Abstract
Hypothalamic glucose detection participates in maintaining glycemic balance, food intake, and thermogenesis. Although hypothalamic neurons are the executive cells involved in these responses, there is increasing evidence that astrocytes participate in glucose sensing (GS); however, it is unknown whether astroglial networking is required for glucose sensitivity. Astroglial connexins 30 and 43 (Cx30 and Cx43) form hexameric channels, which are apposed in gap junctions, allowing for the intercellular transfer of small molecules such as glucose throughout the astroglial networks. Here, we hypothesized that hypothalamic glucose sensitivity requires these connexins. First, we showed that both Cxs are enriched in the rat hypothalamus, with highly concentrated Cx43 expression around blood vessels of the mediobasal hypothalamus (MBH). Both fasting and high glycemic levels rapidly altered the protein levels of MBH astroglial connexins, suggesting cross talk within the MBH between glycemic status and the connexins' ability to dispatch glucose. Finally, the inhibition of MBH Cx43 (by transient RNA interference) attenuated hypothalamic glucose sensitivity in rats, which was demonstrated by a pronounced decreased insulin secretion in response to a brain glucose challenge. These results illustrate that astroglial connexins contribute to hypothalamic GS.
INTRODUCTION
The hypothalamus has a pivotal role in energy homeostasis. In response to energy-related stimuli such as hormones and nutrients, the hypothalamus modulates multiple neuroendocrine responses as well as food intake. 1 Among these energy-related clues, glucose constitutes an important signal. Hypothalamic detection of increased blood glucose level induces a set of rapid physiologic responses, including reduced hepatic glucose production, 2 increased insulin secretion, 3 and increased energy expenditure through thermogenesis. 4 It has been suggested that glucose-sensing (GS) neurons that modulate their electrical activity in response to changes in extracellular glucose levels 5 have a critical role in these physiologic responses. Glucose-sensing neurons are mostly located within specific hypothalamic nuclei. The most studied hypothalamic nuclei are the arcuate (ARC) and ventromedial nuclei (VMN), which together constitute the mediobasal hypothalamus (MBH). 1 Moreover, altered hypothalamic GS is an early defect observed in some animal models of insulin resistance and type 2 diabetes,6, 7, 8 lending support to the idea that this sensing is essential to maintain energy homeostasis.
Astrocytes have been identified as partners of GS neurons in hypothalamic glucose detection. For instance, increasing brain glucose levels by a carotid injection of glucose (thus avoiding the effects of peripheral glucose detection) activates some neurons and astrocytes within the ARC, as revealed by increased c-fos proto-oncogene expression. 9 This ARC activation and the ability of increased hypothalamic glucose to induce insulin secretion were both reversed by a preliminary injection of the xenobiotic amino acid methionine sulfoximine, a glutamine synthetase blocker that alters glial metabolism. 9 In addition, inhibition of the hypothalamic astroglial-specific glucose transporter GLUT1 is associated with a decrease in lowered hepatic glucose production. 10 Moreover, the inhibition of the low affinity GLUT2 in astrocytes was shown to alter insulin and food intake in response to increased central glucose levels.11, 12 Thus, these data support the idea that even though interstitial glucose diffusion occurs, 13 glucose taken up by hypothalamic astrocytes participates in the detection of the increase in blood glucose levels and in the generation of appropriate responses.
