Abstract
Dietary restriction (DR) protects against neuronal dysfunction and degeneration, and reduces the risk of ischemic stroke. This study examined the role of silent information regulator T1 (SIRT1) and arterial baroreflex in the beneficial effects of DR against stroke, using two distinct stroke models: stroke-prone spontaneously hypertensive rats (SP-SHRs) and Sprague-Dawley (SD) rats with middle cerebral artery occlusion (MCAO). Sirt1 knockout (KO) mice were used to examine the involvement of sirt1. Sinoaortic denervation was used to inactivate arterial baroreflex. Dietary restriction was defined as 40% reduction of dietary intake. Briefly, DR prolonged the life span of SP-SHRs and reduced the infarct size induced by MCAO. Dietary restriction also improved the function arterial baroreflex, decreased the release of proinflammatory cytokines, and reduced end-organ damage. The beneficial effect of DR on stroke was markedly attenuated by blunting arterial baroreflex. Lastly, the infarct area in sirt1 KO mice was significantly larger than in the wild-type mice. However, the beneficial effect of DR against ischemic injury was still apparent in sirt1 KO mice. Accordingly, arterial baroreflex, but not sirt1, is important in the protective effect of DR against stroke.
INTRODUCTION
The beneficial effects of dietary restriction (DR), a reduction of energy intake without affecting the nutritional needs, against age-related diseases are well documented. Dietary restriction prolongs the lifespan of many organisms ranging from yeast to mammals. 1 Dietary restriction also delays the occurrence of pathophysiological changes including atherosclerosis, diabetes and cancer in some mammalian species, 2 and lower cardiovascular disease risk. 3 Dietary restriction also protects neurons from dysfunction and degeneration, and reduces the risk of ischemic stroke. 4
The benefits of DR are partly caused by induction of neurotrophic factors, protein chaperones, antioxidant enzymes, uncoupling proteins, and antiinflammatory cytokines. 5 Among these targets, silent information regulator T1 (SIRT1) has attracted much attention. Silent information regulator T1 is activated by DR and has been implicated in various beneficial effects associated with DR. 6 The beneficial effects of DR are multifold and might not be explained by change in one gene, or one protein. Many systems, including the metabolic system,7,8 and the nervous system, 9 are involved.
Arterial baroreflex is the most important regulator of cardiovascular functions.10–12 Decreased baroreflex sensitivity (BRS) is often seen with aging and in many age-related diseases, 13 and an important prognostic factor for long-term outcome in many cardiovascular diseases, including myocardial infarction, heart failure, hypertension, and stroke.11,12 Recently, the work in this laboratory suggested that arterial baroreflex dysfunction is an important risk fact for the development of stroke.14,15
In the current study, we tested the hypothesis that arterial baroreflex is involved in the protective effect of DR against stroke. The interaction of DR, SIRT1, and stroke was also evaluated.
MATERIALS AND METHODS
Animals
Male stroke-prone spontaneously hypertensive rats (SP-SHRs) were provided by the Animal Center of the Second Military Medical University. Male Sirt1 knockout (KO) mice, and littermate wild-type (WT) mice were obtained from the Institute for Nutritional Sciences, Shanghai Institutes for Biological Sciences, the Chinese Academy of Sciences. Male Sprague-Dawley (SD) rats were purchased from Sino-British SIPPR/BK Lab Animals (Shanghai, China). All the animals used in this study received humane care in compliance with institutional guidelines of Second Military Medical University for health and care of experimental animals. And all experiments were approved by Second Military Medical University for health and care of experimental animals.
Dietary Restriction
All diets were purchased from Institute of Naval Medicine of China. The diet consisted of: barley flour (14.3%), wheat flour (31.8%), corn flour (15%), bean pulp flour (14%), wheat bran (18.9%), soybean oil (4.5%), and mineral mix (1.5%, including 0.45% NaCl). Both rats and mice were placed under unrestricted diet (ad libitum, AL) or DR (food intake at 60% of the amount consumed by AL group animals). The mean food intake of AL animals per week was estimated by subtracting the remaining weight of food each week from the initial weight of the previous week.
Sinoaortic Denervation
Sinoaortic denervation (SAD) was performed as described previously. 14 Briefly, rats received a mixture of ketamine and diazepam (50 mg/kg +5 mg/kg, intraperitoneally), plus atropine sulfate (0.5 mg/kg, intraperitoneally) and procaine benzylpenicillin (60,000 U, intramuscularly). The superior laryngeal nerves were transected near the vagus nerves on both sides. The superior cervical ganglia and a small section of the sympathetic trunk were removed. The aortic depressor nerves were also transected on both sides. The carotid sinus baroreceptors were mechanically stripped at the carotid bifurcation and its branches. The external, internal, and common carotid arteries and the occipital artery were treated with 10% phenol (in 95% ethanol). The sham operation included a midline neck incision and bilateral isolation of the neck muscles. Sinoaortic denervation was carried out at 1 month before DR or other treatment.
Middle Cerebral Artery Occlusion
Focal cerebral ischemia in rats or mice was produced by intraluminal occlusion of the left middle cerebral artery (MCA) for 2 hours using a silicone rubber-coated nylon monofilament.16,17 Briefly, the core temperature (rectum) was maintained at 36.5°C to 37.5°C by use of a temperature controller pad (ML312, ADInstruments, Sydney, NSW, Australia) throughout the surgery. Cerebral focal ischemia was produced by intraluminal occlusion of the left MCA using a silicone rubber-coated nylon monofilament. Achievement of ischemia was confirmed by monitoring regional cerebral blood flow in the area of the left MCA. Cerebral blood flow was monitored through a laser Doppler transducer (MNP110XP, ADInstruments) to a laser Doppler computerized main unit (ML191, ADInstruments). The microtip was attached to the skull of the animal through cyanoacrilate glue. Animals that did not show a cerebral blood flow reduction of at least 70% were excluded from the experimental group, as were animals that died after ischemia induction. Two hours after MCA occlusion (MCAO), the occluding filament was withdrawn to allow reperfusion. One day after operation, neurologic deficits was evaluated using a scoring methods as previously described.16,17 The animals were killed and the brain slices (for each brain, seven slices for rats and six for mice) were prepared with brain-cutting matrix (ASI Instruments, Warren, MI, USA). The slices were incubated in 1% TTC (2,3,5-triphenyltetrazolium chloride) solution (Sinopharm Chemical Reagents, Shanghai, China) at 37°C for 30 minutes, and then mounted on dry paper and photographed with a digital camera. The infarct size was quantified with ImageJ software (National Institute of Health, Bethesda, MD, USA), and expressed as the percentage relative to the contralateral hemisphere.
