Abstract
Hemiparesis is the most common deficit after cerebral stroke. Constraint-induced movement therapy (CIMT) is a new neurorehabilitation method that emphasizes task-relevant repetitive training for the stroke hand. Twelve chronic stroke patients were studied with single-photon emission computerized tomography at rest before and after the two-week CIMT period. Increased perfusion was found in motor control related areas. The specific areas with an increase in perfusion in the affected hemisphere were in the precentral gyrus, premotor cortex (Brodmann's area 6 (BA6)), frontal cortex, and superior frontal gyrus (BA10). In the nonaffected hemisphere, perfusion was increased in the superior frontal gyrus (BA6) and cingulate gyrus (BA31). In the cerebellum increased perfusion was seen bilaterally. The brain areas with increased perfusion receive and integrate the information from different sensory systems and plan the movement execution. Regional cerebral perfusion decreased in the lingual gyrus (BA18) in the affected hemisphere. In the nonaffected frontal cortex, two areas with decreased perfusion were found in the middle frontal gyrus (BA8/10). Also, the fusiform gyrus (BA20) and inferior temporal gyrus (BA37) in the non-affected hemisphere showed decreased perfusion. Intensive movement therapy appears to change local cerebral perfusion in areas known to participate in movement planning and execution. These changes might be a sign of active reorganization processes after CIMT in the chronic state of stroke.
Keywords
Introduction
Recently, evidence has accumulated in primate stroke models indicating that structural and functional neural reorganization can occur after extensive training in the early period of rehabilitation after stroke (Nudo et al, 1996; Plautz et al, 2000). In humans, a follow-up study of stroke patients who spontaneously recovered well also suggested reorganization in the cortical activation related to voluntary hand movements (Chollet et al, 1991). During the first 3 months, spontaneous recovery is most active in the affected brain areas. Nevertheless, deficits after stroke are commonly considered chronic 6 months after the insult. Recent evidence suggests that years after the insult functionally meaningful recovery can be acquired (Taub and Uswatte, 2003). Therefore, structured intensive interventions might be justified to alleviate disability caused by cerebral infarction even years after the insult.
Constraint-induced movement therapy (CIMT) belongs to a new family of neurorehabilitation methods which emphasize task-relevant repetitive training. Constraint-induced movement therapy utilizes rigorous exercise of the affected arm and hand, while the movement of the healthy arm is simultaneously restrained with a sling. This method is based on the concept of learned non-use, which is mainly identified by discouragement of the use of the affected upper limb from early on by the nurses and physiotherapists in favor of the healthy upper limb, even if the healthy upper limb was nondominant before the cerebral incident. The reduction of learned non-use through CIMT has been well described first in connection with sensory deafferentation studies in monkeys and later in stroke patients (Taub, 1980; Miltner et al, 1999; Taub and Uswatte, 2003). The affected upper limb in humans with stroke may present with various symptoms, such as spasticity, clumsiness, intention tremor, decreased control of individual fingers, reduced grip force, and also flaccid paralysis. Positive therapeutic effects on paralyzed hand motor control at the behavioral level have been shown after a 2- to 3-week intervention program following CIMT principles (Miltner et al, 1999; Taub and Uswatte, 2003).
The longitudinal effect of CIMT on brain function has been studied with various methods: positron emission tomography (PET) (Nelles et al, 2001; Wittenberg et al, 2003), functional magnetic resonance imaging (fMRI) (Johansen-Berg et al, 2002; Jang et al, 2003; Kim et al, 2004), and transcranial magnetic stimulation (TMS) (Cohen et al, 1998; Liepert et al, 2000). The PET study by Wittenberg et al (2003) showed that before the therapy period patients had increased activation of primary motor cortex, supplementary motor cortex, and cerebellum compared with normal volunteers when movement of affected fingers was contrasted to rest. They also found that in the affected hemisphere motor cortical activation decreased after CIM therapy and that there was no increase in activation in any area. Nelles et al (2001) found that blood flow increased in the premotor, parietal, and primary motor cortex after therapy in a patient who had received arm training compared with control patients. Several fMRI studies have shown that cortical activation increases in the contralateral (affected) motor areas and decreases in the ipsilateral (unaffected) cortical areas after CIMT (Johansen-Berg et al, 2002; Jang et al, 2003; Kim et al, 2004). While PET and fMRI measure task-related perfusion changes, single-photon emission computerized tomography (SPECT) usually measures background perfusion in a single state.
