Abstract
To provide guidance to clinicians about best practices, the Wilderness Medical Society (WMS) convened an expert panel to develop evidence-based guidelines for prevention, diagnosis, and treatment of acute mountain sickness, high altitude cerebral edema, and high altitude pulmonary edema. Recommendations are graded based on the quality of supporting evidence and the balance between the benefits and risks/burdens according to criteria put forth by the American College of Chest Physicians. The guidelines also provide suggested approaches for managing each form of acute altitude illness that incorporate these recommendations as well as recommendations on how to approach high altitude travel following COVID-19 infection. This is an updated version of the original WMS Consensus Guidelines for the Prevention and Treatment of Acute Altitude Illness published in
Keywords
Introduction
Travel to elevations above 2500 m by unacclimatized lowlanders is associated with risk of developing 1 or more forms of acute altitude illness: acute mountain sickness (AMS), high altitude cerebral edema (HACE), and high altitude pulmonary edema (HAPE). Because large numbers of people travel to such elevations, many clinicians are faced with questions from patients about the best means to prevent these disorders. In addition, clinicians working at facilities in high altitude regions or as members of expeditions traveling to such areas can expect to see persons who are suffering from these illnesses and must be familiar with prophylactic regimens, diagnostic approaches, and treatment protocols.
To provide guidance to clinicians and disseminate knowledge about best practices, the Wilderness Medical Society (WMS) convened an expert panel to develop evidence-based guidelines for prevention, diagnosis, and treatment of acute altitude illness. We present preventive and therapeutic modalities and make recommendations on how to approach each type of acute altitude illness. Recommendations are graded based on the quality of supporting evidence and consideration of benefits and risks/burdens associated with each modality. These recommendations are intended to apply to all unacclimatized travelers to high altitude whether they are traveling to high altitude for work, military deployments, tourism, or other activities, including hiking, skiing, trekking, and mountaineering.
Methods
This set of guidelines is an updated version of the original WMS Consensus Guidelines for the Prevention and Treatment of Acute Altitude Illness published in
The original expert panel was convened at the 2009 annual meeting of the WMS in Snowmass, Colorado. The WMS selected members based on their clinical and/or research experience. Additional experts have been added over time at the discretion of the original panel members. The panel identified new relevant articles for this update through the MEDLINE database by keyword search using the terms acute mountain sickness, high altitude pulmonary edema, high altitude cerebral edema, treatment, diagnosis, prevention, acetaminophen, acetylsalicylic acid, acetazolamide, dexamethasone, ibuprofen, nifedipine, tadalafil, sildenafil, and salmeterol. We reviewed English language, peer-reviewed studies that included adults and/or children and related to prevention and treatment of acute altitude illnesses, including randomized controlled trials, observational studies, and case series, and assessed the level of evidence supporting various preventive and treatment modalities. We excluded animal studies and abstract-only studies. We did not consider conclusions from review articles in the formulation of recommendations but have cited them to provide relevant background information on acute altitude illnesses and their management. The panel used a consensus approach to develop recommendations and graded each recommendation according to criteria stipulated in the American College of Chest Physicians statement on grading recommendations and strength of evidence in clinical guidelines (see online Supplemental Table). 4
Defining the Threshold for “High Altitude” and When to Apply These Guidelines
Unacclimatized individuals are at risk of high altitude illness when ascending to altitudes above 2500 m. Prior studies and extensive clinical experience, however, suggest that susceptible individuals can develop AMS and, in rare circumstances, HAPE at elevations as low as 2000 m.5-7 High altitude cerebral edema is typically encountered at higher elevations but has also been reported at around 2500 m in patients with concurrent HAPE. 8 Part of the difficulty in defining a specific threshold at which altitude illness can develop is the fact that the symptoms and signs of AMS, the most common form of altitude illness, are nonspecific, as demonstrated in several studies in which participants developed the same symptoms as those seen in AMS despite no gain in altitude.9-11 As a result, studies assessing AMS prevalence at modest elevations may label individuals as having altitude illness when in fact symptoms are related to some other process, erroneously elevating the reported incidence of AMS at that elevation.
Recognizing the difficulty in defining a fixed threshold, the expert panel recommends an approach to prevention, diagnosis, and treatment of acute altitude illness that does not depend strictly on the altitude to which an individual is traveling. Preventive measures should be considered based on the destination altitude and also account for factors such as a history of acute altitude illness, the planned rate of ascent, and availability of acclimatization days (described in detail below). The diagnoses of AMS, HAPE, or HACE should not be excluded solely based on the fact that an ill individual is below 2500 m. These diagnoses should be strongly considered in the presence of compatible clinical features, with careful attempts to exclude other problems, such as carbon monoxide poisoning, hypoglycemia, hyponatremia, pneumonia, severe dehydration, and viral illness.
AMS and HACE
Information on epidemiology, clinical presentation, and pathophysiology of AMS and HACE is provided in several extensive reviews.12-16 From a clinical standpoint, HACE, unlike HAPE, represents an extremely severe form of AMS. Therefore, we address the prevention and treatment of these 2 disorders simultaneously.
Prevention
Measures considered for prevention of AMS and HACE include the following:
Gradual Ascent
Controlling the rate of ascent, in terms of the number of meters gained per day, is a highly effective means of preventing acute altitude illness.17-20 In planning the rate of ascent, the altitude at which someone sleeps is considered more important than the altitude reached during waking hours.
Recommendation
We recommend gradual ascent, defined as a slow increase in sleeping elevation, for AMS prevention. A specific approach is described further below. Strong recommendation, moderate-quality evidence.
Acetazolamide
Multiple trials have established a role for acetazolamide in the prevention of AMS.17,21-24
Acetazolamide contains a sulfa moiety but carries an extremely low risk of inciting an allergic reaction in persons with sulfonamide allergy. As a result, persons with known allergy to sulfonamide medications can consider a supervised trial of acetazolamide prior to the trip, particularly if planning travel to a location remote from medical resources.25,26 Prior anaphylaxis to a sulfonamide medication or a history of Stevens-Johnson syndrome is a contraindication to acetazolamide.