Blood Pressure and Baroreflex Sensitivity
Systolic blood pressure (SBP), diastolic blood pressure, and heart rate were continuously recorded in conscious, freely moving rats as previously described. 14 Noninvasive blood pressure measurements were made using a tail-cuff system (ALCBIO, Shanghai, China). After 3 days of training, each rat was assessed for a minimum of three times per session. Baroreflex sensitivity was measured using a previously described method.14,15 Phenylephrine (5 to 10 μg/kg, intravenously) was used to raise SBP by about 30 mm Hg. The delay between the rise in blood pressure (stimulus) and the prolongation of heart rate (response) was ~1 second. Heart period was plotted against SBP for linear regression analysis for 2 to 8 shifts; the slope with the largest correlation coefficient (r) was expressed as BRS (ms/mm Hg). The mean of two measurements was taken as the final result.
Behavioral Assessment and Morphological Examination
Male SP-SHRs were placed under AL or DR, starting from 3 months of age. Behavioral signs of stroke including decreasing body weight, paralysis, seizure, bleeding from the nose and/or mouth, changes of respiratory rhythm, coma, and death, were observed daily. Upon death, the brain was removed for examination of hemorrhage, edema, and infarction. The brains without apparent change under gross examination were fixed in 4% paraformaldehyde in phosphate saline buffer (pH 7.4) for 24 hours, sectioned transversely at 5 μm thickness with 200 μm interval from the anterior to the posterior extremity, and stained with hematoxylin and eosin.
For assessment of end-organ damage, the right kidney, thoracic aorta, and heart were dissected quickly after deep anesthesia. Gross examination of the heart (weight and wall thickness), right kidney (appearance, weight, and thickness), and aorta (weight) was performed. The ratio of heart to body weight (mg/g), aortic weight to length (mg/cm), kidney to body weight, and the thicknesses of renal cortex, and medulla were used to reflect cardiac/aortic hypertrophy and kidney damage, respectively. 15
Immunoblotting
Tissue/cell extract was boiled in 4 × loading buffer, subjected to the sodium dodecyl sulfate polyacrylamide gel electrophoresis, and transferred onto nitrocellulose blotting membranes. The membranes were incubated with one of the following antibodies: SIRT1, acetyl-Histone H3 and H4 (Cell Signaling, Boston, MA, USA) before incubation with IRDye800CW-conjugated secondary antibody. The image was captured by the Odyssey infrared imaging system (Li-Cor Bioscience, Lincoln, NE, USA), and analyzed using ImageJ software (NIH). The immunoblotting experiments were performed in two to three representative animal subjects.
Proinflammatory Cytokine
The levels of tumor necrosis factor α (TNFα), interleukin-6 (IL-6), interleukin-1α (IL-1α), or interleukin-1β (IL-1β) in the entire cerebral cortex homogenate or serum were determined with an enzyme-linked immunosorbent assay using an automatic microplate reader (Infinite M200; Tecan Austria GmbH, Grödig, Austria).
Serum Glucose and Lipids
Serum glucose, triglyceride, total cholesterol, low-density lipoprotein-cholesterol, and high-density lipoprotein-cholesterol were measured using a clinical chemistry analyzer (Shanghai Xunda Medical Instruments, Shanghai, China) with corresponding kits from Shanghai Fosun Long March Medical Sciences (Shanghai, China).
Statistical Analysis
The investigators were blinded to the procedures when they assessed the infarct size and neurologic deficient score of MCAO animals. The animals were randomly assigned by using the random permutations table. Data were expressed as the mean ± s.d. Data were analyzed with two-tailed Student's t-test or one-way analysis of variance followed by least significant difference t-test for pairwise comparison. Kaplan–Meier analysis was used to estimate survival probabilities. Log-rank testing was used to evaluate equality of survival curves. P < 0.05 was considered statistically significant.
RESULTS
Dietary Restriction Delayed the Development of Stroke in Stroke-Prone Spontaneously Hypertensive Rats
In a period of 40 weeks after diet manipulation starting from 3 months of age, the body weight increased by 117 and 19 g in the AL and DR groups, respectively (Figure 1A). The animals on DR lived longer than AL group (Figure 1B). Death caused by stroke was also confirmed (Figure 1C). As shown in Figure 1D, DR prevented or delayed the development of lethal stroke. Kaplan–Meier analysis revealed significantly longer survival time in the DR group (491 ± 87 versus 289 ± 84 days in AL group; log-rank test χ2 = 54, P < 0.001).
