Abstract
Although gait changes are considered as reliable indices of stroke severity and efficacy of rehabilitative therapies in humans, data from animal models of focal ischemia are lacking. To determine the effect of stroke on gait function in adult rats with distal middle cerebral artery occlusion (MCAO), we assessed the longitudinal changes in gait using an automated computer-assisted gait analysis system. Four days after MCAO, intensity, maximal area of contact, and paw angle were significantly decreased in the affected forepaw whereas the double support time using ipsilateral paws increased. Relative print positions between the fore and hindpaws and limb coupling were also altered, suggesting that stroke affected sensorimotor status and reduced limb coordination. Except for paw angle, these impairments persisted for at least 5 weeks. However, unlike human stroke patients, stride length and stance were not affected in rats with MCAO. Environmental enrichment for 4 weeks improved paw contact area and relative print position of the affected limb and reduced double support time in ischemic rats compared with controls. Our results indicate that gait analysis provides great insight into various aspects of motor function changes in ischemic quadrupeds, and that environmental enrichment is beneficial in reducing a variety of poststroke gait impairments possibly through enhanced structural plasticity mechanisms within injured cortical neuronal networks underlying locomotor function.
Keywords
Introduction
Gait impairment occurs as a result of many forms of neurodegeneration in humans, including Parkinson's disease, Alzheimer's disease, and ischemic stroke (Mah et al, 1999; Mirek et al, 2007; Scherder et al, 2007). Gait is not only indicative of lower extremity motor function, but also reflects cognitive function, particularly executive function. Intact cognition and attention processes are required to ensure proper gait control, the interplay between gait and cognition being often revealed when patients are evaluated under dual-task conditions (Plummer-D'Amato et al, 2008). Poststroke gait is characterized by temporal asymmetry, reduced walking velocity, and reduced stride length (Mah et al, 1999). For biped species like humans, impaired gait function not only reduces ambulation but also could lead to imbalance and falls, especially in elderly patients experiencing stroke or other neurodegenerative illnesses (Scherder et al, 2007). Although systematic gait analyses have been widely used in the clinical setting as important indices to evaluate the severity of stroke and the effect of intervention or rehabilitation on recovery, the extent of gait changes after stroke in experimental quadrupeds remains to be explored.
Methods of analyzing gait and ambulation in laboratory animals include assessing dynamic changes by scoring spontaneous open field locomotion, and by evaluating specific coordination performance while the animals walk either on a narrow beam, rope, grid, horizontal ladder, rotating rod, or over a runway (for review see Corbett and Nurse, 1998). The static changes of gait have been revealed mainly by analyzing footprints using ink (Johnston et al, 1991), X-ray (de Medinaceli et al, 1982), cinematography (Gruner et al, 1980), or scanner (Grasso et al, 2004). However, these tests are tedious to administer and only measure a narrow spectrum of gait characteristics. In contrast, the CatWalk method provides an automated and comprehensive way to assess gait function with the benefit of measuring a number of locomotor-related parameters simultaneously (Hamers et al, 2006). It can not only detect the dynamic as well as static aspects of gait as the human gait analysis systems, but also the spatial and temporal aspects of interlimb coordination that are particularly valuable for quadrupeds. Although the CatWalk method has been used in a variety of rodent studies for assessing impaired gait function after spinal cord injury (Hamers et al, 2006), pyramidotomy (Starkey et al, 2005), Parkinson's disease (Vlamings et al, 2007), neurotmesis (Deumens et al, 2007), and neuropathy (Gabriel et al, 2007), it has not yet been used in experimental stroke. Assessing gait changes in quadrupeds after experimental stroke would provide complementary information regarding motor function status that is directly relevant to clinical studies.
Beneficial effects of exposure to enriched environment (EE) are well documented and have proven successful in compensating behavioral deficits associated with various neurodegenerative diseases. Postischemic EE facilitates functional recovery, increases dendritic branching and spine density in pyramidal neurons of layers II to III in the contralateral cortex (Johansson and Belichenko, 2002), preserves cortical sensorimotor representation maps (Xerri and Zennou-Azogui, 2003), and enhances ischemia-induced neurogenesis (Komitova et al, 2005; Matsumori et al, 2006). Enriched environment combined with intensive motor training has also been reported to attenuate skilled forelimb reaching impairments and to increase branching of cortical neurons in the undamaged hemisphere after focal ischemic injury (Biernaskie and Corbett, 2001). In addition to ischemic stroke, EE has also shown dramatic beneficial results in many experimental models of brain injury including Huntington's disease, Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis, 1937 epilepsy, traumatic brain injury, spinal cord injury, fragile X syndrome, and Down's syndrome (for review see Nithianantharajah and Hannan, 2006). In the present study, we provide a detailed analysis of the changes in specific gait parameters after experimental stroke using the CatWalk-assisted automated gait analysis system and also show that EE is efficacious in promoting gait function recovery in adult rats.
Materials and methods
Animals and Housing
This study was conducted in accordance with the animal care guidelines issued by the National Institutes of Health and by the San Francisco Veterans Affairs Medical Center Animal Care and Use Committee. Adult male Sprague-Dawley rats (2.5 months of age, 230 to 240 g) from Charles River Laboratories Inc. (Wilmington, MA, USA) were housed in institutional standard cages (41 × 25 × 19 cm; 2 rats per cage) on a 12-h light/12-h dark cycle with ad libitum access to water and food before the experimental procedures. Forty rats were randomly assigned to 2 groups that were subsequently subjected to either focal ischemia (n = 22) or sham surgery (n = 18). One week after surgery, rats in each group were randomly divided into either standard housing (STD) environment or EE, resulting in 4 final experimental groups: sham-STD (n = 8), sham-EE (n = 10), middle cerebral artery occlusion (MCAO)-STD (n = 11), and MCAO-EE (n = 11). Rats assigned to the EE group were transferred to the special EE cages and housed for an additional 4 weeks. Each 2-story EE cage with dimensions of 76 × 56 × 77 cm, was equipped with a running wheel for spontaneous exercise, a three-dimensional labyrinth, bedding, a ladder, a house, chains, a hammock, wooden blocks as well as nylon bones and could accommodate 5 to 6 rats (Matsumori et al, 2006). The arrangement of movable objects was changed once a week. Rats assigned to the STD housing remained in the institutional standard cages for the same period of time as the EE group.
Focal Ischemia Model
Procedures to create the focal ischemia/reperfusion model were performed as described previously (Matsumori et al, 2006). Because a permanent distal ligation of the MCA does not give reproducible infarct in most rat strains, we combined it with a bilateral common carotid arteries occlusion. Anesthesia was induced with 3% isoflurane in a closed chamber and maintained with 2% isoflurane in 30% O2 and 70% N2O administered by a facemask. Rectal temperature was monitored and maintained at 37±0.5°C by a thermal blanket throughout the surgical procedure. The rat was placed in the supine position and a ventral cervical midline skin incision was made. Both the common carotid arteries were carefully isolated from the adjacent vagus nerve. The animal was then placed in the lateral position, and a 1.5-cm scalp incision was made at the midpoint between the left eye and the left ear. The temporalis muscle was separated in the plane of its fiber bundles and retracted to expose the zygoma and squamous bone. A 2-mm-diameter burr hole was made with a dental drill 1 mm rostral to the anterior junction of the zygoma and squamous bone. The dura mater was carefully pierced with a 30-gauge needle. The main trunk of the left MCA was exposed and ligated just above the rhinal fissure with a 10-0 suture. After suturing of the muscle and scalp incisions, the common carotid arteries were occluded for 60 mins by using 4-0 sutures. The suture was then removed to restore blood flow, and the cervical incision was closed. Sham-operated rats did not receive occlusion of either the MCA or the common carotid arteries.
