to proton-MRS (1H-MRS), although there is an acquisition delay that can result in some of the signal being lost [5]. This is particularly problematic for phosphorous-MRS (31P-MRS). The spatial resolution of MRS is poor relative to other major imaging modalities [6]. Magnetic resonance imaging provides a more detailed spatial resolution of brain soft tissue structures, compared to computed tomography (CT), which has a spatial resolution of less than 0.5 mm that is approximately 10 times as large as plain X-ray film resolution. Positron emission tomography (PET) has a spatial resolution of approximately 5 mm and single photon emission computered tomography (SPECT) approximately 7 mm [5]. In contrast, MRS VOIs range from approximately 4 cm3 [13] to 40 cm3 [14,15].
In summary, the presence of a particular biochemical and its quantitative measure (e.g. concentration) is theoretically meaningful within a given defined VOI. But the conceptual links between the theoretical construct and its actual meaning are still tenuous. For example, the concentrations of various metabolites of interest are close to the ‘limit of detectability’ [12]. This ‘limit of detectability’ is dependent on the particular target nucleus of interest, sufficient mobility of the metabolite from which a particular spectrum is sought [16], the strength of the magnetic field, the size of the VOI, and the acquisition time [12]. An example of 1H-MRS in the hippocampus, showing the spectra obtained, is shown in Fig. 1.
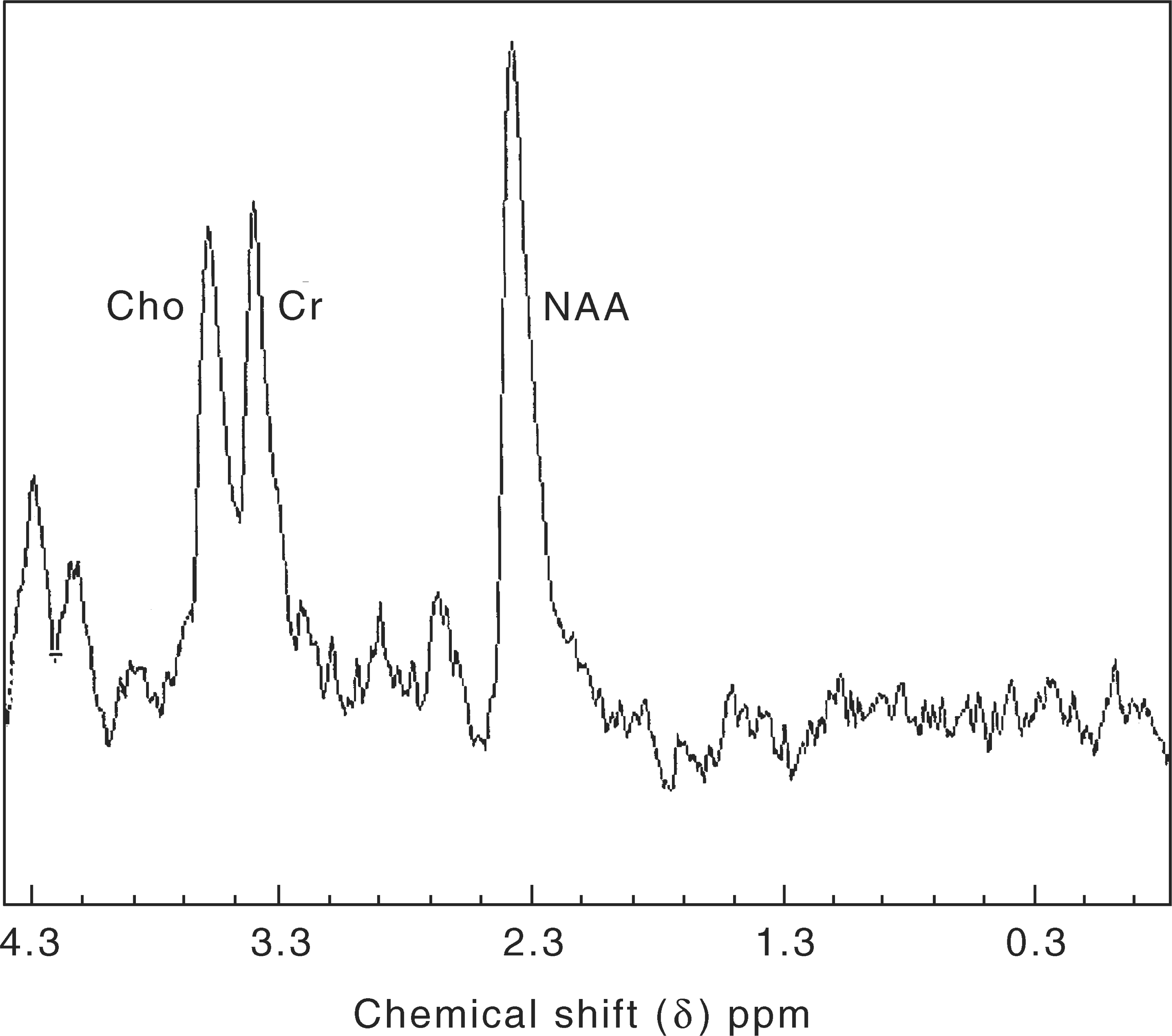
1H-magnetic resonance spectroscopy in a hippocampal VOI. NAA, N-acetylaspartate peak; Cr, creatine peak; Cho; choline peak.
