Abstract
There is still debate about the specific types of changes in cognitive and intellectual functioning that occur over the lifespan of a ‘normal’ individual. Many studies have demonstrated that cognitive change is not unitary, and that some abilities decline more rapidly than others [1–3]. There is much interest and speculation on the age of onset of this change and the brain mechanisms responsible for it. More recently, there has been a greater appreciation of the way in which individuals differ in their rates of cognitive change (i.e., interindividual variability), and in the possible importance of inconsistency in cognitive performance (intra-individual variability) as a predictor of cognitive deterioration. Finally, there is emerging information available about risk factors for cognitive decline.
This paper briefly summarizes recent evidence on the nature of cognitive decline, the variability in individual responses to ageing and the risk factors known to affect the rate of cognitive decline. The paper is divided into three sections. First, data are briefly summarized to examine the question of whether cognitive ageing is unitary, or some abilities decline more rapidly than others. Second, evidence is examined that suggests that there is greater variability in test scores as a function of age, a finding that may indicate the existence of a number of subgroups that age at different rates within the same population group. Finally, the available evidence is assessed for the status of the apolipoprotein E (APOE) ∊4 allele, physical health, activity, education and blood pressure as predictors of normal cognitive change later in life.
Constraints, limitations and definitions
In this paper, ‘normal ageing’ refers to the cognitive performance that is observed in longitudinal studies of community-dwelling elderly people. This essentially side-steps the complex issue of what is normal at a particular age. Longitudinal studies provide the following information that cross-sectional studies cannot provide: estimates of individual rates of decline, risk factors for such decline, and data on correlations between changes in cognitive ability and changes in other cognitive and non-cognitive domains. However, longitudinal studies may underestimate change because of practice effects and selective attrition.
Although there are many methods used to describe or examine cognitive change, this paper characterizes cognitive ability as consisting of three major abilities: crystallized intelligence, memory and cognitive speed. Crystallized intelligence is ‘assumed to be the cumulative end product of information acquired’ by an individual [4], p.69] and is demonstrated on tests of vocabulary, information accumulation and other knowledge-based activities. Memory is commonly divided into short-term and long-term, with both types further fractionated into declarative and procedural memory. Declarative memory refers to conscious recollection and recall, whereas procedural memory (which is memory that does not require the intentional or conscious recollection of an experience) includes priming, classical conditioning and skillbased learning [5]. Only short-term declarative memory is discussed in the present paper. Cognitive speed refers to performance on perceptual-motor tasks that are timed. Examples of such tasks are digit symbol substitution from the Wechsler Adult Intelligence Scale [6] and simple or choice reaction time. In the present paper, measures of crystallized intelligence and memory were created as composite measures of a number of subtests. The validity of the constructs over the three waves of measurement was tested via confirmatory factor analysis [7]. Details of the measures are provided in Appendix I.
Cognitive change is not unitary
The correct answer to the question, ‘Does cognitive performance decline in old age?’ is, ‘It depends on which cognitive ability is assessed’. Crystallized abilities increase up to the sixth or seventh decade, and may only decrease in late old age. In contrast, memory and cognitive speed generally show continuous linear decline during early adulthood, with perhaps further acceleration in late old age. Data from meta-analyses of cross-sectional studies [4] suggest that cognitive speed drops by approximately 20%% at age 40 and by 40–60%% at age 80. The existence of these different developmental trajectories according to the type of mental ability has been well recognized for decades (see [5],[8–10]). To illustrate these findings in longitudinal data, Fig. 1 shows results from the Canberra Longitudinal Study for crystallized intelligence, speed and memory. Four age groups are shown. Age group status was determined on the first occasion of interview. The sample was a probability sample of persons aged 70 years and older drawn from the electoral roll for Canberra and the neighbouring town of Queanbeyan, New South Wales. Data were collected in 1991, 1994 and 1998. The sample and study design have been described previously [11], [12]. Analyses in the present paper were based on the set of participants who completed all cognitive tests at each of three occasions (n = 294). Crystallized intelligence did not decline significantly for any of the four groups, including those older than 85 years at the commencement of the study. Memory declined significantly, and the rate of decline accelerated at older ages. Cognitive speed also deteriorated significantly in all age groups over the 7.5-year period of follow-up.
