Abstract
Peripheral arteriogenesis is distinctly enhanced by increased fluid shear stress. Thus, the aim of this study was to investigate in the rat brain whether increased fluid shear stress can also stimulate cerebral arteriogenesis. To increase fluid shear stress in the cerebral circulation, we developed different shear stress models as the ligature of both common carotid arteries (
Introduction
Besides cardiovascular diseases and cancer, stroke is the third leading cause of death in industrialized countries and frequently produces disability in patients. Clinical studies demonstrated the importance of an adequate hemodynamic compensation
The term arteriogenesis was established by Schaper and his colleagues (Arras et al, 1998) to discriminate between the growth of collateral arteries and angiogenesis—the development of small-calibre capillary vessels in ischemic tissue. However, arteriogenesis describes the remodeling of preexisting arterio-arteriolar anastomoses or arteries outside the ischemic region to functional vessels, which can replace the conductance capacity of a larger artery (Grundmann et al, 2007; Schaper and Scholz, 2003; Scholz et al, 2001).
However, it was shown in animal models that peripheral collateral growth reaches only approximately 40% to 50% of the maximal conductance of the replaced artery (Hoefer et al, 2001; Ito et al, 1997). Our group demonstrated in previous studies in a pig and rabbit hindlimb model that fluid shear stress (FSS) is the pivotal trigger of peripheral arteriogenesis and that long-term FSS-stimulated collaterals can completely restore and even surpass the physiologic function of the occluded artery (Eitenmuller et al, 2006; Pipp et al, 2004).
To chronically elevate FSS after femoral artery ligature, collateral flow was directly drained into the venous system by creating a side-to-side anastomosis [arterio-venous (AV) fistula] between the distal stump of the occluded femoral artery and the accompanying vein. The direct connection of the collateral system to the low venous pressure system provided several beneficial consequences as the minimization of systemic flow resistance and the prevention of the dropping of FSS during later phases of arteriogenesis (Eitenmuller et al, 2006; Pipp et al, 2004).
The morphology of the cerebral arteries is very similar between humans and rats, including anomalies and structure of these vessels as well as their morphologic changes associated with cerebral vascular disease (Lee, 1995). It was demonstrated by Busch et al, 2003 that arteriogenesis can be induced in the adult rat brain. By developing the
On the basis of our previous AV fistula-experiments in the rabbit hindlimb, we established several models to increase FSS in the rat cerebral collateral circulation. Thus, the aim of this study was to determine whether increased FSS is also the pivotal trigger of cerebral arteriogenesis.
We evaluated the effects of shear stress stimulation on the cerebrovascular circulation
Materials and methods
Animal Models
The present study was performed with the permission of the State of Hessen, according to
All surgical procedures were performed under anesthesia with ketamine (100 mg/kg body weight) and xylazine (4 mg/kg) administrated i.p., and buprenorphine (0.03 mg/kg) was applied to prevent pain.
Male Sprague–Dawley rats (250–300 g; Charles River Laboratories, Sulzfeld, Germany) were randomly assigned to
Magnetic resonance imaging analysis and quantitative MRI-flow measurement were performed 1 day (
Magnetic Resonance Imaging-analysis and Quantitative Magnetic Resonance Imaging-flow Measurement
One, 7, and 14 days after surgery, rats were placed in an MRI-scanner (Bruker PharmaScan 7.0T, 16 cm; Bruker Corporation, Ettlingen, Germany;
The magnetic resonance tomograph operates at 300.51 MHz for 1H imaging and is equipped with a 300-mT/m self-shielding gradient system. Thereby, a linear polarized volume resonator (diameter, 35mm) was used. Localizer images were acquired using a spin-echo sequence and correction of slice angulation was performed, if necessary. Rapid acquisition with relaxation enhancement sequences in axial and sagittal slice orientation were used to determine the exact positioning of the rat brain [20 contiguous slices, 1-mm thickness, repetition time (TR) = 2500 ms, time to echo (TE) = 36.7 ms]. Afterwards, a coronal three-dimensional phase contrast angiogram of the whole rat brain was performed to detect the intracerebral, vascular system [TR = 35 ms, TE = 3.9 ms, field of view (FOV) = 70 × 37mm, slab-thickness = 15.00mm, matrix size = 256 × 200 × 64, velocity encoding (venc) = 30 cm/sec]. Three-dimensional reconstruction [maximum intensity projection (MIP)] was made with the use of the Image Processing Tool of the Paravision 4.1 software (Bruker PharmaScan).
The slice orientation of the following flow-quantification sequence was set strictly perpendicular to the target vessel. Parameters of the flow-quantification sequence were TR = 20 ms, TE = 3.5/3.9 ms, flip angle = 30°, venc = 50 to 150 cm/sec (depending on the target vessel). For sagittal orientation, the remaining parameters were NEX= 12, FOV= 70 × 35mm, slab-thickness = 8.06mm, matrix size = 512 × 256 × 16, for axial orientation NEX= 17, FOV= 33 × 35mm, slab-thickness = 5.54mm, matrix size = 257 × 256 × 10, resulting in an in plane resolution of approximately 130 × 130 µm2. Thus, a vessel with a diameter of, for example, 400 µm is covered by 7 to 8 pixels. For estimation of the significance of the partial volume effects on the accuracy of the flow measurement, the following function has to be considered:
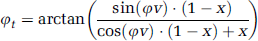
with φv, the signal phase originating from the velocity encoding of the moving spins;
In Paravision 4.1, the slice positioning on projected images is not possible. Owing to the small size of the target vessels and the resulting difficulties in identifying the vessels in the native images, a slice positioning using the native images was not possible in a reliable way. To keep the measurement time in a reasonable range, the number of slices in the following flow measurement was relatively low (16 or 10, respectively) with a slice thickness of approximately 500 µm. Because the covered range of the rat brain was rather small and the density of small vessels in the considered area relatively high, it was difficult to identify the target vessels on the images of the flow map. Therefore, it was necessary to calculate the physical coordinates in the magnet coordinate system from the coordinates of selected points in the MIP of the angiogram.