31P-magnetic resonance spectroscopy
31P-magnetic resonance spectroscopy provides information about membrane phospholipid metabolism and energy metabolism [5–7,10,17–22]. A phospholipid bilayer forms cell membranes. Phosphomonoesters (PMEs) are the precursors and phosphodiesters (PDEs) are the metabolic breakdown products of these membrane phospholipids [5,6,10,17–22]. An in vivo investigation of PMEs and PDEs is allowed by 31P-MRS that indirectly reflects the rates of cell membrane synthesis and degradation. These rates may be different in healthy and disease states in vivo [5,6,10,17–22]. In addition, PDEs include the breakdown products of myelin and may be a marker of its degradation in cell membrane metabolism [18]. The beta moiety of adenosine triphosphate (βATP), phosphocreatine (PCr), and inorganic phosphate (Pi) are indirect indicators of the state of energy metabolism. Adenosine triphosphate is generated from carbohydrate metabolism, via the Kreb's cycle, and functions as an energy store [5,6,10,17–22]; PCr acts to ensure intracellular ATP levels are relatively constant. Again, the rates of in vivo myelin degradation and energy metabolism may differ in healthy and disease states.
The rates of cell membrane synthesis and degradation, myelin degradation, and the state of energy metabolism may affect, or be affected by, changes in neurotransmitter receptors that are membrane bound. These changes in neurotransmitter receptors may be involved in the various phases of the development of schizophrenia [12,23]. In addition, the process of ageing of the central nervous system may be altered in schizophrenia [12]. Feinberg's hypothesis of abnormal synaptic pruning in adolescence is based on this premise of an aberrant ageing process [12,24]. Findings by 31P-MRS of decreased PMEs and increased PDEs suggest decreased synthesis and increased breakdown of cell membranes respectively, and support such an anomaly of synaptic pruning. In turn, this abnormality of synaptic pruning has been used to support the neuro-developmental hypothesis of schizophrenia [25]. Hypofrontality and the purported metabolic asymmetry of the temporal lobes in patients with schizophrenia have been investigated by measuring βATP, PCr, and Pi levels [12]. Increased βATP and PCr and decreased Pi suggest decreased utilisation of βATP as a marker of hypofrontality [12]. Disparity in temporal lobe PCr and the PCr/βATP ratio suggest metabolic asymmetry [12]. However, the assumption that βATP may not be significantly altered in disease states relative to healthy states has yet to be proven.
Summary of 31P-magnetic resonance spectroscopy findings in patients with schizophrenia
The findings of 31P-MRS suggest that there is a relative increase in cell membrane degradation and a reduced level of energy metabolism in prefrontal cortical regions. In addition, the cell membrane degradation seen early in the process of schizophrenia is not as apparent in the chronic phase of the disorder. It is unclear whether this is related to factors associated with the disorder or to the effects of antipsychotic medication.
