Abstract
During intense cerebral activation approximately half of the glucose plus lactate taken up by the human brain is not oxidized and could replenish glycogen deposits, but the human brain glycogen concentration is unknown. In patients with temporal lobe epilepsy, undergoing curative surgery, brain biopsies were obtained from pathologic hippocampus (n = 19) and from apparently ʻnormal' cortical grey and white matter. We determined the in vivo brain glycogen level and the activity of glycogen phosphorylase and synthase. Regional differences in glycogen concentration were examined similarly in healthy pigs (n = 5). In the patients, the glycogen concentration in ʻnormal' grey and white matter was 5 to 6 mmol/L, but much higher in the hippocampus, 13.1 ± 4.3 mmol/L (mean ± s.d.; P < 0.001); the activities of glycogen phosphorylase and synthase displayed the same pattern. In normal hippocampus from pigs, glycogen was similarly higher than in grey and white matter. Consequently, in human grey and white matter and, particularly, in the hippocampus of patients with temporal lope epilepsy, glycogen constitutes a large, active energy reserve, which may be of importance for energy provision during sustained synaptic activity as epileptic seizures.
Introduction
Activation of the brain resets the uptake stoichiometry between oxygen and glucose to values lower than the resting value of ~6:1 (Fox et al, 1988; Madsen et al, 1999). Thus, when the brain is intensely stimulated by exhaustive exercise, and especially if the lactate taken up by the brain is included as an energy source, up to 50% of the carbohydrate taken up by the brain as a whole appears not to be oxidized (Dalsgaard, 2006). Only a minor fraction of this cerebral ʻsurplus' uptake of carbohydrate relative to oxygen can be explained by accumulation of amino acids as glutamate and glutamine, by intermediates in metabolic pathways and by accumulation of lactate (Dalsgaard, 2006; Madsen et al, 1999).
The enhanced need for fast-accessible glucose on cerebral activation is also signified by a decreasing brain glycogen level (Dienel et al, 2002; Madsen et al, 1999) and this would be expected to require an enhanced cerebral oxygen uptake. However, in as much as the oxygen taken up is insufficient to oxidize even the substrate delivered by blood, glycogen breakdown adds to the pool of carbohydrate, the fate of which remains an enigma.
Nevertheless, since the cerebral ʻsurplus' uptake of carbohydrate neither seems to be oxidized, released to the blood, nor accumulated within the brain, it is presumably stored (Dalsgaard, 2006). Glycogen constitutes the brains' largest carbohydrate reserve with concentrations in the rat most likely ranging from 3 to 12 μmol/g depending on the extraction method and handling of the animal (Cruz and Dienel, 2002; Madsen et al, 1999; Siesjö, 1978). Despite the recently introduced and promising assessment of human brain glycogen with 13C NMR (Oz et al, 2003) and few surgical biopsies from patients with different pathology, the concentration in humans is largely unknown. Glycogen appears to be broken down even after the brain stimulus has ceased (Madsen et al, 1999); hence, its replenishment cannot explain the decreasing stochiometry between oxygen and glucose. However, glycogen resynthesis is protracted compared with the duration of the stimulus (Dienel et al, 2002) and may, therefore, eventually establish a carbon balance over the brain when viewed over a time scale of hours. In response to intense cerebral activation, the brain may detain a ʻsurplus' of glucose equivalents of up to ~10 mmol (Dalsgaard, 2006), and we hypothesized that the human brain glycogen store is compatible with such amount.
Methods
The study was approved by the local ethical committee (KF 01-012/03). Written informed consent was obtained from 19 patients (age 37 ± 12 yeas; mean ± s.d.) with medically intractable temporal lobe epilepsy, who were scheduled for curative surgery, that is, hippocampus resection, for medical reasons only. Except for malignancy, there were no exclusion criteria. Anaesthesia was induced by bolus injections of intravenous thiopental (Pentothal, Hospira, Inc. Lake Forest, Illinois, USA) 7 mg/kg and phentanyl (Haldid, Janssen-Cilag, Birkeroed, Denmark) 5 μg/kg and maintained by Sevofluran (Sevoran Abbott, Gentofte, Denmark) 1% with an oxygen fraction of inspired air of minimum 40%. The hippocampal resection was performed according to standard procedures through a 10 mm corticotomy in the superior temporal sulcus, 2.5 cm behind the temporal pole after identification of pathologic spike activity directly on the hippocampus by in situ electroencephalographic recording. Biopsies weighing 15 to 100 mg of apparently ʻnormal' grey and white matter, as determined through the surgical microscope, were obtained in the circumference of the corticotomy, when a temporal lobe resection was necessary. The biopsies were obtained with a precooled cup rongeur (at 0°C) and immediately (within 5 to 10 secs) transferred to liquid nitrogen. Tissue that in situ exhibited signs of ischaemia, for example oedema or bleeding, was not included. All hippocampi removed during the surgical treatment were examined histologically to determine pathology.