The physical coordinates were used for the calculation of the slice orientation and position of the flow measurement sequence. Three points in the MIP of the angiogram were selected, which were situated in maximal possible distance of each other on a straight part of the vessel. The connecting line of the two outer points defined the normal of surface of the new slices and, consequently, the new slice angles, the included third point, the position. The in plane shift was calculated for centring the third point in the new image plane.
For the determination of all three coordinates of a selected image point in the MIP of the angiogram, we got an algorithm from Bruker that not only calculates the MIP but also saves the slice number of the image point with maximal signal used for the construction of the projection. This algorithm functions only perpendicular to the native slices, inhibiting the typical rotation of MIP images but was sufficient for the examined morphology. The calculation of the physical coordinates from the image points and the sequence parameters as well as the calculation of the parameters of the new slices were performed with a program written by one of the authors.
The quantitative flow values were then obtained by integrating across manually drawn regions of interest (ROI) that enclose the vessels (Paravision 4.1 ROI-Tool, Bruker PharmaScan). Blood flow was measured in the left A. cerebri anterior, both A. cerebri posteriores, A. basilaris, left A. carotis interna (to determine flow of the AV fistula) and the right A. carotis interna (only in the Solo-Shunt group) (Figure 2B).
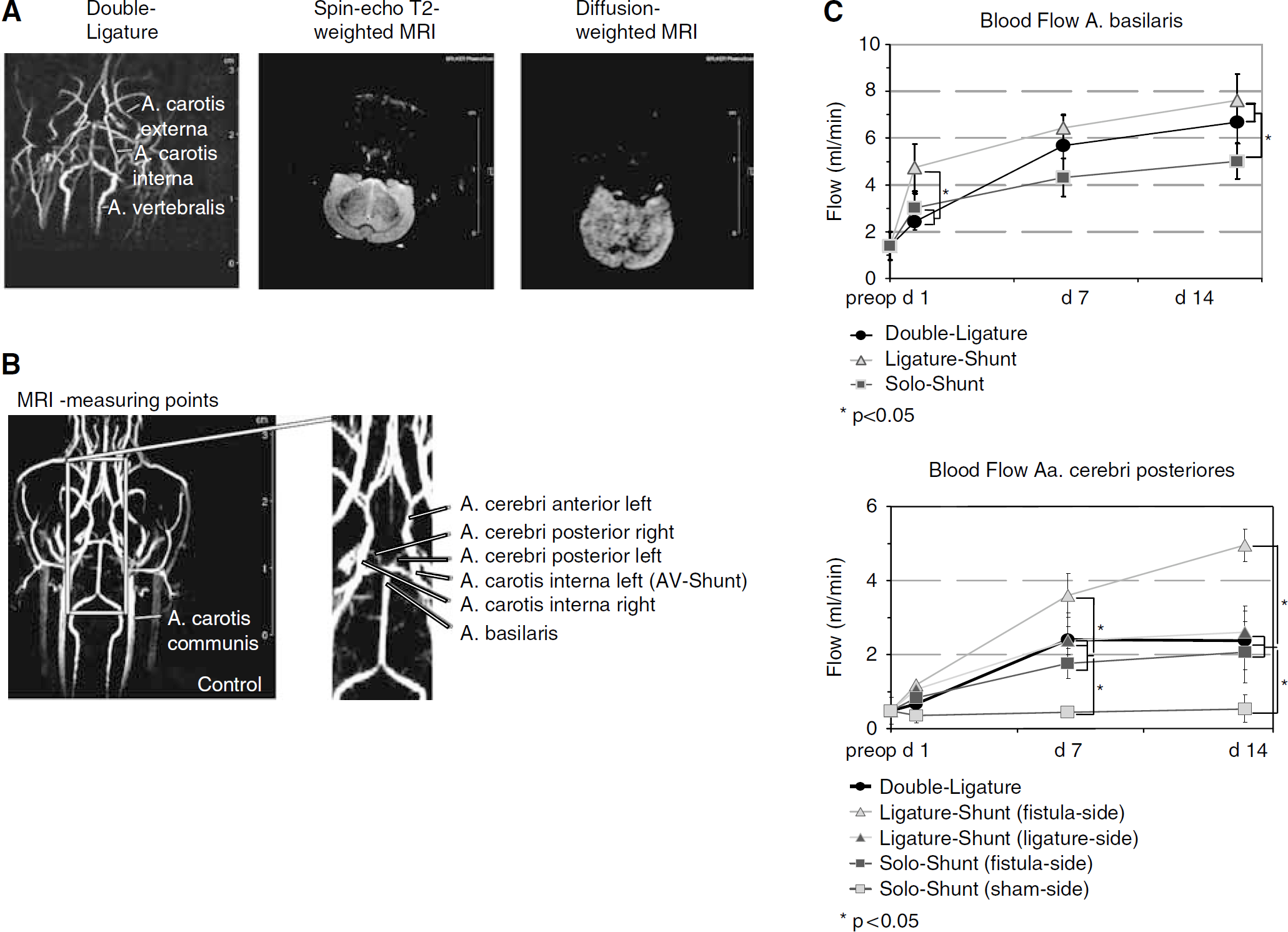
Verification of cerebral integrity and MRI-based blood flow measurement. (
Cerebral integrity was verified 1 day after bilateral carotid ligature or AV fistula-creation. For detection of oedema, a spin-echo Proton- and T2-weighted sequence was used (16 contiguous slices, 2-mm thickness, FOV= 37 × 37mm, matrix size 512 × 256, TR = 3000 ms, effective echo times TE1 = 25.7 ms and TE2 = 77.1 ms, two echoes for proton and four echoes for T2-weighting with an echo spacing of ΔTE = 17.1 ms, imaging time = 12.48 mins). To detect ischemic regions, a diffusion-weighted echo planar imaging sequence was performed (6 contiguous slices, 2-mm thickness, FOV= 32 × 32mm, matrix size 128 × 128, TR = 5000 ms, TE = 46.3 ms, imaging time = 5.3 - mins, diffusion gradient duration δ = 7 ms, diffusion gradient separation Δ = 14 ms, b-value = 1500 s/mm2).