Decreased PME and increased PDE have been reported in a number of studies of newly diagnosed, drug-naive and chronic, medicated patients with schizophrenia [26–33]. Pettegrew et al.'s [26] work is exemplary because they used a categorical and dimensional diagnostic approach to defining schizophrenia, their patient sample was drug-naive with a duration of illness of only approximately 19 months, equivalent numbers of male and female subjects were studied, and raters were blind to the diagnosis in the analysis of the spectroscopic data. Limitations included their small sample size of 11 subjects, their relatively large VOI of 15–20 cm3 and their cross-sectional study design. The general problem of relatively large VOIs that are needed to perform MRS has been noted above. Further limitations of other studies include the use of a categorical approach alone to diagnose schizophrenia, and variation in the duration of illness from 3 to 19 years [28,31,32]. In contrast, Volz et al. [14,15] reported opposing findings of decreased PDE and increased PME/PDE. Both Volz et al. studies have similar advantages of using a categorical and dimensional diagnostic approach to defining schizophrenia, a chronic patient sample that is medicated long-term and with a duration of illness of 8–10 years, equivalent numbers of male and female subjects, and raters who were blind to the diagnosis. A particular advantage is the much larger sample size of 60 subjects, while a particular limitation is their much larger VOI of 39 cm3. They argued that these differing findings were, in part, due to the technical aspects of the VOI selection.
Keshavan et al. [27] have also noted that ‘increased PME/decreased PDE’ spectra were found in one prepsychotic subject who had been a control in the initial study and who later became psychotic [26]. This paper raised the issue that these findings may be a marker of individuals at high-risk for psychosis and also supporting evidence for the neuro-developmental perspective of schizophrenia. Stanley et al. [28] attempted to address these questions by studying the dorsolateral prefrontal cortex (DLPFC) in 11 drug-naive, eight newly diagnosed, medicated, and 10 medicated, chronic patients. They found that PME was decreased in the three groups, PDE was increased in the drug-naive group and Pi was decreased in the newly diagnosed medicated group. A similar pattern of methodological strengths and limitations to Pettegrew et al.'s [26] earlier study was evident. The findings suggested that decreased cell membrane synthesis occurred at all stages of schizophrenia, while cell membrane breakdown was a predominantly early stage process. In contrast, Potwarka et al. [12,34] studied a chronic, medicated group and reported an increase in a phospholipid membrane component of the PDE peak, which would be consistent with abnormal membrane structure rather than increased cell membrane breakdown. Also, Volz et al.'s [35] recent longitudinal study of eight drug-free patients with chronic schizophrenia, pre- and post-antipsychotic medication, demonstrated increased PDE with the use of antipsychotic medication. It remains to be clarified whether increased PDE signifies an abnormal membrane structural process and/or an abnormal membrane breakdown process, while the differentiation of phase of illness and antipsychotic medication effect also needs to be investigated.
While a number of neuro-psychological and functional imaging studies have suggested that patients with schizophrenia have deficits involving the pre-frontal cortex [36–38], the results from structural imaging studies have been equivocal; MRS of the prefrontal cortex, therefore, provides another technique to examine the prefrontal cortex [39–43]. Surprisingly, only two published studies [44,45] thus far have examined the relationship of 31P-MRS measures in the prefrontal cortex to neuro-psychological measures of executive or memory function, especially working memory.
While most of the studies using 31P-MRS have investigated the prefrontal cortex, a few studies have assessed the temporal lobes [46–48] and the basal ganglia [39]. These findings have not been replicated and no consistent hypothesis of temporal lobe biochemical function and/or asymmetry has yet emerged. The significance of these findings is therefore unclear at present, although they need to be considered in the context of recent evidence for structural changes and 1H-MRS studies in medial temporal lobe structures. However, it should be noted that studies using 31P-MRS in this region have proved technically more difficult.
Basal ganglia region findings suggest a similar pattern to that of the frontal regions, although they have not been replicated. Thus, while there are too few 31P-MRS studies of the basal ganglia in schizophrenia to draw any conclusions, such studies may help to elucidate the role of the basal ganglia in schizophrenia. Specifically, they could examine the hypothesis that schizophrenia is due to a problem of frontal–basal ganglia–thalamic circuits [49,50] and investigate the suggestion that deficits of executive function may be due to a functional de-afferentation of these circuits [51]. A summary of the findings of 31P-MRS studies in patients with schizophrenia is shown in Table 1.
