Abstract
Calcium antagonists have been shown to be superior over other antihypertensive drugs to prevent stroke. Because this cannot be fully attributed to blood pressure lowering effects, other mechanisms seem to play a role. Previously we found in patients with subarachnoid hemorrhage that nimodipine enhances fibrinolytic activity. The purpose of this systematic review was to investigate the fibrinolytic effect of calcium antagonists in general, especially in patients with hypertension. We systematically studied the entire PUBMED and EMBASE database with the search terms ‘calcium antagonist’ combined with ‘fibrinolysis’, ‘(euglobulin) clot lysis time’ (ECLT), ‘tissue plasminogen activator’ (tPA), or ‘plasminogen activator inhibitor’ (PAI). Twenty-six prospective studies were identified and 22 manuscripts were included (802 investigated individuals). The results show that calcium antagonists significantly increase fibrinolysis as shown by a reduction of the ECLT standardized mean differences (SMD) −0.58 (95% confidence interval (CI) −1.05 to −0.11)) and an increase of tPA activity (SMD 0.73 (95% CI 0.25 to 1.21)). This increase of fibrinolysis is apparently caused by an increase of the tPA antigen level (SMD 0.16 (95% CI −0.05 to 0.37)) and a decrease of the plasminogen activator inhibitor-1 antigen antigen (SMD −0.36 (95% CI −0.74 to 0.02)). A sensitivity analysis showed that dihydropyridines, but not phenylalkylamines, exert a fibrinolytic effect. This fibrinolytic effect is not only seen in patients with subarachnoid hemorrhage but also in hypertensive patients. In conclusions, calcium antagonists increase fibrinolytic activity. This may add to the beneficial pharmacological effect of calcium antagonists to prevent ischemic events in patients with hypertension and subarachnoid hemorrhage.
Introduction
In the treatment of hypertension, calcium antagonists have been shown to be superior over other antihypertensive drugs to prevent stroke (Angeli et al, 2004; Turnbull 2003; Dahlof et al, 2005). Because the superiority of calcium antagonists cannot be fully attributed to its blood pressure lowering effect, other yet unidentified mechanisms seem to play a role (Turnbull 2003; Dahlof et al, 2005; Poulter et al, 2005).
In the early eighties of last century, the dihydropyridine calcium antagonist nimodipine was introduced in patients with aneurysmal subarachnoid hemorrhage (SAH) to prevent cerebral ischemia. Cerebral ischemia is seen in 30% of the patients with SAH and is the second most important cause of poor outcome after SAH (Roos, 2000). To prevent cerebral ischemia after the hemorrhage, patients with SAH are treated with nimodipine. For many years, the beneficial effect of nimodipine was attributed to an effect on vasospasm, which was considered to be the cause of cerebral ischemia after SAH. However, from a meta-analysis investigating the effect of calcium antagonists in patients with SAH, it could be learned that despite a statistically significant effect on cerebral ischemia and poor outcome, there was no clear effect on angiographically demonstrable vasospasm (Feigin et al, 1998). In a previous study, we showed that the calcium antagonist nimodipine increases fibrinolytic activity in patients with SAH and hypothesized that the beneficial effect of nimodipine on cerebral ischemia is caused by enhanced fibrinolysis (Roos et al, 2001). Because increased coagulation after SAH may lead to the formation of microthrombi and thus result in secondary cerebral ischemia and ischemic stroke, increased fibrinolytic activity after SAH may counteract the activation of the coagulation cascade (Suzuki et al, 1983; Romano et al, 2002; Nina et al, 2001). Indeed, it has been shown that infusion with the thrombolytic agent urokinase into the cisterna magna reduces secondary ischemia after SAH (Hamada et al, 2003).
To investigate whether the fibrinolytic effect of nimodipine in patients with SAH is a pharmacological property of calcium antagonists in general and whether the beneficial effect of calcium antagonists in patients with hypertension could be attributed to enhanced fibrinolysis, we decided to systematically study the available literature on the fibrinolytic activity of calcium antagonists.
Materials and methods
Selection Criteria
Types of studies, participants and intervention: All clinical trials and prospective cohort studies investigating the effect of calcium antagonists on fibrinolytic activity in blood in human subjects were included, regardless of disease, risk factor, or health state. All types of calcium antagonists, administered in any dose, by any route, and for any duration, were included. Excluded were animal and ex vivo studies.
Types of outcome measures: Fibrinolysis was evaluated by studying the effects of calcium antagonists on different fibrinolytic parameters. The fibrinolytic variables during treatment were compared with their pretreatment values.
The main investigated parameters were: (1) (euglobulin) clot lysis time (ECLT), a global indicator of the level of fibrinolytic activity and (2) tissue-type plasminogen activator activity (tPA ac), a measure of free tPA fibrinolytic activity.
To investigate the mechanism by which the ECLT and tPA activity were influenced, the following other fibrinolytic parameters were investigated: (1) tissue-type plasminogen activator antigen (tPA ag), which promotes the conversion of plasminogen to plasmin; (2) plasminogen activator inhibitor-1 antigen (PAI-1 ag), which inhibits tPA and thereby the conversion of plasminogen into plasmin, and (3) plasminogen activator inhibitor-1 activity (PAI-1 ac), a measure of free PAI-1 inhibitory activity.
Search Strategy for Identification of Studies
The search strategy implied electronic searching of the entire PUBMED and EMBASE database up to 2006 (week 12) with the search terms ‘calcium antagonist’ combined with ‘fibrinolysis’, ‘euglobulin clot lysis time’, ‘clot lysis time’, ‘tissue plasminogen activator’, or ‘plasminogen activator inhibitor’. Studies from the Anglo-Saxon, German, and French literature were included. Studies from the literature in other languages were only included when abstracts were written in English, German, or French. If data on fibrinolytic parameters were insufficiently described, authors were contacted for additional information. Manuscripts giving no data on fibrinolytic values were excluded from the review when the contacted authors supplied no further information.
Methods of the Review
One reviewer (MDIV) searched the literature. Manuscripts were combined when results from one study were repeatedly described. When it was unclear whether the degree of dispersity of given parameters were expressed in terms of standard deviations or standard errors and the authors did not respond to our questions, other tables in the same article or manuscripts from the same author on the same subject were checked for which type of measure of spread they had used previously. If other manuscripts were not available, we conservatively assumed the authors had used standard deviations. If only pretreatment values were available with paired delta scores, the measure of spread of the pretreatment values was also used for the values during treatment. In case no data on fibrinolytic values were reported but only figures were available, estimations were made from the figures after copying and enlarging the original figure. When fibrinolysis was measured in rest, during exercise and after exercise, we used the values measured in rest. In case these data were not available, we used the fibrinolytic values measured after exercise. When both free PAI-1 and total PAI-1 were measured, we only used total PAI-1 because in most manuscripts no correction was made for PAI-1 in the tPA/PAI-1 complex. In studies where fibrinolytic variables were measured both before and after 10 mins venous occlusion, only data before venous occlusion were used because in most manuscripts no description was given that fibrinolysis was measured after 10 mins of venous occlusion. When fibrinolysis was measured at different time intervals during the treatment period, fibrinolytic values were used from the end of the treatment period.
Main Analysis and Sensitivity Analyses
The main analysis involved all studies and all patients included. Sensitivity analyses were performed for:
(A) Group of calcium antagonists. Distinction was made between different groups of calcium antagonists; dihydropyridines, benzothiazepines, and phenylalkylamines.
(B) Risk factors and diseases. Distinction was made between the different groups of patients (for example, patients suffering from hypertension, SAH, etc.).
(C) Health state. A dichotomy was made for healthy individuals and patients suffering from hypertension or other diseases (‘non-healthy individuals’).
(D) Methodology. The methodological quality of the included studies was assessed by two authors (MDIV and YBWEMR). Although several clinical trials were randomized, no placebo-controlled studies were performed. In the randomized trials, only the treatment arm was analysed in which patients were treated with a calcium antagonist. Hence, fibrinolytic parameters were compared with baseline values as was done in the prospective cohort studies. The methodological quality of both the randomized and prospective cohort studies was assessed by means of a scale that was originally developed by a workgroup from the Dutch Cochrane Center and others (Offringa, 2003). We used a modification of this validated eight-point scale assessing (a) the definition of study groups (1 point for clearly describing the characteristics of the investigated groups (duration of the illness/risk factor before treatment started) and for having an inception cohort), (b) selection bias (1 point when selection bias can be ruled out), (c) exposure (1 point for clearly defining exposure), (d) outcome (1 point for clearly defining outcome and the criteria used for assessing outcome. Outcome has to be measured in a validated way), (e) masking (1 point for measuring outcome masked for exposure), (f) follow-up (1 point for adequate follow-up period), (g) selective loss-to-follow-up (1 point when all included patients were accounted for. In case it was unclear whether there were dropouts, no points were given, and (h) confounders/prognostic factors (1 point for mentioning which confounders are considered and how these confounders are taken into account in the design and analysis of the study. Or 1 point when all relevant prognostic factors are used in the analysis). For assessing the relation between the study quality and effects size, a dichotomy was made for ‘insufficient’ and ‘sufficient’ quality studies. Studies scoring 0 to 4 points were considered to be of ‘insufficient’ quality; studies scoring 5 to 8 points were considered to be of ‘sufficient’ quality.