These hypothalamic astrocytes, as well as tanycytes lining the third ventricle, express key factors involved in sensing and processing nutritional signals.14, 15 These cells also have high plastic properties depending on their metabolic and hormonal environment 16 but are often considered as single cells. However, several lines of evidence demonstrate that astrocytes are organized into coordinated networks because of a high expression level of gap junction proteins. 17 This networking provides a conduit for the exchange of numerous active molecules, including glucose, 18 and is controlled by multiple factors.17, 19 Astroglial network exchanges comprise gap junction plaques formed by numerous channels, which are made up of connexins 43 and 30 (Cxs). These proteins form hemi-channels embedded in the plasma membrane, establishing an intercellular pathway by facing each other at the interface of two adjacent astrocytes. 17
Recently, it has been demonstrated by
We hypothesized that Cxs expressed in astrocytes have a role
MATERIALS AND METHODS
Animals
Experiments were approved by the Animal Use and Care Advisory Committee of the University of Burgundy (according to the European Communities Council Directive (2010/63/UE). Nine-week-old adult male Wistar rats (250–275 g; Charles River Laboratories, Lyon, France) were housed under standard animal care conditions with
Metabolic Conditions
Anesthesia was performed via an intraperitoneal pentobarbital injection (50 mg/kg, Ceva, Velaine en Haye, France). Anesthetized rats were injected subcutaneously with saline (controls) or a glucose solution (4 g/kg) that induced hyperglycemia for 3 hours. Only rats showing glycemia above 10 mmol/L were used for the study. Another group of animals were fasted for 24 hours (food removed at the beginning of the light phase) and compared with fed rats. At the end of the experiment, the animals were euthanized; the brains were quickly removed; and the MBH or areas of interest (cortex, thalamus, hippocampus, cerebellum, hindbrain) were dissected, snap frozen, and stored at −80 °C until western blot analysis.
Stereotaxic Surgery and Small Interfering RNA Injection
Cranial surgery was performed under isoflurane anesthesia (2% to 2.5%, Abbott, Rungis, France) at a flow rate of 1 L/minute oxygen. Once fully anesthetized, rats received a subcutaneous injection of buprenorphine (0.03 mg/kg, Axience, Pantin, France). The skin on the top of the skull was disinfected and lidocaine injected (10 mg/kg, Ceva, Libourne, France). A bilateral cannula guide (10 mm length, 26-gauge; Plastic-One, Roanoke, VA, USA) was inserted into the arcuate nucleus (coordinates: −3.1 mm posterior to the bregma,±0.4 mm lateral of midline, and −9.0 mm below the skull surface). The cannula guide was adhered to the skull with dental cement. At the end of the surgery, saline was injected subcutaneously to limit dehydration. After 5 days of recovery (monitored through daily food intake and weight gain), small interfering RNAs directed against connexin 43 (siCx43) were injected through the cannula guide in the arcuate nucleus using a transferring agent as described previously.
22
Briefly, siCx43 consisted of double-stranded 21-nucleotide siRNAs (800 pmol) that had previously been demonstrated to knock down rat Cx43 expression
Intracarotid Glucose Injection-Induced Insulin Secretion
We have previously established an
Immunohistochemistry
Anesthetized animals were perfused transcardially with heparinized PBS (Eurobio, Courtaboeuf, France, and 50 U/mL heparin, Sanofi Aventis, Paris, France). Brains were quickly removed, frozen in −25°C isopentane and stored at −80°C until use. After embedding the brain in Tissue-Tek at −20°C (Sakura, Villeneuve d'Ascq, France), the hypothalamus was cut into 20-
Western Blot
Frozen samples from rat brain were homogenized in lysis buffer (150 mmol/L NaCl, 1% Triton X-100, 0.5% sodium deoxycholate, 50 mmol/L Tris, pH8, 10
Biochemical Analysis
Plasma glucose concentrations were measured using the glucose analyzer Performa AccuChek (Roche, Meylan, France). Plasma insulin concentrations were determined using an ultrasensitive ELISA test (AlpCo, Eurobio, France).
