Abstract
Transient occlusion of the middle cerebral artery (MCAO) in rats leads to abnormal accumulation of β-amyloid (Aβ) peptides in the thalamus. This study investigated the chemical composition of these deposits. Adult male human β-amyloid precursor protein (APP) overexpressing (hAPP695) rats and their wild-type littermates were subjected to transient MCAO for 2 h or sham operation. After 26-week survival time, histological examination revealed an overlapping distribution pattern for rodent and human Aβ in the thalamus of hAPP695 rats subjected to MCAO. X-ray microanalysis showed that the deposits did not contain significant amount of iron, zinc, or copper typical to senile plaques. In contrast, the deposit both in hAPP695 and non-transgenic rats contained calcium and phosphorus in a ratio (1.28 ± 0.15) characteristic to hydroxyapatites. Alizarin red staining confirmed that calcium coaccumulated in these Aβ deposits. It is suggested that APP expression is induced by ischemic insult in cortical neurons adjacent to infarct, which in turn is reflected as increased release of Aβ peptides by their corticothalamic axon endings. This together with insufficient clearance or atypical degradation of Aβ peptides lead to dysregulation of calcium homeostatis and coaccumulation in the thalamus.
Introduction
β-Amyloid precursor protein (APP) is a transmembrane protein with a long extracellular N-terminal and short intracellular C-terminal domain. β-Amyloid precursor protein is processed by β- and γ-secretases to release β-amyloid (Aβ), a hydrophobic self-aggregating peptide consisting of 40 to 42 residues. β-Amyloid is a major component of senile plaques, one of the pathological hallmarks of Alzheimer's disease (Braak and Braak, 1997).
Brain insults, including cerebral ischemia, lead to a transient upregulation of APP mRNA and accumulation of APP in cortical areas adjacent to ischemic lesions and in the corpus callosum (van Groen et al, 2005). β-Amyloid and APP staining is also observed in the circuit-connected but nonischemic thalamus after transient occlusion of the middle cerebral artery (MCAO), where it is still evident 9 months after operation (van Groen et al, 2005). Interestingly, N-terminal APP and Aβ staining in the thalamus is diffuse acutely after the infarct, but accumulates, leading to dense plaquelike deposits in the ventroposterior lateral and ventroposterior medial nuclei.
The exact nature of these Aβ deposits in the thalamus is not known. Metal ions such as iron, zinc, and copper are known to promote aggregation of soluble Aβ (Mantyh et al, 1993), and the same metals co-deposit with senile plaques in Alzheimer's disease (Lovell et al, 1998) and in Aβ plaques in transgenic PS/APP mice (Falangola et al, 2005). There is evidence that copper and iron interact with Aβ to facilitate enzymatic production of hydrogen peroxide leading to autooxidation of Aβ and formation of protease soluble and cross-linked Aβ (Bush, 2003). In turn, synaptic zinc reacts with the copper-bound Aβ. Recently it was shown that the presence of calcium can also accelerate Aβ aggregation (Isaacs et al, 2006).
The aim of this study was to characterize further Aβ deposits in the thalamus after MCAO. Given the possible involvement of metals in Aβ accumulation, chemical analysis of thalamic Aβ deposits was carried out by X-ray microanalysis in both human APP overexpressing (hAPP695) and nontransgenic rats.
Materials and methods
Generation of hAPP695 Transgenic Rat
A cDNA construct containing the human amyloid precursor protein 695 isoform (hAPP695) was cloned into the mouse prion promotor (MoPrP.Xho) expression vector at the Sma I site (Borchelt et al, 1996). A 18.7 kb NotI/SnaBI fragment containing hAPP695, the MoPrP promoter, together with its 3′- and 5′-untranslated sequences was microinjected into fertilized (Fisher344XWistar) rat eggs and then implanted into pseudopregnant females. Genomic DNA samples were isolated from the tails of transgenic and non-transgenic rats. Potential founders and transgenic offspring were identified by PCR and by Aβ staining. All the rats used in this study were maintained on a Wistar background. Nontransgenic littermates were used as controls.