Cerebral Postmortem Angiograms
Cerebrovascular anatomy was studied 7 and 14 days after surgery in each study group (
The vasodilator effects of adenosine and adenine nucleotides were first recognized by Drury and Szent-Gyorgyi in 1929. Since then, many studies demonstrated that adenosine has the capability to dilate many vascular beds including heart, skeletal muscle, and brain. Although it was shown for the brain that the intraluminal application of adenosine is less effective than the extraluminal application, the intracarotid adenosine infusion proved as an effective tool for a robust and profound augmentation of cerebral blood flow without any adverse hemodynamic side effects. Increased adenosine levels in the brain were shown to be caused by hypoxia or ischemia, but there were no alterations in the adenosine concentration found when the arterial pressure was manipulated over the normal autoregulatory range in the absence of hypoxia (Collis, 1989; Gordon et al, 2008; Joshi et al, 2002; Miekisiak et al, 2008). Because animals, which had developed cerebral oedema caused by hypoxia or ischemia, were excluded from the study, adenosine-induced vasodilatation and injection of the contrast medium at a constant physiologic pressure ensured demonstration of the correct anatomical vessel size for each of the models.
Immunohistochemistry
Three rats of each study group were euthanized 1, 3, and 7 days after surgery without previous adenosine-dilation. Brains were frozen in 2-methylbutan (precooled in liquid nitrogen) and immunostaining of the A. basilaris was performed on 5 µm-cryosections as previously described (Rodriguez et al, 2005). Primary antibodies: Ki67/MIB-5 (1:20, Dako, Hamburg, Germany) and CD11b (1:100, AbD Serotec, Düsseldorf, Germany). Secondary antibody: Cy3-conjugated anti-mouse IgG (1:300, Chemicon/Millipore, Schwalbach, Germany). Actin was stained using Phalloidin FITC-labeled (Sigma-Aldrich) and nuclei were stained with Draq5 (Alexis Biochemicals, Lörrach, Germany). Sections were viewed with a confocal scanning laser microscope (Leica TCS SP). Confocal images were obtained using concomitant multichannel scanning. Each recorded image was taken using two confocal detectors for reflected fluorescence and consisted of 1024 × 1024 pixels. Series of eight confocal optical sections were taken through the depth of the tissue sample. To improve the image quality and to obtain a high signal/noise ratio, each image from the series was two-fold signal-averaged. After data acquisition, the images were transferred to a Silicon Graphics Indy or Octane workstations (Silicon, Grabsbrunn, Germany) for image restoration and reconstruction using Imaris, the multichannel image processing software (Bitplane, Zürich, Switzerland). The principles of this method have been previously described (Kostin et al, 2004; Polyakova et al, 2008). For determination of the proliferation index in growing basilar arteries Ki67-positive nuclei of endothelial, medial, and adventitial cells were counted and referred to the total number of nuclei of each particular vessel layer.
Statistical Analysis