Statistical Analysis
Statistical analyses were performed with GraphPad Prism 4.0 for Windows (GraphPad Software, La Jolla, CA, USA). Data are presented as the mean±sem. After testing normality, Student's unpaired or paired
RESULTS
Glial Proteins Cx43 and Cx30 are Highly Expressed in the Mediobasal Hypothalamus
We first quantified astroglial Cx43 and Cx30 protein levels in different brain areas, including the MBH, cortex, thalamus, hippocampus, cerebellum, and hindbrain, using western blotting (Figure 1A). The relative MBH Cx43 protein levels from the highest to the lowest (MBH taken as the reference, 100%,
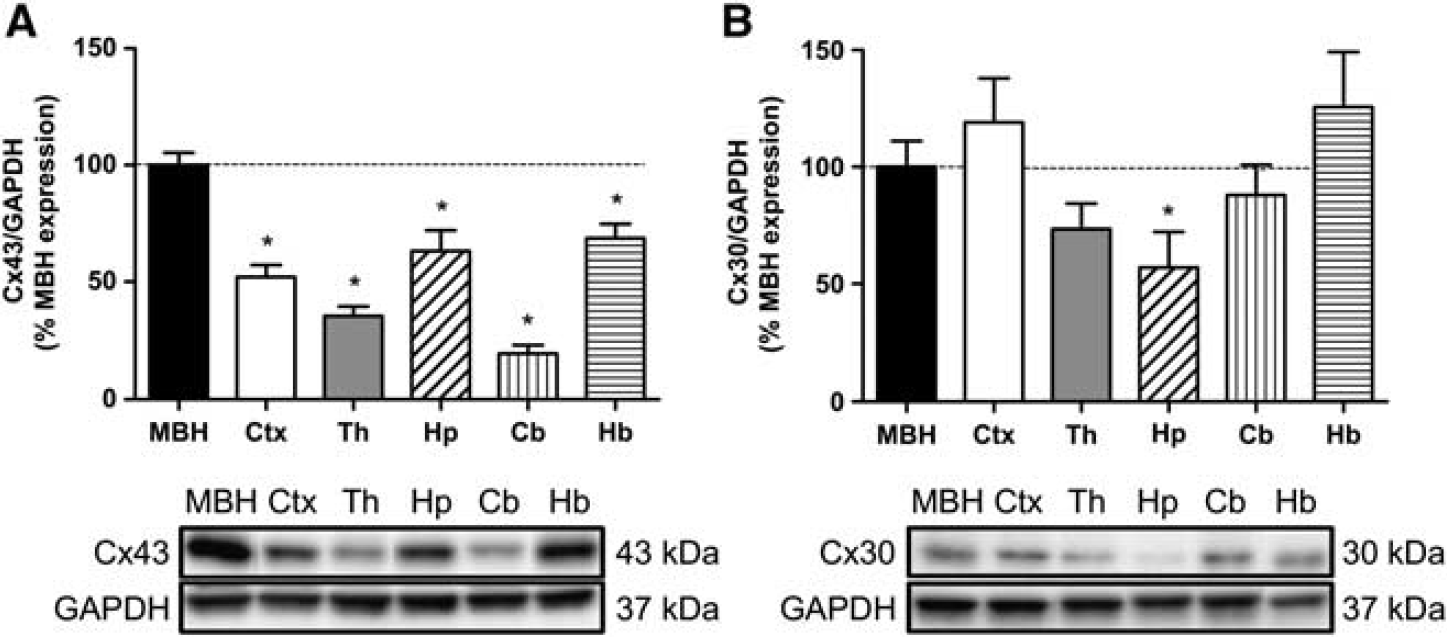
Cx43 and Cx30 protein levels in various brain areas. (
Cx43 and Cx30 are Distributed Differently in the Hypothalamus
To get further insights on the astroglial distribution of proteins Cx43 and Cx30 throughout the hypothalamus, immunohistochemical studies were performed on brain slices containing the hypothalamus. Cx30 and Cx43 proteins exhibited a punctate staining throughout the hypothalamus (Figures 2A and 2C). Nevertheless, the VMN and the ARC of the MBH displayed a high concentration of Cx43-labeled structures, especially on the edge of the medial part of the third ventricle (Figure 2A), whereas Cx30 staining appeared more uniformly distributed throughout the hypothalamus (Figure 2C). The parenchyma exhibited a high density of Cx43 immunoreactivity that appeared to delineate capillary-like structures, particularly in the ARC and VMN (Figure 2A, insets a and a′). Double immunostaining against the von Willebrand Factor, a marker of endothelial cells, revealed that this specific pattern of Cx43 immunoreactivity was located around capillaries (Figure 2B). This observation was specific to Cx43 because Cx30 immunolabeling displayed a uniform punctate staining that was higher in the ARC (Figure 2C, inset) and completely absent from the median eminence and the edge of the third ventricle (Figure 2C).