Some studies, using higher than recommended doses, have suggested that acetazolamide adversely affects maximum exercise capacity,27,28 perceived dyspnea during maximal exercise tests, 29 leg endurance, 30 and respiratory muscle function at high levels of work, 31 although a more recent study with appropriate controls showed no effect on 2-mile exercise performance after 2- or 24-h exposure to 3500 m. 32 If acetazolamide has any detrimental effects on exercise, the observed changes are small and unlikely to affect overall performance in most travelers or the chance of summit success for climbers at moderate and even extreme elevations. Further, the benefits of preventing AMS may outweigh any theoretical decrease in maximal exercise performance.
The recommended adult dose for prophylaxis is 125 mg every 12 h (Table 1). A small study suggested that 62.5 mg every 12 h was noninferior to 125 mg every 12 h, 33 but a larger randomized, controlled trial demonstrated that 62.5 mg was less effective than 125 mg in preventing AMS. 34 Higher doses (eg, 250 mg every 12 h) can be considered for high-risk ascent profiles (Figure 1) up to elevations of 5000 m, although direct comparisons of the 125 mg and 250 mg regimens in high-risk settings have not been conducted. The appropriate dose for ascent to elevations above 5000 m has not been studied. The pediatric dose of acetazolamide is 1.25 mg·kg−1·dose−1 (maximum, 125 mg·dose−1) every 12 h. 35 The medication should be started the night before ascent, but day-of-ascent dosing still yields benefit for those unable to begin the medication the night before. 36
Recommended dosages for medications used in the prevention and treatment of altitude illness
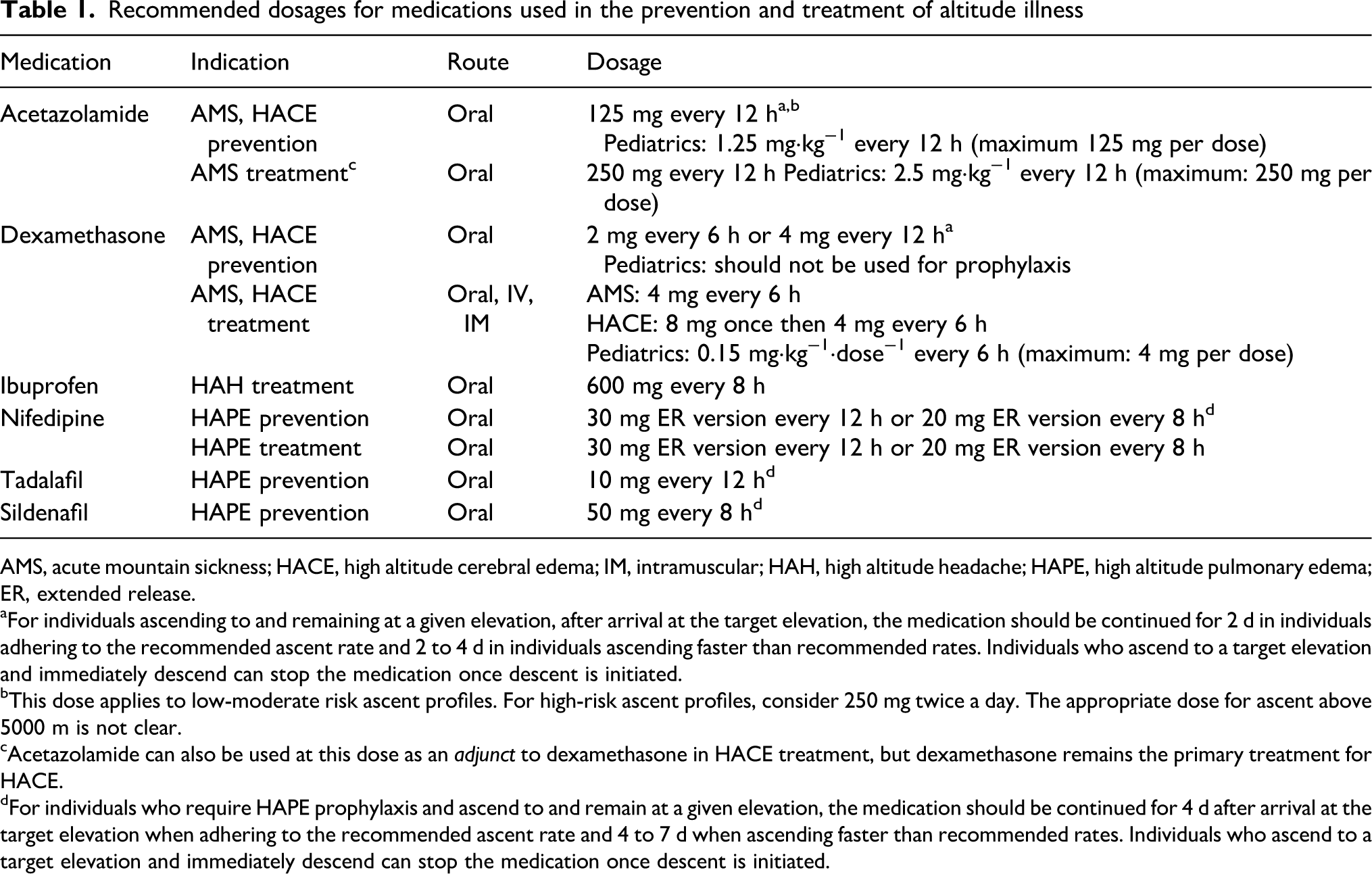
AMS, acute mountain sickness; HACE, high altitude cerebral edema; IM, intramuscular; HAH, high altitude headache; HAPE, high altitude pulmonary edema; ER, extended release.
aFor individuals ascending to and remaining at a given elevation, after arrival at the target elevation, the medication should be continued for 2 d in individuals adhering to the recommended ascent rate and 2 to 4 d in individuals ascending faster than recommended rates. Individuals who ascend to a target elevation and immediately descend can stop the medication once descent is initiated.
bThis dose applies to low-moderate risk ascent profiles. For high-risk ascent profiles, consider 250 mg twice a day. The appropriate dose for ascent above 5000 m is not clear.
cAcetazolamide can also be used at this dose as an
dFor individuals who require HAPE prophylaxis and ascend to and remain at a given elevation, the medication should be continued for 4 d after arrival at the target elevation when adhering to the recommended ascent rate and 4 to 7 d when ascending faster than recommended rates. Individuals who ascend to a target elevation and immediately descend can stop the medication once descent is initiated.