Computer-Assisted Method for Gait Analysis
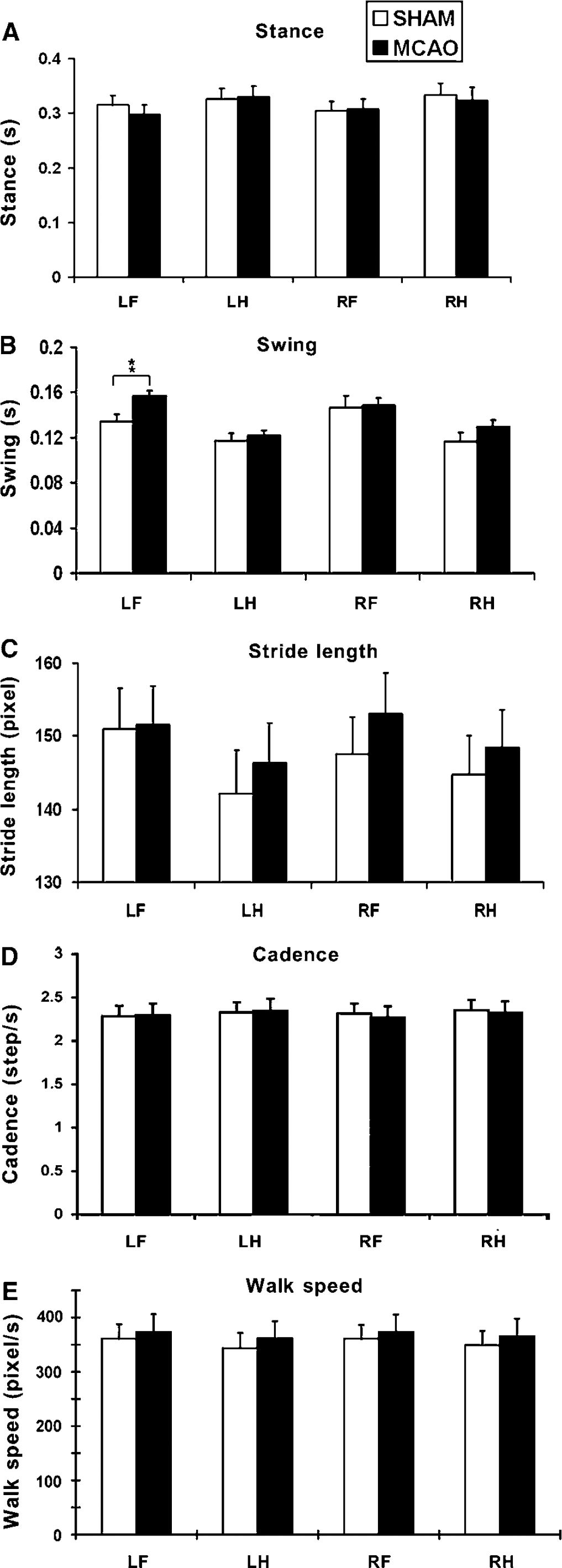
Rats were subjected to gait assessment at 4 days and 5 weeks after MCAO using the CatWalk-automated gait analysis system (Noldus Information Technology, Wageningen, The Netherlands). The apparatus is made of a 1.3-m-long glass plate with dim fluorescent light beamed into the glass from the side. In a darkened environment (below 20 lux of illumination), the light is reflected downward and the images of the footprints recorded by the camera under the walkway when the animal's paws come in contact with the glass surface. Rats were subjected to three consecutive runs of gait assessment at 4 days and 5 weeks after MCAO or sham surgery. The images from each trial were converted into digital signals and processed with a threshold set at 30 arbitrary units (ranging from 0 to 225, meaning all pixels brighter than 30 arbitrary units were used). After the identification and labeling of each footprint, a wide range of gait data was generated including the (1) spatial parameters related to individual paws (intensity, maximum area, print area, box width, box length, and paw angle); (2) relative spatial relationship between different paws (base of support, BOS, relative paw placement, and stride length); (3) interlimb coordination (step pattern, regularity index, and phase lag); and (4) temporal parameters (swing, stance, cadence, and walk speed), as detailed in Table 1.
The terminology and definition for gait parameters relevant to this study
LF, left fore; LH, left hind; RF, right fore; RH right hind.
Tissue Preparation
Animals were anesthetized with ketamine (80 mg/kg; Parke-Davis, Morris Plains, NJ, USA) and xylazine (20 mg/kg; Butler, Columbus, OH, USA) and perfused transcardially with 4% paraformaldehyde in 0.1 mol/L phosphate buffer (PB), pH 7.4. The brains were removed, fixed overnight in 4% paraformaldehyde—phosphate buffer and placed in 20% sucrose for 48 h. Coronal sections were cut at 40 μm on a microtome and collected serially.
Immunohistochemistry Staining and Infarct Volume Assessment
Serial coronal sections (480 μm apart) were immunostained with anti-NeuN antibody as described previously (Matsumori et al, 2006) for evaluation of infarct volume. The area was measured by unbiased stereology in the serial sections, and multiplied by the section interval thickness to obtain volume. Infarct volume was indirectly measured by subtracting the volume of intact tissue in the ipsilateral hemisphere from that in the contralateral hemisphere.
Statistical Analyses
Data were expressed as mean±s.e.m. All statistical tests were performed with StatView 5.0.1 software (SAS Institute Inc., Cary, NC, USA). Statistical significance was evaluated using one-way and two-way analyses of variance followed by post hoc paired comparisons using the Student—Newman—Keuls tests when appropriate. Linear correlations between weight, walk speed, cadence, and the other measures of gait were evaluated by determination of the Pearson's product—moment correlation coefficient using the SPSS 12.0 software (SPSS Inc., Chicago, IL, USA). Values of P <0.05 were considered as significant.
Results
The majority of the gait parameters were expressed for each paw, namely the left fore (LF), left hind (LH), right fore (RF), and right hind (RH) paw. The terminology and definition for gait parameters used in this study are listed in Table 1.