To control for regional differences of normal brain, similar biopsies were obtained from five farm-raised young hogs. After 1 week of acclimatization in stables, the pigs were anaesthetized with bolus injections of intravenous phentanyl 0.2 mg and propofol (Abbott, Denmark) 2 mg/kg and supplemented with propofol 8 mg/kg/h. Care was taken that the craniotomy did not prematurely lesion the dura and that exposure of the hemispheres was sufficient to dissect through to the hippocampus. As in the patients, biopsies were obtained from cortical grey and white matter and from the (in this case, normal) hippocampus as subsequently verified by histology. Biopsies were obtained ~ 90 mins from induction of anaesthesia from both right and left hemisphere.
The effect of anaesthesia on the brain glycogen level was assessed in five awake rats as compared with five anaesthetized rats (female Wistar, weight 200 to 220 g). The brain samples were obtained by the Guillotine Freeze-clamping technique, in which a coronal cross-section of the brain is cut out and freeze-clamped within 30 msecs (Quistorff, 1980). Five of the rats were anaesthetized by pentobarbital intraperitoneal 50 mg/kg, 15 to 25 mins before biopsy, while the five controls were not.
From all experiments, the brain biopsies were analysed for ATP, phosphocreatine, and lactate by spectrophotometry on neutralized methanol—HCl—perchloric acid extracts (Quistorff, 1980). Glycogen was extracted on a separate part of the brain biopsy by boiling (100°C) this sample for 10 mins in 0.4 mol/L KOH. Subsequently, pH of the extract was adjusted to 5.0 by 1 mol/L citric acid. Amyloglucosidase was added (5.2 mg/mL), and the extract incubated for 2 h at 40°C. On this extract, glucose was assayed enzymatically (for further details, see Katz et al, 1976). The enzymatic activity of glycogen phosphorylase was measured radiochemically at 37°C, incubation time 1 h, in the direction of glycogen synthesis (Gilboe et al, 1972) and that of glycogen synthase as described by Golden et al (1977).
Values are presented as mean ± s.d. Results were compared by nonpaired and paired Student's t-test. A P-value < 0.05 was considered statistically significant.
Results
The human brain glycogen level was not significantly different between grey and white matter, but it was 2- to 2.5-fold higher in the hippocampus (Table 1; P < 0.001). Biopsies from these hippocampus on histologic examination were showing loss of pyramidal cells (primarily in CA1 and CA2), astrogliosis, and to a lesser degree microgliosis; one subject had a haemangioma and one expressed nondistinctive pathology.
Brain glycogen and energy metabolites
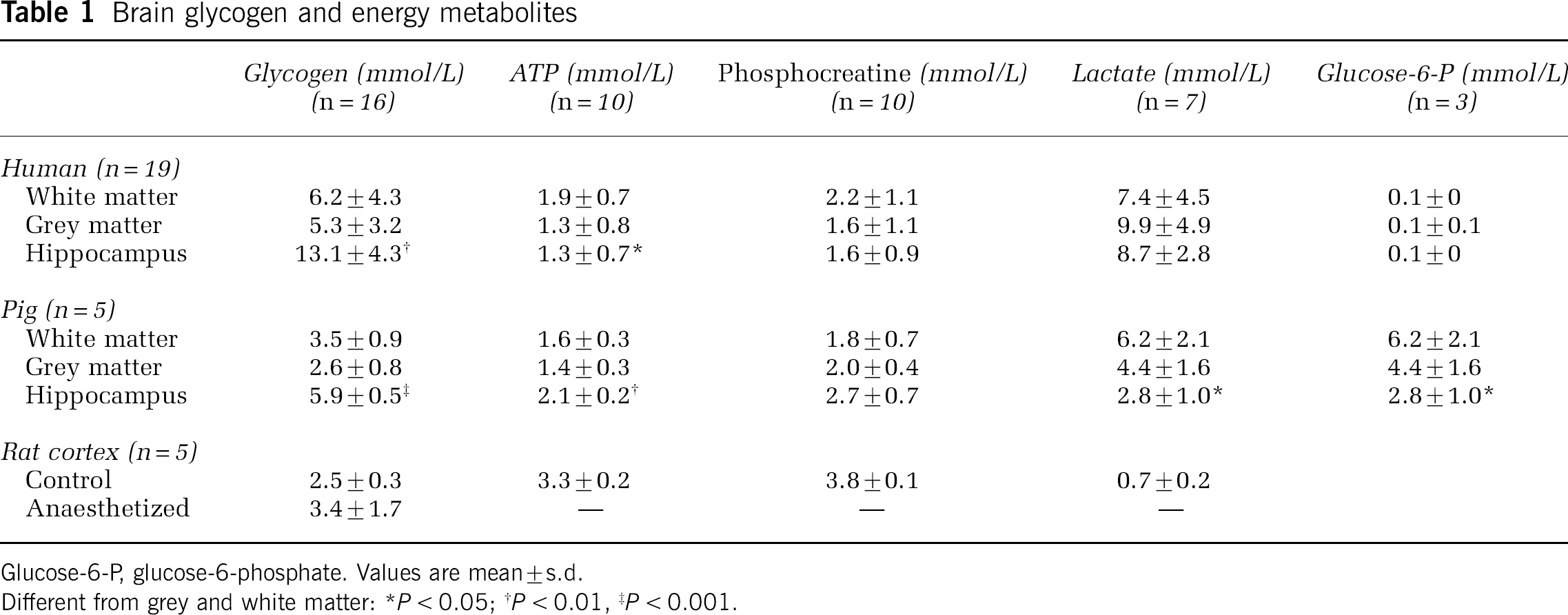
Glucose-6-P, glucose-6-phosphate. Values are mean ± s.d.