The purpose of the present study was to investigate whether chronic stroke patients may benefit from the CIMT program and whether the program induces any changes in their cerebral perfusion at rest as measured by SPECT. The effects of the CIMT program were assessed with a structured motor behavior test and SPECT imaging was performed immediately before and after the intervention program. Our specific aim was to evaluate functional cortical reorganization in resting cerebral blood flow (CBF) in chronic stroke patients.
Materials and methods
Patients
Twelve consecutive chronic stroke patients were enrolled in the 2-week long CIMT rehabilitation program. Entry criteria for the patients were: (1) time since ischemic stroke more than 6 months, (2) the minimum voluntary control of the affected upper extremity in the wrist 10 degrees, and in the fingers 10 degrees (see Miltner et al, 1999), (3) no severe communication problems, and (4) no acute cardiovascular or other diseases preventing participation. Patients were 8 men and 4 women, mean age 48 years (description of the patients is shown in Table 1). The mean time since stroke was 36 months (range 7 to 132 months). Written informed consent was obtained from all patients before the study, and the protocol was approved by the local ethical committee.
Patient characteristics, including the time since stroke (months) and the location and extent of the major lesion in MR images, with the maximum lesion diameter in a representative transaxial MRI slice. Motor behavior of the affected hand was evaluated by the Wolf test as a function of total time (s) and by the total score of functionality of movement (maximum possible score 80) before and after the 2-week therapy
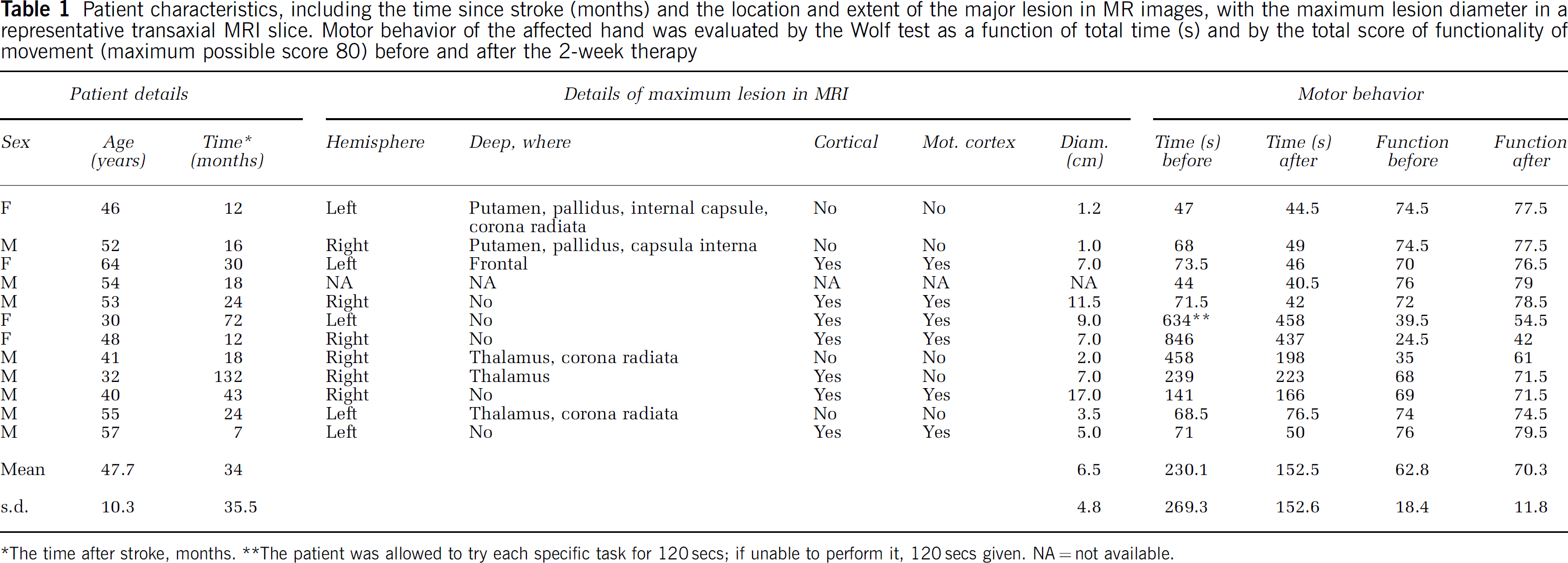
*The time after stroke, months. **The patient was allowed to try each specific task for 120secs; if unable to perform it, 120secs given. NA = not available.