A summary of positive (+) and negative (−) 31P-magnetic resonance spectroscopy (MRS) findings from empirical studies investigating patients with schizophrenia
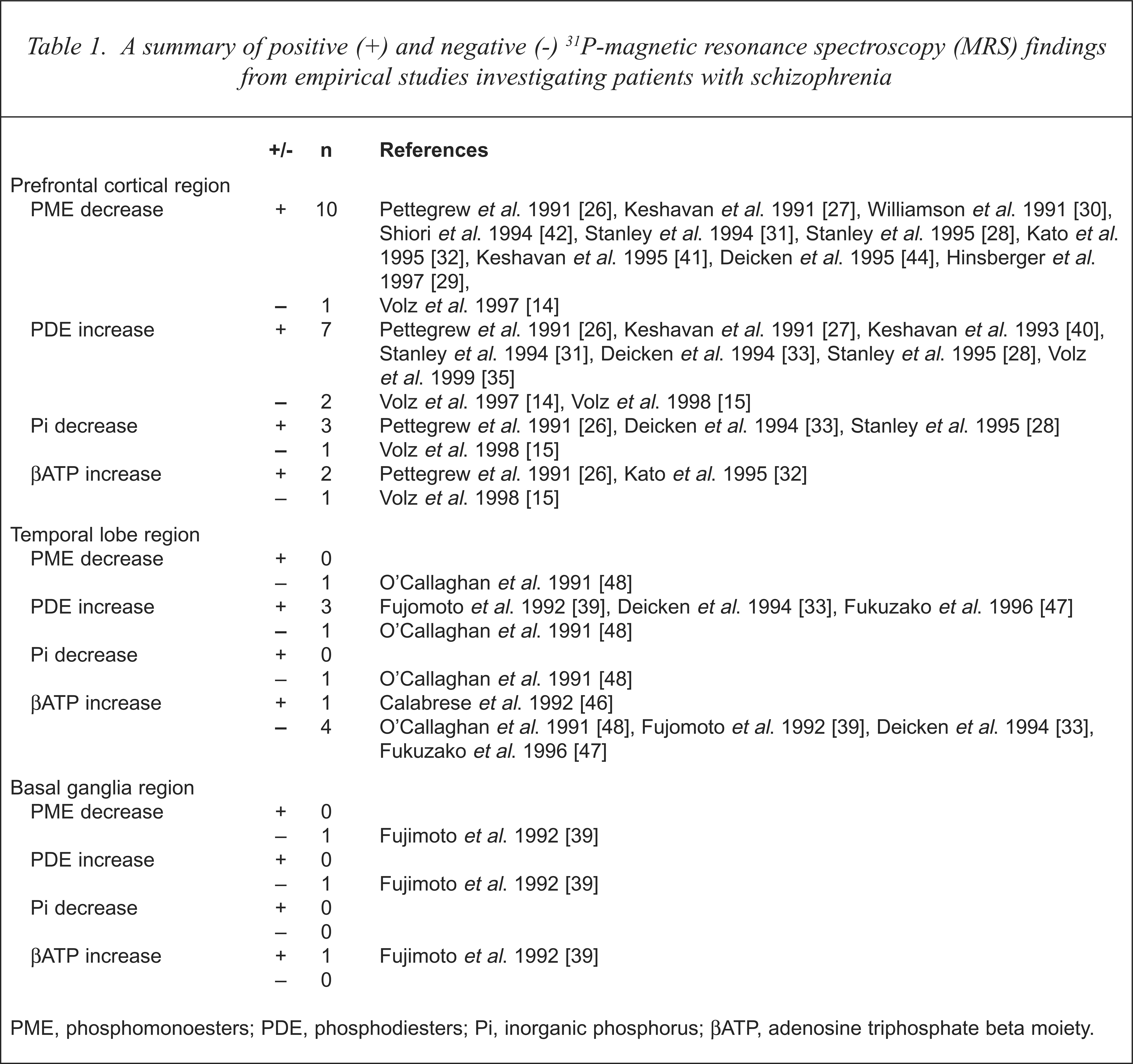
PME, phosphomonoesters; PDE, phosphodiesters; Pi, inorganic phosphorus; βATP, adenosine triphosphate beta moiety.