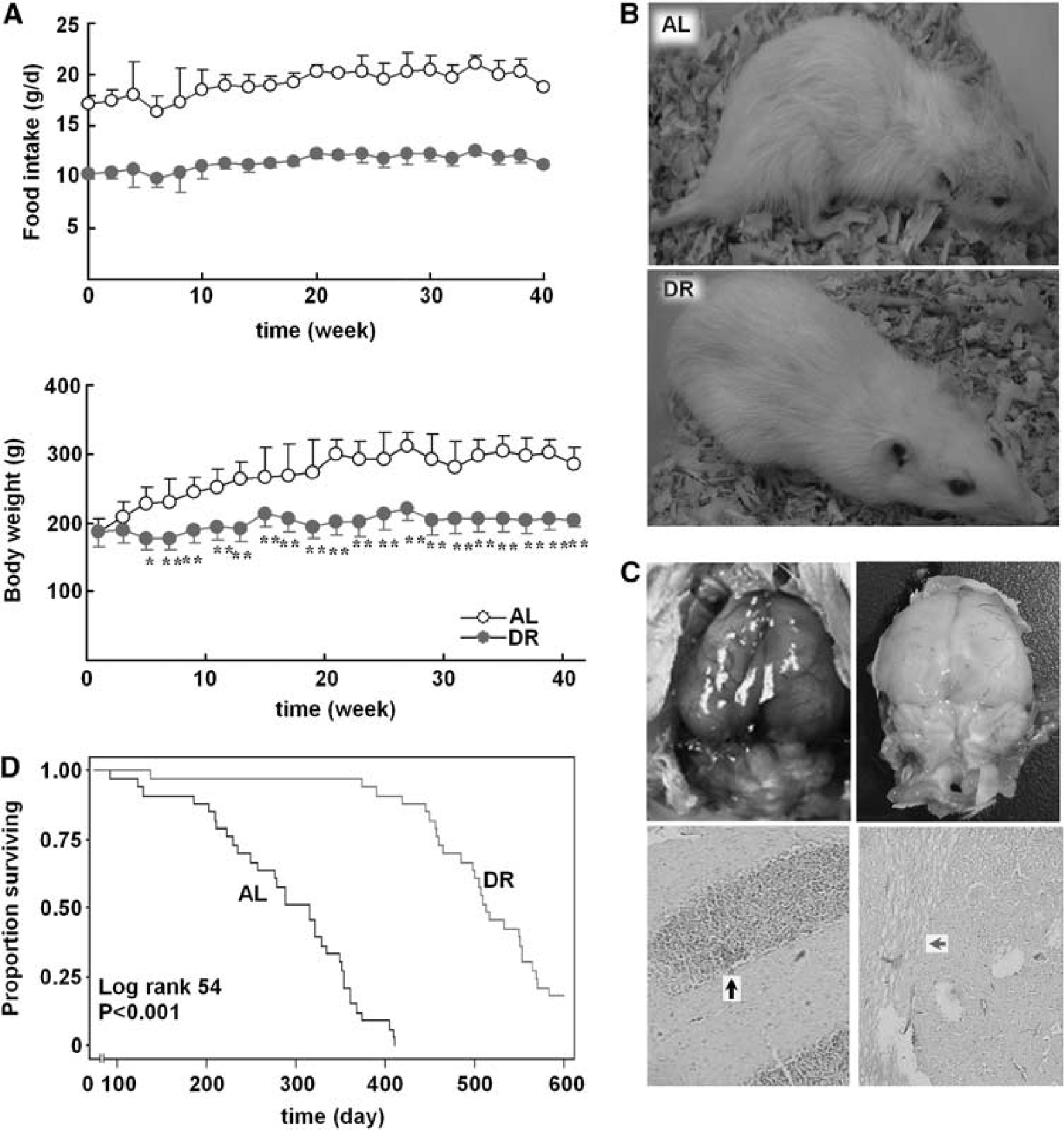
The effect of dietary restriction (DR) on stroke death of stroke-prone spontaneously hypertensive rats (SP-SHRs). Three-month-old male SP-SHRs were randomly divided into two groups and fed standard feed (Control, ad libitum (AL), n = 33) or placed on dietary restricted feed (DR, n = 33). (A) The amount of food in DR rats was adjusted every week according to the food intake in control rats. Body weight was recorded in the two groups every 2 weeks. Data are expressed as mean ± s.d. ∗P < 0.05, ∗∗P < 0.01. Student's t-test. (
Dietary Restriction Decreased Proinflammatory Cytokines and Prevented End-Organ Damage in Stroke-Prone Spontaneously Hypertensive Rats
At 20 weeks after the diet manipulation, the concentration of IL-1β, IL-6, and TNFα in the cerebral cortex of rats under DR was significantly lower than in the AL group (Figure 2A). Dietary restriction also decreased serum level of triglyceride significantly, but did not affect blood glucose, total-, high-density lipoprotein-, or low-density lipoprotein-cholesterol (Figure 2A). Hypertrophy of the heart/aorta and the injury of kidney were evident in all SP-SHRs, but was significantly attenuated by DR (Figures 3A and 3B).