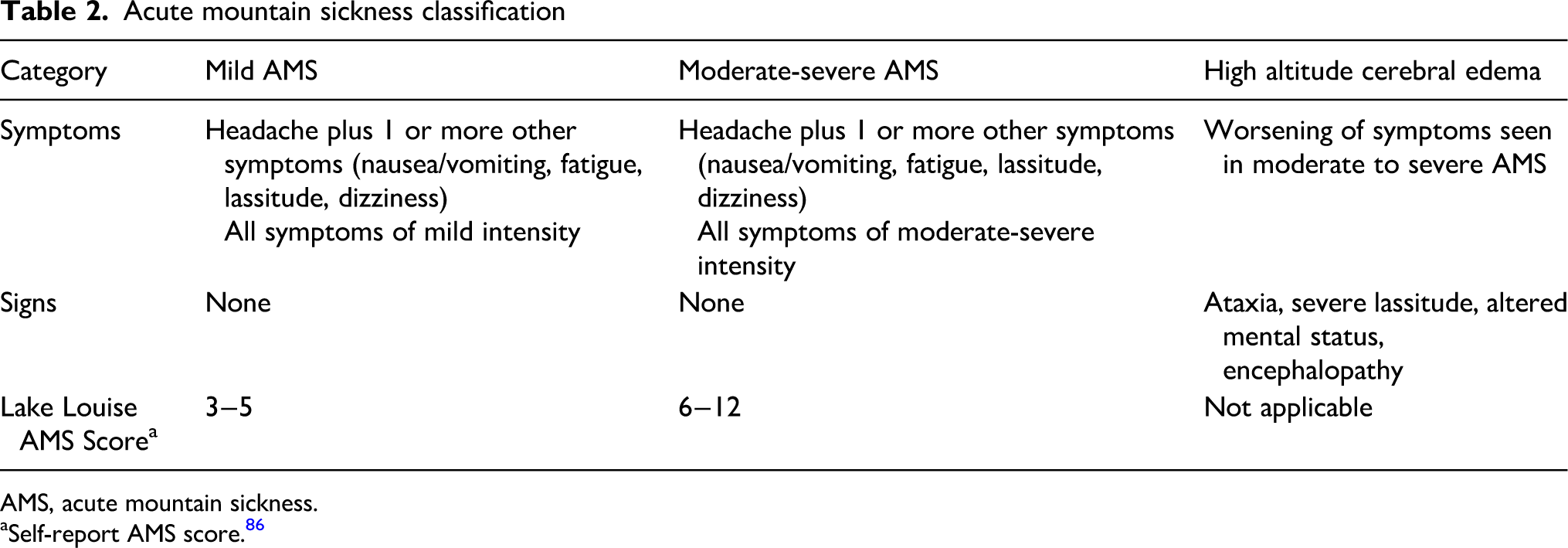
Assessing the risk of acute altitude illness. Medical history and features of the planned ascent can be used to assess the risk of acute altitude illness after ascent. Check marks should be placed in the boxes that best describe the variables in the left-hand column. The risk of a planned ascent is determined by the farthest column to the right in which a check mark is placed. This assessment applies to unacclimatized individuals. Ascent is assumed to start from elevations <1200 m. A history of acute altitude illness does not necessarily reflect high risk with all future ascents, as a slower ascent rate or lower target elevation on subsequent trips may help avoid problems. The risk of travel above any given elevation can be mitigated by ensuring an appropriately slow rate of ascent. The severity of prior AMS can be graded using the information in Table 2. AMS, acute mountain sickness; HACE, high altitude cerebral edema; HAPE, high altitude pulmonary edema.
Recommendation
We recommend that acetazolamide be strongly considered in travelers at moderate or high risk of AMS (Figure 1). Strong recommendation, high-quality evidence.
Recommendation
We recommend that acetazolamide can be used in children for prevention of AMS. Strong recommendation, low-quality evidence.
Dexamethasone
While dexamethasone does not facilitate acclimatization like acetazolamide, prospective trials have established a benefit for dexamethasone in AMS prevention.37-40 The recommended adult doses are 2 mg every 6 h or 4 mg every 12 h. High doses (4 mg every 6 h) may be considered in very high-risk situations, such as military or search and rescue personnel being airlifted to altitudes >3500 m with immediate requirement of physical activity, but should be limited to these circumstances. Prolonged use carries a risk of adrenal suppression. Tapering is not required if dexamethasone use is limited to 5 to 7 d but may be necessary with longer duration. Given the absence of data on the use of dexamethasone for AMS prevention in children and the availability of other safe alternatives–specifically graded ascent and acetazolamide–dexamethasone is not recommended for AMS prevention in children.
Recommendation
We recommend that dexamethasone be used as an alternative to acetazolamide for adult travelers at moderate or high risk of AMS. Strong recommendation, high-quality evidence.
Inhaled budesonide
Two studies indicated that inhaled budesonide 200 micrograms twice daily was effective at preventing AMS when compared to placebo.41,42 These studies were limited by methodological issues, such as timing of the assessment for AMS 41 and number of participants in each study arm. 42 A clear mechanism of action was not apparent in these studies, but small improvements in spirometry and oxygen saturation–both of little clinical significance–were suggested as evidence that the benefit might derive from a direct lung effect. More recent, well-designed, randomized, controlled trials failed to replicate these results.43,44
Recommendation
We recommend that inhaled budesonide not be used for altitude illness prophylaxis. Strong recommendation, high-quality evidence.
Ginkgo biloba
Although 2 trials demonstrated a benefit of
Recommendation
We recommend that
Ibuprofen
Two trials demonstrated that ibuprofen (600 mg 3 times daily) is more effective than placebo at preventing AMS,52,53 while a third, smaller study showed no benefit. 54 Another study claimed to show benefit, but the trial did not include a placebo arm and, instead, compared the incidence of AMS with ibuprofen with historically reported rates from the same region in which the study was conducted. 55 While no studies have compared ibuprofen with dexamethasone, 2 studies have compared ibuprofen with acetazolamide. The first found an equal incidence of high altitude headache and AMS in the acetazolamide and ibuprofen groups, with both showing significant protection compared to placebo. 56 A more recent trial failed to show that ibuprofen was noninferior to acetazolamide (ie, ibuprofen was inferior to acetazolamide for AMS prophylaxis). 57 The above trials all used the medication for a short duration (∼24–48 h). As a result, efficacy and safety (eg, the risk of gastrointestinal bleeding or renal dysfunction) over longer periods of use at high altitude remain unclear. For these reasons, as well as more extensive clinical experience with acetazolamide and dexamethasone, ibuprofen is second-line to either of these medications for AMS prevention with rapid ascent.