1H-magnetic resonance spectroscopy
1H-magnetic resonance spectroscopy provides indirect information about metabolites involved in neuronal and glial energy metabolism and phospholipid metabolism [5,7,11,18]. N-acetylaspartate (NAA) can be considered to be primarily an intraneuronal marker, although glial cells also contain NAA in detectable concentrations [5,7,52]. It has been used as a marker of ‘neuronal mass and integrity’ [5]. Choline includes metabolites involved in the composition of myelin such as phosphorylcholine and glycerophosphorylcholine and phospholipid metabolism such as phosphocholine and phosphoethanolamine [18]. It can be viewed as an indirect marker of myelination and cell membrane metabolism [7,18]. Creatine, including phosphocreatine, is a general marker of energy metabolism [7], Glutamine is localised mainly in glial cells while glutamate is an excitatory neurotransmitter found primarily within glutaminergic neurones [20,53]. There is a dynamic relationship between glutamine and glutamate such that appropriate levels of glutamate are maintained [20,53]. Gamma-aminobutyric acid (GABA) is an inhibitory neurotransmitter which can be perceived as acting as a counterbalance for glutamate-induced neuronal excitation [54].
Decreased NAA and/or decreased NAA/Cr ratio indicates neuronal loss and/or dysfunction while increased choline and/or increased choline/Cr ratio indicates de-afferentation [12,18]. Alterations in glutamine are interpreted as an accompanying change to neuronal loss and/or dysfunction and de-afferentation [12,18].
Summary of 1H-magnetic resonance spectroscopy findings in patients with schizophrenia
A presumed decreased neuronal cell mass in patients with schizophrenia has been reported in 1H-MRS studies of the prefrontal cortex, although it is not yet a robust finding. Components of the PFC, such as the DLPFC, have begun to be investigated, although other significant VOIs, such as the orbitofrontal cortex, are yet to be studied using MRS. The relationship between PFC MRS findings and neuro-psychological and symptom correlates also remains unclear. The extent of MRS-determined biochemical changes in different brain regions and their progression at different stages of the illness require further study using longitudinal study designs [55]. Decreased NAA [56,57] and a decreased NAA/Cr ratio [58,59] have been noted in chronic, medicated and newly diagnosed, medication-naive adult patients [60], medicated adolescents with ‘schizophrenia’ [61] and in children with ‘symptoms of early onset schizophrenia [and]/or schizotypal personality disorder’, termed ‘schizophrenia spectrum disorders’ [59]. These findings have been used to support the neuro-developmental hypothesis of schizophrenia [24,25]. Increased choline and decreased Cho/Cr have been reported as associated findings [56,62] and further evidence for this hypothesis. Raters blind to diagnosis [56,60,61] and equivalent male and female subjects [59] have been methodological strengths of some of these studies, while general methodological limitations include categorical diagnosis alone, small sample size (for example 10–14 subjects in Bertolino et al.[60,61]), poor control of the sample illness duration [56,60,61] and relatively large VOIs studied (for example, Bertolino et al. [60]). In contrast, Brooks et al. [59] studied a relatively small VOI of 8 cm3.
Glutamine has been noted to be increased in chronic, medicated [63,64] and newly diagnosed, unmedicated patients [64]. These studies support an alteration in the glutaminergic function in the left prefrontal cortex, which could indicate glutaminergic under- or over-activity with a commensurate altered availability of the neurotransmitter glutamate. These findings were also used to support the neurodevelopmental hypothesis of schizophrenia [24,25]. Two similar associations of decreased glutamine levels with antipsychotic medication have also been reported in eight first-episode, drug-naive [63] and 34 drug-free, chronic [54] patients. These two findings from independent groups using a prospective study design form an association between decreased glutamine levels and antipsychotic medication. Such altered glutaminergic activity may have important symptomatic correlates, may be related to medication effects or both. This requires further investigation with a first-episode, drug-naive sample, using a longitudinal study design. Stanley et al.'s study [63] is noteworthy because of the methodological strengths of a categorical and dimensional diagnosis of schizophrenia, defined duration of illness, blind raters, and a relatively small VOI (8 cm 3).
