Abstract

Keywords
The Fiftieth Anniversary of the Coulter Principle
Introduced in the mid-1950s, the Coulter Principle became the foundation of an industry responding to the need for automated cell-counting instruments. The industry developed in three acts, as Wallace H. Coulter and his brother Joseph R. Coulter, Jr., developed the simple idea of passing cells through a sensing aperture. In Act I, Wallace's desire to automate the routine erythrocyte count led to a simple idea, the definition of the Coulter Principle, its patenting, its acceptance by the National Institutes of Health, and its description at a national conference. In Act II, the Coulter brothers addressed the practicalities of a commercial instrument and of a business organization to support its manufacture and sale. In Act III, a broad research effort developed regarding volumetric errors originating in functional characteristics of the sensing aperture, and the brothers’ growing organization found solutions permitting introduction of increasingly automated hematology analyzers. Today the industry thrives, with several participants.
Introduction
“You can't patent a hole.” With that opinion, several attorneys dismissed Wallace H. Coulter's hopes of patenting his method for automating the erythrocyte count.
1
For each specimen, a technician spent some 30 tedious minutes at a microscope counting the red cells in a standard chamber, the result being only rarely repeatable. Automating such counts had tantalized Wallace (Fig. 1) since he had read Moldavan's 1934 note proposing photoelectric counting of cells in a suspension as it flowed through a capillary tube mounted on a microscope.
2
Over a decade had elapsed before Wallace discovered a better method, and a second decade was to pass before he was prepared to announce it.
Wallace H. Coulter encountered the routine erythrocyte count during his visits to hospitals for General Electric X-Ray Corporation and began experiments toward automating it after returning to Chicago in 1946. In 1947, he discovered one form of the Coulter Principle.
Act I
After gathering technical experience elsewhere, Wallace returned to Chicago in 1946 and was soon joined in his quest by his brother, Joseph R. Coulter, Jr. (Fig. 2). As he later reminisced about their work in a basement laboratory:
3
“The approach originally was to count blood cells going down a capillary tube, passing through a light beam, like counting people going down a corridor, but we weren't getting very good signals. We asked the question, ‘Is there some way, besides modulating a light beam, to generate electrical pulses by the passage of a cell?’ Although we didn't know it at the time, blood cells are insulators—so we arrived at a solution by modulating an electric current instead of a light beam.”
Joseph R. Coulter, Jr., after his Army service, joined his brother Wallace in a small basement laboratory and helped solve many difficulties while incorporating the Coulter Principle into a practical automated instrument. In 1958, he helped found Coulter Electronics, Inc.
In a characteristically simple solution, by 1947 Wallace had reduced the capillary tube to its minimum length: 1 “When we started we didn't have much money, so we made an aperture by making a small hole with a hot needle in a piece of cellophane from a cigarette package. It didn't hold up long, but we were able to count some cells.” Held onto the end of a glass tube by a rubber band, the punctured cellophane separated two electrodes connected to a source of electric current while cells suspended in ionic medium were passed through it simultaneously with the current. A cell's displacement of liquid equal to its own volume within the aperture was proportionally signaled by a voltage pulse between the electrodes passing the current through the aperture.
The brothers had found electrical contrasts between cells and the suspending medium that were some 10 times the ones obtained photoelectrically, the resultant voltage pulses readily permitting an accurate count of the cells in the volume of cell suspension passing through the aperture. This discovery had prompted Wallace's unexpectedly prolonged search for an attorney willing to prepare a patent application. Finally, in 1948, he was introduced to Irving Silverman who recognized the new method's significant potential, and in August 1949, a patent application was filed.