Recommendation
We suggest that ibuprofen be used for AMS prevention in persons who do not wish to take acetazolamide or dexamethasone or have allergies or intolerance to these medications. Weak recommendation, moderate-quality evidence.
Acetaminophen
A single study demonstrated that acetaminophen 1000 mg 3 times daily was as effective as ibuprofen at preventing AMS in trekkers traveling between 4370 and 4940 m in elevation. 55 Rather than including a placebo arm, the study attempted to establish the benefit of acetaminophen by comparing the incidence rates in this study with those of untreated trekkers from prior studies that used the same ascent profile. Based on these data, acetaminophen is not recommended for use as a preventive agent over acetazolamide or dexamethasone.
Recommendation
We recommend that acetaminophen not be used for AMS prevention. Strong recommendation, low-quality evidence.
Staged Ascent and Preacclimatization
Two studies showed that spending 6 to 7 d at moderate altitude (∼2200−3000 m) prior to proceeding to higher altitude (referred to as “staged ascent”) decreases the risk of AMS, improves ventilation and oxygenation, and blunts the pulmonary artery pressure response following subsequent ascent to 4300 m.19,58 A more recent study has shown that a 2-d stay at 3000 m is also effective at preventing AMS upon further ascent to 4300 m. 20 Many travelers to high altitude visit mountain resorts at more moderate elevations between 2500 and 3000 m. The value of short stays at intermediate elevations of ∼1500 m for decreasing the risk of AMS with such ascents makes sense from a physiologic standpoint, but aside from a cross-sectional study showing a decreased risk of AMS in travelers to resort communities between 1920 and 2950 m when they spent 1 night at 1600 m prior to ascent, 6 this approach has not been studied in a prospective randomized fashion.
A larger number of studies examining the effects of repeated exposures to hypobaric or normobaric hypoxia in the days and weeks preceding high altitude travel (referred to as “preacclimatization”) demonstrate mixed results, with some studies showing benefit in terms of decreased AMS incidence or severity59-61 and others showing no effect.62-65 A significant challenge in interpreting the literature on preacclimatization is variability among studies in the hypoxic exposure protocols as well as the fact that not all studies include evidence that their protocols induced physiologic responses consistent with acclimatization.
Implementation of either staged ascent or preacclimatization may be logistically difficult for many high altitude travelers. In general, short-term exposures (eg, 15−60 min of exposure to hypoxia or a few hours of hypoxia a few times prior to ascent) are less likely to aid acclimatization, while longer exposures (eg, >8 h daily for >7 d) are more likely to yield benefit. Hypobaric hypoxia is more effective than normobaric hypoxia in facilitating preacclimatization and preventing AMS. 66 Because the optimal methods for preacclimatization and staged ascent have not been fully determined, the panel recommends consideration of these approaches but does not endorse a particular protocol.
Recommendation
We recommend that a staged ascent and preacclimatization be considered as a means for AMS prevention. Strong recommendation, low-quality evidence.
Hypoxic Tents
Commercial products are available that allow individuals to sleep or exercise in hypoxic conditions for the purpose of facilitating acclimatization prior to a trip to high altitude. Only 2 placebo-controlled studies have examined their utility. The first 64 demonstrated no effect of 7 nights of normobaric hypoxia on the incidence of AMS or exercise performance the day after sleeping in hypoxia. The other study 67 demonstrated a lower incidence of AMS after 20 h in a hypobaric chamber in persons who spent 14 nights sleeping in a hypoxic tent compared to normoxia, but this difference was not statistically significant. In the latter study, technical difficulties with the system resulted in a substantial number of study participants receiving less than the intended hypoxic dose.
While the systems are marketed to be of benefit and anecdotal reports suggest they are widely used by climbers and other athletes competing at high altitude, there are no data indicating increased likelihood of summit success or improved physical performance. Benefits may theoretically accrue from these systems with long hypoxic exposures (>8 h/d) for >3 wk prior to planned high altitude travel, but such exposures have not been studied. Short and/or infrequent exposures, including exercise training, are likely of no benefit. In addition to the cost of the systems and power needed to run them, individuals face the risk of poor sleep, which over a long period of time could have deleterious effects on performance during an expedition.
Recommendation
We recommend that hypoxic tents should not be relied upon for facilitating acclimatization, preventing AMS, or increasing the odds of summit success. Strong recommendation, moderate-quality evidence.
Other Options
Chewed coca leaves, coca tea, and other coca-derived products are commonly recommended for travelers in the Andes for AMS prevention. Their utility in prevention of altitude illness has not been properly studied, so they should not be substituted for other established preventive measures described in these guidelines. Multiple studies have sought to determine whether other agents, including angiotensin receptor antagonists, 68 antioxidants, 69 iron, 70 dietary nitrates, 71 leukotriene receptor blockers,72,73 phosphodiesterase inhibitors, 74 salicylic acid, 75 spironolactone, 76 and sumatriptan, 77 can prevent AMS, but the current state of evidence does not support their use. “Forced hydration” or “overhydration” have never been shown to prevent altitude illness and might increase the risk of hyponatremia; however, maintenance of adequate hydration is important because symptoms of dehydration can mimic those of AMS. 78 Neither nocturnal expiratory positive airway pressure (EPAP) administered via a single-use nasal strip during sleep 79 nor a regimen of remote ischemic preconditioning is effective for AMS prophylaxis. 80
No studies have examined short-term oxygen use in the form of either visits to oxygen bars or over-the-counter oxygen delivery systems by which individuals inhale oxygen-enriched gas from a small prefilled canister. Due to the small volume of gas (2−10 L/canister) and short duration of administration, these interventions are unlikely to be of benefit and, as a result, have no role in AMS/HACE prevention. Other over-the-counter products, such as powdered drink mixes, patches, and oral supplements, also lack any evidence of benefit.
Suggested Approach to AMS/HACE Prevention
Because the rates of acclimatization and physiologic responses to high altitude vary considerably between individuals, clinicians must recognize that the following recommendations, while generally effective, do not guarantee successful AMS/HACE prevention in all high altitude travelers.