Statistical Analysis
With the exception of two studies (De Luca et al, 2000; Pahor et al, 2002), none of the individual studies presented mean and standard deviation of intra-patient change scores (before and during treatment). Hence, most studies analyzed their observations as unpaired data. Therefore, we treated all data as if they had come from a parallel-group design. Effect size was expressed in standardized mean differences (SMD), which was defined as the difference between two means (pretreatment and during treatment) divided by the pooled standard deviation. Means, standards deviations, and standard errors were derived by calculation or extraction from the available data. Standard errors of the mean were converted to standard deviations as follows: standard deviation = √n multiplied by the standard error. Individual weighted effect sizes were pooled with the generic inverse variance method. In this method, 1/variance of the effect estimate was used as weights. In all analyses, we used random effects models because most studies showed considerable heterogeneity. Statistical uncertainty was expressed in 95% confidence intervals (CIs). All data were analysed in Review Manager 4.2 as supplied by the Cochrane Collaboration.
Results
The search resulted in 26 studies investigating the effect of calcium antagonists on fibrinolytic parameters (Gleerup and Winther 1989, 1991; Gleerup et al, 1991, 1995; Ahaneku et al, 2000a, b, c; Fogari et al, 2003, 2004; De Luca et al, 2000; Pahor et al, 2002; Sengeløv and Winther 1989; Raccah et al, 1994; Ranieri et al, 1996; Smith et al, 1997; Fujinishi et al, 1997; Held et al, 1997; Yin et al, 1998; Sakata et al, 1999; Candela et al, 2001; Chi et al, 2002; Turchetti et al, 2002; Mugellini et al, 2004; Ishikawa et al, 2005; Ruilope et al, 2005; Roos et al, 2001). Fifteen studies were randomized studies (De Luca et al, 2000; Pahor et al, 2002; Gleerup and Winther 1989, 1991; Held et al, 1997; Yin et al, 1998; Sakata et al, 1999; Fogari et al, 2003, 2004; Mugellini et al, 2004; Roos et al, 2001; Sengelov and Winther 1989; Ranieri et al, 1996; Fujinishi et al, 1997; Chi et al, 2002; Turchetti et al, 2002; Ishikawa et al, 2005), and the remaining were longitudinal cohort studies (Ahaneku et al, 2000a, b, c). One longitudinal cohort study and three randomized trials were excluded afterwards because of insufficient data presented (Raccah et al, 1994; Smith et al, 1997; Ishikawa et al, 2005; Ruilope et al, 2005).
An overview of the 22 included studies is described in Table 1. The fibrinolytic effect of calcium antagonists was most often studied in patients with hypertension (Gleerup et al, 1991, 1995; Gleerup and Winther 1991; Ranieri et al, 1996; Chi et al, 2002) (15 studies, 406 patients) (De Luca et al, 2000; Pahor et al, 2002; Gleerup and Winther 1989; Ahaneku et al, 2000a, b, c; Sakata et al, 1999; Fogari et al, 2003, 2004; Mugellini et al, 2004). Other investigations were performed in patients with angina pectoris (one study, 301 patients) (Held et al, 1997), SAH (one study, 27 patients) (Roos et al, 2001), preeclampsia (one study, 14 patients) (Yin et al, 1998), coronary artery disease (one study, 11 patients) (Fujinishi et al, 1997), congestive heart failure (one study, 10 patients) (Turchetti et al, 2002), and systemic sclerosis (one study, seven patients) (Candela et al, 2001). In two studies, the fibrinolytic effect of calcium antagonists was investigated in healthy individuals (26 healthy individuals) (Sengeløv and Winther 1989; Gleerup et al, 1991).
Characteristics of included studies
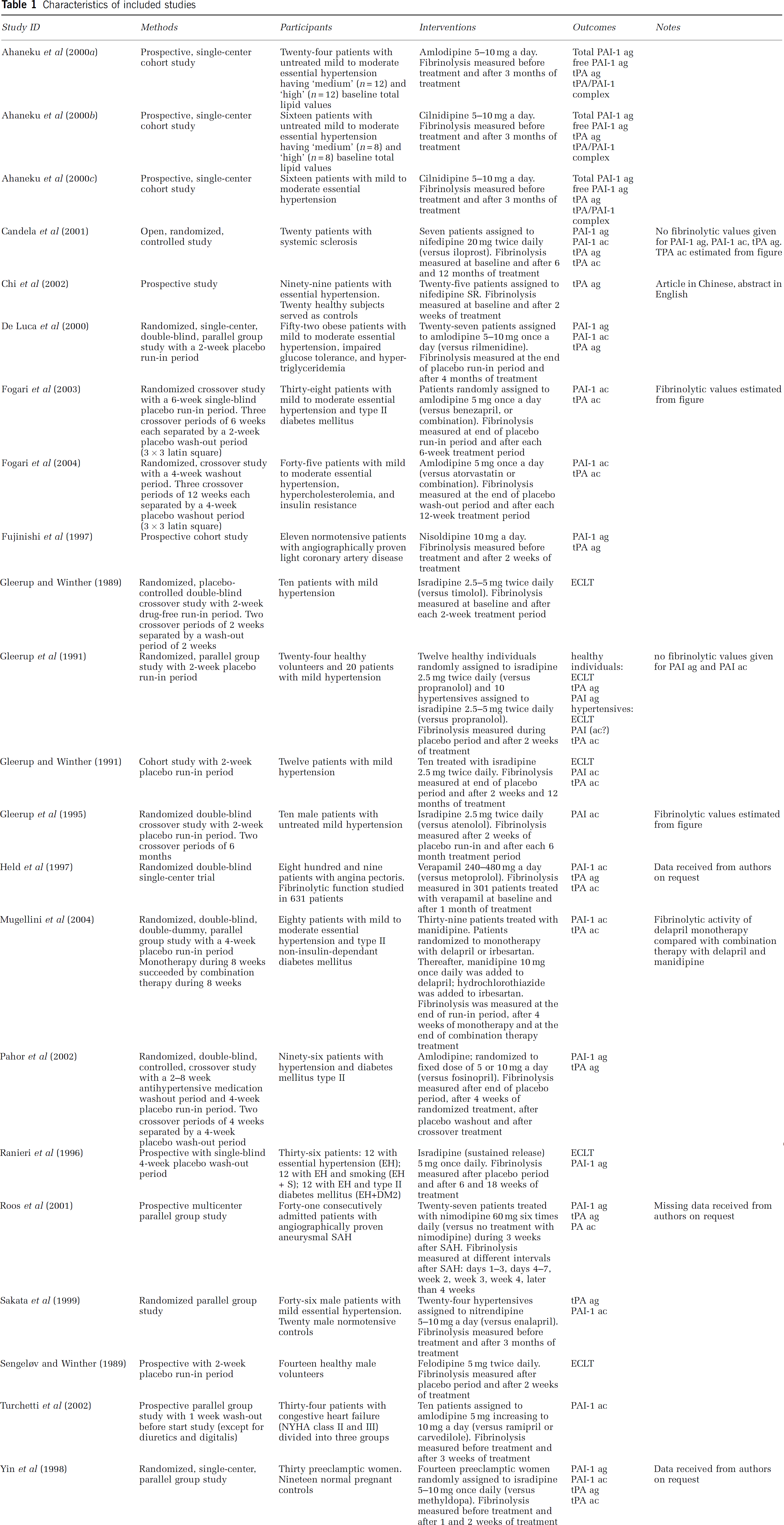
See appendix for abbreviations.
In 21 studies (501 patients), the following calcium antagonists from the dihydropyridine group were studied: isradipine (n = 6) (Gleerup and Winther 1989, 1991; Gleerup et al, 1991, 1995; Ranieri et al, 1996; Yin et al, 1998), amlodipine (n = 6) (Fogari et al, 2003, 2004; Turchetti et al, 2002; De Luca et al, 2000; Pahor et al, 2002; Ahaneku et al, 2000a), nifedipine (n = 2) (Candela et al, 2001; Chi et al, 2002), cilnidipine (n = 2) (Ahaneku et al, 2000b, c), nimodipine (n = 1) (Roos et al, 2001), felodipine (n = 1) (Sengeløv and Winther 1989), manidipine (n = 1) (Mugellini et al, 2004), nitrendipine (n = 1) (Sakata et al, 1999), and nisoldipine (n = 1) (Fujinishi et al, 1997). In one study (301 patients), the effect of the phenylalkylamine verapamil was investigated (Held et al, 1997). No studies were performed with calcium antagonists from the benzothiazepine group. The median treatment period was 5 weeks (range 2 to 52 weeks).
Three papers partially described the same data set (Gleerup and Winther 1989, 1991; Gleerup et al, 1991). We assumed that standard deviations were used in the manuscripts by Candela et al, 2001) and Chi et al, 2002) and standard errors in manuscripts by Pahor et al, 2002) and Gleerup and Winther 1989) and Gleerup et al, 1991). Fibrinolytic values were estimated from figures in three manuscripts (Gleerup et al, 1995; Candela et al, 2001; Fogari et al, 2003). In two studies, only pretreatment fibrinolytic values with delta scores were given with mean changes for individuals (De Luca et al, 2000; Pahor et al, 2002). Data from two studies were only partially used as data on some measured fibrinolytic values were incomplete (Gleerup et al, 1991; Candela et al, 2001). In one case, the original manuscript was written in Chinese and data from an abstract in English were used for the present analysis (Chi et al, 2002).