Ischemia Led to a Persistent Reduction in Paw Pressure and Area of Paw Contact
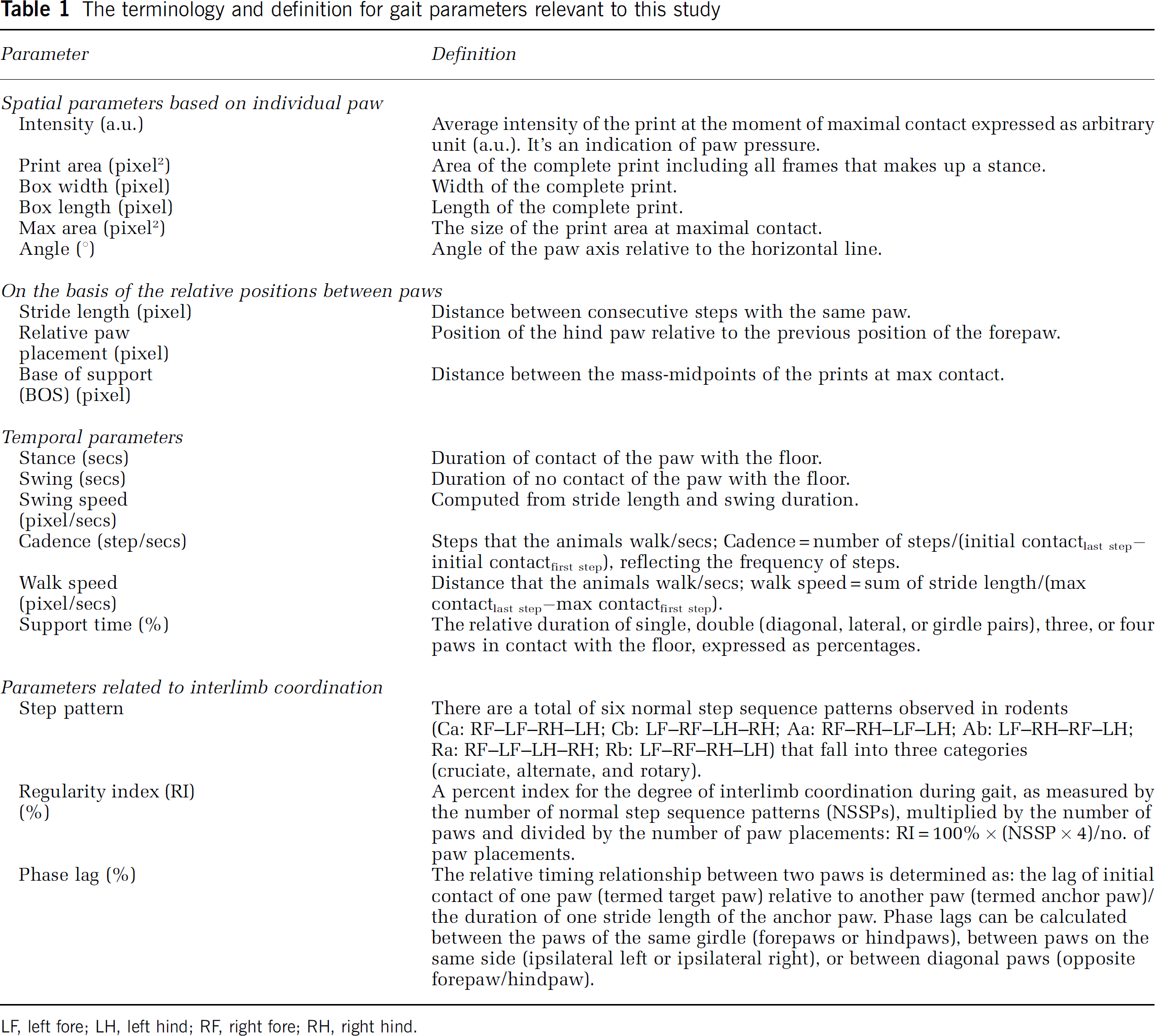
Intensity: Four days after ischemia, the paw pressure during floor contact, as measured by the intensity of the footprint, was significantly affected (treatment effect: F1,112 = 20.61, P < 0.0001; paw effect: F3,112 = 20.35, P < 0.0001). There was no significant treatment × paw interaction (F3,112 < 1), indicating that footprint intensity of all four paws in MCAO rats was similarly affected. Thus, paw pressure was generally lower than that of sham rats, although, a significant decrease in footprint intensity was only detected in the RF (affected) paw (P < 0.01; Figure 1A) contralateral to the ischemic cortical infarct that was systematically induced in the left hemisphere of all the MCAO rats. The intensity of the RF paw in MCAO animals remained decreased 5 weeks after ischemia (P < 0.05), indicating a persistent reduction in RF pressure during walking (Figure 1B).
Effects of ischemic stroke and EE on paw pressure, area of paw contacts, paw angle, and relative paw placement. Gait parameters were assessed at 4 days (
Max area: The size of the print area at maximal contact of all four paws of MCAO rats during floor contact was significantly smaller than that of sham rats at 4 days after ischemia (treatment effect: F1,112 = 49.22, P < 0.0001; paw effect: F3,112 = 5.37, P < 0.01). All four paws were similarly affected by ischemia as shown by a lack of treatment × paw interaction (F3,112 < 1; Figure 1C). Five weeks after ischemia, only the maximal area of the RF paw remained significantly smaller (P < 0.05; Figure 1D).
Print area and dimensions: The results of print area were globally similar to those of max area at 4 days after ischemia. Print areas of all four paws in MCAO rats were significantly smaller than those of sham rats, especially in the affected right side (main effect of treatment F3,112 = 35.02, P < 0.0001; Figure 1E). Five weeks after ischemia, the print area of the RF paw was still significantly smaller in MCAO rats than sham rats (P < 0.05; Figure 1F). Detailed analysis further revealed that the reduction in print area seen after ischemia likely resulted from a decrease in the width (called box width) and/or in the length (box length) of the paw print at either time points (data not shown). Ischemia significantly reduced box width of LH and RF paws 5 weeks after MCAO (P < 0.05 for both paws). It also reduced box length of RH 4 days (P < 0.01) and of RF 5 weeks (P < 0.05) after distal MCAO (data not shown).
Ischemia Altered the Relative Positions Between Paws
Angle: The sign of paw angle is determined by whether the paw stands inwards or outwards relative to horizontal line (Figure 1G). For the right paw if the paw is placed outward, the value is positive, if inward, the value is negative; for the left paw it is the other way around. In sham rats, the angle of the LF paw was negative and that of the RF paw was positive, with an absolute value ranging from 10° to 20° in the forepaws. In contrast, the absolute values of the hindpaws were near zero, suggesting that the sham rats usually walked with outward bilateral forepaws and almost straight bilateral hindpaws. Paw angles were attected differentially by the ischemia treatment as revealed by a significant treatment × paw interaction (F3,112 = 3.55, P < 0.02). Four days after ischemia, only the angle of the affected RF forepaw in MCAO rats was significantly smaller compared with that of the sham rats (P < 0.01; Figure 1G). However, the RF paw angle naturally recovered 5 weeks after ischemia regardless of housing conditions (P > 0.36 for all measures; Figure 1H).
Relative paw placement: In the sham rats, the hindpaws were always placed behind the forepaws of the same cycle, producing a negative value when subtracting the X-coordinates of the mass midpoints of ipsilateral hindpaws from forepaws at maximal contact. Four days after ischemia, the absolute values of relative paw placement were decreased (F1,56 = 9.08, P < 0.01) with a significant effect on the affected right side (P < 0.05), suggesting that the right hindpaw of the MCAO rats advanced in more distance from cycle to cycle compared with that of the normal rats (Figure 1I). Five weeks after ischemia, abnormal paw placement not only persisted in ischemic animals, but also was intensified and affected both left and right paws, even producing positive values for the relative paw placement index (left: F1,36 = 16.78, P < 0.001); right: F1,36 = 15.40, P < 0.001; Figure 1J). There was no significant difference in the distance between the bilateral paws, known as BOS, between the sham (4 days; forepaw: 30.9±1.1; hindpaw: 37.1±1.3) and MCAO rats (4 days; forepaw: 29.9±0.9; hind-paw: 36.20±1.6) at either 4 days (forepaw: P < 0.48; hindpaw: P > 0.72) or 5 weeks after ischemia (data not shown).