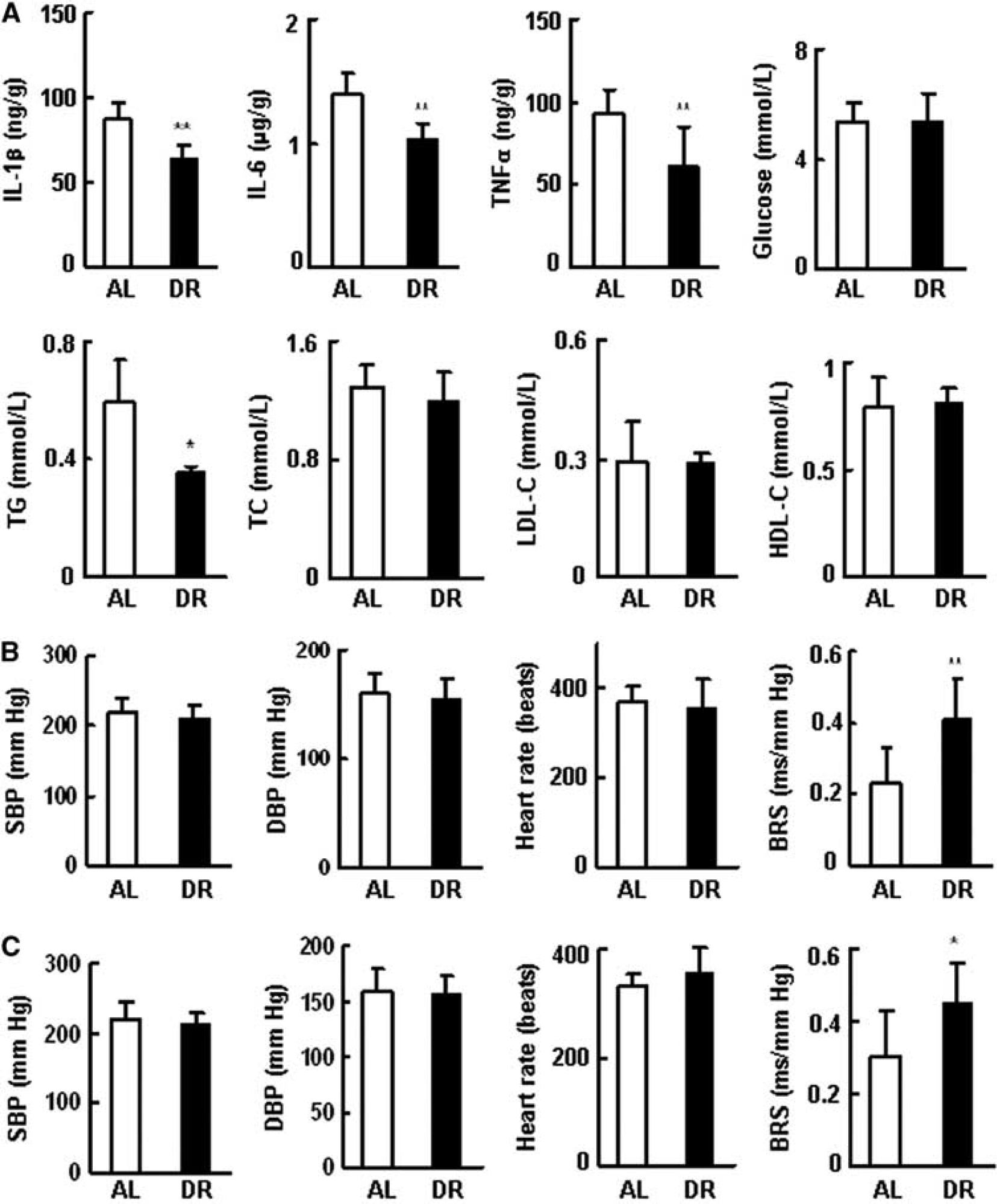
The effect of dietary restriction (DR) on the proinflammatory cytokines, glucose, lipids, and hemodynamic parameters of stroke-prone spontaneously hypertensive rats (SP-SHRs). Three-month-old male rats were randomly divided into two groups and fed control diet (ad libitum (AL)) or placed on DR. (
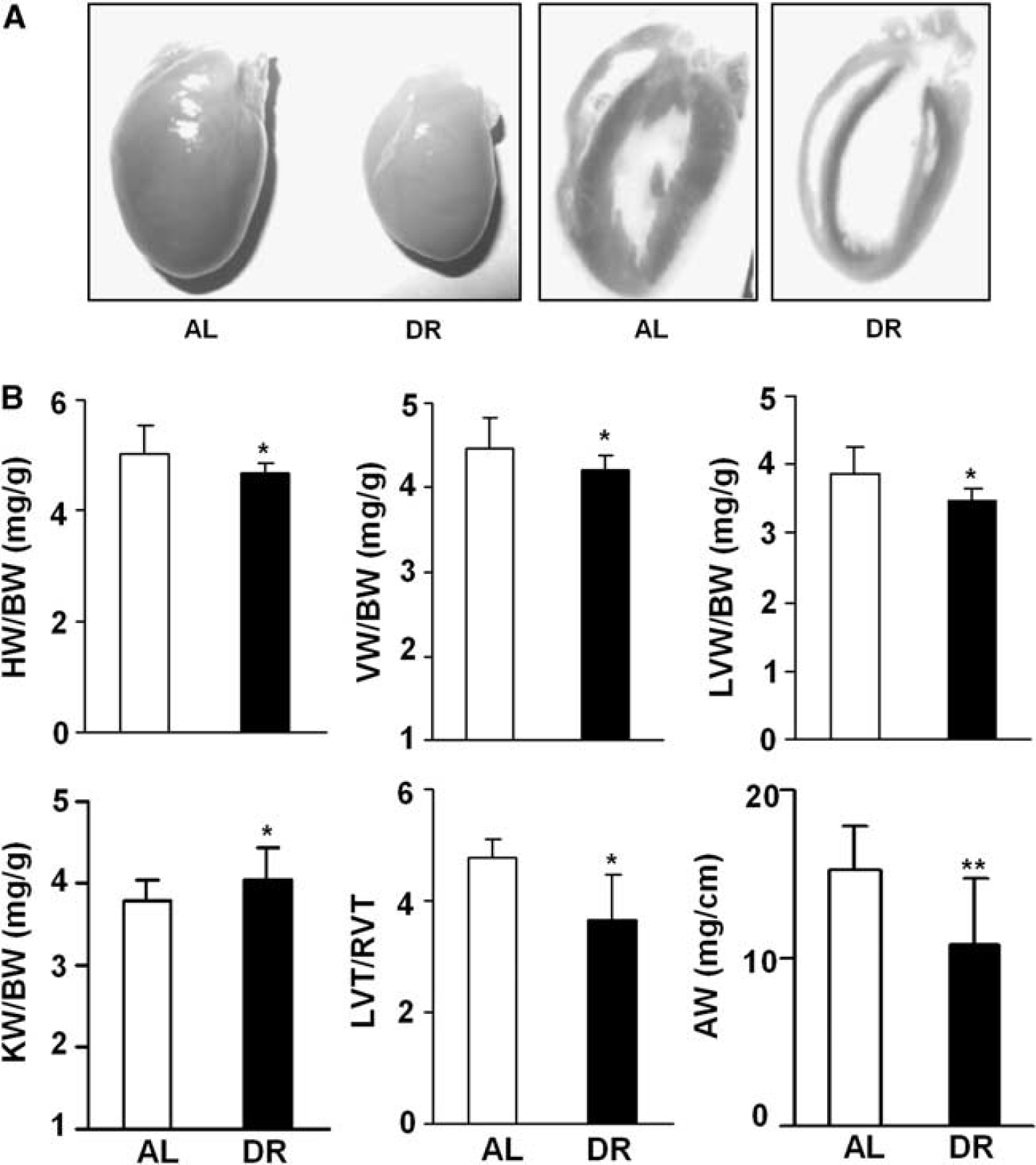
Dietary restriction (DR) reduced end-organ damage in stroke-prone spontaneously hypertensive rats (SP-SHRs). Three-month-old male rats were randomly divided into two groups and fed unrestricted (ad libitum (AL)) or restricted diet for 20 weeks. (
Dietary Restriction Increased Arterial Baroreflex
Dietary restriction for 20 weeks decreased blood pressure (mean change of SBP, –8 mm Hg, P > 0.05) of SP-SHRs. Baroreflex sensitivity was increased by a much larger magnitude (0.45 ± 0.11 versus 0.23 ± 0.09 ms/mm Hg, P < 0.01; Figure 2B). Heart rate was not affected. Similar results were obtained with 10-week DR (Figure 2C).