Main Analysis
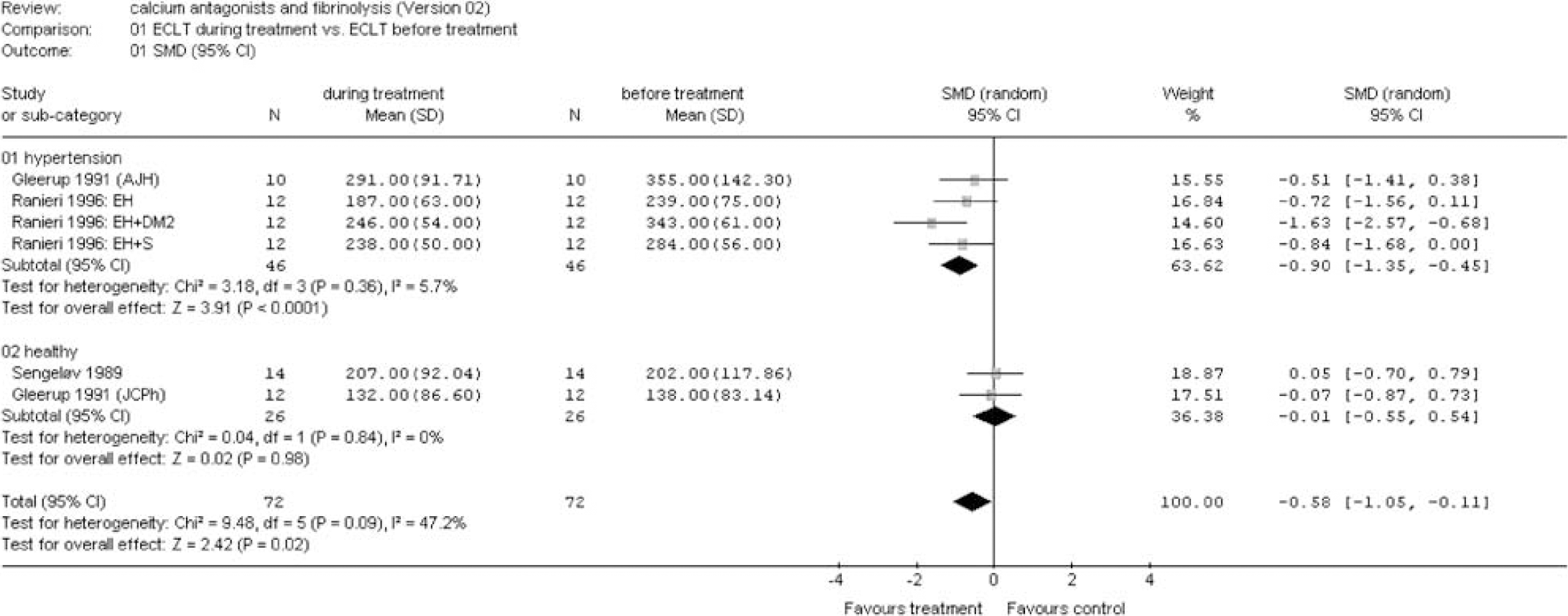
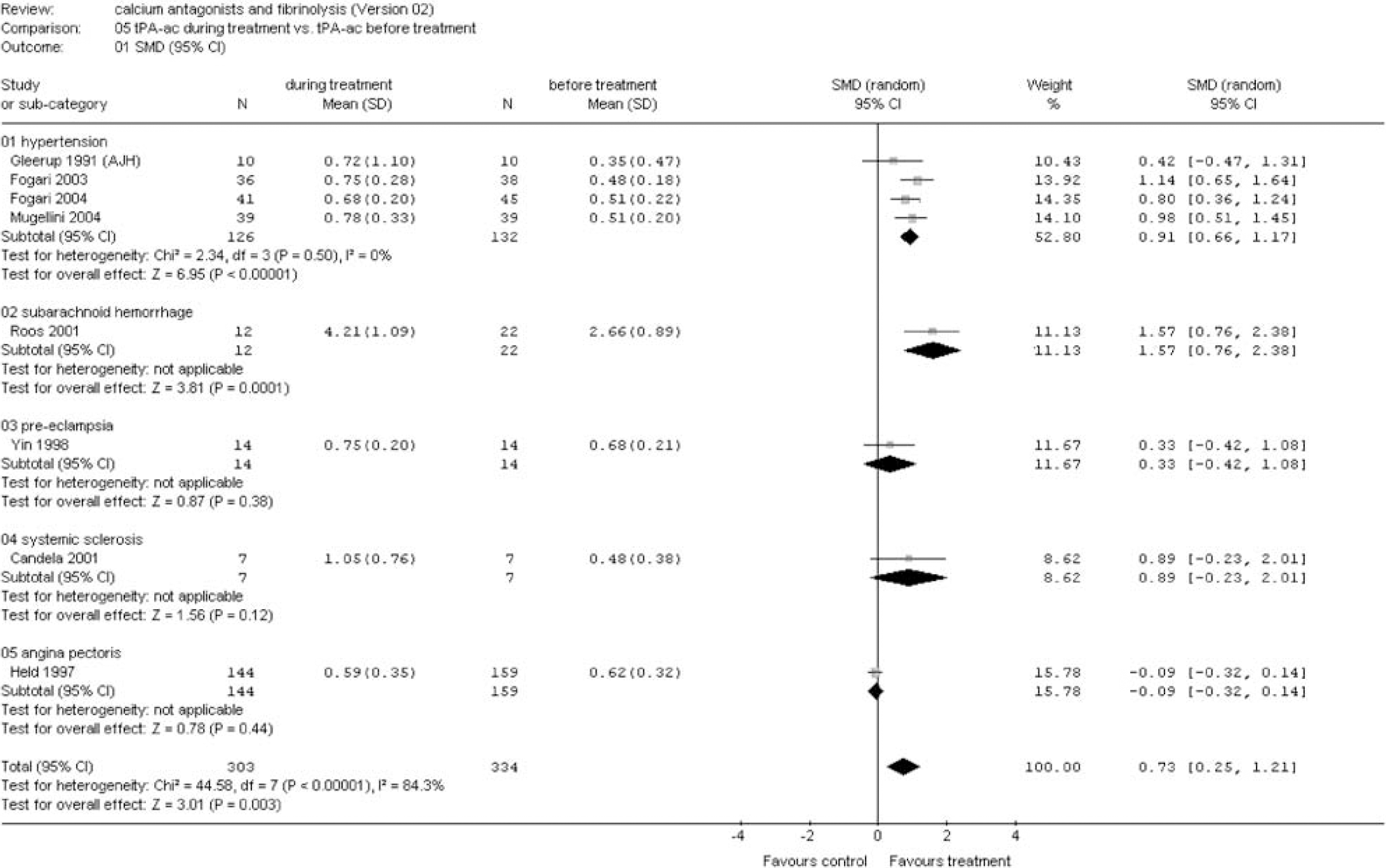
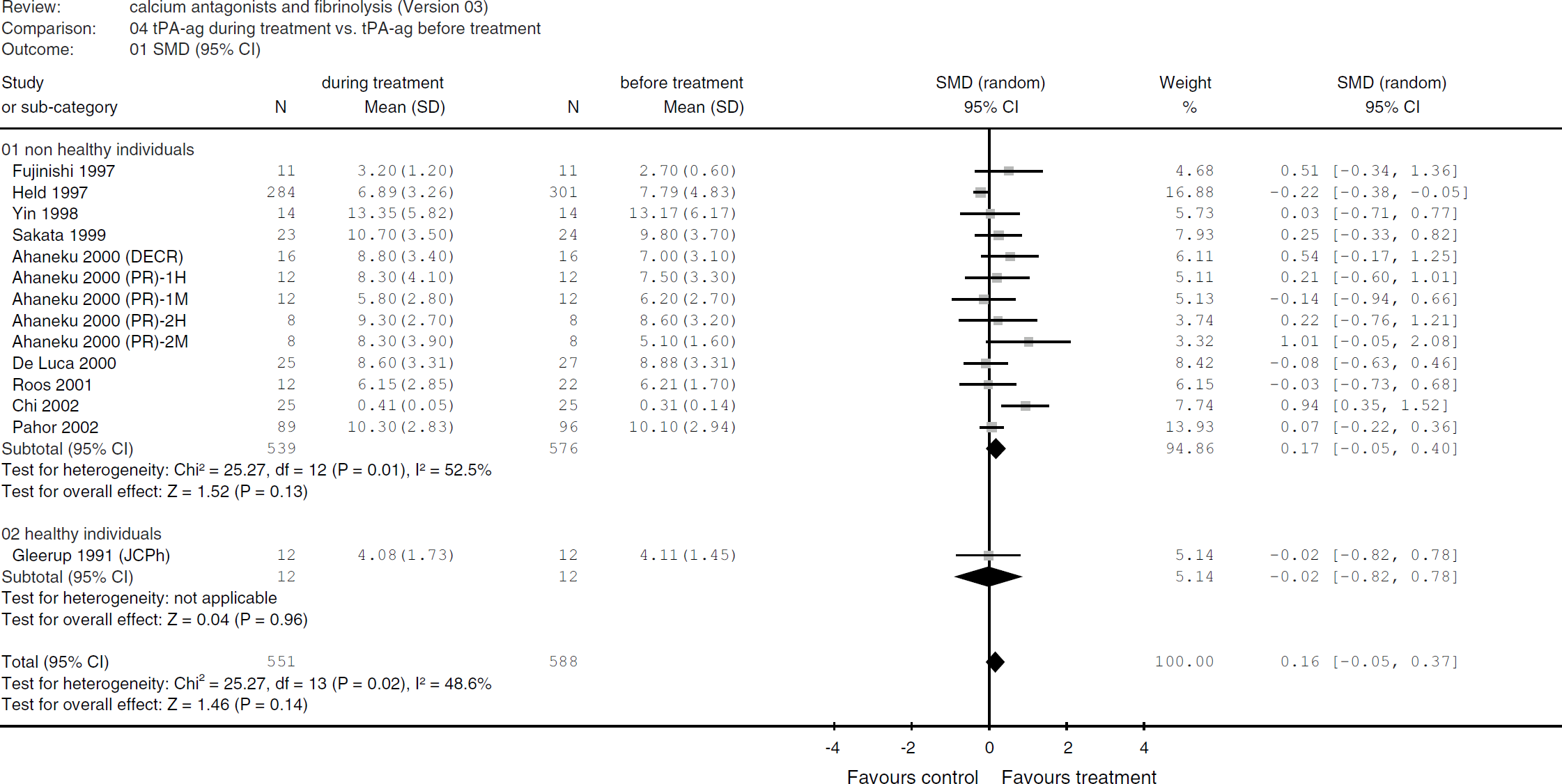
The main analysis was an aggregated analysis across different types of calcium antagonists and different patient populations. Calcium antagonists significantly increased fibrinolysis as shown by a reduction of the ECLT (SMD −0.58 (95% CI −1.05 to −0.11)) and an increase of tPA activity (SMD 0.73 (95% CI 0.25 to 1.21)) (Figures 1 and 2). This increase in fibrinolysis seemed to result from an increase in tPA antigen level (SMD 0.16 (95% CI −0.05 to 0.37)) and a decrease in PAI-1 antigen (SMD −0.36 (95% CI −0.74 to 0.02)) (Figures 3 and 4). No differences were observed for PAI-1 activity (Figure 5).