Ischemia Affected Interlimb Coordination and Increased the Double Support Time
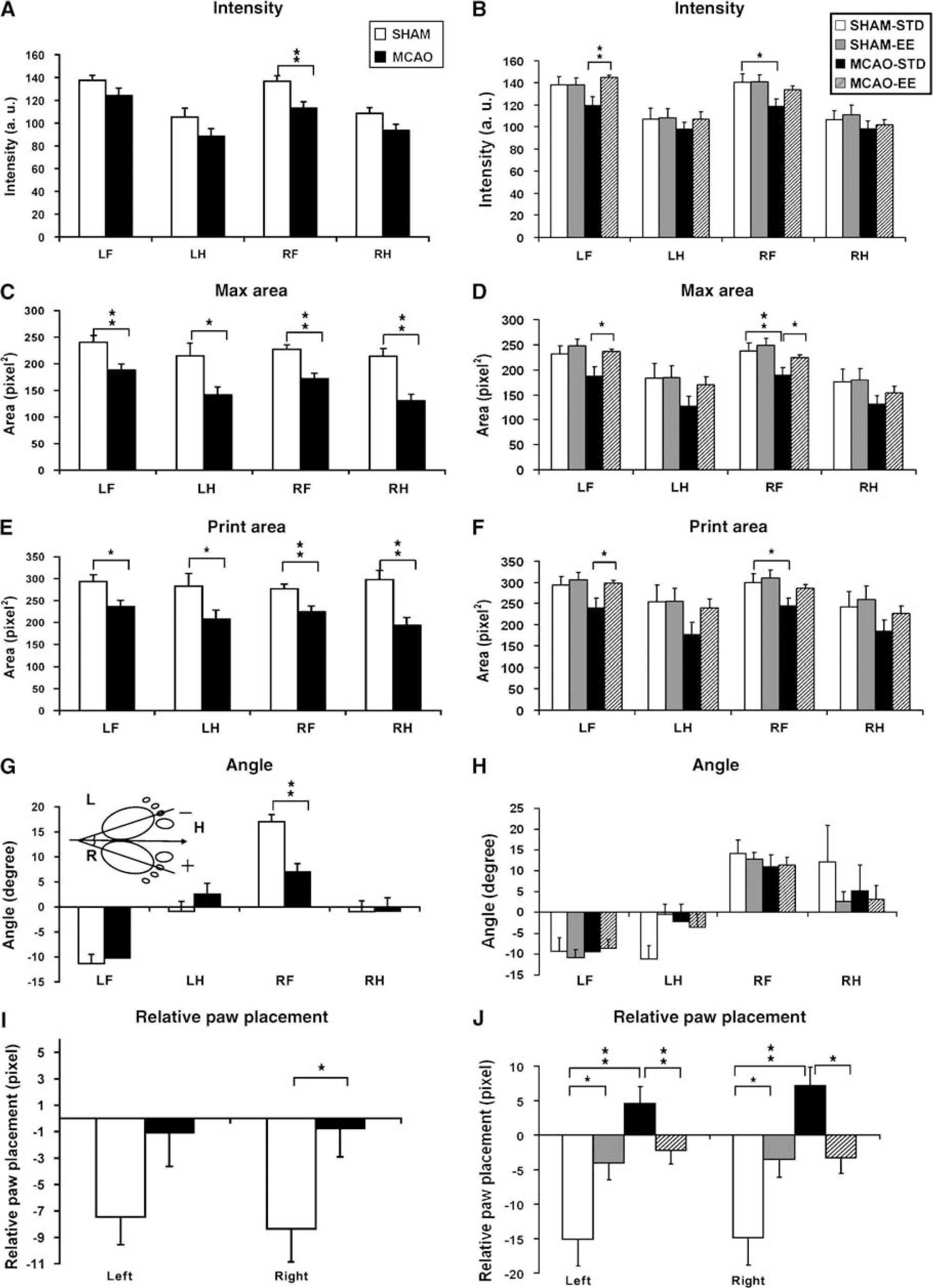
Phase lag: Phase lag is one of the coordination-related parameters that determine the temporal relationship between paw placements. In other words, phase lag, expressed as phase dispersion, reflects the time lag of initial contact of the target paw to the stride length of the anchor paw (Figure 2A). According to the CatWalk method, a total of six pairs of paw placement were compared for time dispersion. Four days after ischemia, the phase lag of girdle pair between RH and LH paws was significantly longer (LHanchor-RHtarget, F1, 28 = 4.34, P < 0.05) in the MCAO rats than the sham rats (Figure 2A), suggesting that the placement of the affected (right) hindpaw relative to unaffected hindpaw was significantly delayed compared with normal rats. This effect was only transient, as it was no longer observed 5 weeks after ischemia (Figure 2B). An opposite temporal pattern was observed for the ipsilateral pairs. Although no significant difference was observed 4 days after ischemia (Figure 2C), the phase lag in the ipsilateral pair (especially the right side) of the MCAO rats became significantly longer from that of sham rats 5 weeks later (F1, 36 = 12.94, P < 0.001; Figure 2D), likely because of an improvement of the timing in RF paw placement during walking over a 1-month period in the ischemic rats. There was no difference in the phase lag for the diagonal pairs at either time points (Figures 2E and 2F).
Effects of ischemic stroke and EE on interlimb coordination and double support time using the lateral pair of paws. Time relationship between two footfalls (expressed as phase dispersions) and sequential step patterns were assessed at 4 days (
Step pattern: There are six step-sequence patterns described in rodents, as indicated in Figure 2G. The most frequently observed step pattern was the alternate pattern Ab, which accounted for 88.7% of all step patterns in the sham rats and 81.3% in the MCAO rats at 4 days after ischemia. The overall patterns in the MCAO rats were more diverse than the sham rats, with a 3.6% in rotary patterns (Ra and Rb) that are considered as normal step sequence patterns but that were never observed in our sham rats. Five weeks after distal MCAO, the frequency of the rotary pattern decreased and frequency of the Ab pattern slightly increased in the MCAO rats (Figure 2H). There was no significant difference in the regularity index (RI), the degree to which the animals use normal step sequence patterns, between the sham and MCAO rats at either 4 days (sham: 99.4±0.30; MCAO: 99.7±0.16; P > 0.37) or 5 weeks after ischemia (sham-STD: 99.45±0.36; MCAO-STD: 99.85±0.15; P > 0.27).
Support time: The longest simultaneous usage of paws during floor contact was with three paws, followed by two (diagonal, lateral, or girdle pairs), then four, paws in both sham and ischemic rats (Figure 2I). Rats rarely supported themselves with only a single paw at any given time during standing. However, MCAO rats were supported longer with 2 ipsilateral paws compared with sham rats at 4 days after ischemia (P < 0.05; Figure 2I). There was no between group difference in support time with either one, three, or four paws (Figure 2I). Five weeks after ischemia, the preference in standing with ipsilateral paws still persisted in the MCAO rats that remained in the STD (P < 0.05; Figure 2J). At both the time points, sham rats tended to be supported more by the girdle pair of paws than did the MCAO rats, although this trend did not reach statistical significance (4 days: P = 0.08; 5 weeks: P = 0.22; Figures 2I and 2J).