All values are expressed as mean±s.e.m. D'Agostino and Pearson omnibus normality test (Prism, GraphPad Software, Inc.) was used to test normal distribution. Group differences were analyzed for statistical significance using one-way ANOVA (Prism, GraphPad Software, Inc.).
Results
Novel Rat Models were Established to Increase Fluid Shear Stress in the Cerebral Circulation
Under conditions of laminar flow, FSS is directly related to blood flow and inversely related to the third power of the growing radius (Schaper and Scholz, 2003; Scholz et al, 2002). Enhanced blood flow in the cerebral collateral circulation can be reached, in principle, by either a reduction of the flow resistance or an increase of the pressure gradient.
Because the main blood supply to the brain is physiologically provided by the two carotid arteries (6.35±1.14 mL/min
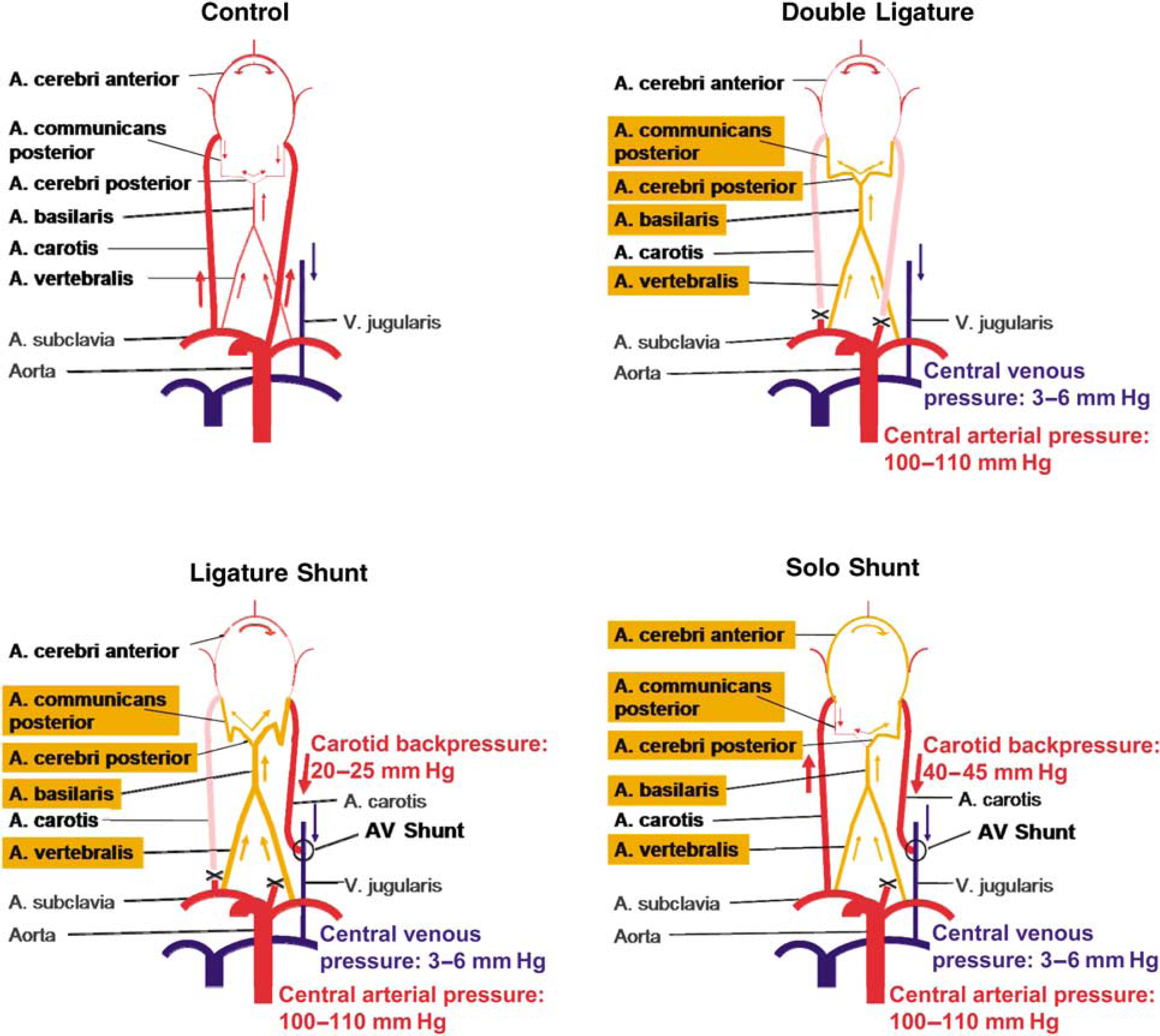
Rat models to increase blood flow in the cerebral circulation. Schematic diagrams of physiologic, cerebral blood flow (Control) and expected flow changes in the different shear stress models. After ligature of both carotid arteries in the Double-Ligature model, a blood flow increase in the posterior cerebral collateral circulation should be the direct consequence. Arterio-venous fistula-creation on the left side, additionally to Double Ligature, was expected to increase blood flow even more because of the steep pressure gradient between the arterial and venous system (Ligature-Shunt model). In the Solo-Shunt model, right carotid artery remained nonligated and an AV fistula was created on the left side that should cause a blood flow increase in the posterior and anterior cerebral collateral circulation. Central arterial pressure was determined in the nonligated carotid artery, central venous pressure in the jugular vein, and carotid backpressure in the cut distal stump of the ligated carotid artery before AV fistula-creation. Red: arteries with intravascular blood flow; pink: ligated arteries with interrupted blood flow; orange: arteries with expected shear stress elevation.
Blood flow changes in the rat cerebral collateral circulation (absolute: ml/min and relative to preoperative: fold changes)
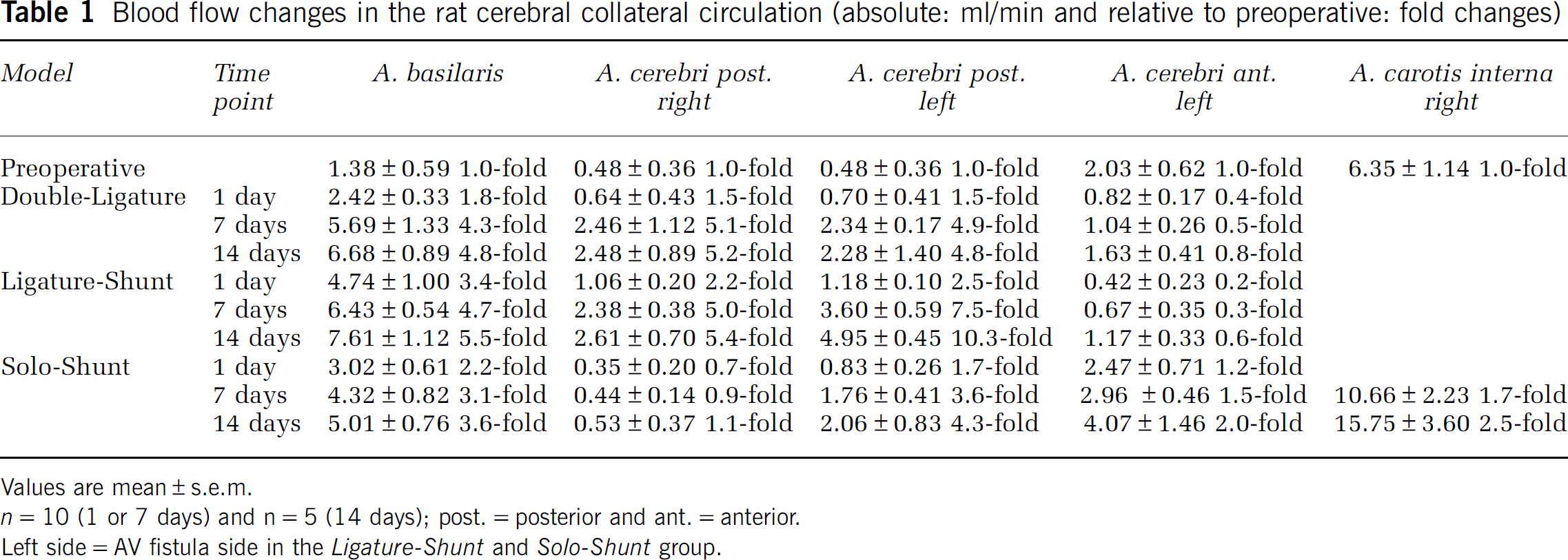
Values are mean±s.e.m.
Left side=AV fistula side in the
Cerebral Integrity as well as Functionality of the Arterio-venous Fistula was Verified by Magnetic Resonance Imaging
Control of cerebral integrity was performed by spine-cho T2- and diffusion-weighted MRI 1 day after bilateral carotid ligature or AV fistula-creation, respectively. Diffusion-weighted MRI is very sensitive to show early ischemic changes in the acute stage after stroke. The cytotoxic oedema visualized as hyperintensity on the diffusion-weighted images can be shown within 5 mins to 1 to 3 h after onset of symptoms (Mitsias et al, 2002; van Everdingen et al, 1998). In our models, cerebral integrity could be proved in all animals of the Double-Ligature and Solo-Shunt group (Figure 2A). None of the rats developed any neurologic symptoms during the further observation period. In the Ligature-Shunt group, 15.8% (3/19) of the animals developed signs of cerebral oedema indicating that the cerebral collateral system in this model comes close to its limits (data not shown).