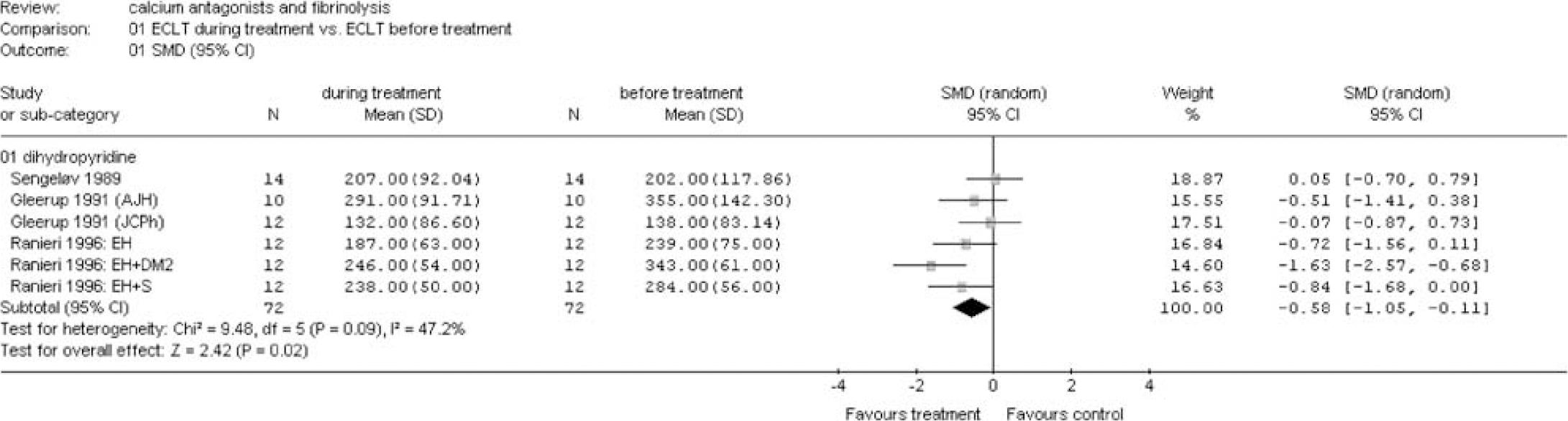
Euglobulin clot lysis time during treatment versus before treatment. Main analysis and sensitivity analysis for group of calcium antagonists (see appendix for abbreviations).
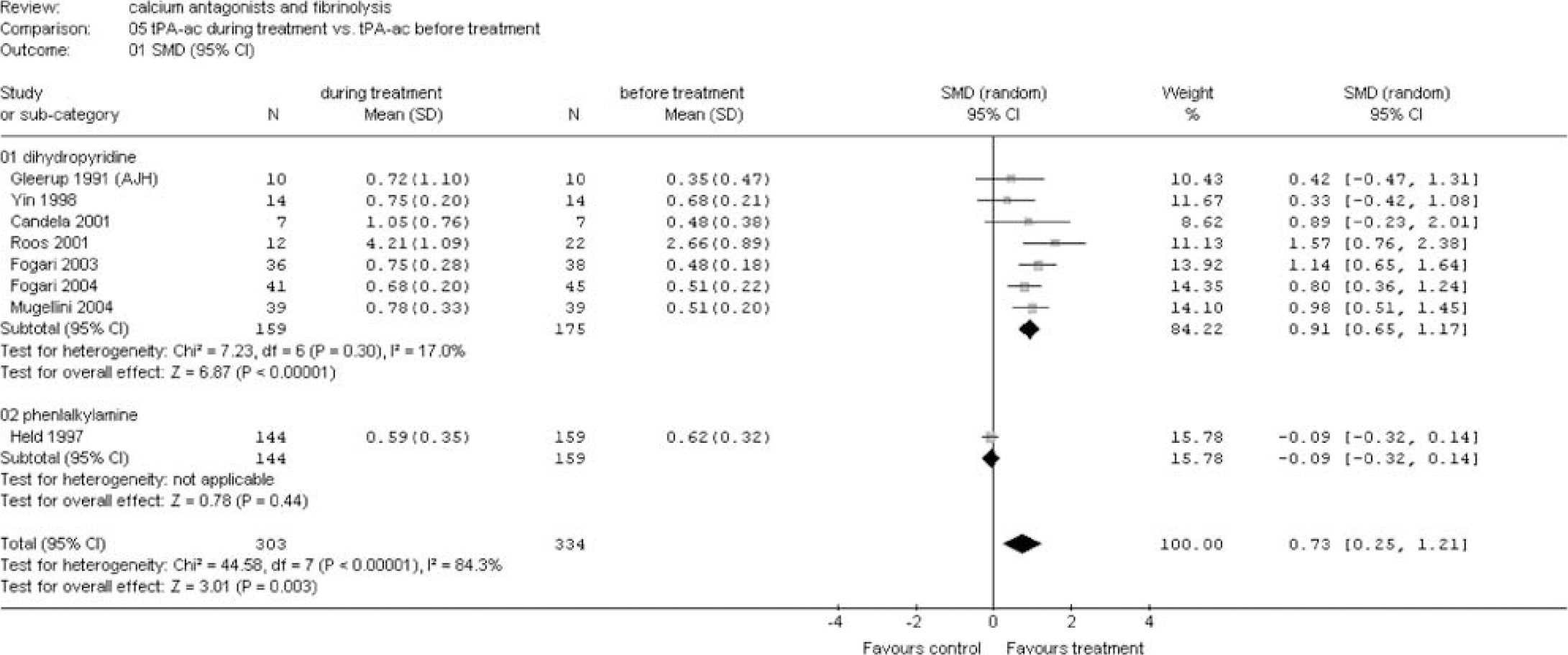
Tissue plasminogen activator activity during treatment versus before treatment. Main analysis and sensitivity analysis for group of calcium antagonists (see appendix for abbreviations).