Intervention
The affected arm and hand therapy followed closely the practice established by Taub et al (1999) and further refined by Miltner et al (1999). Therapy included 10 weekdays of continuously supervised exercises provided within two weeks. The CIMT program was always performed by one therapist simultaneously supervising two patients. Before the exercise program started, a lightweight sling was fitted in the patient's non-affected arm to prevent its use during the exercises and in daily activities during the 2-week period. The sling included a body temperature-activated clock that registered the hours of wear. The hand restriction system was locally tailor-made for the CIMT program (Figure 1). The patients were encouraged to wear the sling for approximately 10h/day. The exercises were individually tailored, increasingly challenging, yet possible to complete. The duration of each exercise was short, and almost all exercises were timed by a stop-watch to keep the motivational level high and to allow comparison of performance from day to day. Exercises were performed for 6h/day.

A light-weight restriction sling for the nonaffected hand and upper extremity. Note the body temperature-activated clock that registered the minutes of sling wear.
Structured Motor Function Test
To assess the motor behavior of the affected arm and hand, a structured motor function test, Wolf test (Wolf et al, 1989), was performed before the start of the therapy and after the 2 weeks. This test examines only the affected hand and arm, and it includes 16 motor tasks, such as dragging of 500g weight a designated distance on the table or picking up a pencil from a predetermined place. Each task is timed and scored in the functionality (score range 0 to 5) and quality (score range 0 to 5) of movement. The maximum score of 80 points in functionality and 80 points in quality of movement is easily attainable with a healthy hand and arm.
Single-Photon Emission Computerized Tomography Imaging
For SPECT imaging a dose of 550 MBq of Tc-99m-ethylcysteine dimer (Neurolite, Dupont Pharma/Durham APS, Kastrup, Denmark) was intravenously injected into the patient's right antecubital vein in a dim and quiet room before and after the therapy. The SPECT scan was performed 45 to 60 mins later, with a three-head Siemens MultiSPECT 3 gamma camera equipped with fan beam collimators (Siemens Medical systems Inc., Hoffman Estates, IL, USA) (Kuikka et al, 1993). The radius of the rotation was 13.8 cm. A total of 5 to 7 million counts were acquired for the entire head using an angular step of 3° over 360° (the matrix size 128 × 128 and 40 projections/camera head and each 35 s). The total imaging time was 25 mins.
Transaxial slices (3-mm thick) were reconstructed using Butterworth filter (a cutoff frequency of 0.5 cm−1 and an order of 6). The Chang attenuation correction with a uniform attenuation coefficient of 0.12 cm−1 was applied. The imaging resolution was 7 to 8mm. The slices were visually surveyed and then the slices were consecutively summarized to the total slice thickness of 6 mm for further analysis. First, all images of those patients whose lesion was in the left hemisphere were mirrored. After mirroring, all lesions were located in the affected ‘right’ hemisphere. The ‘left’ hemisphere was the one nonaffected, that is, healthy, hemisphere. Then all images were realigned into the same position. Furthermore, normalization was performed using bilinear interpolation. Finally, normalized images were smoothed by Gaussian kernel with 11 mm FWHM.
To clarify the details of the lesions, magnetic resonance imaging (MRI) was performed (Siemens Vision, 1.5 T, Erlangen, Germany) with a circular polarized head coil. MRI examination consisted of conventional T1-, T2-, PD-weighted images and fluid-attenuated inversion recovery (FLAIR) images. To exclude any acute ischemic lesion, diffusion weighted imaging (DWI) was also performed. All MR images were examined visually by an experienced neuroradiologist.
The statistical comparisons of the Wolf motor function test times were performed with the paired t-test, and the comparisons of structured motor function test scores were performed with nonparametric Wilcoxon test (SPSS software, version 9.0). Single-photon emission computerized tomography images were analyzed using statistical parametric mapping software (SPM99, Wellcome Department of Imaging Neurosciences, Institute of Neurology, UCL, London, UK, and MATLAB, Mathorks, Natick, MA, USA). After the pre-processing of SPECT images, the statistical group analysis was performed by the paired t-test, comparing the perfusion before the therapy with the perfusion after the therapy. Proportional scaling was performed for the t-test by scaling the global mean to the value of 50 ml/min. Threshold masking was also proportional, 0.8 of the global value. The global value was calculated as a mean voxel value, within per image full mean/8 mask. Resolution in SPM analysis was 2 × 2 × 2 mm3. Voxel-by-voxel statistical testing leads to the possibility of false-positive significance of single voxels because of large number of voxels in one functional volume. Therefore, in the statistical analyses a P-value less than 0.005 was considered significant, and only clusters with P-value ≤ 0.001 and size > 20 voxels (160mm3) were considered.