Sinoaortic Denervation Attenuated the Neuroprotective Effects of Dietary Restriction
Measurement of BRS at 1 month after SAD confirmed the loss of baroreflex in SP-SHRs: 0.08 ± 0.02 versus 0.38 ± 0.08 ms/mm Hg in sham-operated rats (P < 0.01). Blood pressure was not affected (SBP, 211 ± 13 versus 204 ± 7 mm Hg, P > 0.05; Figure 4A). The change of food intake in different four groups is shown in Figure 4B. A 24-week, DR reduced the SBP by about 10 mm Hg in SP-SHRs with or without intact baroreflex (the right panel of Figure 4B). In rats with intact arterial baroreflex, DR increased the lifespan by about 200 days (Figure 4C, left). This prolongation was markedly attenuated by SAD (about 119 days in SAD rats. Figure 4C, right panel).
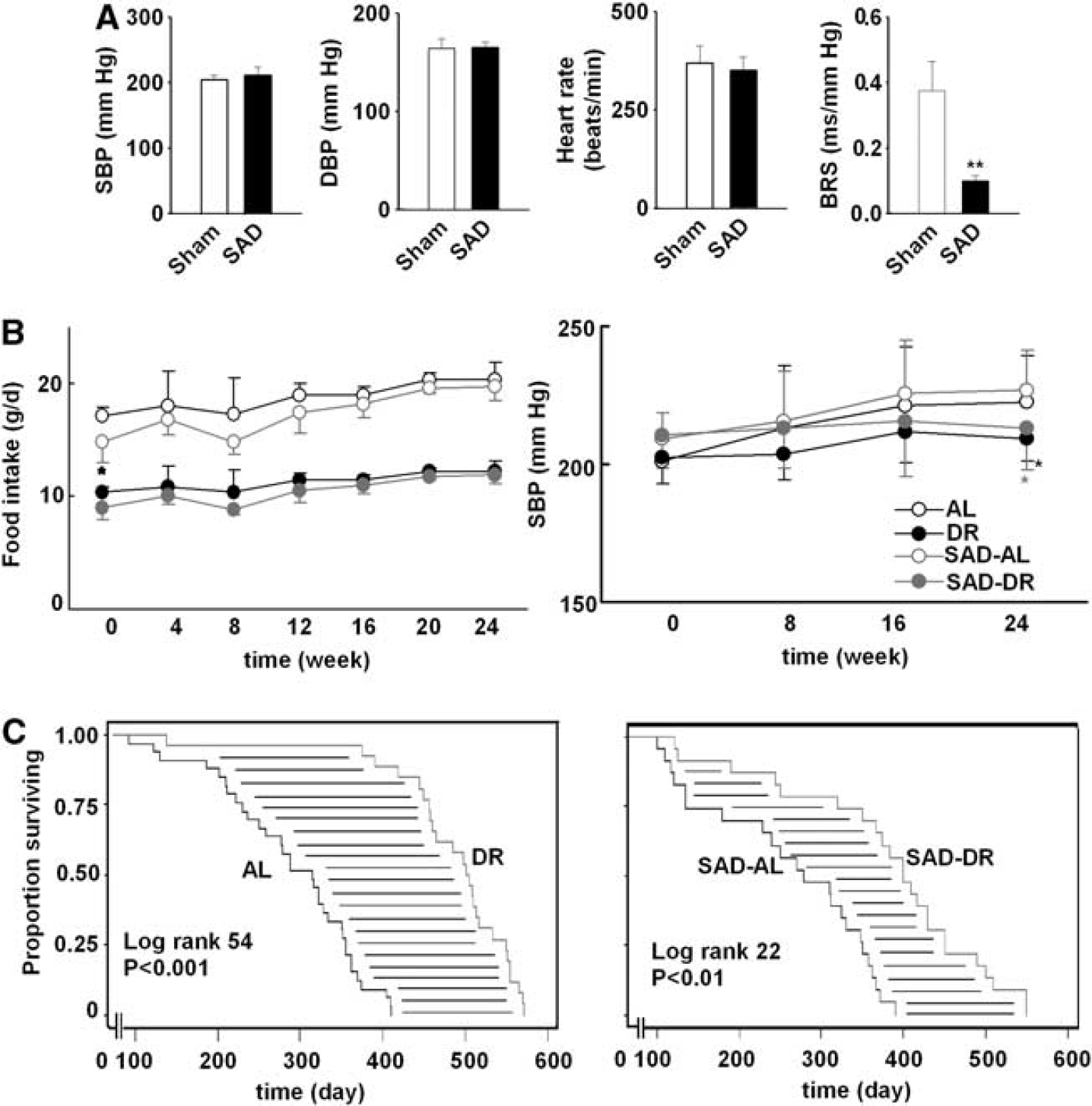
Arterial baroreflex involved in the neuroprotection of dietary restriction in rats. (
Sinoaortic denervation was also successful in SD rats (Figure 5A). The infarct caused by MCAO was much larger in rats subjected to SAD than in the sham-operated rats (40.2% ± 8.3% versus 25.4% ± 8.3%, P < 0.01). Dietary restriction reduced infarct size in SD rats with intact baroreflex (by 66%), and to a much lesser degree in SAD rats (23%; Figure 5B). Dietary restriction improved neurologic function (neurologic score 1.6 ± 0.5 versus 2.6 ± 0.7, P < 0.05) in rats with intact baroreflex, but not in rats with SAD (2.8 ± 0.7 versus 3.4 ± 0.5, P > 0.05).