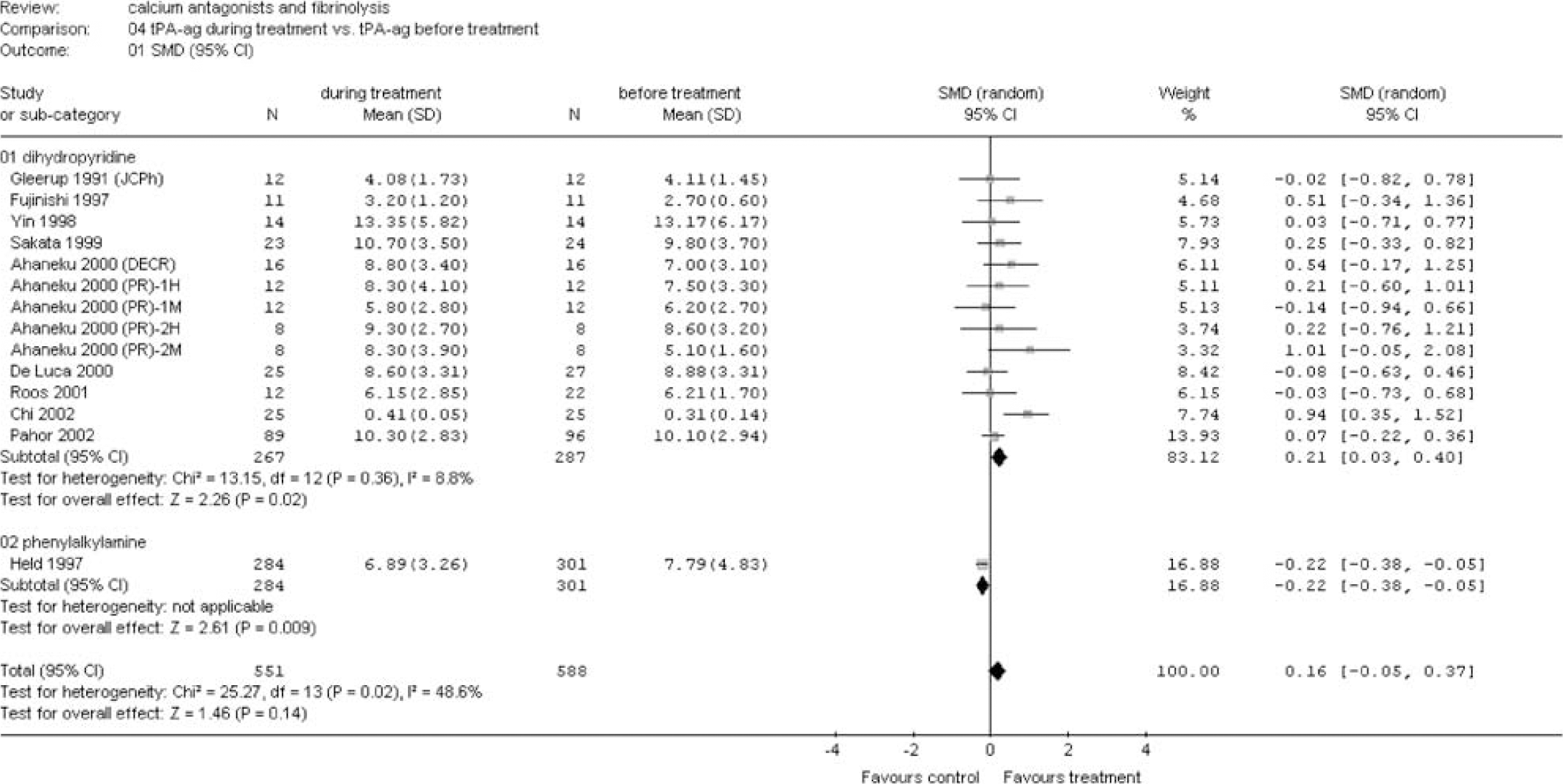
Tissue plasminogen activator antigen during treatment versus before treatment. Main analysis and sensitivity analysis for group of calcium antagonists (see appendix for abbreviations).
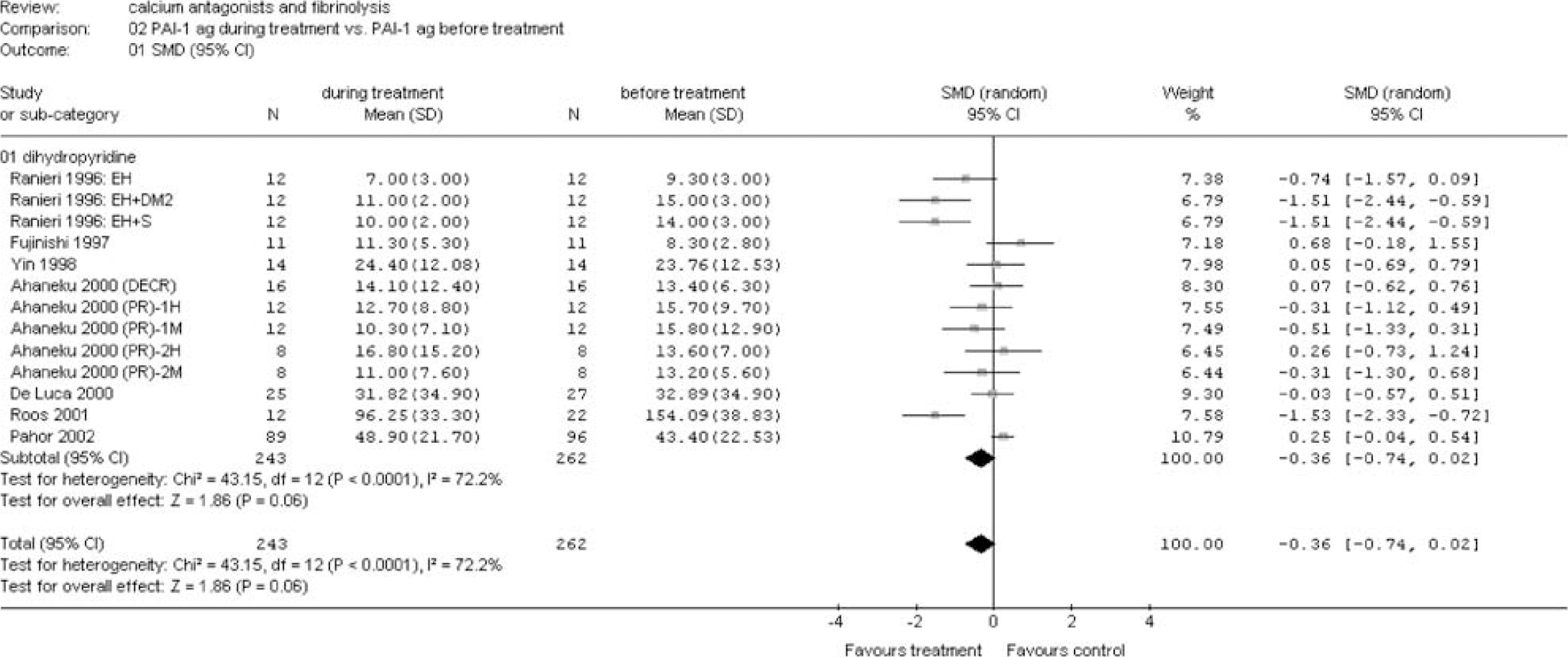
Plasminogen activator inhibitor (PAI) antigen during treatment versus before treatment. Main analysis and sensitivity analysis for group of calcium antagonists (see appendix for abbreviations).
Plasminogen activator inhibitor (PAI) activity during treatment versus before treatment. Main analysis and sensitivity analysis for group of calcium antagonists (see appendix for abbreviations).