But the patent examiner also doubted that a hole could be patented. Fortunately, he also surmised that if an application were to include examples other than an axial current and sensing path through an aperture, a patent might be obtained on the principle of sensing particles in a constricted current path. 1 An analogous path transverse to the aperture suspension flow was described, as well as apertures having non-circular cross sections. An insulated needle sweeping past particles in a stationary suspension was another example, a particle's presence being signaled by a pulse in the current between the moving needle and a second electrode contacting the conductive suspending medium. The new Coulter Principle being thus defined, the seminal patent was issued 50 years agoonOctober20,1953, with the preferred embodiment being a pinpoint aperture formed in the lower wall of a test tube. 4
Meanwhile, the Coulter brothers had continued development, with emphasis on automation where feasible. With volunteer help from Walter Hogg, Joseph's friend from Army days, the brothers assembled an experimental instrument under contract to the Office of Naval Research: a mechanical metering system moved a controlled volume of cell suspension through the aperture while an interface unit provided it an electric current and sensed resultant signal pulses via a voltage amplifier having an adjustable threshold. Signal pulses above threshold triggered a pulse counter (Berkeley Scientific Model 410; Beckman Instrument, [then] Richmond, CA) mounted atop the interface unit. Repeated runs of a sample at successively increasing thresholds permitted manual recording of a cumulative distribution of cell size. The need to detect partial occlusion of the aperture soon became evident, so an oscilloscope was added to allow simultaneous monitoring of the signal pulses and the threshold setting.
Experience also emphasized the need to meter a precise volume of cell suspension through the aperture. Together, the Coulter brothers invented an elegant solution based on a mercury manometer (Fig. 3). As Joseph once remarked:
5
“It was the manometer that made the counter work. It was simple, it was easy to control, and it kept working.” Combined with a microscope focused on the aperture, this metering system formed the basis of a sample stand that would remain essentially unchanged for over 30 years. In 1952, the first two instruments incorporating the new sample stand were built; the interface electronics and oscilloscope were integrated into a single compact console also including decade counters to accumulate the voltage pulses. In 1953, these prototypes went to the National Institutes of Health (NIH) for evaluation, and in May 1956, a patent application was filed on the manometer metering system.
6
Functional schematic of the Coulter sample stand, co-invented by the Coulter brothers.
6
When stopcock 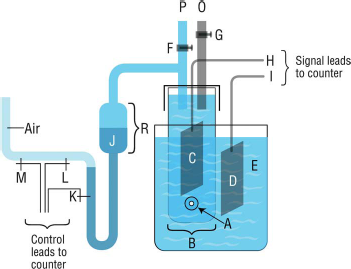
The Coulter Principle was formally announced on October 3, 1956 in Wallace's sole technical paper:
7
“In the new counter individual cells are caused to move through a small constricted electric current path in the suspending fluid and detection is based upon differences in electrical conductivity between the cell and the suspending fluid. The constricted current path is analogous to a light beam of small dimensions in an optical system. In passing thru the small current path in the fluid the individual blood cell changes the electrical resistance in the circuit, and causes a change in the voltage drop appearing across the current path. The electric current path of small dimensions and the flow of cell bearing fluid thru the path is provided for with a very simple structure. The boundary of the current path is the bore of a submerged orifice of small dimensions in the wall of an insulated vessel. ” In Figure 3, this submerged orifice is the central hole in aperture wafer
Shortly thereafter, the two NIH evaluations were published.8, 9 Both noted the improved accuracy, efficiency, and convenience of the Coulter method for counting erythrocytes. The journal issue containing one evaluation 8 also contained the first advertisement for the new Coulter Counter®. The second evaluation 9 attributed skewness in the size distribution to cell coincidence and presented a manual method for correcting the count for coincidence loss. It also included preliminary data on leukocyte counting.
The feasibility of automating significant hematological tasks had been demonstrated, but the work of commercialization yet lay ahead. Reliable mounting of the aperture had proven difficult. 10 The mercury used in the manometer metering the suspension through the aperture was a concern, emphasizing an advantage of the mechanical system used in the experimental counter. 11 The voltage source used to provide excitation current to the aperture produced unacceptable sensitivity to characteristics of both the aperture and the medium used to suspend cells. 12 The need for precise sample dilutions had been recognized in the NIH studies, indicating the value of an automatic diluter. 13 As Joseph later summarized a decade of preparation, 5 “We knew there were problems, but we also knew we had something useful.” As another decade began, solutions to these problems would soon appear in the patents just cited.