Biochemistry
Total Aβ levels from cortex and hippocampus of intact hAPP695 transgenic and nontransgenic rats were assessed using the Aβ1–42 ELISA kit (Innogenetics, Ghent, Belgium). Western blotting was used to analyze the levels of APP full length, APP C83/C99, and GAPDH in total protein lysates (50 μg) extracted from cortex and hippocampus. Anti-APP C-terminal (A8717; Sigma Chemical Co., St Louis, MO, USA) and anti-GAPDH (AB25970; Abcam, Cambridge, UK) antibodies were used in combination with enhanced chemiluminescence detection.
Experimental Groups
Male rats (3 to 4 months old, weighing 375 to 425 g at the beginning of the study) were used. Rats were assigned to the following experimental groups: transgenic MCAO rats (n = 14), nontransgenic MCAO rats (n = 13), and sham-operated rats (n = 8). The animals had free access to food and water, and were housed in individual cages in a temperature-controlled environment (20 ± 1°C) with lights on from 0700 to 1900 hours. Experimental procedures were conducted in accordance with the European Community Council directives 86/609/EEC and the study was approved by the Ethics Committee of the University of Kuopio and the Provincial Government of Kuopio (Kuopio, Finland).
Focal Cerebral Ischemia
Focal cerebral ischemia was induced using the intraluminal filament technique (van Groen et al, 2005). Anesthesia was induced in a chamber using 3% halothane in 30% O2/70% N2O. A surgical depth of anesthesia was maintained throughout the operation with 0.5 to 1% halothane, delivered through a nose mask. To occlude blood flow to the right MCA territory, a heparinized nylon filament (Ø0.25mm, rounded tip) was advanced 1.9 to 2.1 cm into the internal common carotid artery until resistance was felt. The filament was held in place by tightening a suture around the internal common carotid artery and placing a microvascular clip around the artery. Body temperature was monitored and maintained at 37°C using a heating pad connected to a rectal probe. After 120 mins of MCAO, the filament was removed and the external carotid artery was permanently closed by electrocoagulation. Successful MCAO was verified by assessing sensorimotor impairment and by measuring infarct volumes at the end of the study.
Histochemistry
Rats were perfused transcardially 26 weeks after operation with 0.9% NaCl followed by 4% paraformaldehyde in 0.1 mol/L phosphate buffer, pH 7.4. The brains were removed from the skulls, postfixed, and cryoprotected. Frozen sections (40 μm) were cut with a sliding microtome and stored in a cryoprotectant tissue collection solution at −20°C. The thalamic accumulation of Aβ was examined using a rodent-specific antibody (rabbit anti-rodent Aβ3–16, #9151; Covance, Delham, MA, USA) or a human-specific antibody (mouse anti-human Aβ4–10, WO-2; The Genetics Company, Schlieren, Switzerland). The sections were pretreated for 30 mins with hot (85°C) citrate buffer. This series of sections was transferred to a solution containing the primary antibody (rabbit anti-rodent Aβ at 1:5,000 or mouse anti-human Aβ at 1:2,000) and Tris-buffered saline with 0.5% Triton X-100 (TBS-T). After incubation in this solution for 18 h on a shaker table at room temperature (20°C) in the dark, the sections were rinsed three times in TBS-T and transferred to a solution containing the secondary antibody (goat anti-mouse *biotin; Sigma Chemical Co., or goat anti-rabbit Ig*biotin; Chemicon, Temecula, CA, USA). After 2 h, the sections were rinsed three times with TBS-T and transferred to a solution containing mouse ExtrAvidin® (Sigma Chemical Co.), and then incubated for approximately 3 mins with diaminobenzidine or Ni-enhanced diaminobenzidine (van Groen et al, 2005).
Calcium was stained with Alizarin red method. In brief, sections were mounted on gelatinized glass and immersed in 2% (w/v, distilled water pH 4.1 to 4.3) Alizarin Red (Merck, Darmstadt, Germany) for 30 secs followed by a rinse in distilled water. Sections were quickly dehydrated with acetone and xylene and mounted in Depex (VWR International Ltd, Poole, UK).