Functionality of the AV fistula was confirmed by MRI-phase contrast angiography and quantitative MRI-flow measurement 1 day after surgery. Fistula-flow was 1.37±0.61 mL/min in the Ligature-Shunt and 3.28±1.34mL/min in the Solo-Shunt group. During the following 14 days-observation period, fistula-flow did not change significantly (data not shown). AV fistula-reocclusion (10.5% or 2/19) was only seen in the Ligature-Shunt group.
Magnetic Resonance Imaging-Analysis Demonstrated Significantly Increased Blood Flow after Surgery
One, 7, and 14 days after bilateral carotid ligature or AV fistula-creation, blood flow changes in the A. basilaris, Aa. cerebri posteriores, A. cerebri anterior, and A. carotis interna were monitored by quantitative MRI-flow measurement (Figure 2B; Table 1). Quantitative MRI-flow measurement was recently introduced as a noninvasive method for detecting blood flow insufficiency in patients who may require revascularization (Conway et al, 2008; Rutgers et al, 2000). Therefore, we first had to adapt this technique to the rat model (vessel diameter > 0.5mm).
When compared with the preoperative flow, MRI-analysis verified in all models a significant blood flow increase for the A. basilaris at every point of time (Figure 2C; Table 1). Significant differences in the basilar flow between the different models were observed 1 and 14 days after surgery (Figure 2C; Table 1). In both Aa. cerebri posteriores significant flow increase, compared with the preoperative flow, was confirmed in all models 7 and 14 days after bilateral carotid ligature or AV fistula-creation, respectively (Figure 2C; Table 1). MRI-flow measurement also revealed in these two collateral arteries a significant flow difference between the different treatment options ‘AV fistula—ligature—sham’ at 7 and 14 days, even in one animal (Figure 2C; Table 1). Further, in the Solo-Shunt group, blood flow increase was demonstrated during the 14 days-observation period in the left A. cerebri anterior toward the AV fistula by 2.0-fold and in the nonligated, contralateral carotid artery by 2.5-fold (Table 1), which was consistent with the existing studies using the carotid-jugular AV fistula (Tohda et al, 1992).
Increased Fluid Shear Stress Enhances Cerebral Arteriogenesis
Stimulation of vascular growth by the different shear stress models was analyzed postmortem by contrast agent-angiography at maximum vasodilatation as well as
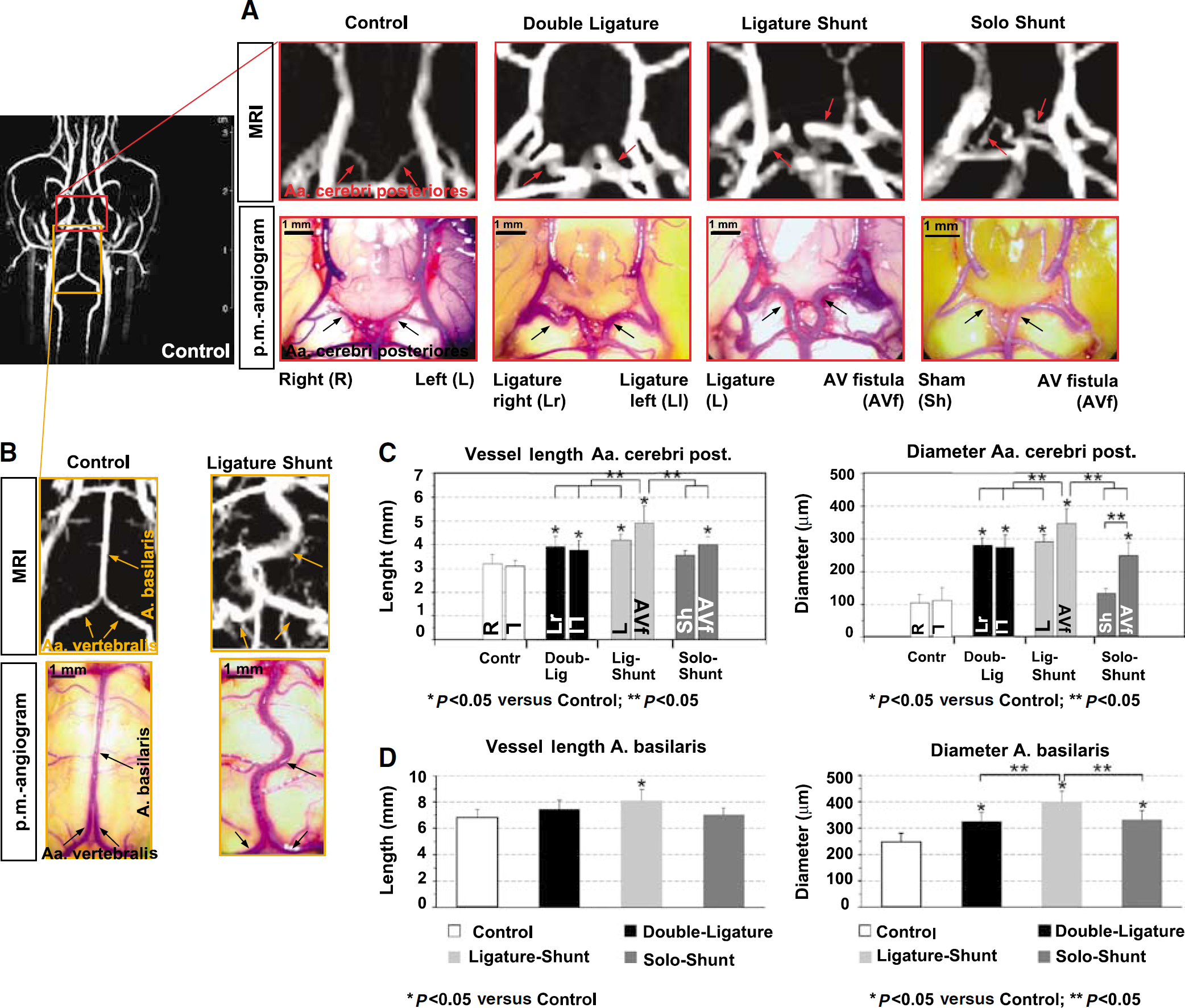
Growth evaluation after 14 days of shear stress stimulation. Representative MRI-phase contrast angiographies and postmortem angiograms 14 days after indicated treatment. MRI was performed
A considerable, FSS-dependent enlargement of the Aa. cerebri posteriores and A. basilaris in length and especially in diameter appeared in all shear stress models 7 days (Table 2) and 14 days after surgery (Figures 3A–3D; Table 2). Maximum increase of length and diameter of the A. basilaris was found in the Ligature-Shunt group (Figures 3B and 3D; Table 2). Concerning the Aa. cerebri posteriores, analysis demonstrated a significant difference in vessel length and diameter between the different treatment options ‘AV fistula—ligature—sham’ (Figures 3A and 3C; Table 2). Enlargement of the A. cerebri anterior was only seen ipsilateral to the AV fistula in the Solo-Shunt group (Table 2).