Act II
The sensing aperture (the hole in wafer
By 1958, the Coulter brothers were ready to found Coulter Electronics, Inc., together with its sales organization, Coulter Sales Corporation. Two of the parent company's first fulltime employees were long-time volunteers Joseph R. Coulter, Sr., and Walter Hogg. The Coulters’ father had served as weekend secretary and accountant from the beginning, but now at age 68, he retired as a railroad telegrapher and began a second career working with his sons, only going into partial retirement in 1971. Walter would become the first employee to reach 20 years of service and the only employee to be named as an inventor on more U.S. patents than Wallace (95 and 82, respectively).
In hindsight, 1958 was a year of significant beginnings. Kilby demonstrated the first integrated circuit, an oscillator. Soon afterward, Noyce and Hoerni developed the planar process that enabled the microelectronics explosion. Townes and Schawlow described requirements for masers to function at optical frequencies, and within two years, Maiman demonstrated the first laser. All of these developments would play significant roles in the future of the new company.
At the beginning, replications of the prototypes, now known as the Model A Coulter Counter®, were assembled by Ernie Yasaka as Wallace could sell them. For industrial use,
15
a stirrer was added to the sample stand (Fig. 4). Of immediate concern to the new company was a paper demonstrating a direct correlation between pulse amplitude and particle volume.
16
By coupling a Coulter Counter® to a single-channel pulse-height analyzer (PHA) having dual variable pulse thresholds, Kubitschek had generated the first differential size distribution, thus highlighting two disadvantages of the Model A: First, its single threshold required generation of a cumulative size distribution via multiple sample runs at successively increasing thresholds,14, 16 requiring substantial time and calculation to manually produce a differential size distribution. The need for automated sizing was apparent. Second, the voltage source used to provide aperture current made pulse amplitudes sensitive to the dimensions of the particular aperture, the resistivity of the particular suspending medium, and temperature-induced variation in the latter,14, 15 all of which would thus complicate automation of accurate counting and sizing.
The Model A Coulter Counter® with the industrial version of the sample stand (Fig. 3) shown at the right. The round black object at the upper right of the stand is the stirrer motor used to keep heavy industrial particles in suspension; the stirrer was not used on the stand for blood cell counting. The console contains, from left to right, the mechanical totalizer used for the slowly accumulating high-value digits, the three decade counters used for the rapidly accumulating low-value digits, and the oscilloscope display tube. Controls for the single threshold and the aperture current appear below the display tube.