Different from grey and white matter: *P < 0.05; †P < 0.01, ‡P < 0.001.
In all biopsies from human and pig brain, the concentrations of ATP and phosphocreatine were somewhat decreased than the levels in optimally frozen rat brain and that of lactate was higher (Table 1). The activity of glycogen-related enzymes (Table 2) displayed approximately the same pattern of change as the glycogen concentration, that is, in the hippocampus, the activities of glycogen phosphorylase a-form and synthase a-form were greater than in white matter by 1.7- (P < 0.05) and 2.3-fold (P < 0.01), respectively, and compared with grey matter by 1.9- and 1.3-fold, respectively. The degree of enzyme activation in hippocampus was not significantly different from that in grey and white matter.
Enzyme activities in human brain
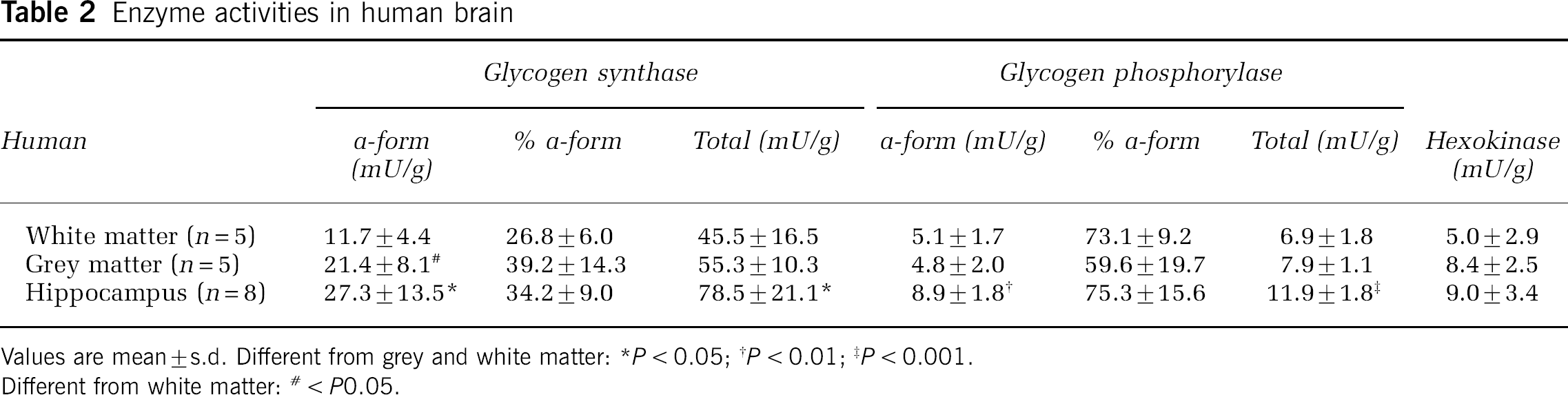
Values are mean ±s.d. Different from grey and white matter: *P < 0.05;†P < 0.01;‡P < 0.001.
Different from white matter: # < P0.05.
The study on normal pigs confirmed that hippocampus glycogen is higher than in grey and white matter. The glycogen concentration in the anaesthetized rats was ~35% larger than in the awake animals (P < 0.05).
None of the patients had seizures the day before surgery; within the last week the frequency of seizures was: five patients had no seizure, two had one, one had two, four had four, and the remaining seven patients experienced four to nine seizures. At follow up, approximately 1 year after surgery of the last patient, almost all patients had been cured.
Discussion
The main finding of this study is that the glycogen level in the hippocampus of these patients with temporal lope epilepsy was two to three times higher than in grey and white matter and that this was confirmed in normal pigs.