Growth of rat cerebral collateral arteries after 7 and 14 days of shear stress stimulation (length: mm and diameter: µm)
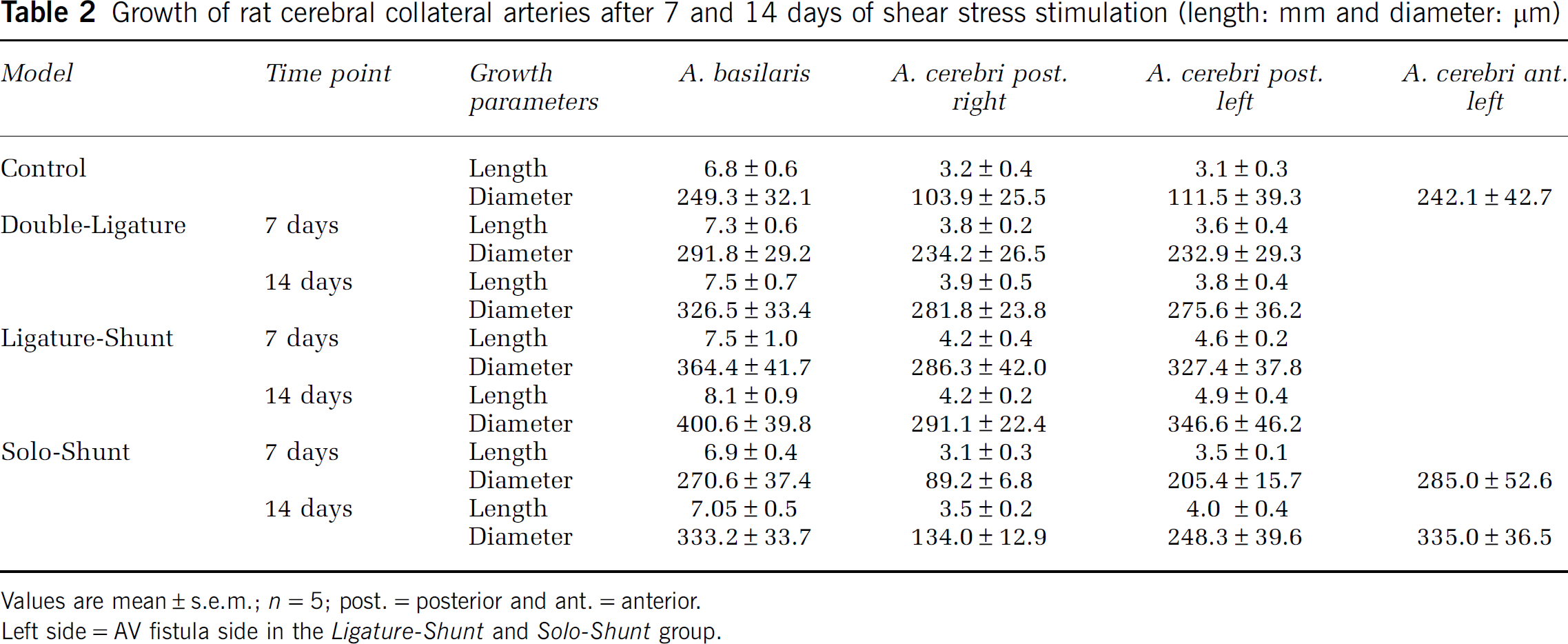
Values are mean±s.e.m.;
Left side=AV fistula side in the
For the Aa. cerebri posteriores, a correlation was found between vessel length/external diameter and blood flow (Figure 4). The enlargement of the vessel length followed a linear and the enlargement of external diameter a potential trend line, both with high correlation (coefficient of determination
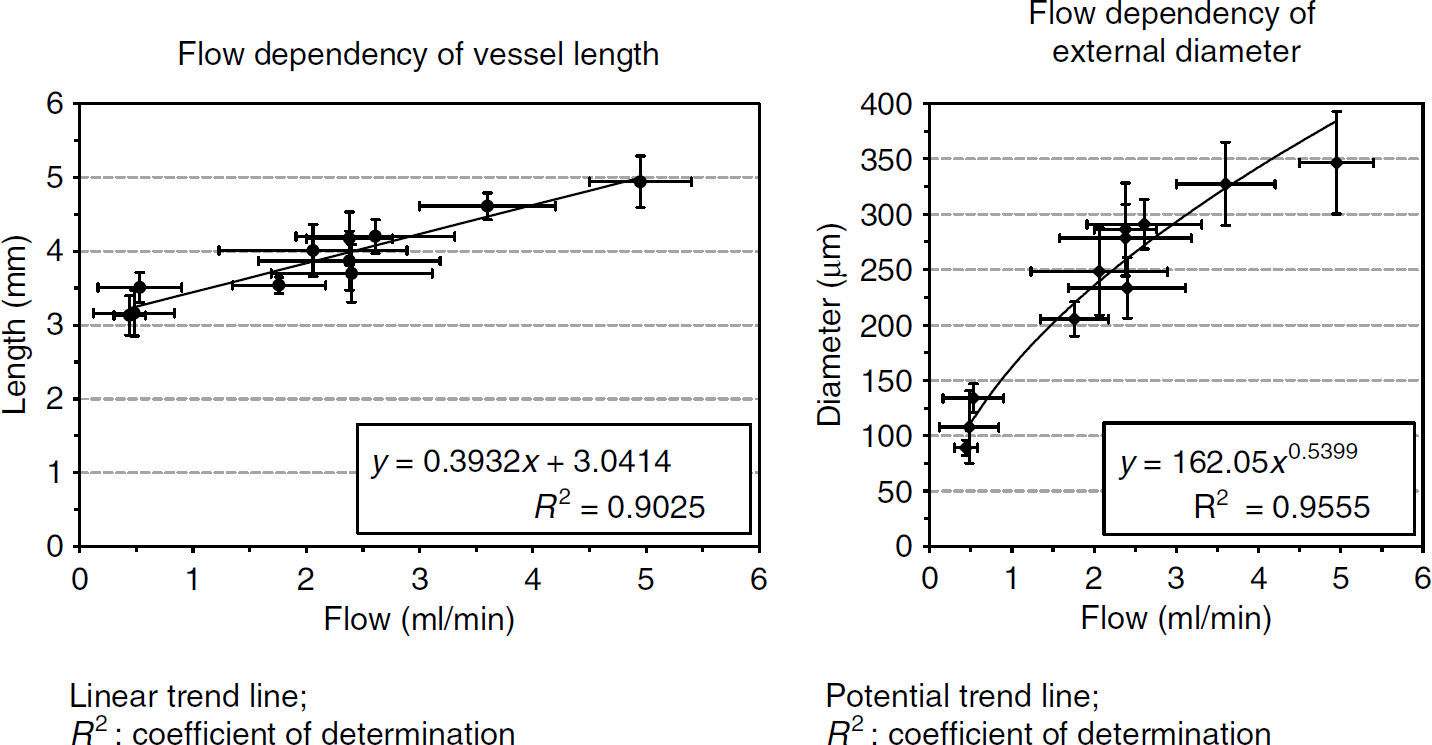
Flow-dependent growth of cerebral collaterals. Charts of vessel length (mm) and external diameter (µm) of the Aa. cerebri posteriores versus blood flow (ml/min) at equal points of time. A correlation was found between vessel length/external diameter (analyzed on postmortem angiograms by ImageJ 1.38p software at 7 and 14 days) and blood flow (determined