Scanning Electron Microscopy and Chemical Microanalysis
Scanning electron microscopy (SEM) and chemical microanalysis was performed in a XL30 ESEM (FEI Company, Eindhoven, The Netherlands) equipped with Röntec EDS microanalyzer (Röntec GmbH, Berlin, Germany). Brain sections were thoroughly rinsed in water and deposited on high-purity aluminium stubs using sticky tabs (Agar Scientific, Cambridge, UK). Scanning electron microscopy was used to localize mineral deposits in the thalamus before obtaining detailed X-ray spectra and maps. Win-Shell and ScanVision software (Röntec GmbH) were used for chemical analysis. The detection sensitivity of scanning electron microscopy with energy-dispersive X-ray spectroscopy is 10−15 g.
Results
Enzyme-linked immunosorbent assay measurements of adult, intact hAPP695 transgenic rats showed that total Aβ levels were increased in the cortex and hippocampus approximately twofold compared with non-transgenic littermates. Consistent with the Aβ result, a similar increase was observed with APP C99 fragment levels in both cortex and hippocampus of intact hAPP695 transgenic rats. Although the total Aβ levels were increased, amyloid plaques typically seen in Alzheimer's disease brain were not observed in aged hAPP695 transgenic rats.
The primary infarct in this transient MCAO model included the parietal cortex extending to the anteroposterior level used for histological staining of Aβ and calcium. The striatum was usually completely damaged. The thalamus is not in the vascular territory of the middle cerebral artery, meaning that it was not directly affected by ischemia. Mean infarct volume was 70.1 ± 7.4 mm3 for the cortex and 28.7 ± 1.2 mm3 for striatum. Middle cerebral artery occlusion rats were also impaired in sensorimotor functions as assessed by the limb-placing test on postoperative day 2 (scores, from a maximum of 10, for MCAO rats 2.5 ± 0.3 and for sham-operated rats 9.1 ± 0.3).