A similar presumed decrease in neuronal elements within the hippocampus and medial temporal structures is a replicated finding. These studies support the growing evidence from structural neuroimaging studies in schizophrenia that have identified smaller size of the hippocampi bilaterally, most particularly on the left [65,66]. Advocates of the neurodevelopment hypothesis have suggested that the site of the lesion in schizophrenia is the hippocampus and that other deficits, such as executive function impairments and hypofrontality, can be understood as a consequence of such a lesion [37,38]. However, no studies to date have used 1H-MRS to investigate such a relationship. Decreased NAA has been reported separately in the right [67] and left [13,68] hippocampus, while only one study reports bilateral reductions in both [69]. These different results have occurred with increased sample size and increased specificity of location of the VOI, while similar findings of decreased NAA/Cr ratio have been reported [58,60,70–72]. Nasrallah et al. [67] studied 11 chronic, medicated patients with schizophrenia and determined a VOI of 12 cm3, while Maier et al. [13,68] investigated 25–26 similar patients and defined VOIs of 4–9 cm3 [13] and 5–7 cm3 [68]. Recently, decreased NAA/Cr ratio has been noted in adolescents with ‘schizophrenia’ [61] and in the ‘unaffected siblings’ of patients with schizophrenia [73]. These latter results have also been used to support the neurodevelopmental hypothesis of schizophrenia [24,25]. Decreases in choline in the right [67] and left [13,68] hippocampus have been noted, along with a finding of increased Choline/Cr ratio in the left medial temporal lobe [70]. Maier et al. [68] noted that an increase in choline concentration with increasing age, which occurs in normal subjects, does not occur in patients with schizophrenia. This was interpreted as a failure of continuing myelination with increasing age that has been reported in postmortem studies [74]. The complex relationship between choline levels determined by 1H-MRS and myelin metabolism in vivo is noted by the authors. The significance of these different results remains unclear. However, they have been interpreted to implicate the hippocampus as a site of a trait marker for schizophrenia [73]. Again, the size of the sample studied and the specificity of the location of the VOI are crucial variables, which determine the results obtained.
The current findings in the basal ganglia region need to be extended by investigating the effect of antipsychotic medication and the nature of such metabolites in the basal ganglia at different stages of the illness, most particularly in drug-naive patients [75–78]. Prospective, longitudinal studies are, again, required. A summary of the findings of 1H-MRS studies in patients with schizophrenia is shown in Table 2.
A summary of positive (+) and negative (–) 1H-Magnetic resonance spectroscopy (MRS) findings from empirical studies investigating schizophrenia
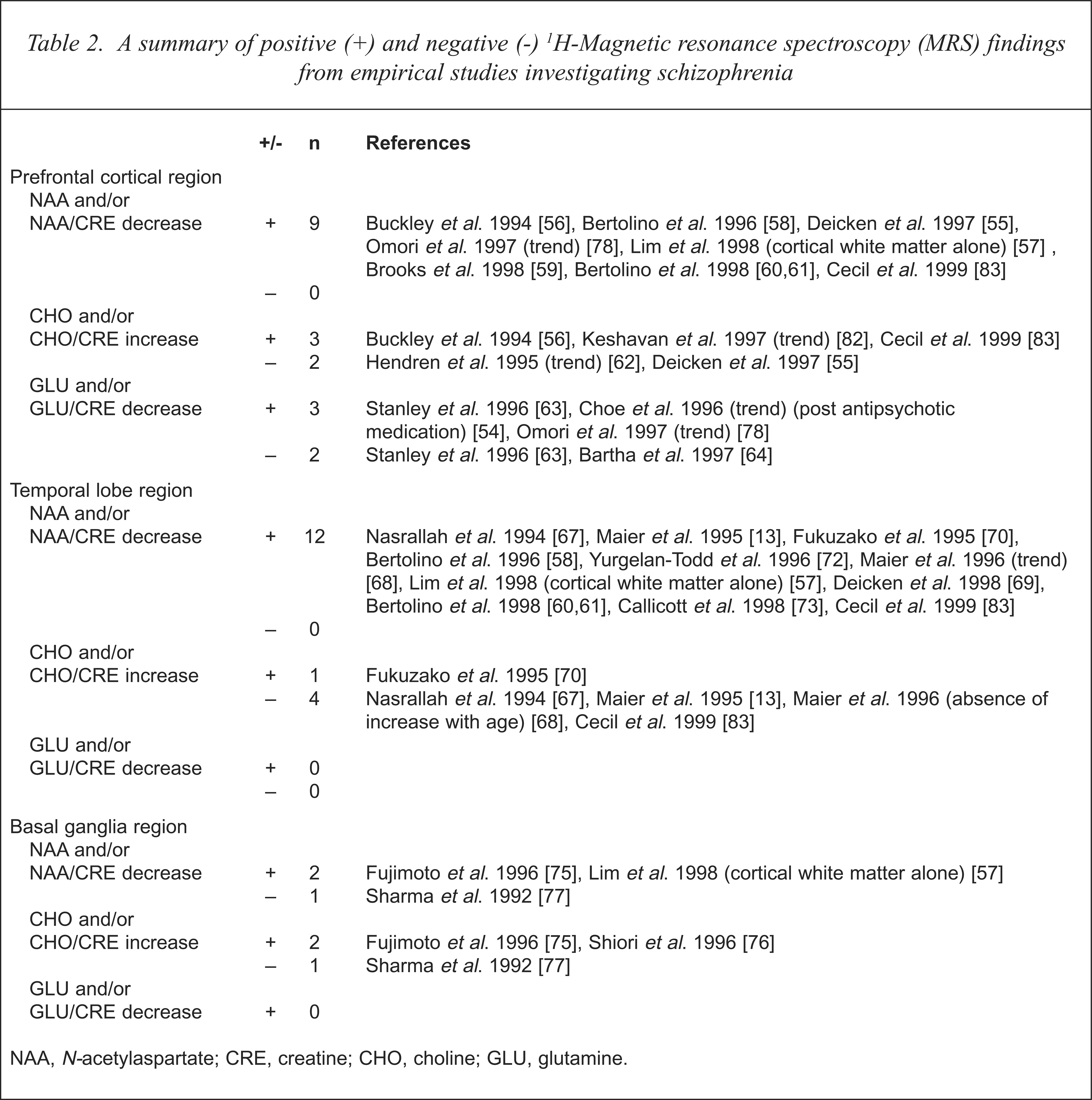
NAA, N-acetylaspartate; CRE, creatine; CHO, choline; GLU, glutamine.
19F-magnetic resonance spectroscopy