Immunohistochemistry Showed Typical Signs of Arteriogenesis
We performed Ki67-immunostaining of the A. basilaris at multiple sections to ensure that the observed enlargement of the vessels is really because of arteriogenesis and verified active proliferation 1, 3, and 7 days after surgery in all shear stress models. Proliferation started from 1 day in adventitial cells and was subsequently followed by endothelial (from 3 days) and smooth muscle cells (from 7 days) (Figures 5A and 5B).
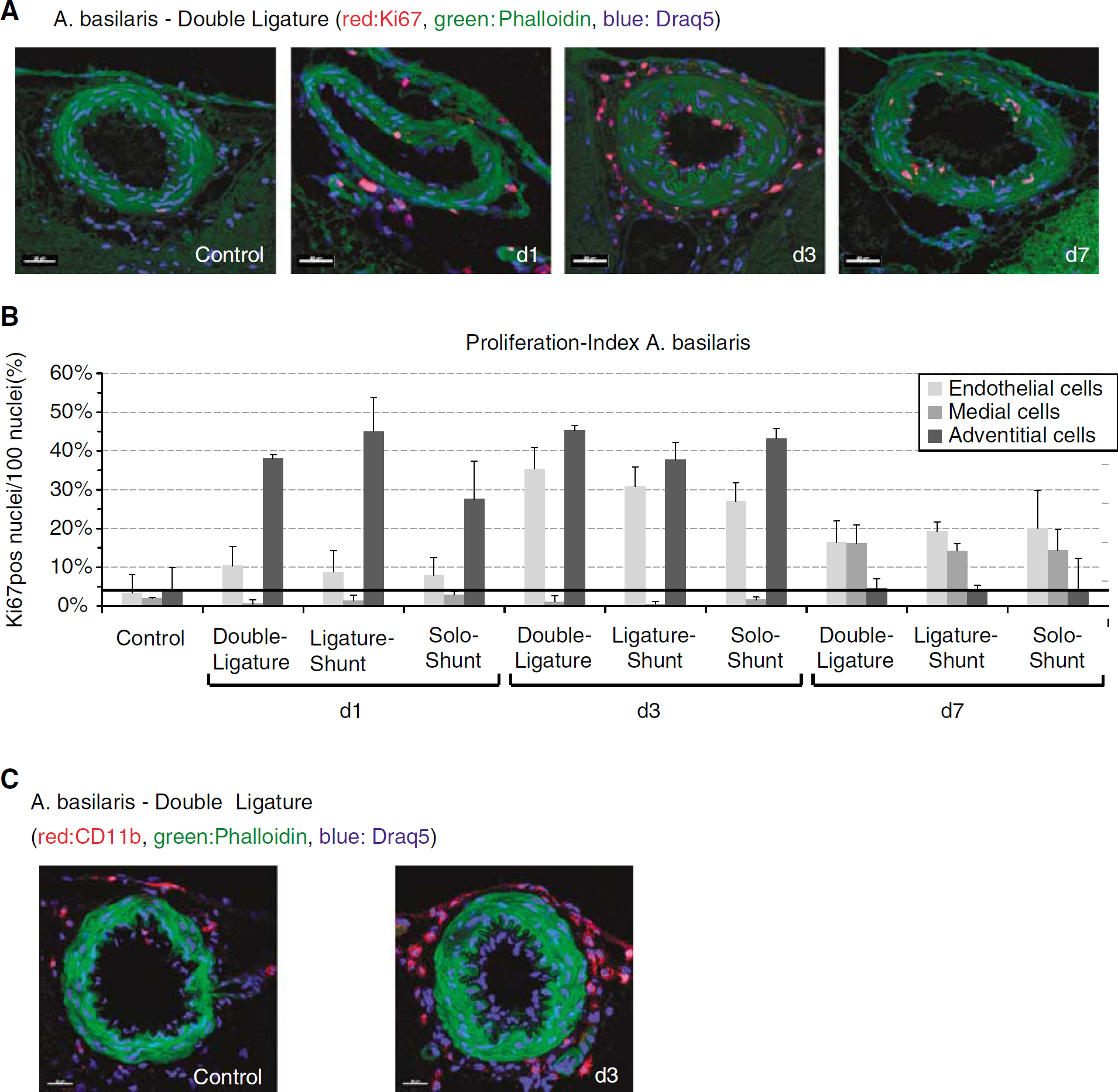
Increased proliferation activity and perivascular accumulation of mononuclear cells. (
Immunohistochemical evaluation of monocyte accumulation showed a considerable increase of mononuclear cells perivascular to the growing cerebral collaterals (Figure 5C).