Mean scores on measures of (a) crystallized intelligence; (b) speed; and (c) memory for four age groups across three occasions. Age groups are: –▵–, 70–74 years;…□…, 75–79 years; –▴–, 80–84 years; —▪—, 85+ years at time of testing.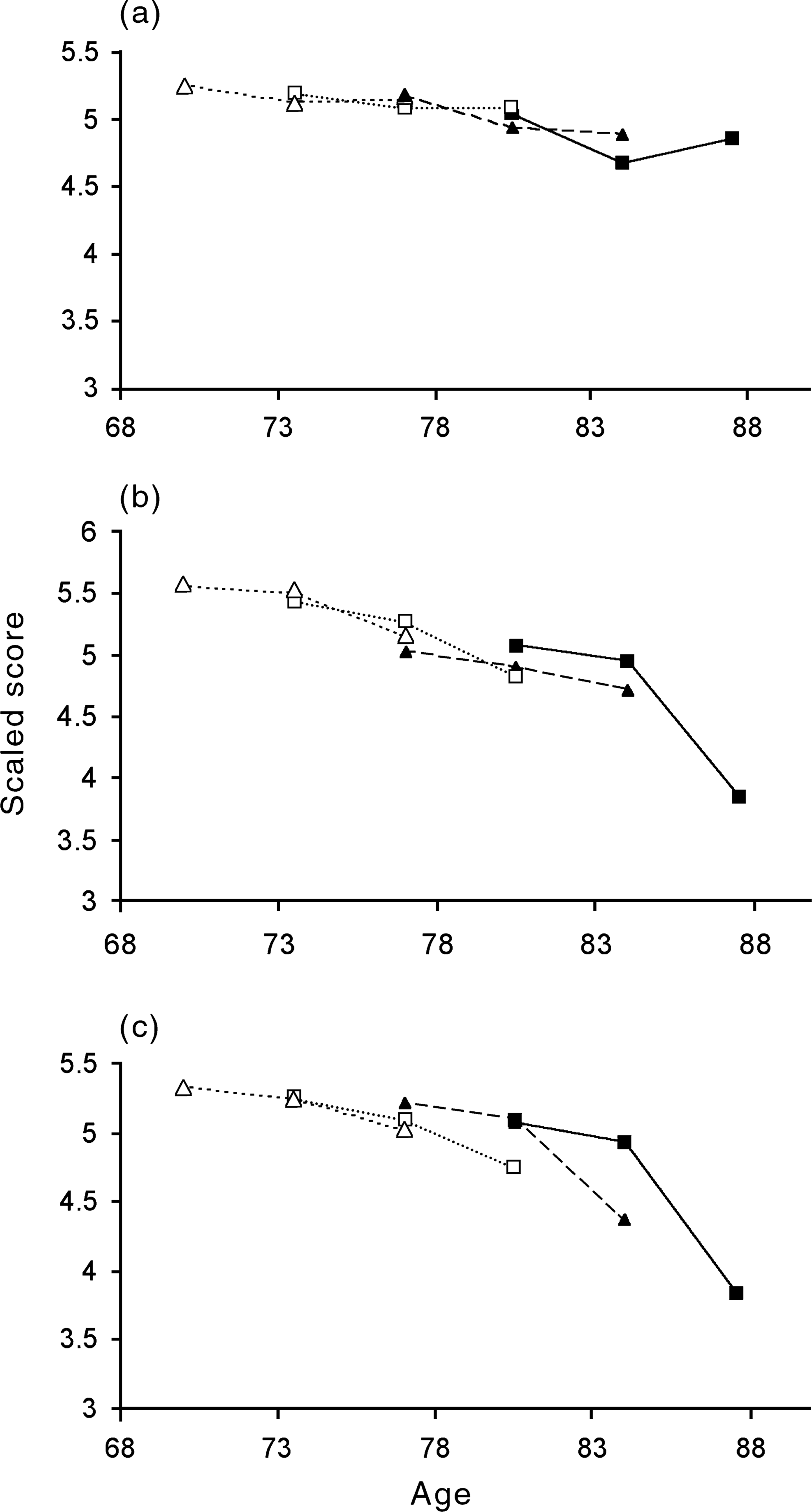
The finding of a significant deterioration in memory and cognitive speed over the lifespan is also a feature of other longitudinal data sets, such as the Einstein Ageing Study [13]; Victorian Longitudinal Study [3], the Seattle Longitudinal Study [2] and other Australian longitudinal studies [14].