Sensitivity Analyses
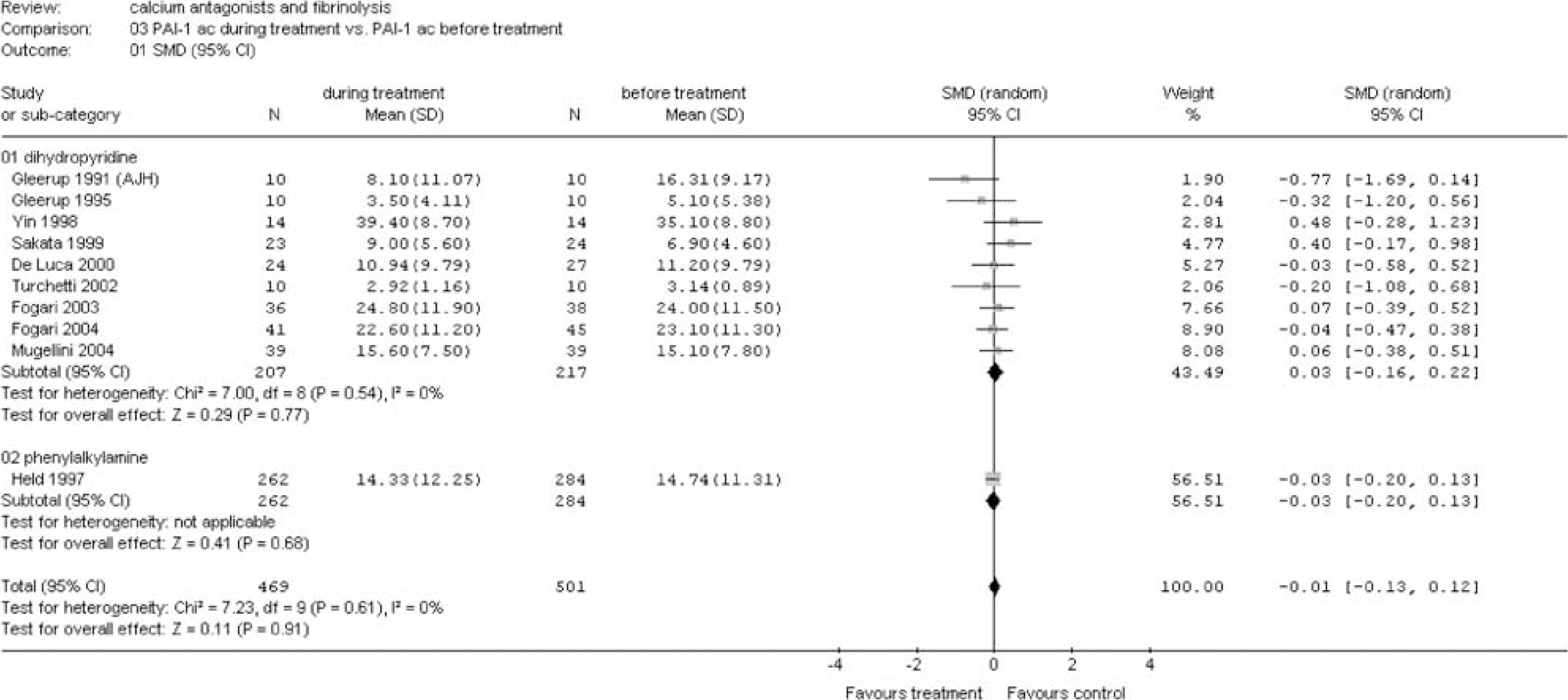
(A) Group of calcium antagonists: Calcium antagonists from the dihydropyridine group significantly increased fibrinolysis as shown by a reduction of the ECLT (SMD −0.58 (95% CI −1.05 to −0.11)) and an increase in tPA ac (SMD 0.91 (95% CI 0.65 to 1.17)) (Figures 1 and 2). This increased fibrinolytic activity was apparently the result of an increase in tPA ag level (SMD 0.21 (95% CI 0.03 to 0.40)) and a decrease in PAI-1 ag (SMD −0.36 (95% CI −0.74 to 0.02)) (Figures 3 and 4). No differences could be demonstrated for PAI-1 ac (Figure 5).
In one large-scale study investigating the effect of the phenylalkylamine verapamil, no significant effect was observed for tPA ac and PAI-1 ac (Figures 2 and 5). However, a significant reduction of tPA ag was found (SMD −0.22 (95% CI −0.38 to −0.05)) (Figure 3).
(B) Risk factors and diseases: A significant fibrinolytic effect was seen in patients with hypertension as measured by ECLT (SMD −0.90 (95% CI −1.35 to −0.45)), tPA ac (SMD 0.91 (95% CI 0.66 to 1.17)), and tPA ag (SMD 0.27 (95% CI 0.02 to 0.53)) (Figures 6, 7 and 8). A trend was found for PAI-1 ag (SMD −0.37 (95% CI −0.77 to 0.02)) (Figure 9). No effect was observed for PAI-1 ac (Figure 10). A strong fibrinolytic effect was also demonstrated in patients with aneurysmal SAH as measured by tPA ac (SMD 1.57 (95% CI 0.76 to 2.38)) and PAI-1 ag (SMD −1.53 (95% CI −2.33 to −0.72)) (Figures 7 and 9). In patients with angina, pectoris no fibrinolytic effect could be demonstrated as measured by tPA ac and PAI-1 ac (Figures 7 and 10). However, a significant reduction in tPA ag was seen (SMD −0.22 (95% CI −0.38 to −0.05)) (Figure 8). No effect was observed in the studies investigating the fibrinolytic activity of calcium antagonists in patients with preeclampsia, coronary artery disease, congestive heart failure, and systemic sclerosis (Figures 6–10).
Euglobulin clot lysis time during treatment versus before treatment. Sensitivity analysis for risk factors and diseases (see appendix for abbreviations).
Tissue plasminogen activator activity during treatment versus before treatment. Sensitivity analysis for risk factors and diseases (see appendix for abbreviations).
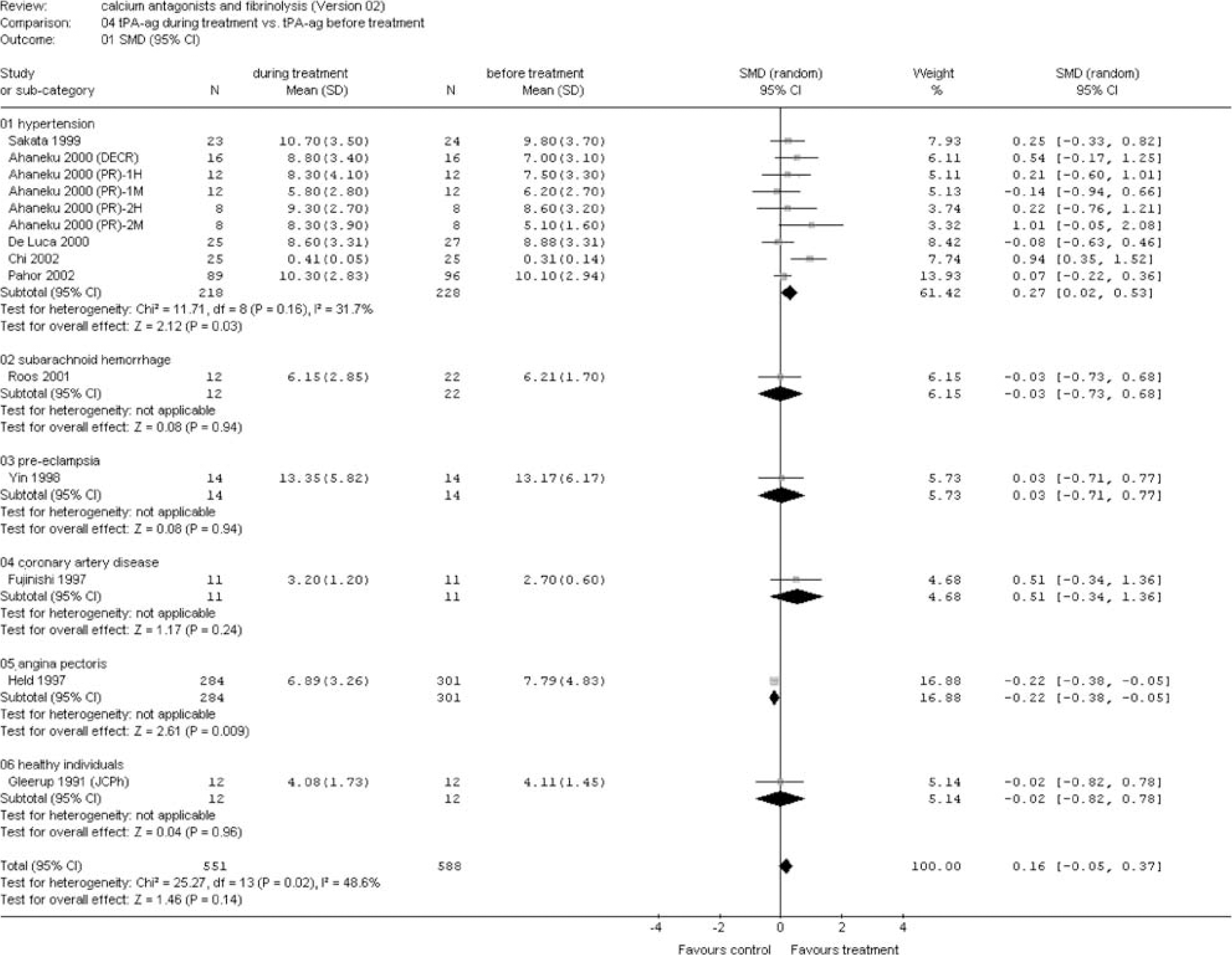
Tissue plasminogen activator antigen during treatment versus before treatment. Sensitivity analysis for risk factors and diseases (see appendix for abbreviations).