Ischemia Had Little Effect on Stance, Stride Length or Gait Velocity
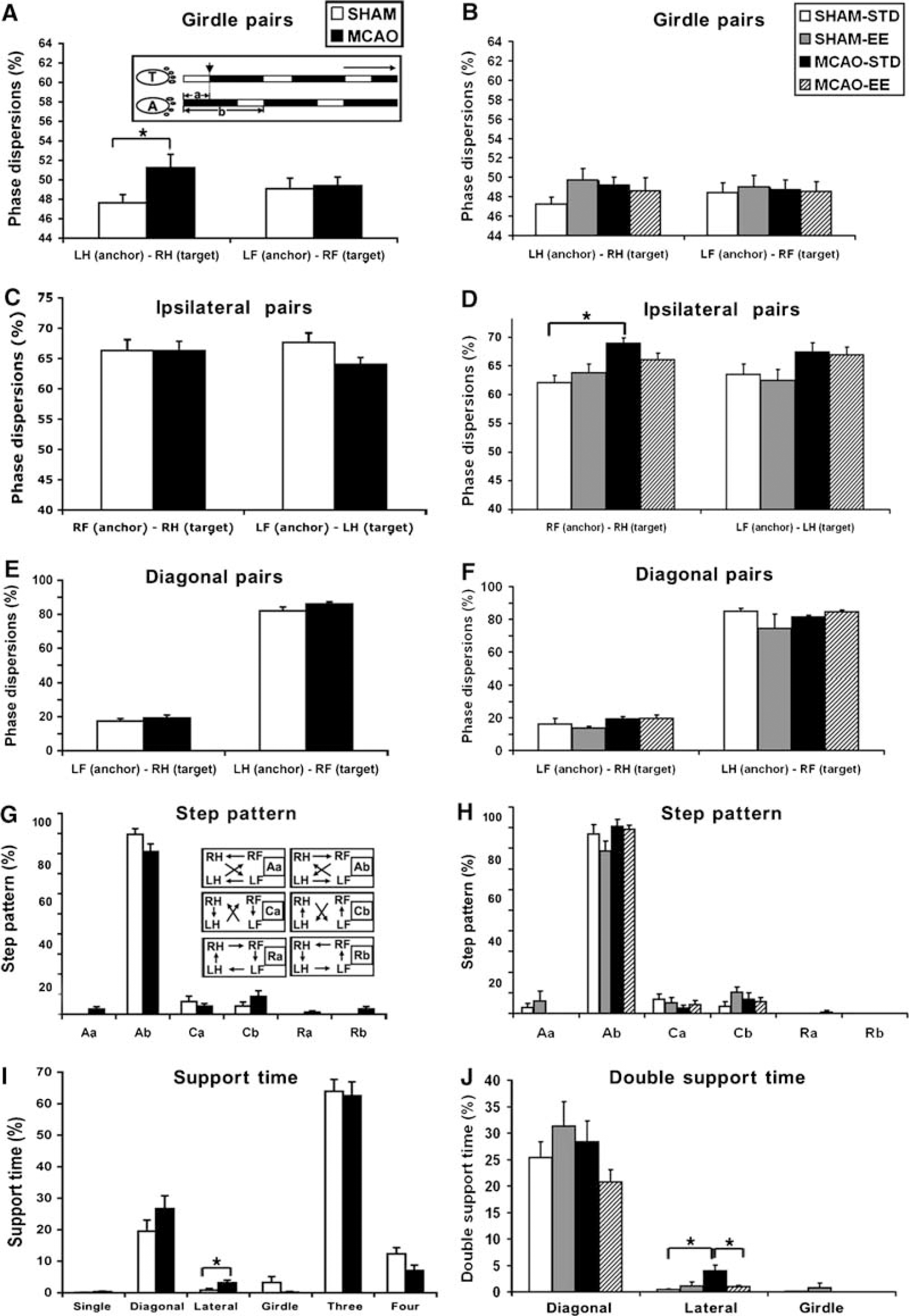
Temporal parameters: The duration of the stance did not change significantly for either forepaws or hindpaws in the MCAO rats at either 4 days (F1, 112 = 0.73, P = 0.39; Figure 3A) or 5 weeks (F1, 36 = 1.60, P = 0.21, data not shown). The swing phase was increased only in the LF paw at 4 days after ischemia (P < 0.01) (Figure 3B) but recovered 5 weeks later (P = 0.69, data not shown).
Stroke did not significantly affect temporal gait parameters. There was no significant difference in stance (
Stride length: Contrary to finding in stroke patients (Mah et al, 1999), there was no significant difference between MCAO and sham rats in the distance between successive placements of the same paw during maximal contact, known as stride length, at 4 days (F1, 112 = 0.32, P = 0.57; Figure 3C) or 5 weeks after ischemia (F1, 36 = 3.52, P = 0.07, data not shown).
Velocity: Because gait parameters are influenced by the speed of walking, we determined the effect of ischemia on cadence (steps/secs) and walk velocity (distance moved/secs). Unlike the findings reported in human stroke patients, there was no significant difference in either cadence (F1, 112 = 0.02, P = 0.88) or walk velocity (F1, 112 = 0.45, P = 0.51) between sham and MCAO rats at 4 days (Figures 3D and 3E) or 5 weeks after ischemia (cadence: F1, 36 = 1.43, P = 0.24; walk speed: F1, 36 = 2.71, P = 0.11, data not shown).
Enriched Environment Reduced PostStroke Gait Impairment
Consistent with our previous finding (Matsumori et al, 2006), there was no significant difference in the size of infarct between MCAO rats housed in either environment (infarct volume in STD: 66.36±6.52 mm3; EE: 63.4±7.03 mm3; P > 0.76). The infarct was mainly located in the somatosensory cortex with occasional damage to the white matter underneath or to the lateral caudate—putamen. The infarct volume was proportional to the duration of occlusion in our previous experiment (90 mins occlusion; STD: 92±12.5 mm3; EE plus learning: 80±14.8 mm3; P > 0.38) and the current experiment (60 mins occlusion).
Four weeks of EE was globally efficacious in reducing poststroke gait impairments but did not significantly improve gait parameters in sham animals. Enriched environment improved the intensity of the paw pressure during floor contact (housing effect: F1, 44 = 4.87, P < 0.03). Footprint intensity of the LF paw (P < 0.05) was increased with a borderline effect on the RF paw (P = 0.06; Figure 1B). Enriched environment was beneficial for the ischemic rats but failed to increase footprint intensity in sham rats exposed to EE, most likely because of a ceiling effect. However, the housingx treatment interaction failed to reach significance (F1, 144 = 2.95; P = 0.08). Enriched environment also reversed the ischemia-induced impairment in the maximal area of paw contact (housing effect: F1, 144 = 6.54, P < 0.02). The size of the print area of the RF paw at maximal contact in the MCAO-EE group was significantly higher than that of the MCAO-STD group (RF: P < 0.05; Figure 1D). Maximal area of LF paw contact was also increased in the MCAO-EE group (P < 0.05). The print area of the MCAO-EE group was higher than the MCAO-STD group, with a borderline statistical significance (P = 0.05; Figure 1F). Enriched environment improved both the print width and length of the RF paw in the MCAO rats (box width: P < 0.05; box length: P < 0.05, data not shown). In addition, there was also a significant improvement in the LH box length after housing in the EE (P < 0.05). Paw angles were not affected by EE (Figure 1H).
Contrary to the results of the rest of experimental groups, the bilateral hindpaws of the MCAO rats housed in the STD environment were placed in front of the forepaws, suggesting that the change in paw placement was progressively increased without rehabilitation. After housing in the EE, this change was significantly reduced bilaterally (left: F1, 20 = 4.61, P < 0.05; right: F1, 20 = 9.06, P < 0.01; Figure 1J).
Regarding the temporal relationship of paw placement, the impairment in girdle phase lag no longer existed 5 weeks after ischemia in the MCAO-STD rats (P = 0.09) or in the MCAO-EE rats (P = 0.54) when compared with respective sham rats (Figure 2B). However, the ipsilateral phase lag in the affected (right) side was greater in the MCAO rats than in the sham animals and it was not improved by EE housing (Figure 2D). Finally, EE reduced the double support time using the ipsilateral paws in MCAO rats (P < 0.05; Figure 2J). The EE by itself did neither has any significant effect on BOS, RI, or step patterns (data not shown), nor did it affect swing, stance, stride length, or gait velocity (data not shown).