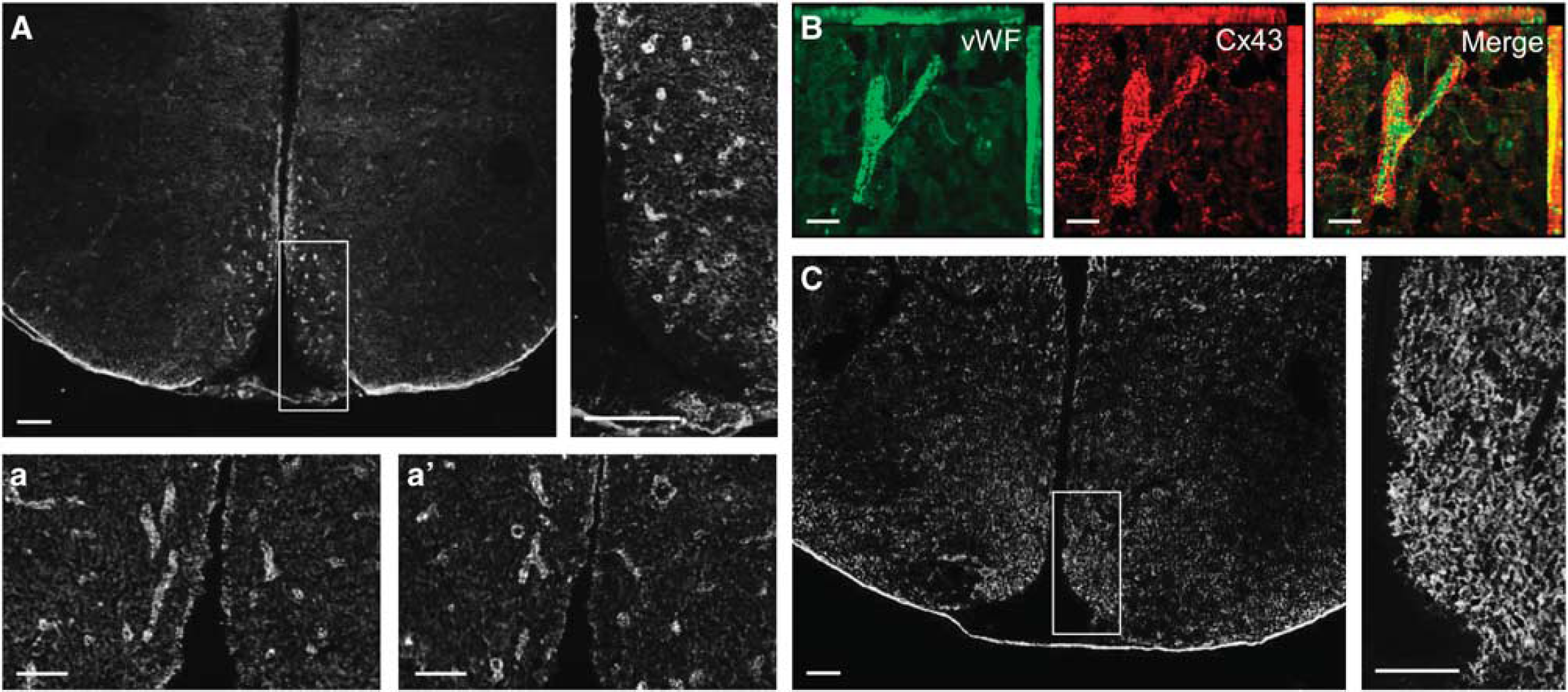
Hypothalamic Cx43 and Cx30 distribution. (
Fasting Decreases and Hyperglycemia Increases Cx43 in the Mediobasal Hypothalamus
We next examined whether the expression levels of both Cx43 and Cx30 were affected by changes in the metabolic status. We tested the effect of 24-hour fasting and 4-hour refeeding after overnight fasting. Although Cx30 expression levels were not affected by these conditions, MBH Cx43 protein levels significantly decreased (−29.4±3.4%,
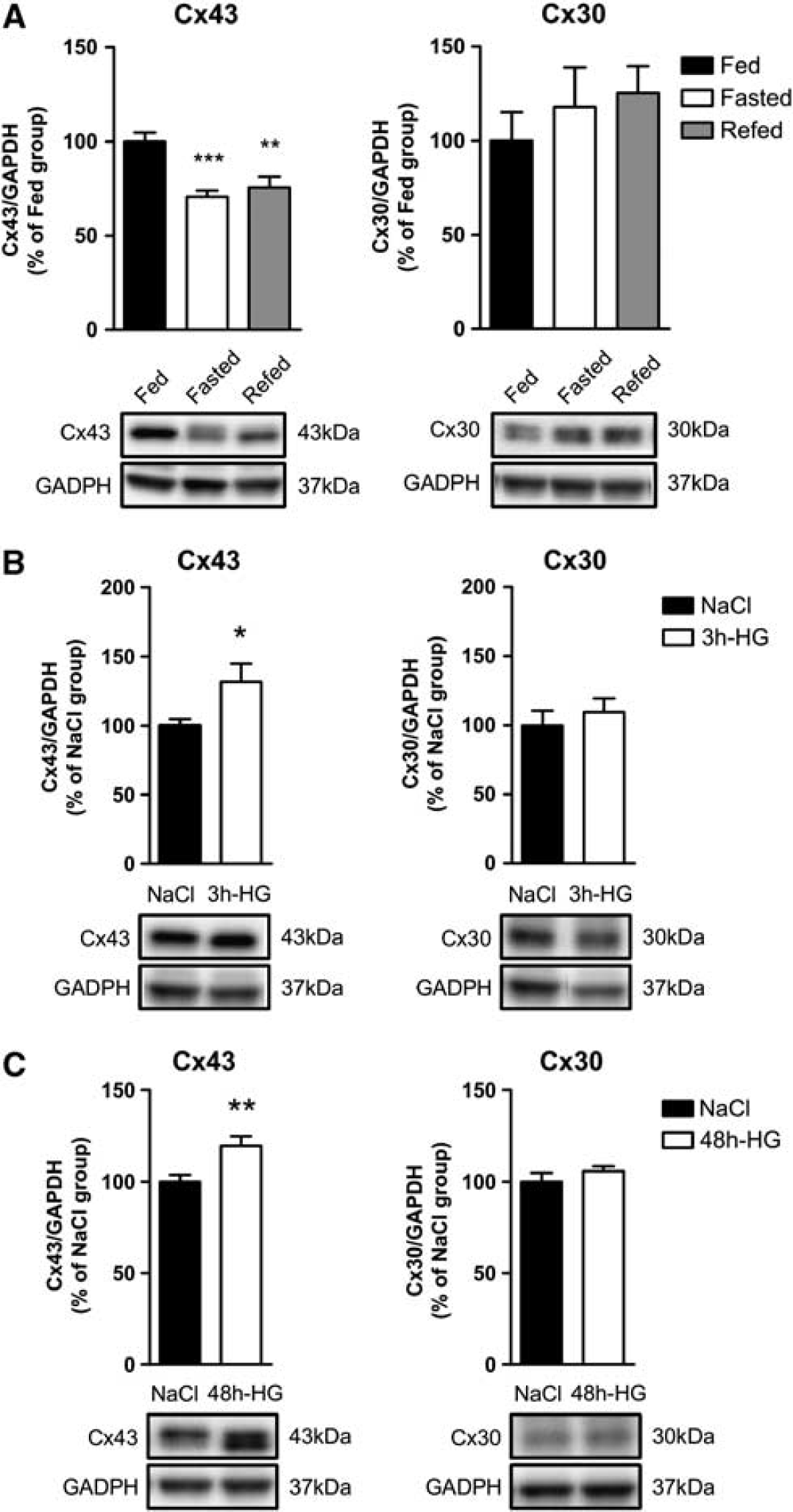