The approach to the prevention of AMS and HACE should consider the risk profile of the individual traveling to high altitude. Unfortunately, predicting the risk of acute altitude illness in individuals is challenging. While several models have been put forth, including one that utilizes the individual's history in conjunction with parameters obtained during exercise testing in hypoxia 81 and another that looks at the cumulative dose of hypoxia experienced with a planned ascent, 82 various factors limit their wide applicability. For this reason, we recommend a less formal approach that takes into account an individual's prior experiences at high altitude, if known, as well as aspects of their ascent profile (Figure 1). This method has not been systematically examined in research studies but is a practical approach that can be applied by a diverse group of travelers. The most conservative approach is to define risk based on the highest column with a check mark (ie, the column furthest to the right in the table with a check mark). It should be noted that a history of AMS does not necessarily reflect high risk with all future ascents, as a slower ascent rate or lower target elevation on subsequent trips may help avoid problems. Similarly, the lack of problems on prior trips does not necessarily imply a low risk of problems on future trips as faster ascent rates or higher target elevations may predispose to acute altitude illness.
The first priority for all individuals should be ensuring gradual ascent to the target elevation. Travelers can lower their risk by sleeping 1 night at an intermediate altitude. For example, sea-level residents traveling to Colorado resort areas over 2800 m can spend 1 night in Denver (1600 m). A large number of people will travel directly by car or plane to commonly visited mountain high altitude locations, often located between 2500 and 3000 m, and may be unable to ascend gradually due to various logistical factors. In such situations, pharmacologic prophylaxis may be considered. After the initial gain in elevation, maintaining a sufficiently slow ascent rate with further ascent should be a priority.
With travel above 3000 m, individuals should not increase the sleeping elevation by more than 500 m/d and should include a rest day (ie, no ascent to higher sleeping elevation) every 3 to 4 d. While only a single report has specifically shown the value of rest days in reducing the risk of AMS, 83 the concept is thought to be beneficial as it slows the overall ascent rate, thereby providing time for acclimatization. In many areas, terrain and other logistical factors prevent strict adherence to this approach and mandate larger gains in sleeping elevation over a single day. In such cases, acclimatization days should be strongly considered before and/or after these large gains in elevation and elsewhere in the itinerary to ensure, at the very least and as an approximation of properly controlled ascent, that the overall ascent rate averaged over the entire trip (ie, total elevation gain divided by the number of days of ascent during the trip) falls below the 500 m/d threshold.
Prophylactic medications are not necessary in low-risk situations but should be considered in addition to gradual ascent for use in moderate- to high-risk situations (Figure 1). Acetazolamide is the preferred medication; dexamethasone may be used as an alternative in adults with prior history of intolerance of or allergic reaction to acetazolamide. Concurrent use of acetazolamide and dexamethasone has only been studied in a single small, randomized trial 84 and should not be used outside of very limited, high-risk circumstances, such as climbers or rescue teams that must ascend rapidly to and perform physical work at >3500 m.
Pharmacologic prophylaxis should be started the day before ascent but still has beneficial effects if started on the day of ascent. 36 For individuals ascending to and staying at the same elevation for more than several days, prophylaxis may be stopped after 2 d at the highest altitude. Individuals ascending faster than the recommended ascent rates may consider continuing preventive medication for 2 to 4 d after arrival at the target altitude, but there are no data to support this approach. For individuals ascending to a high point and then descending toward the trailhead (eg, descending from the summit of Mount Kilimanjaro), in the absence of AMS/HACE symptoms, preventive medications should be stopped when descent is initiated.
Suggested Approach to Diagnosis of AMS and HACE
The diagnosis of AMS is made in a very specific clinical context, that of an unacclimatized lowlander who becomes ill within several hours to 3 d following ascent to high elevations, generally >2500 m. Diagnosis is based solely on reported symptoms, as there are no characteristic findings on physical exam or laboratory studies. The Lake Louise Acute Mountain Sickness Score85,86 and the Environmental Symptoms Questionnaire 87 are long-standing and well-known scoring systems used to diagnose AMS, but their use is limited to case identification in the research setting. Nevertheless, the symptoms evaluated in these scoring systems inform diagnosis in clinical settings; these are headache, anorexia, nausea, fatigue, and light-headedness or dizziness. Debates exist as to whether headache, long regarded as the cardinal feature of AMS, is required for diagnosis88,89 as well as whether disturbed sleep should be included in the diagnostic criteria.90-92 In line with the updated Lake Louise Acute Mountain Sickness Score, 86 these guidelines emphasize the role of headache and de-emphasize the role of sleep disruption in the diagnosis of AMS. Given the lack of a diagnostic gold standard, however, the best approach to diagnosis is to consider the traveler's well-being and functional status. 93 Individuals who feel ill and/or must reduce their daily activities several hours to 3 d following ascent to elevations >2500 m likely have AMS.
The diagnosis of HACE is heralded by signs of encephalopathy including ataxia–which is often the earliest clinical finding–and altered mentation. Other signs include apathy, irritability, lassitude, and inability to provide self-care, all of which can be subtle. Untreated HACE can progress to coma.13,94,95 Focal neurologic deficits are unusual and should prompt consideration of other diagnoses. Heel-to-toe walking can be used to gauge ataxia, and papilledema can confirm cerebral edema. Optic nerve sheath diameter measured by ultrasound may change with gains in elevation but is not reliable for diagnosis of AMS or HACE.96,97 In well-resourced settings, diagnostic testing including head imaging and laboratory studies can be used to rule out alternative diagnoses such as cerebrovascular accident, carbon monoxide poisoning, hypoglycemia, hyponatremia, infection, or traumatic brain injury. With HACE, magnetic resonance imaging (MRI) often shows a characteristic increased FLAIR and T2 signal in the corpus callosum as well as microhemorrhages consistent with blood-brain barrier disruption on susceptibility-weighted images, also mainly in the corpus callosum. 8 Because microhemorrhages can still be seen on MRI up to 120 mo following an episode of HACE,8,98 MRI scanning can be used to retrospectively evaluate cases in people seeking evaluation following a return to sea level.
Treatment
Potential therapeutic options for AMS and HACE include:
Descent
Descent remains the single best treatment for AMS and HACE. However, it is not necessary in all circumstances (discussed further below). Individuals should descend until symptoms resolve, unless terrain, weather, or injuries make descent impossible. Symptoms typically resolve following descent of 300 to 1000 m, but the required altitude decrease varies between persons. Individuals should not descend alone, particularly if they are suffering from HACE.