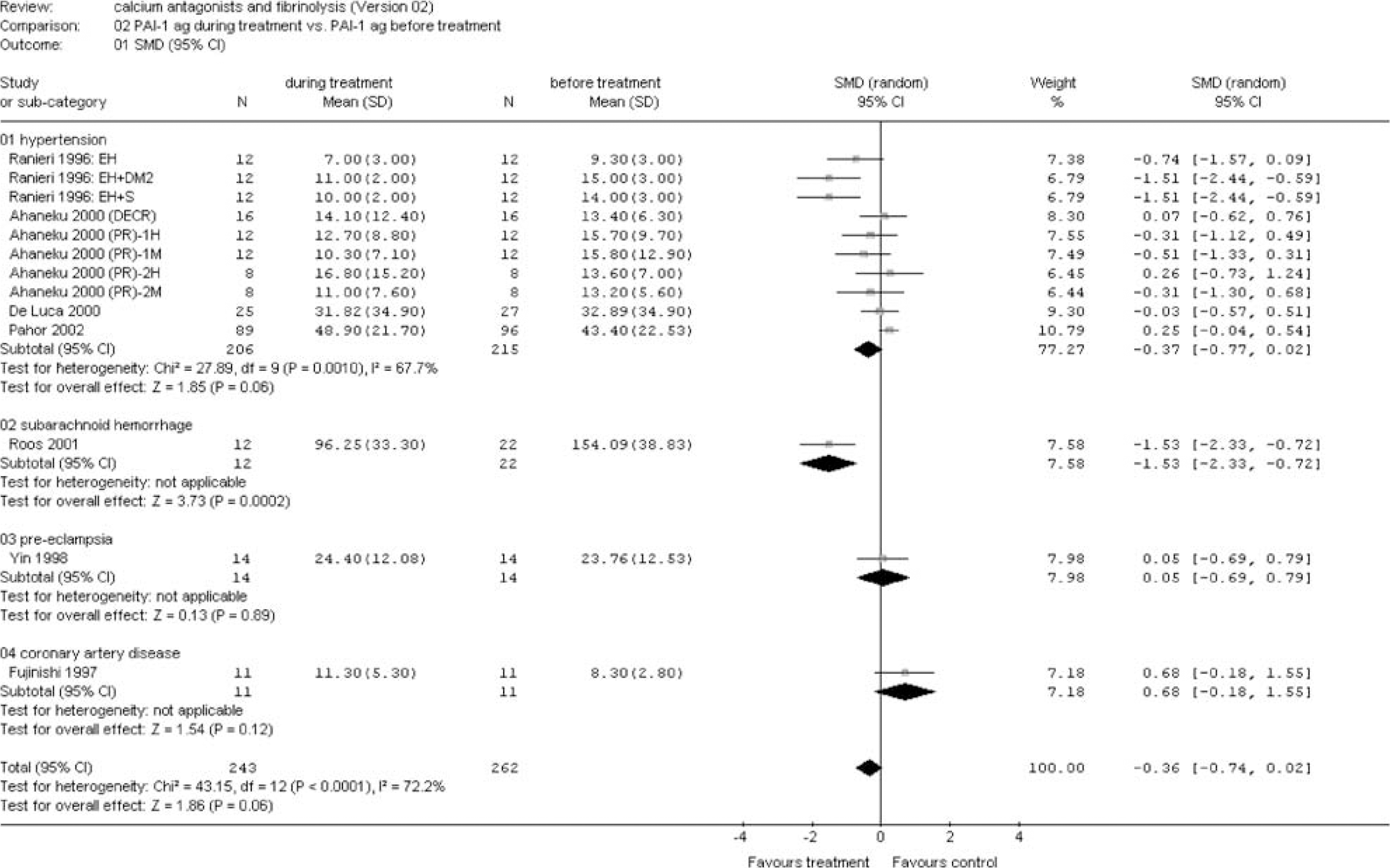
Plasminogen activator inhibitor (PAI) antigen during treatment versus before treatment. Sensitivity analysis for risk factors and diseases (see appendix for abbreviations).
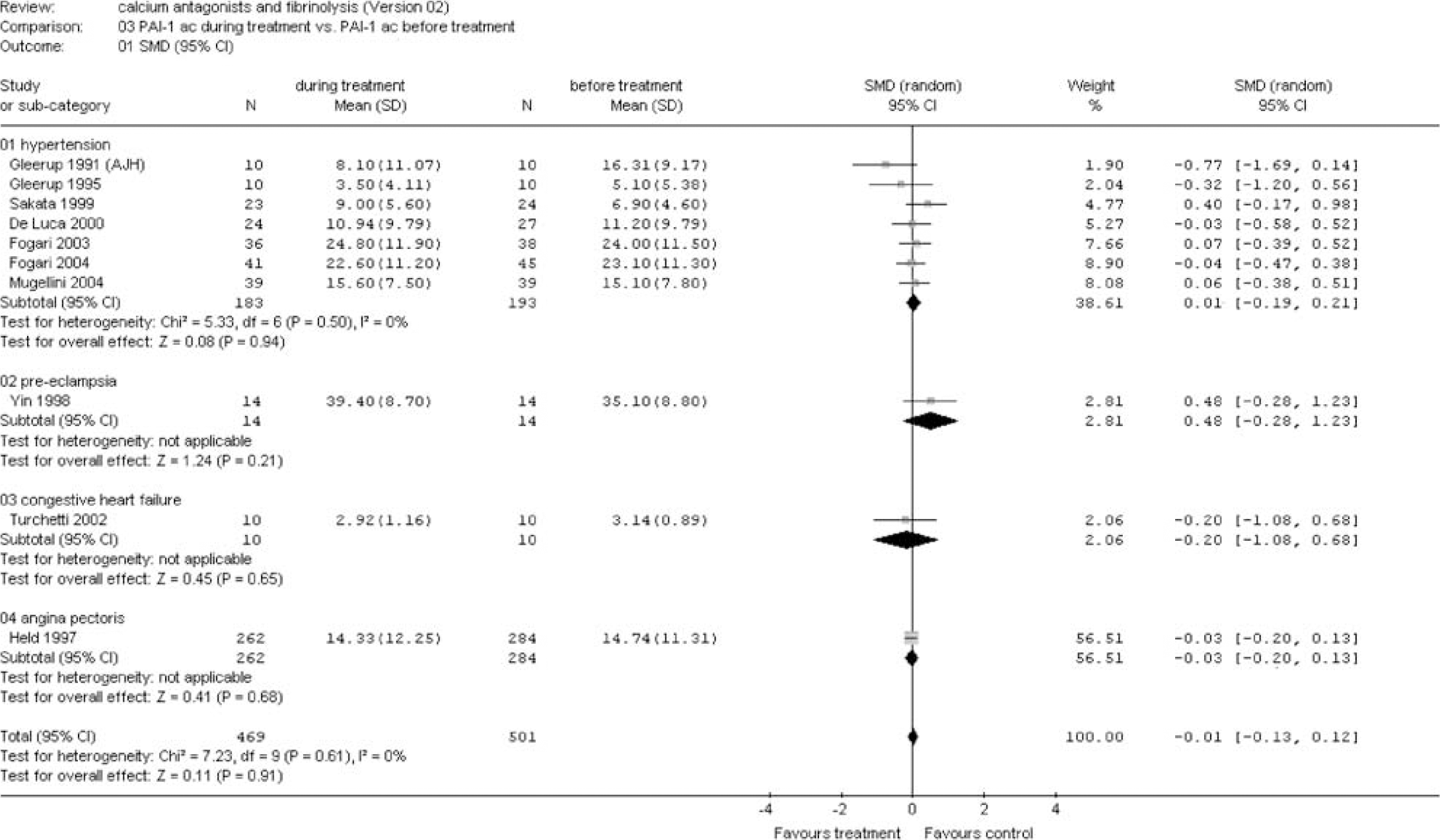
Plasminogen activator inhibitor (PAI) activity during treatment versus before treatment. Sensitivity analysis for risk factors and diseases (see appendix for abbreviations).
(C) Health state: In two studies, the effect of calcium antagonists in healthy individuals (n = 26) was investigated (Sengeløv and Winther 1989; Gleerup et al, 1991). In this group, no effect on fibrinolysis was observed, as measured by ECLT and tPA ag (Figures 6 and 8). In the group of non-healthy individuals, a significant reduction in ECLT was found (SMD −0.90 (95% CI −1.35 to −0.45)), but there was no increase in tPA ag (Figures 6 and 11).
Tissue plasminogen activator antigen during treatment versus before treatment. Sensitivity analysis for health state.
Comparisons between healthy and non-healthy individuals could not be made for the fibrinolytic parameters tPA activity, PAI-1 activity, and PAI-1 antigen as no such measurements were performed in healthy individuals.
(D) Methodology: The methodological quality of the included studies was sufficient in 16 studies (De Luca et al, 2000; Pahor et al, 2002; Gleerup and Winther 1989; Sengeløv and Winther 1989; Ahaneku et al, 2000a, b, c; Held et al, 1997; Yin et al, 1998; Sakata et al, 1999; Candela et al, 2001; Fogari et al, 2003, 2004; Turchetti et al, 2002; Mugellini et al, 2004; Roos et al, 2001), and insufficient in six studies (Gleerup et al, 1991, 1995; Gleerup and Winther 1991; Ranieri et al, 1996; Fujinishi et al, 1997; Chi et al, 2002). The median score was 5 (range 1 to 7). In studies with sufficient methodological quality, a significant increase of fibrinolytic activity was observed as measured by tPA activity (SMD 0.77 (95% CI 0.25 to 1.29)), but no effect could be demonstrated in terms of ECLT, PAI-ag, PAI-ac, and tPA ag (figures available on request). In studies with insufficient methodological quality, an increase of fibrinolytic activity was only reflected in ECLT (SMD −0.72 (95% CI −1.21 to −0.24)) (figures available on request).
Discussion
The reason to perform this systematic review was to investigate the effect of calcium antagonists on endogenous fibrinolysis. Our results show that calcium antagonists significantly increase endogenous fibrinolytic activity, as measured by ECLT and tPA activity. Further analysis shows that this increase in fibrinolysis is associated with an increase in tPA antigen and a decrease in PAI-1 antigen. In a sensitivity analysis, dihydropyridines, but not phenylalkylamines, increase fibrinolytic activity. Another sensitivity analysis showed a significant fibrinolytic effect in patients with hypertension and SAH.