Changes in metabolic status and blood glucose concentration alter mediobasal hypothalamus (MBH) Cx43 and Cx30 protein levels. Relative protein level quantification (upper panel) and representative western blots (lower panel) of MBH Cx43 (left panel) and Cx30 (right panel) in panel
Astroglial Cx43 Transient Inhibition in the Mediobasal Hypothalamus is Associated with Alterations in Hypothalamic Glucose Sensing
To determine whether Cx43 is involved in hypothalamic GS, we transiently inhibited MBH Cx43 expression using RNA interference. Hypothalamic glucose sensitivity was assessed 72 hours post injection when animals fully recovered from siRNA injection (Supplementary Figure S1). At this time, Cx43 protein levels were significantly decreased by 34.2±4.1% (
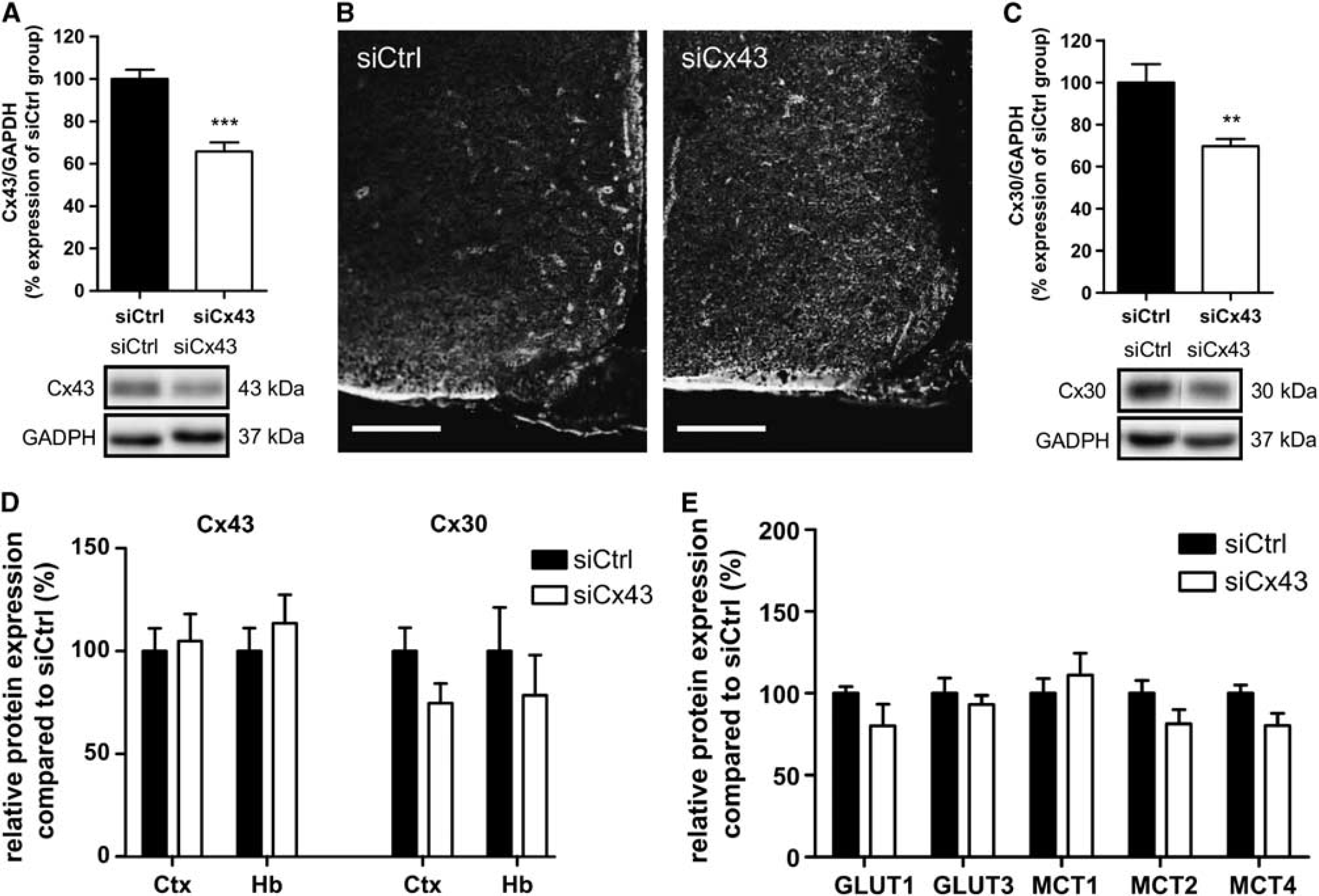
Transient mediobasal hypothalamus (MBH) Cx43 inhibition decreases protein levels of astroglial connexins Cx43 and Cx30, without altering main glucose and lactate transporters expression. (
We next evaluated the impact of siCx43 inhibition on hypothalamic GS. We quantified the insulin secretion in response to an intracarotid glucose load toward the brain that does not alter peripheral blood glucose levels (and therefore beta cell stimulation by glucose).