Recommendation
We recommend that descent is effective for any degree of AMS/HACE and is indicated for individuals with severe AMS or HACE. Strong recommendation, high-quality evidence.
Supplemental Oxygen
Oxygen delivered by a nasal cannula or mask at flow rates sufficient to relieve symptoms provides a suitable alternative to descent. An SpO2 (oxygen saturation measured by pulse oximetry) of >90% is usually adequate. The use of oxygen is not required in all circumstances and is generally reserved for mountain clinics and hospitals where supply is abundant. It should also be used when descent is indicated but not feasible or during descent in severely ill individuals. Supplemental oxygen should be administered to target an SpO2 of >90% rather than a specific fraction of inspired oxygen (FIO2). This is because the inspired oxygen fraction varies significantly between oxygen delivery systems, including nasal cannulas, simple facemasks, Venturi masks, or nonrebreather masks. Use of low-flow oxygen (1−2 L/min) for ≥2 h has much greater benefit than short bursts (several minutes) of large amounts of oxygen. Short visits to oxygen bars or use of over-the-counter oxygen cannisters has never been studied for AMS treatment and should not be relied on for this purpose.
Recommendation
We recommend that, when available, ongoing supplemental oxygen sufficient to raise SpO2 to >90% or relieve symptoms can be used while waiting to initiate descent or when descent is not practical. Strong recommendation, high-quality evidence.
Portable Hyperbaric Chambers
Portable hyperbaric chambers are effective for treating severe altitude illness99,100 but require constant tending by care providers and are difficult to use with patients who are claustrophobic or vomiting. Symptoms may recur when individuals are removed from the chamber, 101 but this should not preclude use of the chamber when indicated. In many cases, ill individuals may improve sufficiently to assist with their evacuation and descent once symptoms improve. Use of a portable hyperbaric chamber should not delay descent in situations where descent is required.
Recommendation
We recommend that, when available, portable hyperbaric chambers should be used for patients with severe AMS or HACE when descent is not feasible or must be delayed and supplemental oxygen is not available. Strong recommendation, moderate-quality evidence.
Acetazolamide
Only 1 study has examined acetazolamide for AMS treatment. The dose studied was 250 mg every 12 h; whether a lower dose might suffice is not known. 102 No studies have assessed AMS treatment with acetazolamide in pediatric patients, but anecdotal reports suggest it has utility. The pediatric treatment dose is 2.5 mg·kg−1·dose−1 every 12 h up to a maximum of 250 mg·dose−1.
Recommendation
We recommend that acetazolamide be considered for treatment of AMS. Strong recommendation, low-quality evidence.
Dexamethasone
Dexamethasone is very effective for treating AMS.103-105 The medication does not facilitate acclimatization, so further ascent should be delayed until the patient is asymptomatic without the medication. Although systematic studies have not been conducted, extensive clinical experience supports using dexamethasone in patients with HACE. It is administered as an 8-mg dose (intramuscular, IV, or oral administration), followed by 4 mg every 6 h until symptoms resolve. The pediatric dose is 0.15 mg·kg−1·dose−1 every 6 h. 35
Recommendation
We recommend that dexamethasone be considered for treatment of AMS. Strong recommendation, low-quality evidence.
Recommendation
We recommend that dexamethasone be administered for treatment of HACE. Strong recommendation, low-quality evidence.
Acetaminophen
Acetaminophen has been shown to relieve headache at high altitude 106 but has not been shown to improve the full spectrum of AMS symptoms or effectively treat HACE.
Recommendation
We recommend that acetaminophen can be used to treat headache at high altitude. Strong recommendation, low-quality evidence.
Ibuprofen
Ibuprofen has been shown to relieve headache at high altitude 106 but has not been shown to improve the full spectrum of AMS symptoms or effectively treat HACE.
Recommendation
We recommend that ibuprofen can be used to treat headache at high altitude. Strong recommendation, low-quality evidence.
Continuous Positive Airway Pressure
Continuous positive airway pressure (CPAP) carries theoretical benefit in acute altitude illness by virtue of its ability to increase the arterial partial pressure of oxygen (PO2). This impact is not due to increases in barometric pressure, as application of 15 cm H2O of CPAP, for example, yields only an 11 mm Hg increase in barometric pressure. Instead, CPAP works by increasing transmural pressure across alveolar walls, thereby increasing alveolar volume and improving ventilation-perfusion matching and gas exchange. Two reports have demonstrated the feasibility of administering CPAP to treat AMS,107,108 but this has not been studied in a systematic manner. Logistical challenges to use in field settings include access to power and the weight and bulk of these systems.
Recommendation
No recommendation can be made regarding use of CPAP for AMS treatment because of lack of data.
Suggested Approach to AMS/HACE Treatment
Care should be taken to exclude disorders whose symptoms and signs resemble those seen with AMS and HACE, such as carbon monoxide poisoning, dehydration, exhaustion, hypoglycemia, hypothermia, and hyponatremia.
People with AMS should avoid moving to higher sleeping elevations. Those with mild-moderate illness (Table 2) may remain at their current altitude and should be closely observed for progression of symptoms. It is sufficient to treat symptoms such as headache with nonopioid analgesics and nausea and vomiting with antiemetics. Descent should be initiated for AMS if symptoms worsen or fail to improve after 1 to 3 d of appropriate interventions. Persons with severe AMS should cease ascending and descend to lower elevation.
Acetazolamide and dexamethasone are generally not necessary for treatment of mild AMS but may be useful in moderate-severe cases. In these cases, dexamethasone should be strongly considered along with descent. While acetazolamide facilitates acclimatization, it is likely better for the prevention rather than the treatment of AMS. Nonetheless, it may also be considered for treatment in moderate-severe cases along with dexamethasone or if dexamethasone is unavailable. Both medications may facilitate descent by improving symptoms and allowing the individual to descend under their own power. Individuals with AMS may resume ascending once symptoms resolve. Further ascent or reascent to a previously attained altitude is contraindicated if there are ongoing symptoms. After resolution of AMS, prophylactic doses of acetazolamide should be used during reascent.
Individuals developing HACE in locations with access to hospitals or specialized clinics should receive supplemental oxygen sufficient to achieve an SpO2 of >90%. In remote areas away from medical resources, descent should be initiated in any suspected HACE victim. If descent is not feasible, supplemental oxygen or a portable hyperbaric chamber should be used. All persons with HACE should also be started on dexamethasone. There are no systematic data or case reports about reascent during the same trip or expedition following resolution of HACE. The prudent course is to avoid reascent in this situation.