The magnitude of fibrinolytic activity of calcium antagonists that was observed in this systematic review is only modest compared with the fibrinolytic activity exerted by well-known fibrinolytics such as urokinase and recombinant tissue plasminogen activator (rtPA). Our results show that dihydropyridine calcium antagonists are not fibrinolytic in healthy individuals. Probably, the magnitude of fibrinolytic activity of dihydropyridines is limited to an improvement of impaired fibrinolysis as is present in patients with hypertension and aneurysmal SAH. By improving the impaired fibrinolytic system, treatment with dihydropyridine calcium antagonists may lead to a decreased incidence of ischemic stroke. The results imply that calcium antagonists may be superior to antihypertensive drugs without fibrinolytic properties in other groups of patients with impaired fibrinolysis, such as in smokers and in patients with obesity or insulin resistance (Newby et al, 2001; Simpson et al, 1997; Rosito et al, 2004; Stegenga et al, 2006).
The enhancement of fibrinolytic activity by calcium antagonists may theoretically also increase the risk of bleeding. Indeed, an increased incidence in systemic hemorrhagic events as a side effect of calcium channel blockers has been reported previously (Gore et al, 1991; Wagenknecht et al, 1995; Pahor et al, 1996). In a study reporting on patients who underwent systemic thrombolysis for myocardial infarction receiving calcium antagonists, an increased incidence of intracerebral hemorrhage was observed (Gore et al, 1991). A randomized placebo-controlled trial, investigating a possible neuroprotective effect of nimodipine in patients undergoing cardiac valve replacement, was preliminarily stopped after an interim analysis because of the occurrence of major surgical bleeding and increased mortality in the group treated with nimodipine (Wagenknecht et al, 1995). In a prospective cohort study, a significantly higher risk of gastrointestinal hemorrhage was found in patients treated with a calcium antagonist, irrespective of the type of calcium antagonist (Pahor et al, 1996). In other randomized placebo-controlled trials, the results were inconclusive: in some trials, an increased incidence of bleeding events was reported, whereas in others no significant relation could be demonstrated (Black et al, 2003; The ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group 2002; National Intervention Cooperative Study in Elderly Hypertensives Study Group 1999; Staessen et al, 1997). The results of the present analysis may also implicate that patients with SAH treated with nimodipine may have an increased incidence of rebleeding. In contrast, however, a Cochrane analysis studying the effect of calcium antagonists in SAH showed a tendency towards a reduced incidence of rebleeding (Rinkel et al, 2005). The authors suggested that this might be a confounding effect as a result of protection against ischemia, which allows earlier intervention to secure the aneurysm by clipping or coiling, and consequently eliminate the risk of rebleeding. In 1998, a review on the effect of antihypertensive drug treatment on fibrinolytic activity included only two studies investigating the effect of calcium antagonists and was therefore inconclusive (Lottermoser et al, 1998).
It is unknown why in some of the above-described studies, an increased incidence of hemorrhagic events was observed, while in other studies this effect was not seen. Because none of the manuscripts that was included in the present systematic review made notice of an increased incidence of systemic hemorrhages, it was not possible to investigate the relation between tPA and PAI-1 levels in plasma to the risk of hemorrhage. It could be that calcium antagonists only lead to hemorrhagic complications when they interact with other medications. As described previously, in one study, an increased incidence of hemorrhage was observed in patients with myocardial infarction receiving systemic thrombolysis and calcium antagonists. In another study, an increased rebleeding incidence was observed in patients receiving cardiac valve replacement and nimodipine therapy. As patients with cardiac valve replacement are mostly heparinized, the results of these latter two studies suggest that calcium antagonists only lead to hemorrhagic complications when combined with anticoagulation or fibrinolytic therapy. Indeed, in an animal model, increased bleeding was observed in rabbits receiving the combination of rtPA and the calcium antagonist diltiazem daily for 3 days, compared with rabbits receiving rtPA or diltiazem alone (Becker et al, 1993). Future studies have to elucidate whether anticoagulation or fibrinolytics are contra-indicated in patients receiving calcium antagonists and whether calcium antagonists are contra-indicated in patients with a bleeding tendency, such as in patients with hepatic dysfunction.
Presently, not much is known on the mechanism by which calcium antagonists increase fibrinolytic activity. Compared with healthy individuals, hypertensive patients have impaired fibrinolytic activity because of impaired function of the endothelium (Tomiyama et al, 1998; Jastrzêbska et al, 2003; Poli et al, 2000). Because PAI-1 is mainly produced by the endothelium and tPA is released by the vascular endothelium, endothelial dysfunction results in changes in serum levels of tPA and PAI-1 antigen. Because endothelial dysfunction is not only associated with hypertension but also with thrombosis, the fibrinolytic effect of dihydropyridine calcium antagonists is probably exerted by direct effects on the endothelium (Rubanyi, 1993). By lowering blood pressure, endothelial function improves. However, it is not likely that the blood pressure lowering effect of antihypertensive agents per se is responsible for the pro-fibrinolytic effect. Another explanation for the fibrinolytic effect of calcium antagonists may be an increased secretion of tPA from the endothelium as it has previously been shown that the major determinant of tPA availability in the vascular bed of an organ is the capacity of the endothelium to increase tPA release when required (Hrafnkelsdottir et al, 2004). It is unknown whether the L-type calcium channel, which is blocked by calcium antagonists causing vasorelaxation and a decreased blood pressure, is involved in the fibrinolytic effect of calcium antagonists. In case the fibrinolytic effect of calcium channel blockers results from a direct effect on the endothelium, the L-type calcium channels are not involved as the macrovascular endothelium does not express L-type calcium channels. However, as PAI-1 is not only expressed in platelets, hepatocytes, and monocytes but also in smooth muscle cells, the role of the L-type calcium channel cannot be excluded. Apart from effects on the endothelium, other factors probably play a role as well. One of these factors may be activation of platelets which results in secretion of large amounts of PAI-1, a calcium-dependant process (Brogren et al, 2004; Lang and Schleef 1996). Presumably, if calcium influx is inhibited, the secretion of PAI-1 will decrease and as a consequence fibrinolytic activity may increase. Other sources of PAI-1 include the liver and adipose tissue, so these sites may also be involved in the fibrinolytic effect of calcium antagonists.
Calcium antagonists are often considered to be one separate group. However, because of differences in pharmacological properties, a distinction can be made between dihydropyridines, phenylalkylamines, and benzothiazepines. Dihydropyridines and phenylalkylamines for example differ in selectivity for the alpha-1 subunits of the L-type calcium channel (Morel et al, 1998). Such differences in pharmacodynamics probably result in different effects of dihydropyridines and phenylalkylamines on fibrinolytic function. Indeed, in a meta-analysis, it was shown that only dihydropyridine calcium antagonists and not non-dihydropyridine calcium channel blockers are superior to other antihypertensive drugs (Angeli et al, 2004).
We do realize that this systematic review has some limitations. Four studies, demonstrating no significant differences in fibrinolytic activity, could not be included as the measured values were not available (Raccah et al, 1994; Smith et al, 1997; Ishikawa et al, 2005; Ruilope et al, 2005). Similarly, fibrinolytic values from other studies could only partially be used (Gleerup et al, 1991; Candela et al, 2001). Not surprisingly, considerable heterogeneity was observed among studies as a result of (a) various patient populations, (b) different types of calcium antagonists, and (c) multiple techniques and a variable specificity of several ELISA tests to study the fibrinolytic parameters (Declerck et al, 1993). However, homogeneity was observed in our main sensitivity analyses for type of calcium antagonist and in patients suffering from hypertension. In these groups with homogeneity among studies, a significant fibrinolytic effect of calcium antagonists was observed. Although in several analyses, the effects of PAI-1 activity and PAI-1 antigen seem not to be concordant, these differences can be explained by the fact that the antigen assay measures both active PAI-1 as the inactive form of PAI-1 in the tPA/PAI-1 complex.
In agreement with the results of our previous study in which we showed that nimodipine increases fibrinolytic activity in patients with SAH, this systematic review shows that an increased fibrinolytic activity is also present in other calcium antagonists from the dihydropyridine group and in patients with other disorders such as hypertension. This fibrinolytic effect may add to the beneficial pharmacological effect of calcium antagonists to prevent ischemic events in patients with hypertension and SAH.
Appendix
The abbreviations used are: ac, activity; ag, antigen; AJH, American Journal of Hypertension; CI, confidence interval; DECR, Drugs under Experimental and Clinical Research; DM, diabetes mellitus; ECLT, euglobulin clot lysis time; EH, essential hypertension; JCPh, Journal of Cardiovascular Pharmacology; NYHA, New York Heart Association; PAI-1, plasminogen activator inhibitor-1; PR-1H, Pharmacological Research 2000;41:75–79. High baseline lipid values; PR-1M, Pharmacological Research 2000;41:75–79. Medium baseline lipid values; PR-2H, Pharmacological Research 2000;41:81–84. High baseline lipid values; PR-2M, Pharmacological Research 2000;41:81–84. Medium baseline lipid values; S, smoking; SAH, subarachnoid hemorrhage; SMD, standardized mean difference; SR, slow release; tPA, tissue plasminogen activator.