19F-MRS has a potential role in investigating differential concentrations of fluorinated psychotropic medications in particular brain regions, glucose metabolism, cerebral blood flow, and neurotransmitter ligands [5]. Three single case reports have been published to date which described the detection of fluorinated fluphenazine in the frontal lobe region after intramuscular injection [79–81]. The potential advantages of this technique over similar positron emission tomography (PET) studies of psychotropic agents include their long-term pharmacokinetics and the biochemical correlates arising from the spectra generated [12]. However, at present more carefully structured investigations of the technique in normal volunteer subjects may be useful to further understand and refine the methodology prior to rigorous investigation of patients with schizophrenia. Improved homogeneity of the magnetic field and improved surface coil design will aid better localisation of particular brain regions [12].
Limitations and future directions
The main advantage of MRS is its provision of in vivo quantitative biochemical information which is superior to data arising from in vitro models. Its non-invasiveness and its lack of known side-effects make it a preferred technique for longitudinal studies [9]. MRS also allows an empirical integration of cerebral function with cerebral structure.
The primary disadvantages include its insensitivity, as only certain elements are amenable to study [5–7,9–12]. In addition, the concentrations of various metabolites of interest which contain these elements are close to the ‘limit of detectability’ [12]. As noted earlier, this ‘limit of detectability’ is dependent on the particular target nucleus of interest, sufficient mobility of the metabolite from which a particular spectrum is sought [13], the strength of the magnetic field, the size of the VOI and the acquisition time [12]. A homogenous magnetic field is also required which is much stronger than that needed for MRI. In addition, the strength of the generated MRS signal is much smaller than that of the MRI signal. Segmentation of different cerebral tissues types, within a given voxel, is not routinely performed. Normal values of given metabolites signified by MRS peaks, within given tissue types and within particular cerebral regions, are not known. Hence, at present, MRS is unable to reliably determine quantitative biochemical information in a particular brain region that is stable over time within a given individual. Current techniques that address the reliable quantification of various metabolites include the use of creatine ratios [9,10,12] and metabolite concentrations relative to those within a particular ‘stable’ cerebral region, such as the centrum semiovale [58,61]. In addition, the effect on in vivo cerebral biochemistry of an applied strong magnetic field is not known as yet.
The determination of the VOI is a critical and difficult aspect of the technique. The gradient-localisation techniques currently result in delayed acquisition times, which diminish the strength of the spectra obtained and the information subsequently available [5]. Also, the complex cyto-architecture of various brain regions does not readily lend itself to clear definition. Hence, the focus on the DLPFC or the hippocampus in one study might not equate to these same named regions in another study. These VOI issues have been used to explain different results obtained [14]. Nevertheless, it is a promising clinical research technique whose utility continues to be explored.