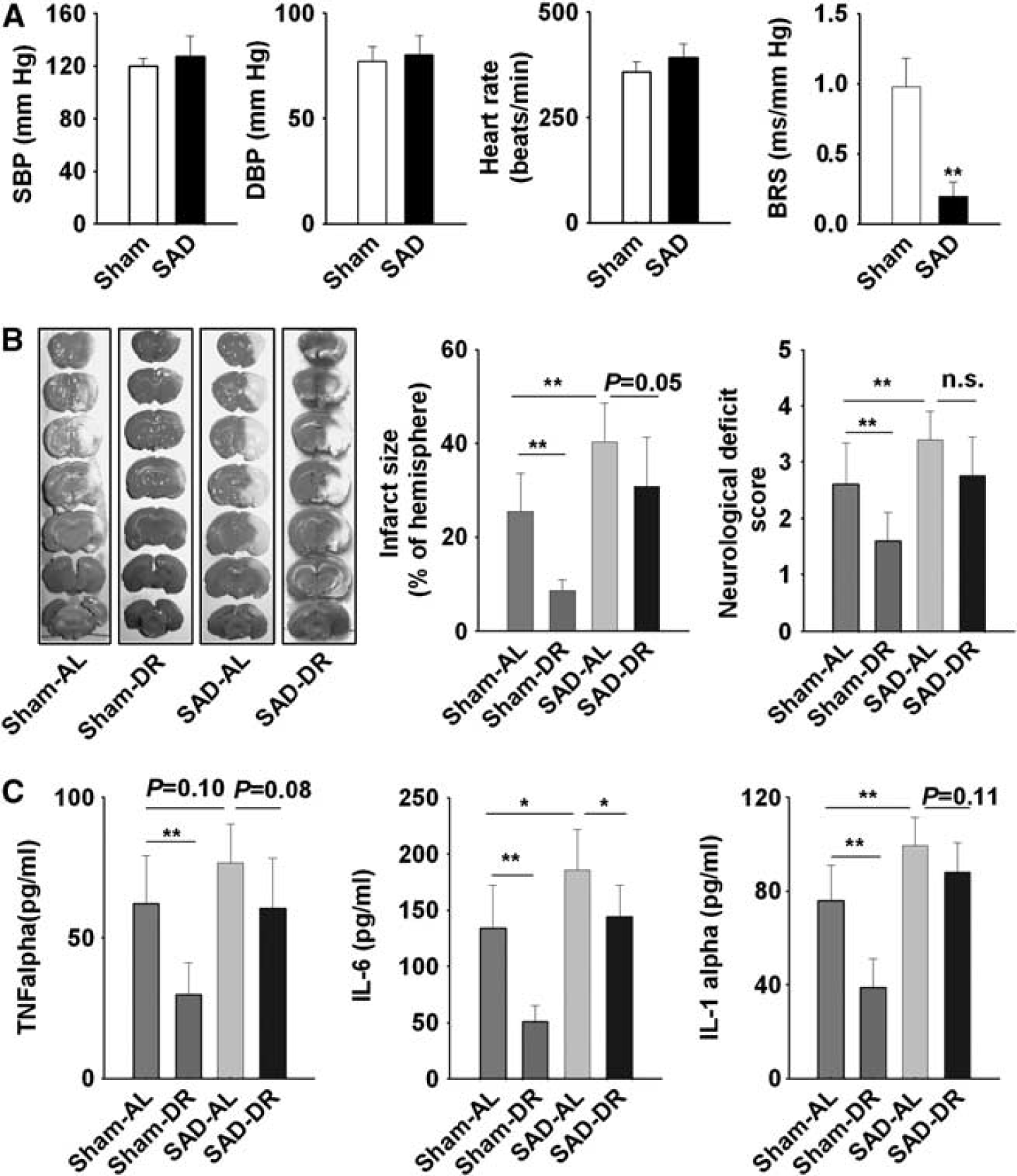
Sinoaortic denervation (SAD) attenuated the beneficial effect of dietary restriction (DR) on ischemic stroke. The animals were placed on DR for 8 weeks 1 month after SAD. (
Dietary restriction reduced the concentration of TNFα, IL-6, and IL-1α in the serum of rats with MCAO. In contrast, SAD increased the concentration of IL-6 and IL-1α, and attenuated the reduction of proinflammatory cytokines induced by DR (Figure 5C).