Encouraged by favorable results on automated leukocyte counting, 17 the company introduced an improved instrument 18 in 1960, just as broad interest in accurate cell and particle sizing was emphasized. 19 A current source for aperture excitation replaced the original voltage source, and a dual-threshold current-sensitive amplifier for sensing resultant particle pulses replaced the original single-threshold voltage amplifier. 12 Consequently, the Model B Coulter Counter® was practically insensitive to the factors limiting the original counter, and with its thresholds interlocked to form a movable channel controlled by a sequencing four-second timer, it allowed an accessory Model H distribution plotter 20 to automatically accumulate a 25-channel differential size distribution from 100-s sample runs. 18 Development of the Model C Coulter Counter was also advancing—the prototype included a 12-channel PHA. Not only did its more than 350 vacuum tubes contribute significantly to heating the company's facility, the resulting bulk also required disassembly for it to be moved. By 1961, when the company relocated from Chicago to Hialeah, Florida, a tabletop Model C was available to industry. 21
During the 1960s, Model A and Model B counters proved useful for the counting and sizing of both erythrocytes and leukocytes17,18, 22–28 (see reviews25,27) and were gaining application in microbiology16,29,30 and industrial particle analysis. 31 Meanwhile, worries arose. In 1959, a competitive instrument based on the Coulter Principle was described. 32 In 1960, the skewed distributions seen in the NIH study 9 were confirmed. 23 The cumbersome diluter 13 provoked a customer to design his own 22 and a better Coulter design. 33 In 1962, the length-to-diameter ratio of the sensing aperture was reported to affect sizing resolution. 34 A future competitor patented a derivative form of the Coulter Principle, 35 and a principal of Coulter Sales Corporation patented a derivative sample tube on his own, 36 later leaving to become another competitor. 37 Further, early efforts at platelet counting encountered unexpected interference, apparently from small particles not seen by phase microscopy. 38 Lushbaugh et al. raised the sizing ante by coupling a 100-channel PHA to a Model A Coulter Counter®,39,40 and soon, several groups were coupling purpose-built Coulter particle sensors to commercial PHAs29, 41 48 providing as many as 512 channels. 29 The increasing availability and sophistication of volumetric tools increasingly made artifacts apparent in size distributions for a variety of cells and particles. In Wallace's snapshot phrasing, 1 “Challenges are good, and we sure had our share of good.”
Act III
The sensing aperture (the hole in wafer A, Fig. 3) was the heart of the Coulter Principle, and designing a Coulter Counter® to automatically compensate for its functional characteristics now became the main priority. One of the NIH studies had noted the aperture's sensitive volume being about three times that of the geometric aperture, a consequence of the electric field established by the excitation current throughout the volumes of suspending medium in the sample vessel and sample tube (Fig. 3). Cells (or particles) interacted with this electric field as they were carried through the sensing aperture by the analogous hydrodynamic field produced by the metering system. For both aperture fields, the significant particle interactions occurred in a sensitive volume containing the sensing aperture and extending semi-elliptically outward from its entry and exit orifices approximately three or four aperture diameters.
Thus, by displacing volumes of the conductive suspending medium equal to its own, each cell distorted the electric field throughout its extent—but most significantly while passing through the sensing aperture. 49 As a result, the volume of the cell was compared to that of the aperture. A change in the electrical resistance of the aperture, typically of about 1 part in 50,000, and the accompanying small change in ionic current through the aperture, produced the signal pulses that enabled counting and sizing of the cells. Accurate counting and repeatable sizing thus required extremely smooth suspension flow through the aperture.
Unlike the ionic current flow, due to the suspending medium's mass and viscosity, suspension flow was influenced by both inertia and a boundary layer at the surface of the sensing aperture, respectively. Consequently, these reacted to the microgeometry of the aperture and its two orifices, 50 producing a kinetic flow field that was asymmetric about the midpoint of the aperture axis. The toroidal recirculating flow pattern at the aperture's exit orifice carried particles back into the aperture's sensitive volume, thereby generating secondary pulses that erroneously contributed to the particle count. For example, Walter Hogg found that the phantom particles encountered in an early platelet study 38 were erythrocytes recirculating into the sensitive volume, their secondary pulses causing them to also be counted as platelets. Auxiliary flow sweeping the particles away from the exit orifice prevented both particle recursion and secondary pulses. 51
It was always expected that coincident passage of cells through the aperture's sensitive volume would reduce cell counts through masked particle pulses,4, 7 but count loss was statistically predictable from cell concentration in the suspension.9, 52 60 Thus, single-channel counts were automatically correctable by suitable circuitry, 61 and a late version of the Model D Coulter Counter® introduced this approach. However, atypical pulses resulting from coincident particle passage also induced broadening in size distributions, 45 and typical erythrocyte size distributions demonstrated skewness.39,41,44,62 The skewness, attributed in one of the NIH studies 9 to coincidence, was soon found in size distributions for other cell types 38 and particles.31, 63, 64 With improvements in volumetric accuracy, size distributions proved to be bi-modal for erythrocytes.40, 41, 65 All such distribution artifacts reduced sizing resolution.