31P-magnetic resonance spectroscopy and 1H-MRS studies have both involved small sample sizes in the determination of differences between patients and controls. This is consistent with the relative newness and the procedural complexity of the imaging technique in schizophrenia research, as reliability and validity issues are addressed.
The vast majority of studies have used a case–control design that limits the interpretation of the data, compared with prospective, longitudinal studies which report within-subject and within-group differences. This latter form of study is now required to clearly demonstrate developmental or phase-specific differences in patients with schizophrenia. Such longitudinal studies have the additional problems of matching voxel placement and replicating MRS spectra obtained in a given individual, whether suffering from schizophrenia or a normal control subject. 1H-magnetic resonance spectroscopy studies have primarily investigated medicated, chronic patients while 31P-MRS studies have also focused on drug-naive patients who are in the early phase of their illness. This limits the ability of the available 1H-MRS studies to adequately address the neuro-developmental hypothesis of schizophrenia as particular critical phases of the illness such as the first episode of illness, the prodrome of illness and the possibility of prepsychotic markers are not clearly defined in their patient sample. One group [59,62] has studied two interesting child samples who were defined by the ‘symptoms of early onset schizophrenia’ or ‘schizotypal personality disorder’. However, important developmental definitional issues were not addressed, given the major developmental factors that form a context for determining phenomena in 8 to 12-year-old patients. For example, the notion of ‘personality disorder’ in this age group is debatable[82].
19F-magnetic resonance spectroscopy is a promising technique that is in its infancy. Its non-invasiveness and its suitability for longitudinal studies are definite advantages over other functional neuroimaging techniques such as PET for investigating the pharmacodynamics and pharmacokinetics of antipsychotic medication. However, fundamental technical problems of MRS signal reliability need to be overcome.
Only a few studies have examined the relationships between MRS-derived measures and symptom profiles (only in 31P-MRS studies) or neuro-psychological variables. These correlations have been noted between symptom profile in 31P-MRS studies and neuro-psychological variables in both 31P-MRS and 1H-MRS studies. These correlations have attempted to provide supporting evidence for current theoretical hypotheses in schizophrenia research, such as the neuro-developmental hypothesis and the hypofrontality hypothesis of schizophrenia. However, the current technical demands in reliably and validly producing spectra of particular brain regions suggest careful interpretation of such correlations, as do the patient selection and study design methodological issues, mentioned above. Nevertheless, future studies should systematically examine these relationships in longitudinal studies, as they may help to shed light on some current hypotheses to explain the symptoms and neurocognitive deficits in schizophrenia.
Conclusion
Despite the limitations noted above, MRS studies in patients with schizophrenia are producing findings that provide tentative support for previous neuroimaging, neuropathological and neuropsychological work. For example, 31P-MRS studies have been reported as suggesting a relative increase in cell membrane degradation and a reduced level of energy metabolism in prefrontal cortical regions, while 1H-MRS studies have noted a presumed decrease in neuronal cell mass in the hippocampal region. Yet, it should be noted that these same findings from 31P-MRS and 1H-MRS studies may support an interpretation of abnormal cell membrane structure [34] and neuronal disruption [74], respectively.
These findings are compatible with current ideas about the nature and aetiology of schizophrenia, but they also raise further questions about the biochemical nature of the disorder and its course. Longitudinal studies will provide answers to some of these, but current methodological limitations make the reliable interpretation of MRS data difficult. The presence of a particular biochemical and its quantitative measure (e.g. concentration) is theoretically meaningful within a given, defined VOI, but the conceptual links between the theoretical construct and its actual meaning are still tenuous. Recent developments, such as chemical shift imaging, have attempted to improve this conceptual link, by allowing slices rather than relatively large VOIs to be studies. However, currently a clear understanding of the methodology and its reliable interpretation is yet to emerge.
Acknowledgements
The authors would like to thank Maria A. Ron, National Hospital for Neurology and Neurosurgery, Queen Square, London, United Kingdom, for her helpful comments on this manuscript. This project was supported by a National Health and Medical Research Council Grant (no. 981112). Alasdair Vance was supported by the 1997 Eli Lilly Psychiatry Fellowship.