The ‘ageing’ of the brain, and pathology resulting from age-associated insults to the brain, are assumed to underlie the deficits in speed and memory performance. However, the biology of cognitive ageing is not well understood. Deary in his recent book, Looking down on intelligence, noted (p. 262): The ‘biology of human intelligence differences’ is not a coordinated filed of study. It is a collection of pragmatic stabs at uncovering correlations between putative brain parameters and mental ability test differences. The brain parameters are rarely arrived at via detailed theory; rather they have been alighted on by convenience and common sense. Five main strands of research cover associations between mental ability scores test differences and brain size, nerve conduction velocity, eventrelated potentials (ERPs) and electroencephalography, COGNITIVE CHANGES IN NORMAL AGEING 770 functional brain scanning and molecular genetics. In no area is there a large effect size that combines with a clear mechanistic story. [15]
To these statements, we might add that the investigations of associations between cognitive changes in ageing and brain changes across the lifespan are also in their infancy. Cross-sectional investigations have revealed substantial age differences in ventricular or sulcal cerebrospinal fluid (CFS), brain, hippocampal, frontal and temporal lobe volumes [16]. It is known from specific lesion studies that memory deficits, particularly short-term declarative deficits, are associated with hippocampal damage and the medial temporal lobe region [5]. Cognitive speed deficits may be associated with white matter hypersensitivies in the frontal lobes and other brain regions [17]. However, data showing that changes in hippocampal structures and white matter lesions are correlated with changes in memory and cognitive processes are in short supply. Schmidt et al. [18] recently reported that white matter lesions increased in approximately 18%% of 273 older participants over 3 years, but lesion progression had no effect on cognitive test performance. In contrast, Swan et al. [19] reported an association between increased white matter hyperintensities and neurobehavioural functioning, whereas Jack et al. [20] reported that hippocampal atrophy was associated with cognitive status 3 years later. Crystallized intelligence is presumably represented in the brain in areas or processes that do not deteriorate until late in life, or do not deteriorate until a threshold of functional loss has been reached. Studies of individuals who have the APOE ∊4 allele, and hence may be more vulnerable to cognitive decline, have been found to have brain atrophy, ‘perhaps preferentially affecting temporal lobe structures such as the hippocampus, and also with alterations in brain glucose metabolism’ [15]. Further research correlating changes in cognitive functioning with changes in brain processing is needed to help identify the brain mechanisms underlying cognitive change. To quote Deary 15, p. 309] ‘Medical researchers often take the neuropsychological approach, sometimes glibly assuming that different tests test not only distinct cognitive abilities but also distinct geographical regions of the brain. These assumptions are largely derived from work with animals and humans with experimentally or traumatically induced, relatively discrete lesions. Linkage among construct, test and region is often strong under these special circumstances. However, in more diffuse and perhaps less severe processes, such as seen in early Alzheimer's disease (AD), and also in looking at cognitive abilities in the normal range in healthy people, these linkages are likely to be less strong.’