The human-specific Aβ antibody stained cortical neurons (layer 5) both in sham-operated and ischemic hAPP695 rats. Human Aβ-positive deposits were observed in the thalamus in hAPP695 rats only after MCAO (Figure 1G). The rodent-specific antibody for Aβ showed only light staining in sham-operated nontransgenic or transgenic hAPP695 rats. However, rodent Aβ-positive deposits were found in the thalamus both in hAPP695 and nontransgenic rats after MCAO (Figures 1E and 1H). Both rodent and human Aβ staining in the thalamus varied from small granules to large formations exceeding several 100 μm. While the location of deposits varied, the most affected nucleus was ventroposterior medial/ventroposterior lateral. There was partial overlap of rodent and human Aβ staining in hAPP695 rats subjected to MCAO (Figures 1G and 1H).
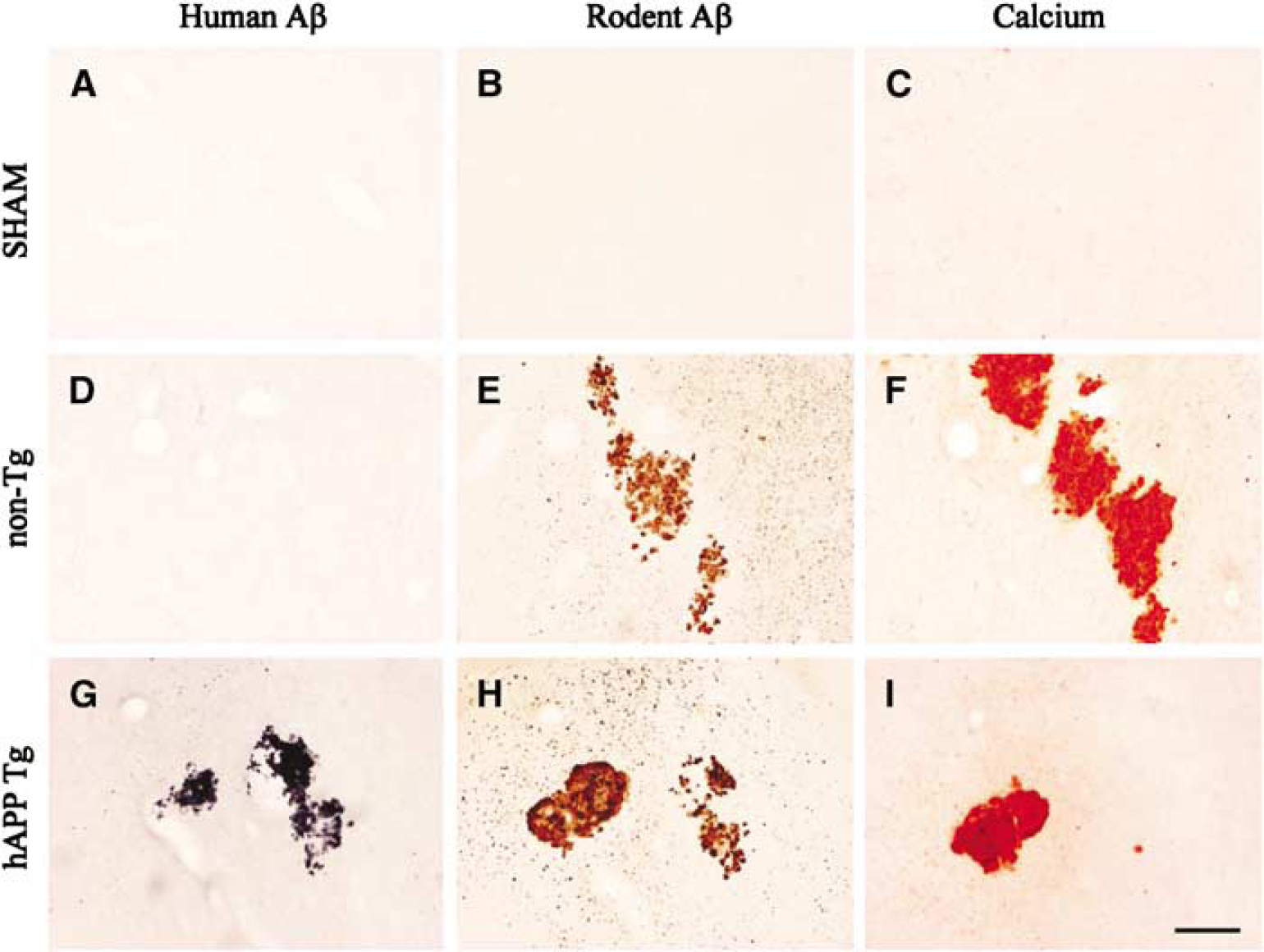
Coronal sections showing staining for human and rodent Aβ and calcium in the thalamus 26 weeks after middle cerebral artery occlusion (MCAO). (
Scanning electron microscopy showed large mineral deposits in the thalamus with coral-shaped protrusions both in hAPP695 and non-transgenic rats after MCAO (Figure 2D). Seven rats with large deposits (> 100 μm) were selected for a detailed chemical analysis. X-ray analysis showed no evidence of iron, zinc, or copper in deposits. This was confirmed by histological staining for zinc and iron (data not shown). However, calcium and phosphorus were detected in all deposits with a ratio of 1.28 ± 0.15 characteristic of hydroxyapatites. The distribution of calcium and phosphorus was completely overlapping within the deposit (Figures 2A and 2B). Interestingly, the signal for carbon in the deposit showed a decrease compared with surrounding areas (Figure 2C).