Protective Effect of Dietary Restriction Against Stroke Is Independent on Silent Information Regulator T1
Dietary restriction increased SIRT1 expression in the cerebral cortex, hypothalamus, and medulla of brain (Figure 6A); also in the heart and kidney of SP-SHRs (Figure 6B). To investigate the effect of SIRT1 on stroke, resveratrol, an activator of SIRT1, was used in two doses (5 and 15 mg/kg per day). The activation of SIRT1 was confirmed by the measurement of acetylated Histone H3 and H4 (Figure 6C). Resveratrol was mixed into feeding stuff, and did not increase the lifespan of SP-SHRs (Figure 6D).
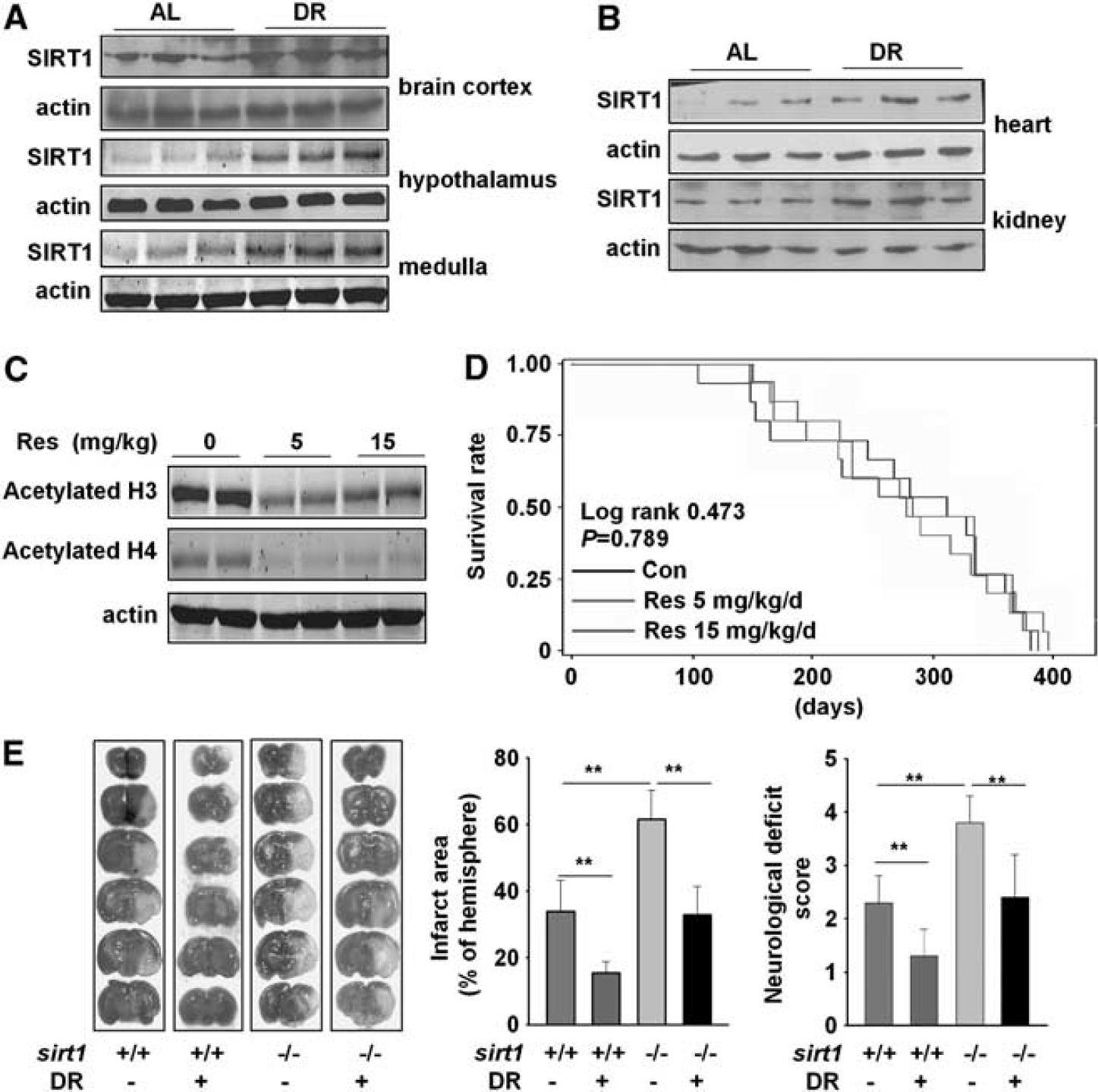
The protective effect of dietary restriction (DR) against stroke is independent on silent information regulator T1 (SIRT1). Stroke-prone spontaneously hypertensive rats (SP-SHRs) were fed unrestricted or restricted diet. Representative Western blotting of SIRT1 in: (
Infarct induced by MCAO was significantly larger, by ~81%, in sirt1 KO mice than in WT littermate controls (62% ± 8.5% versus 34% ± 9.2%, P < 0.01; Figure 6E). The sirt1 KO mice also had significantly worse overall neurologic outcomes (neurologic score 3.8 ± 0.5 versus 2.3 ± 0.5 in WT control mice, P < 0.01). Dietary restriction reduced infarct size in both the sirt1 KO mice (33% ± 8.4% versus 62% ± 8.5%, decreased by 46%, P < 0.01) and WT mice (15.5% ± 3.4% versus 34% ± 9.2%, decreased by 54%; Figure 6E), and significantly improved neurologic function in both sirt1 KO mice (neurologic score 2.4 ± 0.8 versus 3.8 ± 0.5, P < 0.05) and WT mice (1.3 ± 0.6 versus 2.3 ± 0.5, P < 0.05).