Results
The accomplished hours of exercise were 53.4 ± 1.8 h (mean ± s.d.) and the mean duration of the sling wear was 80.0 ± 14.2 h. Two weeks of CIM therapy resulted in an improvement in the voluntary hand motor control in the chronic stroke patients. The mean total time obtained in the Wolf test (total of 16 timed tasks) decreased significantly after therapy (P = 0.035) and the mean functionality and quality scores improved significantly (P = 0.02 and 0.02, respectively) (Table 1). Some patients were able to decrease their test time by almost 50% and to improve their functionality and quality scores by almost 30%, while some showed only a minor change in their behavioral scores and a minor decrease in performance time. The individual variability of the improvements in the Wolf test did not correlate with the amount of exercise; furthermore, those improvements did not correlate with the time lapse since the insult. Interestingly, the improvements in the Wolf test correlated significantly with the functionality (Pearson correlation coefficient −0.963; P < 0.001) and the quality scores (Pearson correlation coefficient −0.951; P < 0.001) obtained at the start of the therapy.
All patients, except one with claustrophobia, underwent MRI of the brain. Five patients had the maximum lesion in the left hemisphere and six in the right hemisphere according to the MR images. Five patients had only cortical lesions, six patients had deep striatocapsular lesions and two of them had also an additional cortical lesion. None had acute ischemic lesions. Eleven lesions were ischemic, and two showed sequelae of hemorrhage. The patient without MR images had a lesion in the right hemisphere, with paresis of the left hand.
Cerebral perfusion at rest changed in both hemispheres after the 2-week long therapy when compared with the pre-therapy perfusion. Areas of both increase and decrease were encountered in the perfusion analysis. Perfusion increased in four cerebral areas and in two areas in the cerebellum. The specific areas with perfusion increase in the affected hemisphere were in the precentral gyrus (Brodmann's area 6 (BA6); P < 0.001) and in the superior frontal gyrus (BA10; P < 0.001). In the nonaffected hemisphere, perfusion was increased in the superior frontal gyrus (BA6; P < 0.001) and in the cingulate gyrus (BA31; P < 0.001). In the cerebellum, there was one area with a perfusion increase in both hemispheres (P < 0.001) (Table 2, Figure 2). All these areas were near the midsagittal line, within a distance of 23 mm from the midline.