The Correlation Among the Parameters
To determine whether body weight, velocity, and cadence affected the other measures of gait, we conducted Pearson's correlation analyses at the 4-day time point. Body weight did not affect intensity of paw pressure or size of the print area at maximal paw contact in shams or MCAO rats (Figure 4). Moreover, there was no significant correlation between body weight and all the other gait parameters in each limb (not shown). However, some of temporal parameters were interdependent with walk speed for both shams and MCAO rats. Stance showed a strong negative correlation but stride length and cadence showed a strong positive correlation with walk speed (Figure 5 and Table 2). Swing duration had the tendency to decrease when the walk speed increased, but correlation was only significant for the LF paw. The support time with diagonal double limbs showed a positive correlation with walk speed, whereas triple-limb support time had a negative correlation with walk speed (Table 2). There was no correlation between walk speed and other temporal or spatial parameters such as phase lags, BOS, relative paw placement, and step pattern (data not shown).
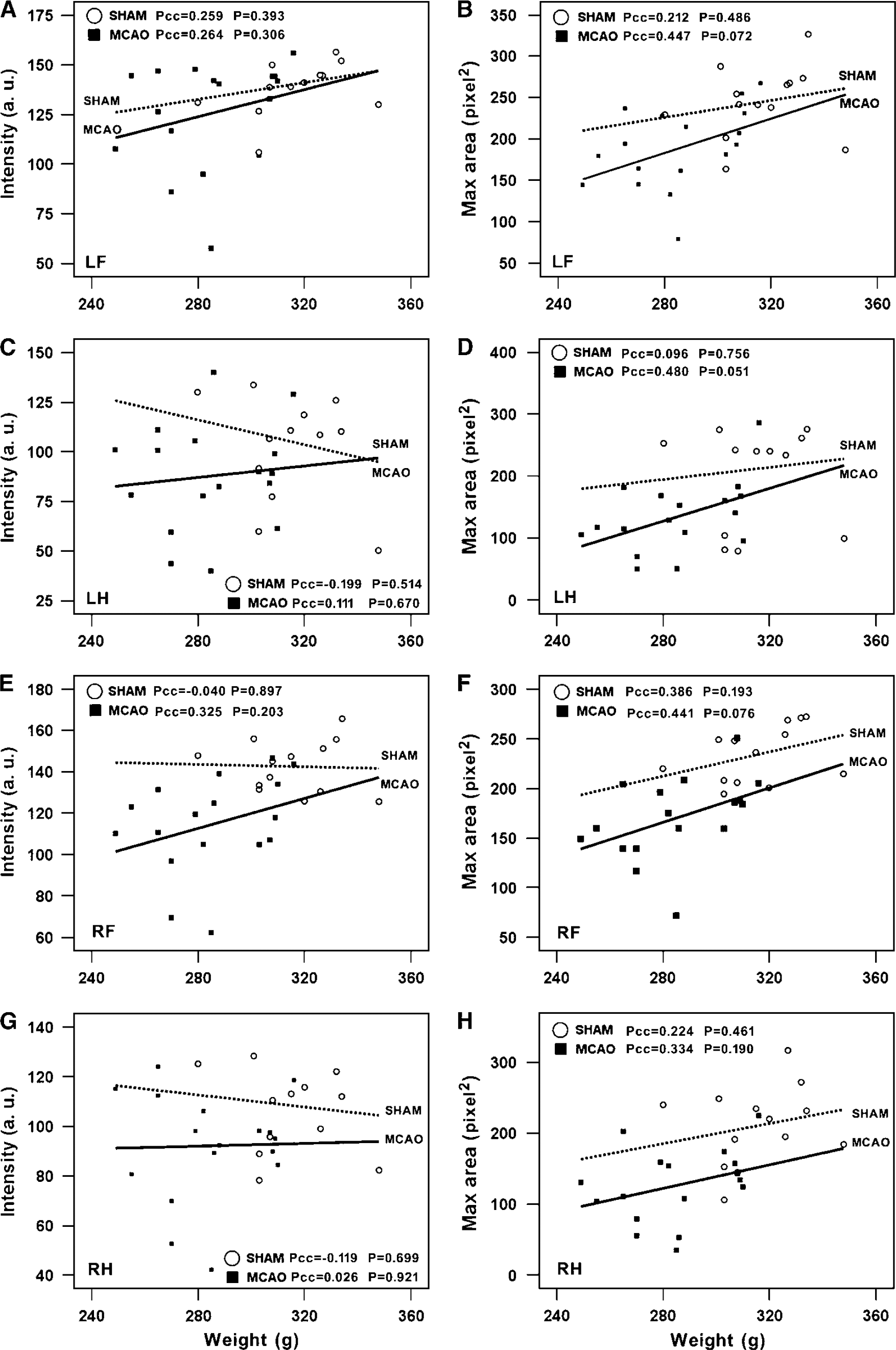
Lack of correlation between body weight and intensity (
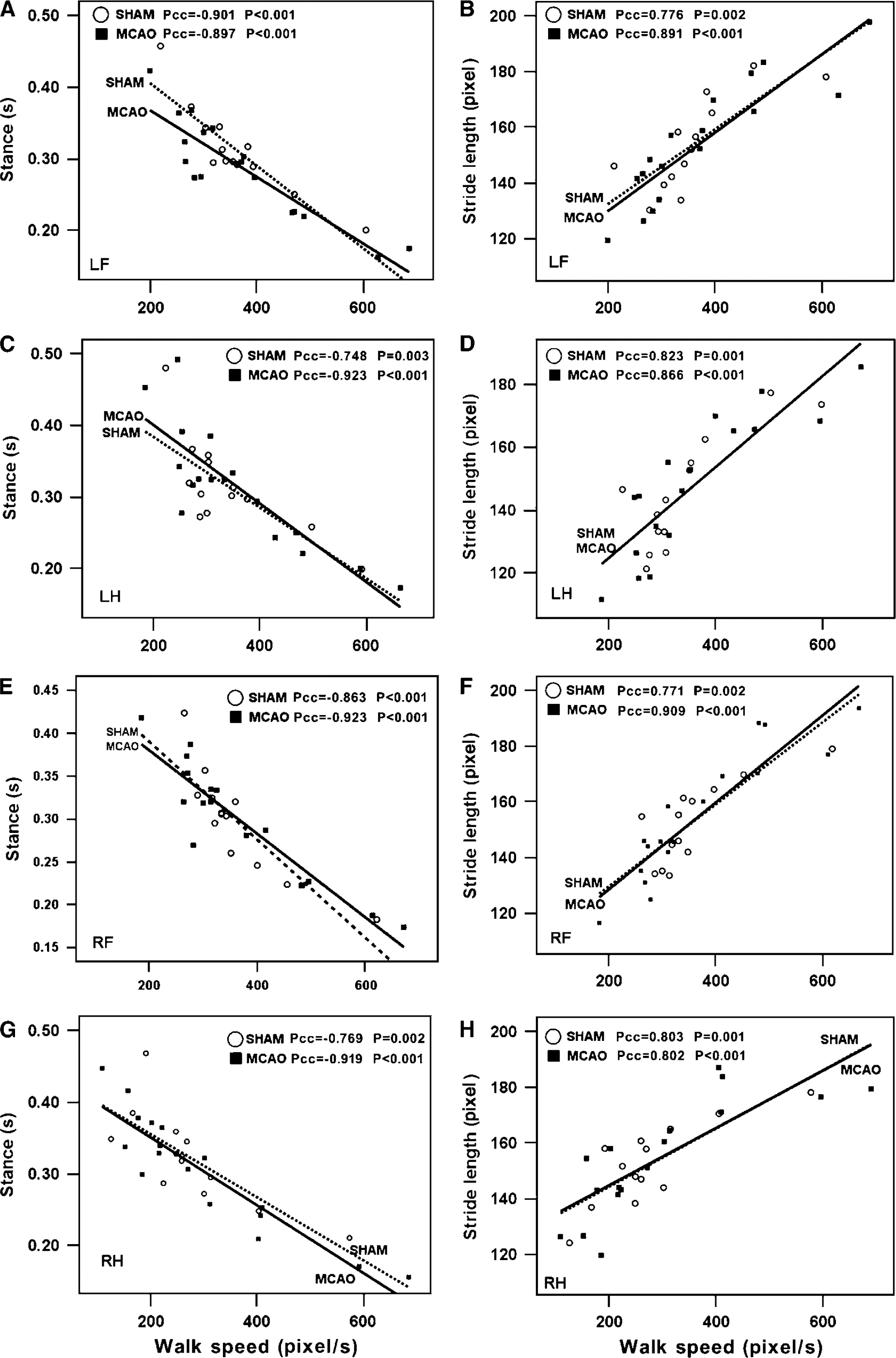
Significant correlations between walk speed and stance (
Relationship between walk speed and stance, stride length, swing, cadence, support time, and step pattern
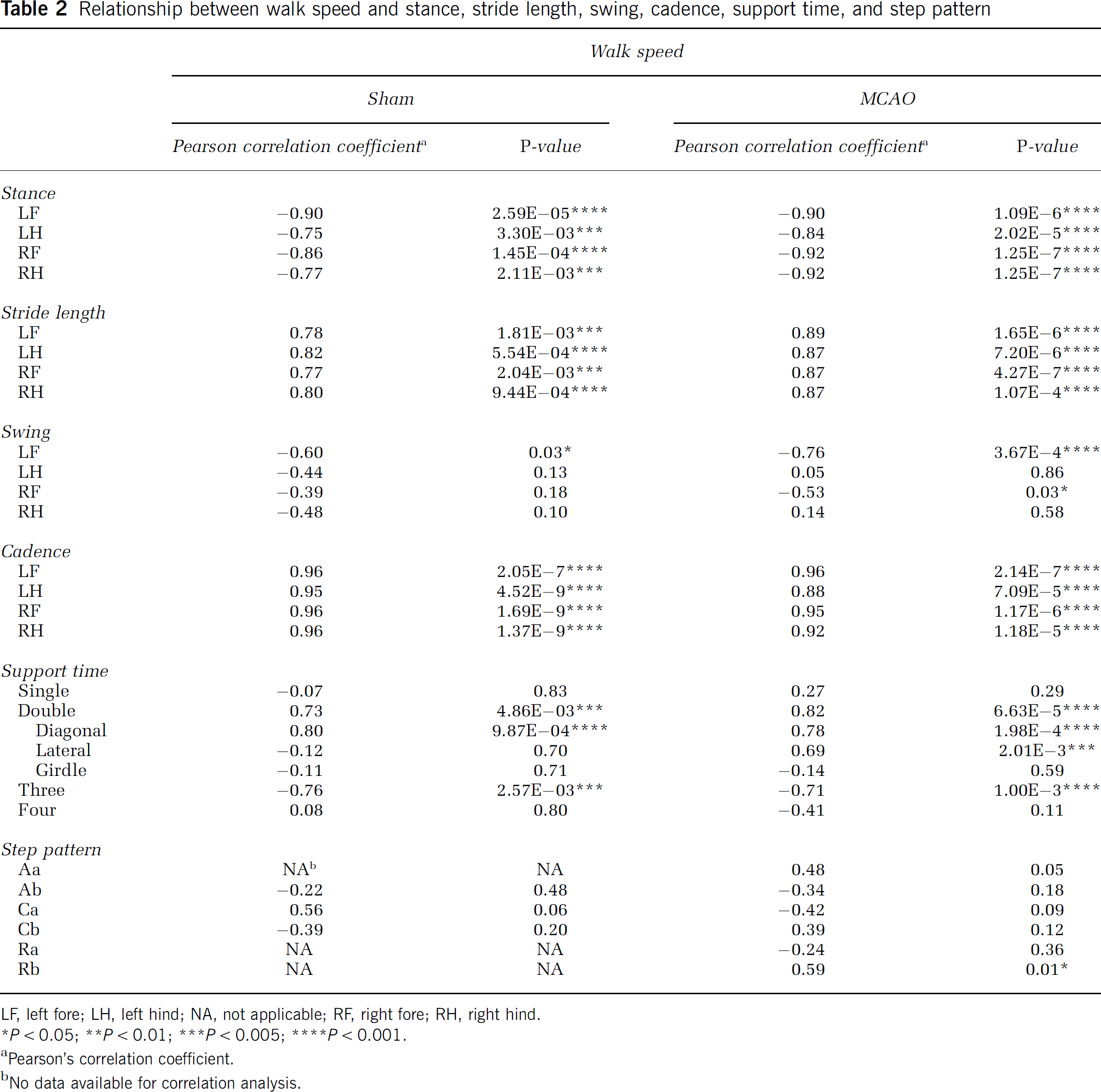
LF, left fore; LH, left hind; NA, not applicable; RF, right fore; RH, right hind.
P < 0.05;
P < 0.01;
P < 0.005;
P < 0.001.
Pearson's correlation coefficient.
No data available for correlation analysis.
Discussion
The present study was aimed at providing a thorough analysis of gait function in rats after stroke and rehabilitation using the CatWalk-automated gait analysis method. Most of the spatial parameters related to individual paw placements and inter-limb coordination were altered in rats with focal ischemia either temporarily or chronically. These gait impairments were not because of autonomy as there was no difference in gait velocity between the sham and MCAO groups. In contrast, the temporal parameters such as stance, swing, or stride length remained largely unchanged. Environmental enrichment was efficacious in reducing a variety of gait impairments, reflecting improvement in both sensorimotor status and interlimb coordination.