HAPE
Information on the epidemiology, clinical presentation, and pathophysiology of HAPE, the majority of which comes from studies in adults, is provided in extensive reviews.14,15,109,110 While some of the prophylactic and therapeutic modalities are the same for HAPE as for AMS and HACE, important differences in the underlying pathophysiology mandate certain specific prevention and treatment approaches.
Prevention
Potential preventive measures for HAPE include the following:
Gradual Ascent
No studies have prospectively assessed whether limiting the rate of increase in sleeping elevation prevents HAPE; however, a clear relationship between rate of ascent and HAPE incidence exists.17,111-113
Recommendation
We recommend that gradual ascent be utilized to prevent HAPE. Strong recommendation, moderate-quality evidence.
Nifedipine
Given the role that excessive hypoxic pulmonary vasoconstriction plays in the development of HAPE, 110 pulmonary vasodilators have long been viewed as important for HAPE prevention. A single, randomized, placebo-controlled study 114 and extensive clinical experience have established a role for nifedipine in HAPE prevention in susceptible individuals. The recommended dose is 30 mg of the extended release preparation administered every 12 h. Hypotension was not noted in the study cited above 114 and is generally not a concern with the extended release version of the medication but may occur in a limited number of individuals.
Recommendation
We recommend that nifedipine be used for HAPE prevention in HAPE-susceptible people. Strong recommendation, moderate-quality evidence.
Salmeterol
In a single, randomized, placebo-controlled study, the long-acting inhaled beta-agonist salmeterol decreased the incidence of HAPE by 50% in susceptible individuals. 115 This study used very high doses (125 micrograms twice daily) that are often associated with side effects, including tremor and tachycardia. Clinical experience with salmeterol at high altitude is limited.
Recommendation
We suggest that salmeterol should not be used for HAPE prevention. Weak recommendation, moderate-quality evidence.
Tadalafil
In a single, randomized, placebo-controlled trial, 10 mg every 12 h of tadalafil prevented HAPE in susceptible individuals. 116 The number of individuals in the study was small, and 2 of the 10 subjects in the tadalafil group developed incapacitating AMS and had to be withdrawn from the study prematurely. Clinical experience with tadalafil is lacking compared to nifedipine. As a result, further data are necessary before tadalafil can be recommended over nifedipine.
Recommendation
We recommend that tadalafil be used for HAPE prevention in known susceptible individuals who are not candidates for nifedipine. Strong recommendation, low-quality evidence.
Dexamethasone
In the same study that assessed the role of tadalafil in HAPE prevention, dexamethasone (8 mg every 12 h) also prevented HAPE in susceptible individuals. 116 The mechanism for this effect is not clear, and there is very little clinical experience using dexamethasone for this purpose. Further data are necessary before it can be recommended for HAPE prevention.
Recommendation
We recommend that dexamethasone can be used for HAPE prevention in known susceptible individuals who are not candidates for nifedipine and tadalafil. Strong recommendation, low-quality evidence.
Acetazolamide
Despite evidence that acetazolamide hastens acclimatization and blunts hypoxic pulmonary vasoconstriction in animal models117-119 and a single study in humans, 120 no data support a role in HAPE prevention. A randomized, placebo-controlled, double-blind study of 13 healthy unacclimatized lowlanders with a history of HAPE found no significant reduction in the incidence of HAPE or pulmonary artery pressure after rapid ascent to 4559 m in those taking acetazolamide compared with placebo despite reductions in AMS and improved oxygenation. 121 Clinical observations suggest that acetazolamide may prevent re-entry HAPE, 122 a disorder seen in individuals who reside at high altitude, travel to lower elevation, and then develop HAPE upon rapid return to their residence.
Recommendation
We recommend that acetazolamide not be used for HAPE prevention in those with a history of the disease during prior trips to high altitude. Strong recommendation, moderate-quality evidence.
Recommendation
We suggest that acetazolamide be considered for prevention of re-entry HAPE in people with a history of the disorder. Weak recommendation, moderate-quality evidence.
Preacclimatization and Staged Ascent
No study has examined whether preacclimatization strategies are useful for HAPE prevention. Staged ascent, with 7 d of residence at moderate altitude (∼2200 m), has been shown to blunt the hypoxia-induced increase in pulmonary artery pressure. 58 However, uncertainty remains as to the magnitude and duration of moderate altitude exposure necessary to yield benefit, and no study has specifically investigated whether the strategy is of benefit in HAPE-susceptible individuals. Although the risks of preacclimatization and staged ascent are likely low, feasibility is a concern for many high altitude travelers. Because the optimal methods for preacclimatization and staged ascent have not been fully determined, the panel recommends consideration of these approaches but cannot endorse a particular protocol for implementation.
Recommendation
We recommend that, when feasible, staged ascent and preacclimatization be considered for HAPE prevention. Strong recommendation, low-quality evidence.
Suggested Approach to HAPE Prevention
As noted earlier, because the rates of acclimatization and physiologic responses to high altitude vary considerably among individuals, the recommendations that follow, while generally effective, do not guarantee prevention in all high altitude travelers. A gradual ascent profile is the primary method for preventing HAPE; the recommendations provided above for AMS and HACE prevention also apply to HAPE prevention. Pharmacologic prophylaxis should only be considered for individuals with a history of HAPE, especially multiple episodes. Nifedipine is the preferred drug in such situations. It should be started the day prior to ascent and continued either until descent is initiated or the individual has spent 4 d at the highest elevation, perhaps up to 7 d if the individual ascended faster than recommended ascent rates. Note that these durations are longer than those for use of acetazolamide for AMS prevention and that systematic studies have not examined the 4- versus 7-d recommendation. For individuals ascending to a high point and then descending toward the trailhead (eg, descending from the summit of Kilimanjaro), prophylactic medications should be stopped when descent is initiated. Further research is needed before tadalafil or dexamethasone can be recommended over nifedipine for prevention. Acetazolamide facilitates acclimatization and prevents AMS but should not be relied upon for HAPE prophylaxis in known HAPE-susceptible individuals.