Two common questions that are asked of these changes in cognitive performance with age are: (i) ‘Will it happen to me if I'm healthy?’; and (ii) ‘Are these changes inevitable?’ There is a consensus among researchers that serious health problems may be detrimental to cognitive functioning (see later discussion) but that less serious health problems and self-reported measures of health problems do not account for the bulk of cognitive ageing. To quote Arbuckle et al. [21] p. 664]: Crosssectional and longitudinal studies of the relation between health and cognitive functioning have generally shown that poorer physical health is associated with poorer cognitive functioning [2], [22] but that age-associated illnesses explain only part of the of the age-related variance in cognition [2], [3], [24].
Are these changes inevitable?
This question is difficult to answer conclusively. Many of the changes in cognitive functioning are gradual and develop from early adulthood, suggesting that the cognitive changes may be a normal developmental process. Studies of specific elderly groups suggest that cognitive decline may be unavoidable in old age. For example, highly educated academics with good health, and a lifetime of exposure to complex and stimulating environments, nevertheless show cognitive change in late adulthood, even without the diagnosis of dementia [26], [27]. In the longitudinal phase of one study, individuals showed at least some deterioration on one or more of the four core outcome measures, and most showed decline on two or more [27]. Our conclusion is that these changes may be inevitable, but that there may well be large individual differences in the ages at which these cognitive changes begin.
Inter-individual and intra-individual variability increases with ageing
Older age is associated with greater interindividual (diversity) and intra-individual differences.
Inter-individual differences (diversity)
Although the average performance on most cognitive tasks declines with age, studies have suggested that many older individuals change very little, whereas others deteriorate dramatically, leading to a greater diversity of cognitive scores [28–31]. In a review of six longitudinal and 48 cross-sectional studies, Nelson and Dannefer reported increased variability with age, not only for cognitive variables but also for personality and biological indices [28]. There have also been recent studies reporting greater individual differences in cognitive change [3], [32], findings that indicate greater diversity in the rates of change at older ages. Most, but not all studies, suggest that the interindividual differences in speed and memory increase with increasing age [33].
Data illustrating increased interindividual differences in change from the Canberra Longitudinal Study are displayed in Fig. 2. These data illustrate individual paths for participants with full data sets over the three occasions of measurement. A measure of individual diversity was calculated by regressing cognitive change scores on age, determining the absolute residuals of all change scores and then correlating these residual scores with age. We found a positive correlation between residuals scores and age for both speed and memory (R = 0.23, P < 0.05, n = 426, R = 0.18, p < 0.05, n = 426), indicating greater diversity in these cognitive abilities at older ages. This association was not found for crystallized intelligence (R = 0.06, ns).