Comparing Gait Impairments After Ischemic Stroke with Other Models of Central Nervous System Injury in Rodents
Ischemic stroke, unilateral spinal cord injury, or pyramidotomy all share a compromised corticospinal tract with various degrees of hemiparalysis and distinct gait features. In rats with pyramidotomy, the stride length of all four paws is typically reduced as compared with sham controls, possibly because of a general decrease in walk speed (Starkey et al, 2005). After unilateral spinal cord injury, the maximal contact area of the attected forepaws is often reduced most likely because of reduced forelimb weight bearing (Gensel et al, 2006). Stride length has also been reported to decrease in the unaffected, but not the affected, forepaw, pointing to compensatory phenomena in the affected limbs. Although commonly monly examined in ischemic patients, it is surprising that detailed gait changes in quadrupeds commonly used in animal models of stroke are lacking. Using the CatWalk-automated gait analysis method, we found that focal ischemia led to a persistent reduction in paw pressure and maximal area of paw contact. These gait impairments are likely because of an altered use of the plantar surface of the paw, a pattern commonly observed in other models of central nervous system injury including sciatic nerve resection, spinal cord injury, Parkinson's disease, and pain (Gabriel et al, 2007; Gensel et al, 2006; Vlamings et al, 2007; Vogelaar et al, 2004). The absolute values and impairment in foot print intensity and paw contact area were always greater in the forepaws than hindpaws of ischemic rats, possibly because the forepaws play a more important role in supporting the body weight during walking. However, there was no direct correlation between body weight and any of these parameters. The intensity and contact area of the unaffected paws of ischemic rats tended to decrease as well, though not reaching a statistically significant difference.