3
Neither basal glycemia nor basal insulinemia were modified in siRNA-treated rats (Supplementary Figure S2). Both siCtrl- and siCx43-injected animals displayed stable glycemia during the test (Figure 5A). siCtrl-injected rats exhibited a characteristic increase in insulin secretion 1 minute after the glucose load (+40.9±6.2
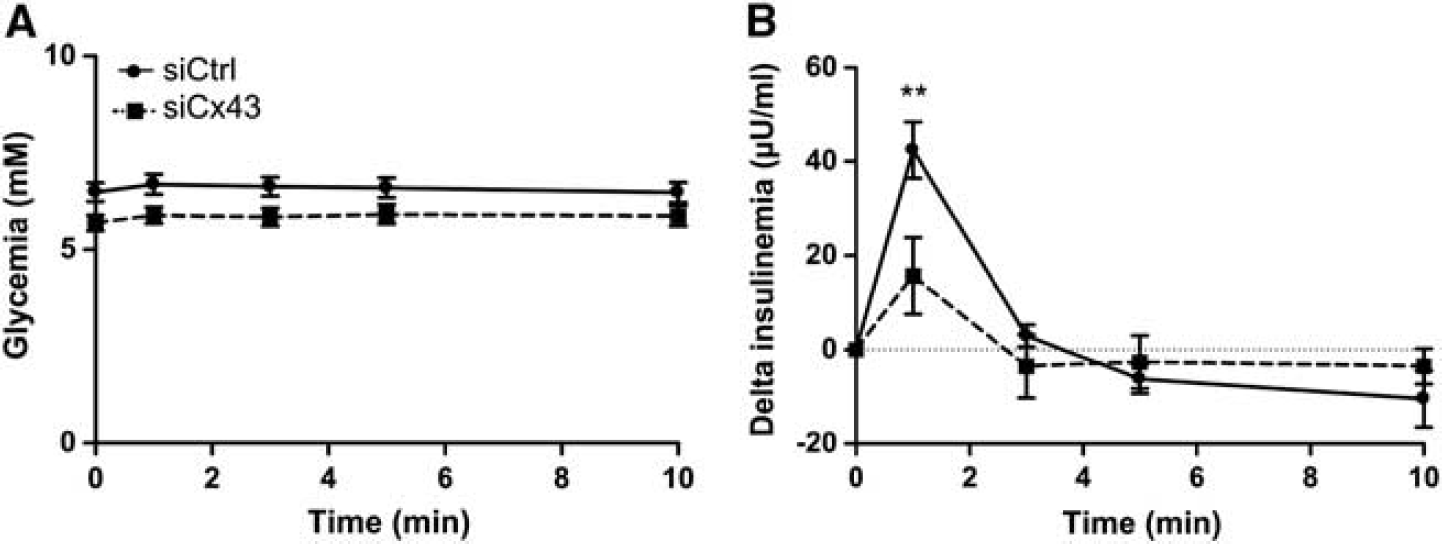
Transient downregulation of mediobasal hypothalamus (MBH) Cx43 inhibits hypothalamic glucose sensing
DISCUSSION
Over the last few years, there has been a significant improvement in the knowledge of cellular and molecular mechanisms involved in hypothalamic glucose sensitivity. We and others have identified astrocytes as being important factors in hypothalamic GS.2, 9, 10, 11 Likewise, astroglial gap junctions formed by Cx43 and Cx30 are necessary for glucose transfer through astroglial networks, as recently demonstrated in the hippocampus. 20 Thus, we hypothesized that Cxs-dependent astroglial networks have a role in the hypothalamic glucose detection. Here, we show that (1) the MBH is highly enriched in Cx43; (2) changes in blood glucose levels modulate Cx43 protein levels; and importantly, (3) decreased MBH Cxs expression inhibits the increase of brain glucose-induced insulin secretion.