The cerebral areas with changed perfusion in SPET after 2 weeks of CIMT
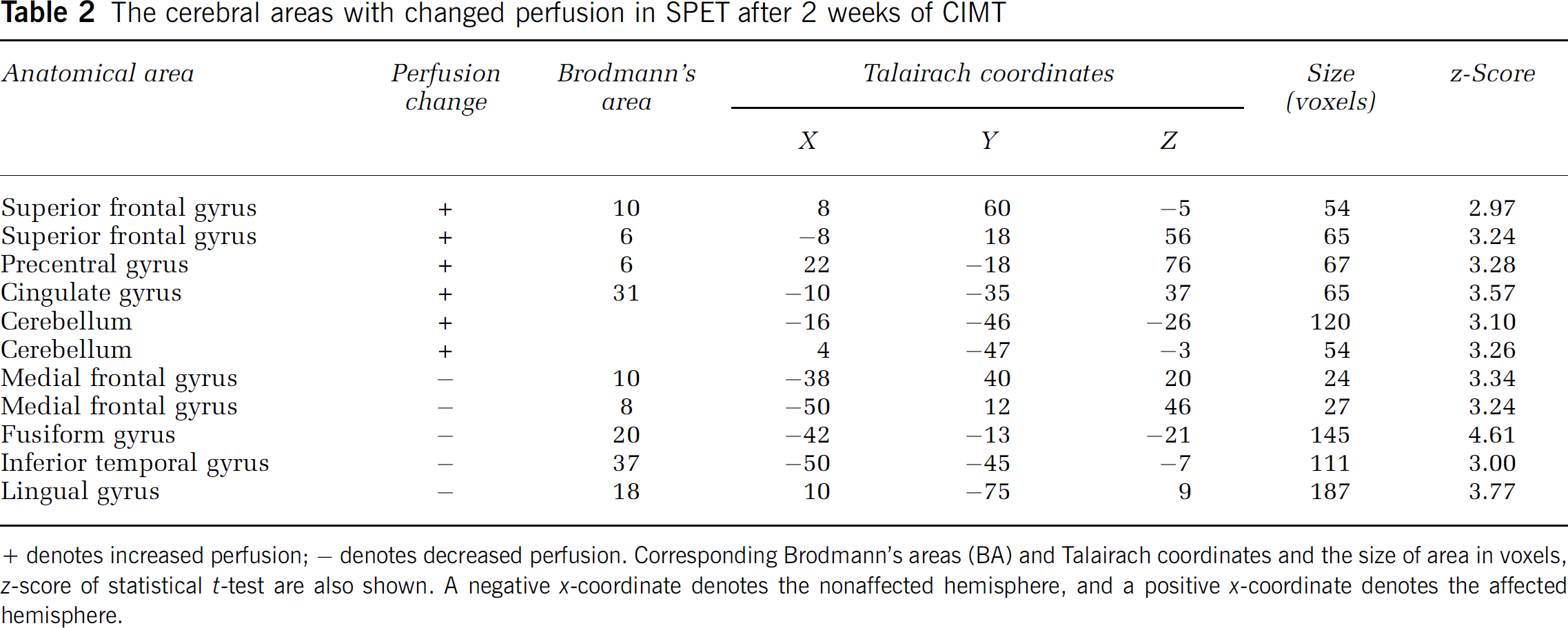
+ denotes increased perfusion; − denotes decreased perfusion. Corresponding Brodmann's areas (BA) and Talairach coordinates and the size of area in voxels, z-score of statistical t-test are also shown. A negative x-coordinate denotes the nonaffected hemisphere, and a positive x-coordinate denotes the affected hemisphere.
Perfusion at rest decreased in four cerebral areas in the nonaffected hemisphere, but only in one area in the affected hemisphere after the rehabilitation program (Table 2, Figure 2). The perfusion decrease in the affected hemisphere was detected in the lingual gyrus (BA18; P < 0.001). In the nonaffected frontal cortex, two areas with decreased perfusion were found in the middle frontal gyrus (BA8, P < 0.001; BA10, P < 0.001). Also, an area in the fusiform gyrus (BA20, P < 0.001) and an area in the inferior temporal gyrus (BA37, P < 0.001) showed decreased perfusion. In the nonaffected hemisphere, all areas with decreased perfusion were located more laterally than those areas with increased perfusion.
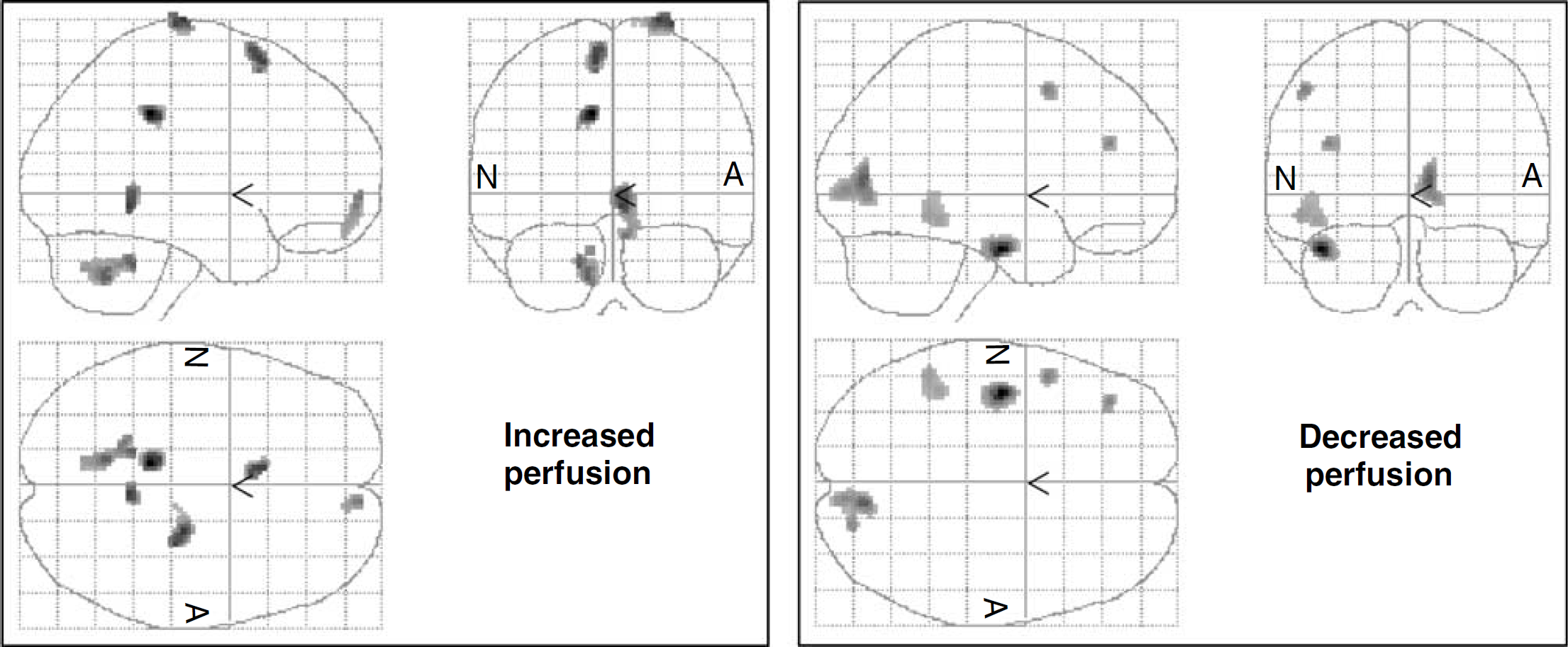
Cerebral areas with changed perfusion: areas of increased perfusion in the left panel and areas of decreased perfusion in the right panel. Letter A denotes the affected hemisphere, and N denotes the nonaffected hemisphere (SPM99, Wellcome Department of Imaging Neurosciences, Institute of Neurology, UCL, London, UK).
Discussion
Constraint-induced movement therapy is an exercise rehabilitation approach that has been shown to further improve motor recovery of the affected upper extremity in chronic stroke patients with mild to moderate hemiparesis (Taub et al, 1993, 1994; Kunkel et al, 1999; Miltner et al, 1999). Our patients experienced significant improvement in motor function in their paretic upper extremities after CIM therapy. The mean total time obtained in the Wolf test decreased significantly after therapy, and the mean functionality and quality scores improved significantly. Individual differences in changes in motor behavior were obvious. The improvements in the Wolf test were neither related to the time lapse since the insult nor to the amount of practice hours, but correlated with the functionality and quality scores at the start of the therapy. It appeared that those patients whose hand functional ability was rather low at start benefited most. Patients were highly motivated to do the exercises and wear the sling during the 2-week CIMT period. The variability in the sling wear hours was mainly due to the functional level of the lower limbs, because patients with a more impaired gait could not wear the sling as long as the others for safety reasons.