Phase lags are typically used to describe interlimb coordination by assessing the timing relationship between two footfalls (Deumens et al, 2007). In the current study we found that the phase lag of LH(anchor)–RH(target) in the stroke group was significantly greater than the sham group, indicating that the affected hindpaws were placed later relative to contralateral hindpaws. Corroborative findings have been reported in other models of nerve injury including sciatic nerve resection and spinal cord injury (Deumens et al, 2007; Koopmans et al, 2005). In the inflammatory pain model, in which carrageenan was injected into the right knee, a late placement of the affected (right) hindpaw was also observed (Gabriel et al, 2007). Similarly, after sciatic nerve resection in the left leg, the affected hindpaw was placed relatively later in experimental animals than in controls. In ischemic rats, the affected forepaws also displayed a similar trend, although there was no statistically significant difference between the two groups. Five weeks after surgery, the timing in placing the affected forepaw was better than that of the hindpaw, and this differential recovery led to the emerging difference in phase lag in ipsilateral RF(anchor)—RH(target) between the MCAO-STD and sham-STD groups.
Step pattern is another relevant parameter reflecting interlimb coordination. The ‘Ab’ alternate pattern is the most common walking pattern, constituting 80% to 95% of the total step cycles in intact rats and mice (Cheng et al, 1997), and remains predominant in animals with nerve injury (Hamers et al, 2006). This is further supported by our current results, in which the Ab pattern was observed in 88.7% and 81.3% of steps in the sham and MCAO groups, respectively. After experimental stroke, rare step sequences, such as the rotary pattern, transiently appeared. Although rotary patterns have been previously classified as normal patterns for rodents, they are very rare in intact animals and were never seen in our sham rats.
Regularity index is a common, but not a very sensitive parameter for assessing interlimb coordination. Theoretically, the value of RI is 100% in healthy (fully coordinated) animals, and the loss of interlimb coordination leads to a decrease in RI. Because only extra or missing paw placement by one or multiple limbs are defined as ‘irregular’ steps (Vrinten and Hamers, 2003), resulting in almost certainly normal RI in most cases as long as all the four paws were used in single step cycle no matter what the step sequence was or how the paws were placed. This likely explains why abnormal RI has rarely been detected, except in some cases of spinal cord injury models in which RI was temporarily decreased during the early phase after surgery, followed by a full recovery at a later time point (Hamers et al, 2006). The RIs of all groups in the current study were more than 98%, making it unlikely that we would detect differences among groups.
Correlations Among the Gait Parameters
Previous reports suggest that body weight might affect the value of intensity, contact area, and print length. Koopmans et al (2007) compared the differences of gait parameters among different strains of rats of the same age. They found differences in stride length, BOS, stance duration, and even step cycle because of differences in body weight among strains. Dellon and Dellon, 1991 reported a significant increase in paw print length, toe spread, and intermediate spread with increasing body weight, but only when the body weight exceeded 300 to 350 g. In our study, we failed to find significant correlations between body weight and any gait parameters, including intensity and contact area, possibly because of a small variation in body weight among our cohorts and a lack of difference in strain, gender, or age. Velocity is also a very important determinant that can potentially influence several parameters of gait function (Gorska et al, 1998). Consistent with a previous finding (Gillis and Biewener, 2001), our results show that both walk speed and cadence directly affected stride length and temporal parameters including stance, swing, and support time. However, ischemic injury did not affect velocity.
Enriched Environment Effects on Postishemic Gait Function
Motor experiences, including rehabilitation interventions, considerably influence poststroke functional recovery (Schaechter, 2004). Enriched environment has been reported to enhance gait function recovery after spinal cord contusion injury in rats by increasing swing duration, BOS, and maximal area of paw contact (Van Meeteren et al, 2003). In this study, we found that the EE increased the maximal area of paw contact and paw pressure in stroke animals and decreased the impairment in relative paw placement. Enriched environment also reduced the increased double support time with lateral paws caused by stroke. Our data suggest that the beneficial effects of poststroke EE on gait function can be differentially analyzed by temporal and spatial parameters in a more comprehensive and refined fashion compared with conventional methods in used to assess motor function such as the ladder and rotor rod tests (Liu et al, 2007). Activity-dependent neuroplasticity can be regulated at least at two levels. First, synaptic efficacy at individual synaptic junctions can be modified by motor activity (Cooper, 2005). Second, neural plasticity can be induced by rehabilitation involving reorganization of cortex around the lesion site and also in the undamaged contralateral cortex or efferent pathways (Dancause, 2006; Nudo et al, 1996). However, the cellular and molecular mechanisms underlying the beneficial effects of EE on gait function remain unclear. One candidate mechanism involves upregulation of neurotrophic factors leading to structural changes in synaptic connectivity (Kleim et al, 2006). Indeed, EE has been reported to stimulate brain-derived neurotrophic growth factor release (Rossi et al, 2006) and to increase dendritic spine density in pyramidal neurons of somatosensory cortex contralateral to a cortical infarct in distal MCAO rats (Johansson and Belichenko, 2002); thus making synaptogenesis a plausible mechanism for driving recovery of gait function. This can be further supported by the fact that brain-derived neurotrophic growth factor is not only involved in experience-dependent modification of corticospinal output and motor map topography in humans (Kleim et al, 2006), but also promotes connections of corticospinal neurons onto spared descending interrneurons in rats with spinal cord injury (Vavrek et al, 2006). Besides, EE enhances periinfarct neuronal density in the ipsilateral cortex possibly through a sparing effect (Matsumori et al, 2006), suggesting that EE might also directly enhance the integrity of corticospinal tract that leads to improved gait function.
Comparing Gait Impairment Between Human and Experimental Stroke
Gait analysis is not only more routinely used to monitor motor impairment and recovery in humans than animals, but the technology in measuring gait is also much more advanced for humans. There are a variety of systems that are commercially available for measuring gait and posture changes in humans, including GAITMAT or GAITRite (Ng and Hui-Chan, 2005) and the three-dimensional gait analysis and plantar pressure distribution measure system (Chen et al, 2005). Certain systems allow physicians to analyze not only footprint, but also joint angle changes from pelvis to ankles and plantar pressure distribution when a patient walks, with the option of attaching an electromyography device on the patient's extremities to obtain feedback electro-physiologic data in the changes of muscle activity (Woodford and Price, 2007). In terms of conceptual design, the GAITRite is very similar to the CatWalk system used in the current study. It has an electronic walkway that is made of a carpet with several embedded pressure sensors. As the subject walks over the carpet, the sensors close under pressure, enabling collection of data on spatial and temporal gait parameters. Unlike the results of our experimental stroke study, velocity, cadence, stride length, and BOS of stroke patients tend to decrease while stance increases. One simple explanation is that perhaps the kinetic mechanisms in gait and body weight support in quadrupeds are different from humans. Another possibility is that the distal MCAO model used in the current study limits the infarct within the sensorimotor cortex, whereas the injury in hemiparetic human stroke often involves the internal capsule, resulting in damage to thalamo-cortical or extrapyramidal tracts in addition to the corticospinal tract. Future studies investigating how EE contributes to gait function recovery by improving the corticospinal tract at the neuroanatomical level are warranted.