A previous analysis of Cxs-rich structures in the brain reported high levels of Cxs protein in the hypothalamus.
26
Here, we show that in hypothalamic areas, strong immunostaining for Cx43 was restricted to the MBH. This labeling exhibited a pattern, confirmed by co-staining of endothelial cells, that suggested that astroglial endfeet are enriched with Cx43, as previously described.20, 26, 27 Endfeet are specialized astroglial processes apposed to the brain capillaries. They are part of the blood–brain barrier and constitute a highly regulated diffusion barrier between vasculature and neuronal tissue.28, 29 Mathematical simulations based on their
Changes in metabolic status have been shown to alter MBH glucose sensitivity.8, 10, 33, 34 Here, we examined whether Cxs protein levels were differently affected by changes in blood glucose levels. We found a positive correlation between blood glucose concentration and MBH Cx43 protein levels. A fasting-induced decrease in blood glucose levels attenuates MBH Cx43 expression, whereas short- (3 hours) or long-term (48 hours) hyperglycemia increases MBH Cx43 expression. Nevertheless, it is noteworthy that by opposition to the 3-hour hyperglycemia associated with increased Cx43 protein levels, mild hyperglycemia obtained after 4-hour refeeding was not sufficient to increase Cx43 protein levels. Blood glucose levels after 4 hours of refeeding did not exceed 7.5±0.2 mmol/L (
Because only MBH Cx43 expression levels were affected by metabolic status, we aimed to determine whether inhibition of MBH Cx43 expression alters glucose sensitivity. MBH Cx43 protein levels were significantly decreased 72 hours after MBH siRNA injection, as confirmed by western blot and immunohistochemistry. When using Cx43-deficient mice, a compensatory upregulation of Cx30 has been reported.
37
Surprisingly, we found that a decrease in siCx43-induced MBH Cx43 levels is associated with a decrease in Cx30 protein levels. The siRNA sequence we used does not match that of the Cx30 RNA sequence, suggesting that a decrease in Cx43 may indirectly affect Cx30 expression by a signaling pathway yet to be determined. A recent study by Ezan
Under basal conditions, astroglial MBH Cxs inhibition did not affect glycemia and insulinemia of siCx43-injected rats (Supplementary Figure S2). In contrast to siCtrl animals, siCx43 animals exhibited a strong decrease in increased insulin secretion in response to an intracarotid glucose injection toward the brain, suggesting that glucose detection was incomplete. In this experiment, even if gap-junction activity might be modified to compensate for decreased Cxs protein levels, it is not sufficient to ensure a complete sensing of increased hypothalamic glucose levels. Although it is not possible to infer this reduction to one or the other Cx because both Cx43 and Cx30 expression were inhibited in siCx43-treated animals, this result suggests that the MBH astroglial networks overall are necessary for GS.
With regard to the underlying molecular mechanisms by which Cxs are needed for hypothalamic glucose detection, tanycyte glucose detection has been shown
Finally, this study highlights the importance of the astroglial connexins to ensure proper hypothalamic GS and suggests that astroglial networks form a complex metabolic-sensing unit with GS neurons. Collectively, these
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
CA conducted and designed
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