In the present study, we identified alterations in regional cerebral perfusion related to intensive movement therapy. Cerebral function is supported primarily by oxidative metabolism, and regional blood flow continuously regulates the supply of oxygen and glucose locally. Single-photon emission computerized tomography is a well-established method to study regional blood flow. Changes in regional cerebral blood flow (rCBF) reflect changes in the underlying neuronal activity. Single-photon emission computerized tomography was performed at rest before and after the completion of the therapy and several brain areas with significant perfusion changes were detected in both hemispheres. These studies are typically performed with the patient in a single state, usually at rest, to minimize confounding effects of nonwanted activity. The pattern of rCBF distribution represents actual perfusion at the time of injection.
In the affected hemisphere, perfusion was increased in the precentral gyrus (BA6). Two somatotopically organized motor maps exist in area 6: the premotor area (PMA) in the lateral region and the supplementary motor area (SMA; somatotopy is mostly in the caudal SMA) in the medial region. Experimental work on primates and recordings in humans have suggested that area 6 plays an important role in the planning of the execution of movement, particularly in complex movement sequences of the distal musculature. Electrical and metabolic activity in both the PMA and SMA are known to increase before the execution of voluntary movements, especially before more complex self-paced movements, and even during the mental imagery of grasping movements (Ikeda et al, 1992; Grafton et al, 1996; Winstein et al, 1997). In the present study, activation of area 6 was observed both in the affected and nonaffected hemispheres. The two intact hemispheres are closely linked via the corpus callosum, which might be shown by our finding of bilateral perfusion increase in BA6. The increase in the healthy hemisphere may also suggest bilateral participation of primary motor cortices in voluntary movement execution after the therapy. Increased ipsilateral motor cortex involvement in movement execution has been reported after CIMT in stroke patients (Schaechter et al, 2002).