Observed individual trajectories for survivors on measures of (a) crystallized intelligence, (b) speed and (c) memory across three occasions (each line represents one individual's test scores).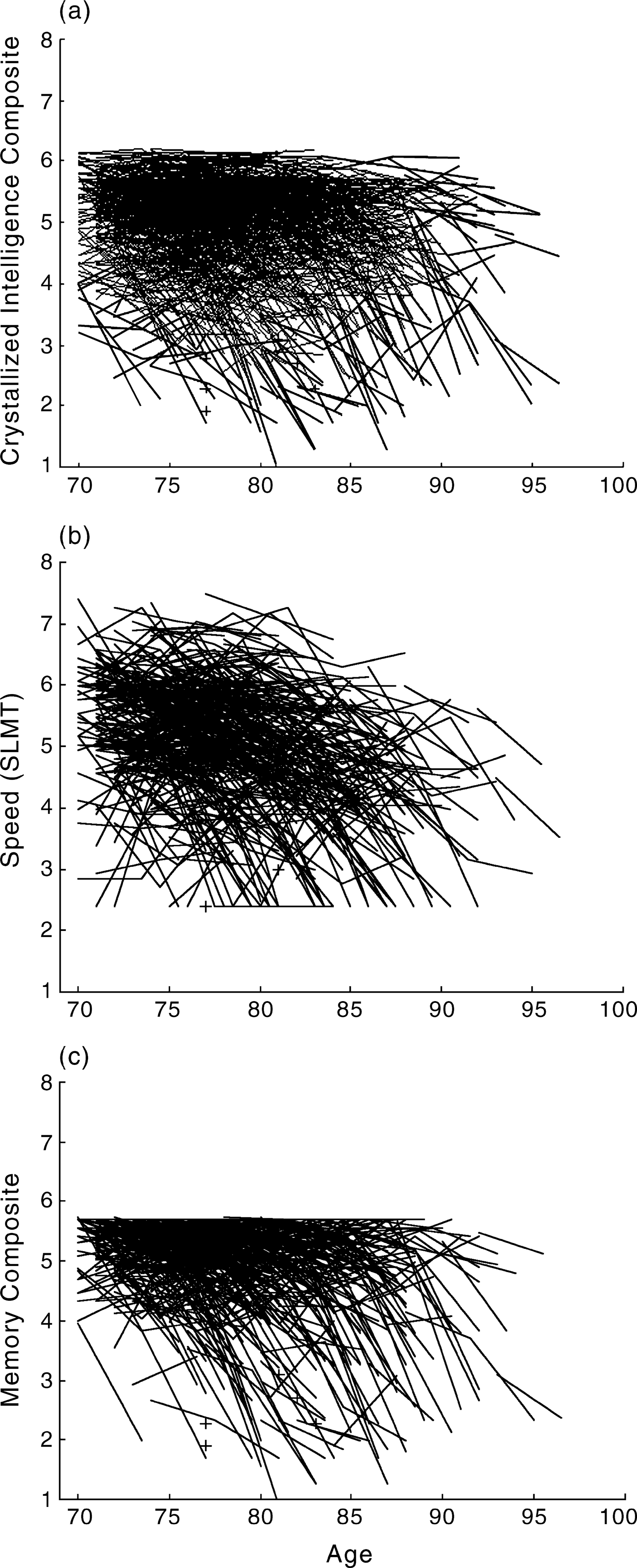
Intra-individual differences (short-term and long-term individual differences)
Greater intra-individual change with older age has also been reported. Nesselroade made a distinction between individual change that is more or less durable or systematic (developmental changes) and individual change, which is transient and short-term (the ‘wobble’ about the developmental change) [34]. Both these types of withinperson change have been examined in a variety of studies [35–37]. In the original research of this kind, Hertzog et al. [38] tested memory for sentences fortnightly in seven women over 2 years, testing performance on equivalent forms of the test. Individuals varied in the extent to which they were consistent in their level of remembering, and also had different trajectories, with either steady improvement or decline over the 2-year period. Recent studies of intra-individual variability show that the wobble increases with age for memory [36] reaction time [35]and sensori-motor tasks [36]. The extent of deviation from the average is also a predictor of poorer levels of performance [37]. This variability seems to occur across different tasks (such as sensori-motor and reaction time) and, hence, may be a relatively stable characteristic of people. Li and Lindenberger [36] have speculated that transient intra-individual variability indicates impaired neurobiological functioning, based on the finding that people diagnosed with dementia show approximately twice as much wobble as those not so diagnosed [37].
Predictors of cognitive change
Many factors have been investigated as potential risk factors for cognitive decline. Following Kraemer et al., these can be conveniently identified as either marker variables or risk factor variables [39]. Marker variables are those that are essentially unchangeable, such as education (schooling), APOE ∊4 allele and gender. Risk factor variables, such as physical activity, age and so on, develop and change with the individual, In a recent review of 34 studies [25], we examined the status of education, activity, health, blood pressure and the APOE ∊4 allele as predictors of cognitive change in old age. These variables were chosen because they have been widely examined in cross-sectional research. Table 1 summarizes the outcomes for each of the risk factors.
Results from studies of risk factors from longitudinal studies as a function of cognitive domain
Lower education
The outcomes of these studies appeared to be related to the types of cognitive tests included. Follow-up was between 1 and 28 years after baseline measures were taken. All seven studies (not shown in table) that included a screening test, such as the Mini Mental State Examination (MMSE), found a protective effect of education. Of those including memory, five of seven showed faster decline in those with poor education, and three of four found faster decline for measures of crystallized intelligence. In comparison, three studies failed to find an effect for speed measures [25]. These findings suggest that the protective effects of higher education may be restricted to crystallized intelligence, memory and mental state measures.