Discussion
In analogy to the observations in the coronary and peripheral circulation, cerebral collaterals that maintain perfusion beyond the site of an occluded artery have long been appreciated as a critical factor to reduce cerebral ischemia and stroke risk in the setting of cerebrovascular disease. Besides the leptomeningeal anastomoses, the circle of Willis, a low-resistance connection between the four main supplying arteries of the brain, is of great importance for brain integrity in patients with carotid or vertebral artery disease (Bisschops et al, 2003; Busch et al, 2003; Hendrikse et al, 2001, 2002; Liebeskind, 2004; Proweller et al, 2007; Todo et al, 2008).
As our group demonstrated in previous studies that FSS is the pivotal trigger of peripheral arteriogenesis leading to a distinctly increased collateral growth (Eitenmuller et al, 2006; Pipp et al, 2004), we tested the hypothesis that increased FSS is also a major stimulus of cerebral arteriogenesis.
According to the resulting blood pressure conditions, creation of an AV fistula distally to the ligature was the critical step to chronically elevate FSS in the peripheral collateral circulation (Eitenmuller et al, 2006; Pipp et al, 2004). Formation of an AV fistula between the common carotid artery and the jugular vein has already been described in the literature but was so far only applied to study the effects of blood flow increase on the remodeling of the common carotid artery (Tohda et al, 1992; Tronc et al, 1996). Moreover, these fistulas were created between the open common carotid artery and the jugular vein exhibiting a much higher shunt flow (Tohda et al, 1992; Tronc et al, 1996). The fistulas in the Ligature-Shunt and Solo-Shunt model on the contrary were created between the distal stump of the ligated carotid artery and the jugular vein. Because we could adapt the quantitative MRI-flow measurement to the rat model, our group was, to our knowledge, the first using the AV fistula for investigations on the effect of an increased blood flow on the remodeling of intracerebral vessels.
Arteriogenesis is defined as the remodeling of preexisting arterio-arteriolar anastomosis or arteries outside the ischemic region to functional vessels, which can completely replace the conductance capacity of a larger artery (Grundmann et al, 2007; Schaper and Scholz, 2003; Scholz et al, 2001). In the rat brain, this definition applies best to the two Aa. cerebri posteriores, arteries with marginal blood flow under physiologic conditions that enlarge after bilateral carotid ligature to provide sufficient brain perfusion. Blood flow increased significantly in all models reaching maximum intravascular flow rates in the Ligature-Shunt model after 14 days. However, carotid backpressure dropped to 40 to 45mmHg after ligature of one and to 20 to 25mmHg after ligature of both common carotid arteries indicating that the observed growth effects were caused by alterations of blood flow but not pressure (Korff et al, 2008).
It has already been proved in former studies that arterial remodeling is in principle related to hemodynamics (Langille, 1996; Tohda et al, 1992; Tronc et al, 1996). For the Aa. cerebri posteriores, a correlation was found between external diameter and blood flow according to a potential trend line. In addition, intracerebral collaterals not only increased in diameter but also flow dependently in length (linear trend line) and developed the typical corkscrew formation of collateral arteries. Both, length and tortuosity are known to contribute to the total resistance to flow limiting a more intense blood flow increase (Schaper and Scholz, 2003; Scholz et al, 2001, 2002).
Meanwhile, it is a well-known fact that collaterals (coronary, peripheral, and cerebral) grow by DNA synthesis and mitosis of endothelial and smooth muscle cells—cells that are quiescent in normal adult arteries but can rapidly convert to G1 under abnormal conditions, as well as that the remodeling of the adventitia is essential to create the necessary space for the enlarging collaterals (Busch et al, 2003; Grundmann et al, 2007; Pipp et al, 2004; Schaper et al, 1971; Schaper and Ito, 1996; Scholz et al, 2002). Immunohistochemical evaluation showed the typical phases of arteriogenesis for all growing collaterals in our shear stress models as well as the accumulation of mononuclear cells in the adventitia, a typical sign of arteriogenesis (Arras et al, 1998; Busch et al, 2003; Grundmann et al, 2007; Pipp et al, 2004; Scholz et al, 2002).
Evidence that arteriogenesis can be induced in the circle of Willis of adult rats was already supplied in previous studies by Busch
Although we chose a different endpoint for growth evaluation (2 weeks instead of 3) all four models, 3-VO, Double-Ligature, Ligature-Shunt, and Solo-Shunt, proved as highly suitable to study cerebral arteriogenesis. In addition, with our new cerebral shear stress models, we could provide evidence that FSS is not only the pivotal trigger of peripheral and coronary (Eitenmuller et al, 2006; Grundmann et al, 2007; Pipp et al, 2004; Schaper and Scholz, 2003; Scholz et al, 2001) but also of cerebral arteriogenesis, and that cerebral arteriogenesis can be further enhanced by increased FSS.
One limitation of this study is the lack of the investigation of the leptomeningeal anastomoses as important preexisting collateral arterioles. Furthermore, we did not include functional investigations on the cerebral hemodynamic reserve capacity. However, no serious neurologic symptoms appeared as a consequence of the different interventions.
In conclusion, we demonstrated that the growth of cerebral collaterals correlates with rising intravascular flow rates. We showed evidence for the first time that FSS is not only a major stimulus of peripheral but also cerebral arteriogenesis. So our shear stress models offer several applications for further studies, for example, concerning FSS-dependent gene expression or FSS-activated pathways in cerebral arteriogenesis.
Footnotes
Acknowledgements
The authors like to thank Mrs U Eule and Mrs B Matzke for expert technical assistance.
The authors declare no conflicts of interest.