Glycogen is emerging as being integral to brain energy metabolism, but its specific role remains elusive (Brown, 2004). Except for glycogen, the brain depends on relatively low quantities of phosphocreatine for fast ATP production (Siesjö, 1978). In this context, glycogen would be of value especially within the astrocyte processes surrounding the synaptic clefts, which are devoid of mitochondria and, therefore, rely on anaerobic metabolism to fuel, at high rate, extracellular clearance of potassium and neurotransmitters; similar physical limitations may also apply to the most peripheral parts of the neuron. The present findings not only confirm the presence of glycogen in the human brain in vivo as demonstrated by nuclear magnetic resonance spectroscopy (<2 μmol/g; Oz et al, 2003) but also quantifies the in vivo glycogen level to match the highest in the rat of 5 to 12 μmol/g (Cruz and Dienel, 2002), that is, it is three- to four times higher than originally thought (Madsen et al, 1999; Siesjö, 1978). This underscores that within the human brain, glycogen constitutes an important energy reserve. Although we carefully minimized the time interval of ʻwarm ischaemia' by the use of ice-cooled tongs to but a few seconds, ischaemia-induced glycogen degradation may have occurred before metabolism was fully quenched on freezing. Consequently, the glycogen concentration could theoretically be higher. Since the lactate concentration in all samples from human brain was approximately 8 to 10 mmol/L (Table 1), the in vivo glycogen concentration could at most have been 4 to 5 mmol/L higher and correspondingly 2 to 3 mmol/L higher in the pig. From a functional perspective, however, given that glycogen is confined mainly to the astrocytes (Cataldo and Broadwell, 1986), its subcellular concentration may be as high as in skeletal muscle fibres, further augmenting its value as a ʻbuffer' for glycolytic substrate. Unfortunately, we were unable to preserve glycogen in the samples examined histologically despite the employment of gentle water extraction and different methods for colouring. Presumably, this is because glycogen granules in the cytoplasm are readily water soluble and/or because in these tissue blocks, glycogen was degraded before freezing. Thus, the actual cellular localization of glycogen could not be identified.
Conversely, an astrocytic preference for glycogen implies that in these patients with temporal lobe epilepsy, the very high glycogen concentration in the hippocampus compared with ʻnormal' grey and white matter could be a result of the pathologic condition with increased number of glial cells relative to neurons (gliosis) as revealed during histologic examination in this study. Notwithstanding, hippocampus glycogen in healthy pigs was comparably elevated which, therefore, seems to characterize this structure in line with its high synaptic density (Choi and Frahm, 1999).
While brain glycogen decreases during neuronal activity (Dienel et al, 2002; Madsen et al, 1999), it increases when neuronal activity is attenuated as in hibernation (Koizumi, 1974) and with denervation (Laatsch and Cowan, 1966). Thus, in these pathologic hippocampus, glycogen could have increased because of synaptic degeneration and slowing of energy turnover (Laatsch and Cowan, 1966), although higher activity of glycogen phosphorylase and synthase in hippocampus than in grey and white matter contradicts this notion. Alternatively, lowering of neuronal activity by the anaesthesia may have influenced the glycogen level as known to occur, particularly, with the depressant agent barbiturate (Koizumi, 1974; Phelps, 1972). Yet, in the present study, the difference between awake and barbiturate anaesthetized rats was only ~30% in line with observations by Phelps (1972). It should, however, be noted that sevofluran rather than barbiturate was the anaesthetic in our patients, and propofol was used in the pigs, but neither agent is known to inhibit glycogen-related enzymes. In fact, it seems that the halogenated anaesthetics used for patients cause a lesser degree of glycogen accumulation than barbiturate (Morgenthaler et al, 2006), that is a 30% increase because of anaesthesia is probably an overestimation.
After skeletal muscle contractions, the glycogen concentration may reach a level above that at rest, or in other words, ʻsupercompensate' (Bergström and Hultman, 1966), a phenomenon also demonstrated in the brain of rats after hypoglycaemia (Choi et al, 2003). Such glycogen supercompensation could occur also after neuronal activation and would not only prepare the neuronal region for a repetitive bout of intense activation but also eventually account for the mysterious, cerebral ʻsurplus' carbohydrate uptake. Neither of the patients exhibited seizures on the day before surgery but more than half had > 4 seizures during the last week, but whether this influenced the glycogen level remains obscure. Furthermore, the functional properties of glycogen metabolism may be illustrated in patients with medically intractable epilepsy, in whom fasting and glucose-restricted diets appear beneficial (Greene et al, 2003). In the rat such diets decrease brain glycogen and, given that astrocyte glycogen is crucial for intense axon potentials, we propose that glycogen is a prerequisite for sustained neuronal activity as in epileptic seizures.