Presence of APOE ∊4 allele
In four studies that included a measure of crystallized intelligence and three studies that included measures of fluid intelligence (not shown), no effect was found. In contrast, three of four studies that included a measure of cognitive speed found an effect of APOE ∊4, and four of six studies found an effect on memory. Only three of eight showed an effect on mental status [25]. Therefore, it appears that the effect of APOE is most reliable for memory and processing speed.
Poor health
Health as a predictor of cognitive decline has been addressed earlier in this paper. Briefly, the findings suggested that the association between poor self-rated health and cognitive deterioration was not consistent, with three studies reporting no significant association (for example [40], [41]), one study reporting that poor health at baseline predicted greater cognitive deterioration [42] and one study noting that high self-reported health was associated with cognitive improvement [43]. A stronger association was found between objective measures of health function, such as lung function and poorer cognitive outcome (for example [40], [43]). Cerebrovascular disease predicted increased risk and amount of cognitive decline.
Activity
Four studies were identified, and the results from them are contradictory [40–43]. It is likely that the effect of activity is small, and that higher activity will only be found to be protective in large samples.
Blood pressure
Ten studies examined the relationship between cognitive change and blood pressure. Different indices of blood pressure were used in the 10 studies. Blood pressure affected change in performance on measures of mental status, speed, memory and visuospatial ability (not shown), but there was not enough evidence to conclude that crystallized abilities were affected. Although there are relatively few studies for each predictor, these findings suggest that higher education, lower blood pressure, absence of the APOE ∊4 allele, and good health are protective of subsequent cognitive decline. The evidence from activity levels is not yet clearly established. An interesting finding is that risk factors may be specific to the type of cognitive task, with, for example, education exerting an effect on crystallized and memory measures, and APOE ∊4 allele affecting memory and speed to a greater extent than crystallized intelligence. Clearly, there is no guarantee that levels of particular factors at time one (i.e., predictors) of change will also change with cognitive decline, or that variables which decline with age will correlate with other variables that also decline. Predictors of cognitive decline may be causal factors in cognitive decline, but a minimum condition is the demonstration that such factors decline conjointly with changes in cognition.
Conclusions
Cognitive ageing is an exciting area of investigation. We know that on average, older people perform more poorly in areas of memory and cognitive speed than younger people. There is evidence that cognitive diversity increases with increasing age, and that the extent of cognitive change can be predicted to some degree by a number of risk factors, including health, APOE ∊4 and disability. Much is known about predictors of cognitive change, but the new challenge is to identify causal relationships. A next step in this process is to investigate factors that change with cognitive change over time in large longitudinal studies. A recent boost in cognitive ageing is a renewed attempt to relate changes in more fundamental biological processes, such as forced expiratory volume (FEV) and grip strength, and brain indices such as white matter lesions, and brain and hippocampal volumes to changes in cognitive performance. An understanding of the relationships among these biological, brain and cognitive variables informs discussion about the nature of the processes that produce cognitive decline. For example, if changes in FEV and grip strength are strongly associated with cognitive change, and share common risk factors, it implies that the processes underlying lifespan changes in intelligence and memory may be very broad indeed. Proposed brain or cell processes capable of mediating such diverse but related changes include cerebral blood flow and decreased metabolic rate, mitochondrial dysfunction, age-related myelin degeneration and dopaminergic neurotransmitter depletions.
Footnotes
Acknowledgements
This study was supported by grant 973302 from the National Health and Medical Research Council and by the Australian Rotary Health Research Fund. Thanks are due to Kaarin Anstey, A. J. Mackinnon, S. Hofer, P. A. Jacomb, A. F. Jorm, A. E. Korten, A. S. Henderson, K. Dear and B. Rodgers for their contributions.
Appendix 1
Crystallized intelligence was a composite measure of three tests.