By the late 1960s, studies related to origins of volumetric artifacts were burgeoning,28,30,44,45,49,65–78 and several groups were building experimental systems based on the Coulter Principle.28–30,41,45–47,73,79 The troublesome sizing artifact arose when particles passed through the sensing aperture at different radial distances from its axis. 65 At low effective particle concentrations, apertures having lengths several times their diameter were shown to improve sizing resolution,34,41,67,80 as did an auxiliary flow surrounding a smaller suspension stream so as to hydrodynamically focus particles through the aperture near its axis.81–85 At typical particle concentrations, sampling signal pulses at aperture midpoint 67 or selecting them according to duration 79 substantially improved volumetric accuracy (see overviews65,67,78,86–88).
A very significant result of the investigation into aperture functional properties was the first cell sorter,48,70, 89–91 invented and built to determine whether bimodal erythrocyte distributions40, 41 were fact or artifact. Fulwyler combined the Coulter Principle with inkjet technology, and used the result to sort cells from a single distribution mode. When the sorted cells were resized, the resulting distribution demonstrated the original bimodal form and, thereby, the artifactual nature of such distributions.
During this investigative explosion, models of the Coulter Counter® followed advances in electronics, the transistorized Model F being developed concurrently with the Model C, to replace the Model A. The sample stand was adapted to flow-through use92, 93 and the Model J distribution plotter replaced the Model H plotter. In the latter part of 1968, the first automated hematology analyzer, 94 the seven-parameter Coulter Counter® Model S, was introduced.95, 96 Simultaneously, the transistorized Model T Coulter Counter® replaced the Model C for industrial applications. Based on integrated circuitry, the Z family of counters was released in 1970, and the Channelyzer® volume analyzer appeared in 1972, simultaneously with the industrial TA family, which unitized counter and 16-channel PHA circuitry. Soon after, instruments came to rely on microprocessors.
Wallace often commented,
1
“If it's useful, people will buy it.” Sales had increased with each level of improvement, but there was unprecedented demand for the Model S Coulter Counter® . Although a solid foundation for an industry was now in place, volumetric artifacts originating in particle interactions with the fields in the vicinity of the aperture82,83 still required solutions that neither increased particle coincidence nor reduced the effective particle concentration within the aperture. Characteristics of the artifactual pulses were sufficiently distinctive that these pulses could be automatically edited from the pulse-data stream by specialized circuitry, and numerous such pulse-editing circuits were developed. Some of these enabled the performance of the automated nine-parameter Model S-Plus and the S-Plus II two-part differential analyzers introduced in 1977 and 1980, respectively. Others were used in the Coulter STKRTM, with its fully automated walkaway sample handling system, or with the VCS flow-cell technology introduced in 1986 and 1987, respectively. This design philosophy continues in the current Coulter LH 700 Series (Fig. 5).
The latest descendant of the Model A Coulter Counter®, the LH 750 analyzer, is a fully automated instrument that determines 26 reportable hematological parameters. When combined with the LH Slide Maker and the LH Slide Stainer to form the LH 755 Hematology System, it also automates preparation of microscope slides from selected whole-blood samples.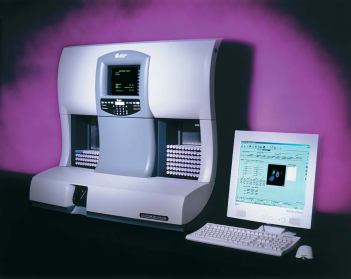
Summary
The hole that could not be patented inspired a principle that could. Originating in Wallace's desire to automate a tedious routine, the Coulter Principle provided a repeatable physical measurement of cell or particle volume when appropriately calibrated and, in turn, has inspired a broad range of automated instrumentation of increasing sophistication and complexity. The broad acceptance of instruments incorporating Wallace's Principle has prompted development of similar instruments by a number of companies. The Coulter Principle remains the method of choice for volume measurement of microscopic particles and has become incorporated in numerous standards in a variety of scientific fields. 31 By the late-1980s there were some 50,000 Coulter Counters® in operation in the United States alone. Publications describing practical applications or theoretical design aspects now number in the thousands, and related patents now number in the hundreds.