Also, the superior frontal gyrus (BA10) in the affected hemisphere showed an increase in perfusion. This prefrontal tertiary motor area (BA10) receives and integrates information from the different sensory systems and participates in motor preparation. Increased perfusion also appeared in the posterior cingulate gyrus in the healthy hemisphere (BA31). The cingulate gyrus is known to have direct connections to the primary motor area. The cerebellum has a primary role in the co-ordination of skilled voluntary movement and in the control of muscle tone and posture. In its motor regulatory role, the cerebellum is assumed to adjust motor outputs on the basis of afferent input relating to planned motor action and reafferent input from executed motor acts. Perfusion was increased in both hemispheres of cerebellum, suggesting increased activation in one or more of the roles of cerebellum.
In the nonaffected hemisphere, perfusion was decreased in four different cortical areas, but only in one area in the affected hemisphere. In the nonaffected hemisphere, two areas were in the medial frontal gyrus (BA8 and BA10) and two areas in the temporal lobe, namely in the fusiform gyrus and in the inferior temporal gyrus. In the affected hemisphere perfusion decreased in the lingual gyrus. Indeed, the perfusion was decreased in the lateral brain regions and in regions not primarily involved with motor control. Omori et al (1999) investigated the neuronal substrates participating in attentional set-shifting for motor selection rules with fMRI. They found activation in fusiform and lingual gyri, which might have reflected the increased attentional demand for visual processing in the light of their task for motor selection. It might be presumed that those cerebral areas of our patients, which showed decreased perfusion after CI therapy in areas primarily involved in nonmotor processes, indicated normalized perfusion. These areas might have been hyperperfused before the therapy in an effort to compensate for disturbed activity of the affected hemisphere with possibly attentional drive from the nonaffected hemisphere. Thus, the decreased perfusion might be considered an indication of change towards relative inactivity in the mentioned areas.
Before statistical SPM analysis, we mirrored the images of those patients whose lesion was in the left hemisphere. After this all lesions were in the right hemisphere and the left hemisphere was the non-affected hemisphere. Variability, asymmetry, and effects of age and gender have been studied in healthy adults in Tc-ECD brain perfusion SPECT (Van Laere et al, 2001). They reported an overall increase in the right/left asymmetry with age, which was most pronounced in the frontal and temporal neocortex. In the present study, statistical analysis was performed using the paired t-test comparing the perfusion before and after therapy, that is, each person served as his or her own control. Thus, the effects of asymmetry, age, and gender can be ignored. Nonetheless, the anatomical variability and the variability of the injuries have a diluting effect on our results.
It is clear that motor skill learning plays a role in shaping functional organization in the primary motor cortex. Repetitive motor behavior can produce changes in representational maps in the primate motor cortex (Nudo et al, 1996). Primate studies also showed that repetitive exercise alone did not enhance cortical reorganization after experimental stroke, but increasingly difficult task-relevant training did enhance reorganization (Plautz et al, 2000). The CIMT provided to our patients an increasingly difficult motor challenge, and thus provided activation in the brain that may enhance reorganization related to motor control. The increased perfusion was found in motor control-related areas. The specific areas with an increase in perfusion in the affected hemisphere were in precentral gyrus, premotor cortex (BA6), and in the frontal cortex in superior frontal gyrus (BA10). For the present subjects, there is no follow-up available and thus we cannot know how enduring the obtained changes were; however, the changes in motor behavior after CIMT have been shown to be endured at least six months by us and others (Miltner et al, 1999; Tarkka et al, in press; Taub and Uswatte, 2003).
In the present study, the patients were investigated as a single group. Certainly, the effects of CIMT on motor behavior and the cerebral perfusion varied individually, yet it appears that intensive movement therapy can change local perfusion in areas known to participate in movement planning and execution. These changes might be a sign of active reorganization processes occurring with CIMT in the chronic state of stroke.