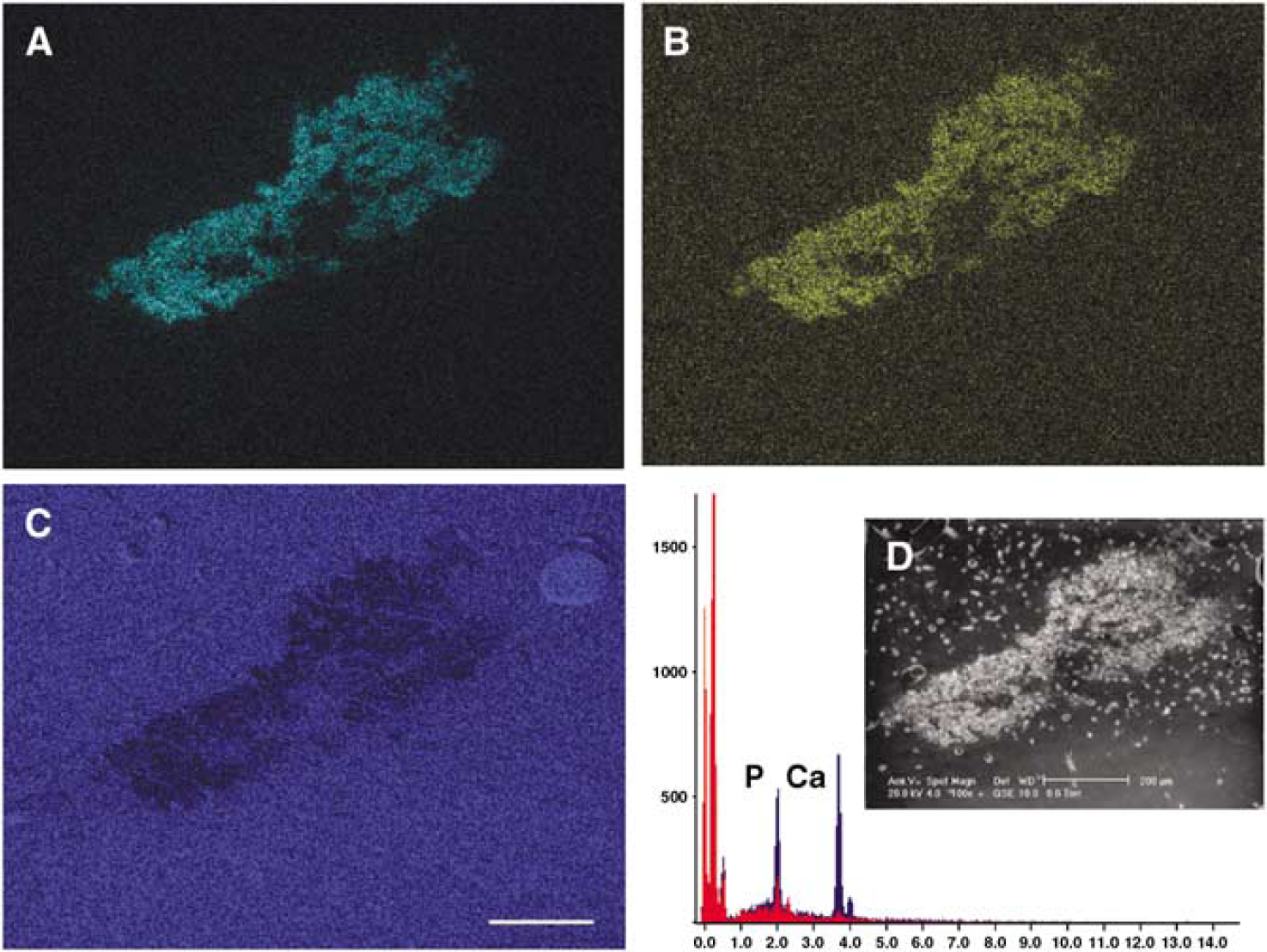
Chemical composition of deposits in the thalamus after middle cerebral artery occlusion (MCAO) in rats. Note a partial overlap with calcium (
Alizarin red staining for calcium from sections adjacent to those stained for rodent and human Aβ showed a similar distribution pattern in the thalamus indicating coaccumulation in the large deposits (Figures 1F and 1I). Similar to the Aβ staining, the size, number, and location of calcium deposits in the thalamus varied between rats. Calcium deposits were not observed in any other brain areas.
Discussion
This study confirmed the abnormal accumulation of endogenous Aβ peptides in the thalamus after MCAO in rats. Furthermore, data from hAPP695 rats showed a similar processing of human APP leading to thalamic deposition. In neither case did the Aβ deposits contain metals typical to senile plaques in Alzheimer's disease. In contrast, Aβ deposits showed a partial overlap with calcium and phosphorus, a finding which has not been reported previously.