When Beckman Instruments, Inc., acquired Coulter Corporation in late 1997, the Coulter brothers’ company had grown to some 5000 employees, with revenues in the hundreds of millions of dollars annually.3, 108 Acceptance of their instruments enabled support of automation in other technical areas, most notably flow sorters and flow cytom-eters, which owe major debts to the Coulter sensing aperture and its apparent simplicity. 86 Among other applications of laboratory automation the brothers supported was pattern-recognition microscopy, 109 experience from which contributed to the Coulter LH Slide Maker (Fig. 5).
Wallace's basic patent 4 defined his principle via two forms of sensing aperture, both in dielectric material. He preferred the axial excitation/sensing version due to its ease of manufacture and linear volume response. Interestingly, due to the electric double layer that forms on conductive materials exposed to an electric field in ionic media, this form can be made to work even if the aperture is formed in some conductive materials.110, 111 In addition to particle volume, the resulting signal pulses are responsive to the electrochemical properties of the particle, the material containing the sensing aperture, and the suspending media. The axial form also can function if the sensing aperture is made in a composite structure comprising Wallace's original aperture wafer between two conductive elements.87, 88 The resulting size distributions can have minimal artifact due to particles either recirculating at the aperture exit orifice or passing through the aperture away from its axis. 112
The axial implementation of the Coulter Principle has by far received the greatest attention, but attempts to scale it downward in size encounter a limit due to thermal noise. In principle, the second form using excitation and sensing transverse to the suspension flow may offer the ability to sense smaller particles, but at expense of requiring a volume linearization based on the selected geometry of aperture and electrodes.35, 113–115
Will Act IV cast the Coulter Principle in a role including one of these, or will it feature a totally new combination of emerging science and familiar technology?
Epilogue
While Wallace was still looking for the insight that led him to the Coulter Principle, his father wrote a poem for the brothers: 1 Why not direct it, / It's within your control. / You can nourish and guide it, / You can reach your goal! / Make use of this gift, / Let it labor for good. / Your thoughts are your life; / Geniuses use them—you should!
This play in three acts suggests that both he and Joseph understood well their father's advice. Wallace saw few problems but many opportunities. He was widely recognized in the scientific community for his inventive insights. Joseph acknowledged problems but saw them as challenges. He implemented much, in both technical and business areas. Both men believed that science should serve humanity, and the two combined their complementary strengths to successfully initiate or foster numerous developments in today's scientific armamentarium. The tremendous humanitarian value of their efforts needs no comment.
Despite their many achievements, the Coulter brothers (Fig. 6) remained both modest and enthusiastic about innovative ideas. They were devoted to family, which to both included employees, and were dependable friends. Both were always ready to unselfishly share their time, experience, and knowledge. Both expected a self-reliant best effort but were supportive when a goal honestly pursued proved unreachable. When one of the author's well-intentioned efforts proved not only counterproductive but also costly, Wallace met an apology with, “People who don't try, don't make mistakes,” to which Joseph added, “Some things are more important than money.”
Wallace (left) and Joseph Coulter in the mid 1990s. Wallace was born February 13, 1913 and died August 7, 1998. Joseph was born August 18, 1924 and died November 27, 1995. In life, both acted on a belief that science should serve humanity, and their legacy is the humanitarian value of their efforts.
It has been remarked that a person is fortunate to have one good teacher. Those who were privileged to work with the Coulter brothers had two. This brief retrospective is dedicated to the memory of these exceptional brothers who both enjoyed and encouraged the quest to do better.