DISCUSSION
Dietary restriction delays the onset of age-associated pathologies.1–4 A study using rhesus monkeys showed that DR could reduce the incidence of diabetes, cancer, cardiovascular disease, and brain atrophy, and ultimately promotes the survival. 18 Reduced incidence of stroke in response to DR may be a result of improved health of the cardiovascular system. 3 A study in both young and aged rhesus monkeys did not show survival benefit of DR, but it is worthy to point out that the control subjects did not receive true AL (to prevent obesity). 19 Dietary restriction, even in slight degree, could produce beneficial effect. 20 The current study demonstrated that 20% and 40% of DR produce similar effects against stroke injury in MCAO rats (data shown in Supplementary Figure S1).
Dietary restriction reduces the risk factors for ischemic stroke, including proinflammatory cytokines, body fat, blood pressure, and serum lipid and lipoprotein levels.3,21 In the present study, hypertrophy of the heart, the thickening of aortic wall, and renal injury all were ameliorated by DR. It should be noted that the changes in lipids levels and blood pressure of SP-SHRs were statistically significant but minimal. Reported effects of DR on blood pressure are variable, with DR effects ranging from significant reduction of blood pressure22,23 to transient and slight reduction.24,25A recent report showed that the depressor effect of DR wanes as pathogenesis progresses, and suggested that the early lasting effect on cardiac hypertrophy and function is independent of a sustained depressor effect. 25
Baroreflex sensitivity is an important determinant of many cardiovascular diseases. Patients with reduced BRS exhibit shorter survival after myocardial infarction, heart failure, 11 or stroke. 12 We previously reported that arterial baroreflex dysfunction promotes the development of atherosclerosis in rats, and decreases survival time in lipopolysaccharide-induced lethal shock. 26 Intact arterial baroreflex function is also necessary to prevent aconitine-induced ventricular arrhythmias. 26 Impaired BRS has also been repeatedly shown to be present in acute stroke.27,28 Robinson et al 12 reported poor prognosis in poststroke patients with impaired BRS (<5.0 ms/mm Hg). We previously demonstrated that BRS is an important factor in determining survival time in SP-SHRs. The result showed that death after stroke was significantly delayed in rats with high BRS than those with low BRS (time to 50% death was 1.47-fold longer for the high BRS group than the low BRS counterparts). 15
Used MCAO rats and SP-SHRs, we now demonstrate that baroreflex function is important in the DR-mediated protection against stroke. The release of proinflammatory cytokines is an important mechanism involved in the development of stroke. Data show that C-reactive protein, IL-6, leukocyte elastase, lipoprotein (a), intercellular adhesion molecule −1, E-selectin, and other hemostatic and rheological factors are consistently higher in people who developed stroke compared with those who remained free of stroke and heart diseases. 26 And the deficiency of E-selectin can alleviate the cerebral injury. 29 Interleukin-6 and TNFα are also reported to be associated with risk of recurrent ischemic stroke independently of conventional risk markers. 26 So the reduction of proinflammatory cytokines might be beneficial for stroke treatment. In our experiments, DR significantly reduced the release of proinflammatory cytokines, which was also confirmed by other clinical study.21,24 We also concluded that arterial baroreflex dysfunction increased the release of proinflammatory cytokines in MCAO rats, and attenuated the beneficial effect of DR on proinflammatory cytokines. But how does arterial baroreflex control these cytokines has not been fully understood.
Recently, ’cholinergic antiinflammatory pathway’ become as a critical regulator of inflammation. Activation of cholinergic signaling inhibits the overproduction of TNF and other proinflammatory cytokines through α7 nicotinic acetylcholine receptor (α7nAChR). 30 Our recent data showed that arterial baroreflex dysfunction lead to a decrease not only in reflex activity but also in tonic activity of vagal nervous system. 31 The endogenous ligand for α7nAChR is acetylcholine. It was found that the expression of vesicular acetylcholine transporter markedly decreased in the brain of SAD animals. These results indicate that the dysfunction of arterial baroreflex related an inactivation of α7nAChR. 31
Dietary restriction also alleviate age-related diseases by affecting a variety of molecular and physiological targets beyond arterial baroreflex. 1 Among these targets, sirtuins may be the most important.1,6 Many studies, most performed in lower organisms, such as Saccharomyces cerevisiae and Drosophila, show that activation of sirtuins is a critical mechanism of DR-induced prolongation of lifespan.32,33 In Caenorhabditis elegans, extra copies of sir-2.1 were shown to increase lifespan. 34 Sinclair's group reported that resveratrol could extend the life in worms and flies via sirtuins.35,36 Mammalian species have seven sirtuins (SIRT1–SIRT7); of these, SIRT1 is activated by DR, and has been implicated in the beneficial effects of DR. 6
An earlier study demonstrated that DR could increase the expression of SIRT1, and prevent neuropathology in an animal model of Alzheimer disease. 37 The overexpression of SIRT1 also attenuated β-amyloid content in Alzheimer disease. But the authors did not provide a direct link between the beneficial effect of DR in Alzheimer disease and SIRT1 activity. In the present study, we showed, by using sirt1 KO mice and a representative SIRT1 activator, that the protective effects of DR against stroke are independent of SIRT1. Recently, several other investigators have questioned the role of sirtuins in lifespan prolongation or in mediating the beneficial effects of DR.38–40 Guarente's group reported that the overexpression of SIRT1 in mice did not increase the lifespan, although the mice were apparently healthier upon aging. 38 Burnett et al 39 showed that DR could increase the lifespan of flies via a sirtuins-independent mechanism. Harrison and colleagues 40 failed to show that resveratrol could alter the lifespan in mice. It appears that the function of SIRT1 and its relationship with DR is more complicated than expected.
In conclusion, the present work indicates that the beneficial effects of DR against stroke are SIRT1-independent, and the arterial baroreflex is involved in the protective effects of DR against stroke.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict interest.
Supplementary Information accompanies the paper on the Journal of Cerebral Blood Flow & Metabolism website (http://www.nature.com/jcbfm)
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