We previously showed that the accumulation of endogenous Aβ peptides in the thalamus might be related to alterations in descending corticothalamic fibers and that cortical damage only affects those thalamic regions, which are connected with these cortical areas (van Groen et al, 2005). Since human Aβ is stained primarily in the cortex in hAPP695 rats, the thalamic accumulation of human Aβ further points to the involvement of corticothalamic projection neurons. We suggest that increase in APP expression in corticothalamic neurons by cerebral insults results in an overflow of Aβ release in the thalamus leading to Aβ accumulation due to insufficient clearance. One should note that the deposits do not contain Aβ1–42 or Aβ1–40, but shorter N-terminal peptides (van Groen et al, 2005) indicating that the deposit are not identical with traditional amyloid deposits, and thus atypical processing of APP, probably, is involved. Rodent-specific antibody stained only short Aβ peptides and, thus, it is not full-length APP that accumulates in the thalamus. Interestingly, the prion promotor of hAPP695 is similarly activated by ischemic insults as the ‘normal’ APP promotor and human APP is similarly processed in the thalamus despite structural differences.
Surprisingly, Aβ deposits in the thalamus after MCAO did not contain metals typical to senile plaques (Lovell et al, 1998; Falangola et al, 2005). Instead, a consistent finding was a calcium coaccumulation in the deposits. Previously, calcium precipitation has been observed in a number of acute and chronic brain diseases related to excitotoxic cell death and neurodegeneration (Rodriguez et al, 2000; Ramonet et al, 2006). Impaired calcium homeostasis has also been reported following focal cerebral ischemia (Shirotani et al, 1994; Watanabe et al, 1998). In these studies a significant 45Ca accumulation was noted 3 days after ischemia, persisting up to 1 month in the ventroposterior medial/ventroposterior lateral nuclei on the lesion side. Disrupted calcium homeostasis was suggested to initiate secondary degeneration in the thalamus. Our data suggest that the process is more long lasting, leading eventually to severe forms of calcification. In addition to glutamate-driven excitotoxicity, thalamic hypoperfusion and hypometabolism (Dijkhuizen et al, 1998; Barbelivien et al, 2002), which are typical consequences of MCAO, may also contribute to calcification. Hypometabolism in the thalamus leads to reduced energy production, the energy needed to keep calcium at basal cytoplasmic level, and hypoperfusion reduces the pH, which favors precipitation of calcium with phosphorus. Interestingly, thalamic calcification also occurs following brain trauma and global ischemia and seizures (Kato et al, 1995; Pierce et al, 1998), indicating that focal cortical damage per se is not needed for deposition. Together these data suggest that disturbed cortical electrical activity leads to dysregulation of thalamic neuronal calcium homeostasis and to calcification possibly through multiple mechanisms.
More puzzling is the thalamic overlap between Aβ staining and calcium in MCAO rats. There is evidence for a complex interaction between Aβ and calcium. Released Aβ has been suggested to be incorporated into neuronal membranes forming calcium-permeable channels (Bhatia et al, 2000; Kawahara and Kuroda, 2000), which would allow uncontrolled calcium influx elevating intracellular calcium concentrations. This is hypothesized to be one of the mechanisms of the neurotoxicity of Aβ peptides. Another possibility is increased calcium influx through NMDA receptors (Kelly and Ferreira, 2006) or generalized increase in membrane permeability (Demuro et al, 2005) by Aβ oligomers. In addition, extracellular calcium, similar to aluminum, iron, and zinc, can accelarate aggregation of released Aβ (Mantyh et al, 1993; Isaacs et al, 2006). A detailed electron microscopy study combined with immunohistochemistry after MCAO is needed to understand the temporal sequence of accumulation and its mechanisms, which shares characteristics with the pathophysiology of cerebral ischemia, Alzheimer's disease, traumatic brain injury, and brain calcification.
In conclusion, the present data show the multi-faceted chemical composition of Aβ deposits in the thalamus after focal cerebral ischemia in rats. The deposits did not contain metals typical to senile plaques, but contain calcium and phosphorus. The functional meaning of this deposition is not known, but immediate APP activation in neurons in the cortical penumbral areas is probably to be a protective rather than a deleterious response to ischemic insult (Masliah et al, 1997). Furthermore, thalamic calcification may have a protective role by removing excessive Ca2+ (Rodriguez et al, 2000).
Footnotes
Acknowledgements
We thank Nanna Huuskonen, Pasi Miettinen, Petra Mäkinen, and Marja-Leena Soininen for excellent technical assistance, and Arto Koistinen, MSci, for assisting in chemical analysis of deposits.