Suggested Approach to the Diagnosis of HAPE
The diagnosis of HAPE requires a very specific clinical context–an unacclimatized lowlander ascending to elevations ≥2500 m–and relies on a characteristic set of symptoms, including dyspnea on exertion out of proportion to previous experiences at high altitude or that experienced by other individuals at the same elevation. Nonproductive cough, fatigue, weakness, and gurgling sensation in the chest may also be present. With progression, individuals become dyspneic with mild exertion or at rest and may develop cyanosis and cough productive of pink frothy sputum.
The setting in which the individual presents for evaluation influences the diagnostic approach. In the field environment, where diagnostic resources are limited, diagnosis may be made on the basis of history alone. If available, pulse oximetry can confirm the presence of hypoxemia out of proportion to that expected for a given elevation, 123 a key feature for distinguishing HAPE from other sources of dyspnea, such as anxiety or poor physical conditioning, although care must be taken to avoid some of the pitfalls of such measurements at high altitude. 124 Identification of B-lines on portable ultrasound is a sensitive but nonspecific diagnostic tool,125,126 but there is currently no accepted threshold for the number of B-lines necessary to confirm diagnosis. Individuals presenting to well-resourced health facilities should undergo pulse oximetry, plain chest radiography, and electrocardiography. The presence of hypoxemia and either unilateral or diffuse bilateral alveolar opacities on plain chest radiography is sufficient to confirm the diagnosis in the appropriate clinical context. Chest computed tomography scanning and echocardiography are generally only warranted when the initial evaluation is unrevealing or other problems remain high on the differential diagnosis.
Consideration should be given to other causes of respiratory symptoms at high altitude, such as asthma, bronchospasm, mucous plugging, pneumonia, pneumothorax, pulmonary embolism, viral upper respiratory tract infection, heart failure, or myocardial infarction.
Treatment
Therapeutic options for HAPE include the following:
Descent
As with AMS and HACE, descent remains the single best treatment for HAPE. Individuals should try to descend at least 1000 m or until symptoms resolve. They should exert themselves as little as possible while descending (eg, via motor vehicle, helicopter, or animal transportation or, if they must travel under their own power, without a pack) because exertion can further increase pulmonary artery pressure and exacerbate edema formation.
Recommendation
We recommend that descent is initiated for individuals with HAPE. Strong recommendation, high-quality evidence.
Supplemental Oxygen
Oxygen delivered by a nasal cannula or mask at flow rates sufficient to achieve an SpO2 of >90% provides a suitable alternative to descent, particularly in settings where patients can be closely monitored.127-129 As noted earlier in the section on AMS/HACE treatment, providers should target an SpO2 of >90% rather than a particular FIO2. Short-term use in the form of visits to oxygen bars or use of over-the-counter oxygen cannisters has no role in HAPE treatment.
Recommendation
We recommend that, when available, supplemental oxygen sufficient to achieve an SpO2 of >90% or relieve symptoms be used while waiting to initiate descent when descent is not feasible and during descent in severely ill patients. Strong recommendation, high-quality evidence.
Portable Hyperbaric Chambers
As for AMS and HACE, portable hyperbaric chambers can be used for HAPE treatment. They have not been systematically studied for this purpose, but their use for HAPE has been reported in the literature. 130 Use of a portable hyperbaric chamber should not delay descent in situations when descent is feasible.
Recommendation
We recommend that when descent is not feasible or delayed or supplemental oxygen is unavailable, a portable hyperbaric chamber may be used to treat HAPE. Strong recommendation, low-quality evidence.
Nifedipine
A single, nonrandomized, unblinded study demonstrated the utility of nifedipine (10 mg of the short-acting version, followed by 20 mg slow release every 6 h) for HAPE treatment when oxygen or descent is not available. 131 Although participants in this study received a loading dose of the short-acting version of the medication, this initial dose is no longer employed due to concerns about provoking systemic hypotension. While hypotension is less common with the extended release preparation, it may develop when nifedipine is given to patients with intravascular volume depletion. A prospective, cross-sectional study of individuals with HAPE evacuated to and treated at lower elevation demonstrated that the addition of nifedipine (30 mg sustained release every 12 h) to descent, oxygen, and rest offered no additional benefit in terms of time to resolution of hypoxemia and radiographic opacities or hospital length of stay. 132
Recommendation
We recommend that nifedipine be used for HAPE treatment only when descent is impossible or delayed and reliable access to supplemental oxygen or portable hyperbaric therapy is unavailable. Strong recommendation, low-quality evidence.
Beta-Agonists
Although there are reports of beta-agonist use in HAPE treatment 133 and the risks of use are likely low, no data support a benefit from salmeterol or albuterol in patients suffering from HAPE.
Recommendation
No recommendation can be made regarding beta-agonists for HAPE treatment due to lack of data.
Phosphodiesterase Inhibitors
By virtue of their ability to cause pulmonary vasodilation and decrease pulmonary artery pressure, there is a strong physiologic rationale for using phosphodiesterase inhibitors in HAPE treatment. While reports document their use for this purpose,133,134 no systematic study has examined the role of tadalafil or sildenafil in HAPE treatment as either monotherapy or adjunctive therapy. Combined use of nifedipine and sildenafil or tadalafil should be avoided due to a risk of hypotension.
Recommendation
We suggest that tadalafil or sildenafil be used for HAPE treatment if descent is impossible or delayed, access to supplemental oxygen or portable hyperbaric therapy is impossible, and nifedipine is unavailable. Weak recommendation, low-quality evidence.
CPAP
As noted above, positive airway pressure works by increasing transmural pressure across alveolar walls, thereby increasing alveolar volume and improving ventilation-perfusion matching and, as a result, gas exchange. A small study demonstrated that EPAP, in which a mask system is used to increase airway pressure during exhalation only, improved gas exchange in HAPE patients. 135 However, while several reports document use of CPAP for management of HAPE in hospital and field settings,7,108 there is no systematic evidence that CPAP or EPAP improves patient outcomes compared to oxygen alone or in conjunction with medications. Given the low risks associated with the therapy, CPAP can be considered an adjunct to oxygen administration in a medical facility, provided the patient has normal mental status and can tolerate the mask. While lithium battery−powered devices and decreased size and weight of CPAP machines have increased feasibility of field use, logistical challenges remain and currently limit overall utility in this setting.
Recommendation
We suggest that CPAP or EPAP be considered for treatment of HAPE when supplemental oxygen or pulmonary vasodilators are not available or as adjunctive therapy in patients not responding to supplemental oxygen alone. Weak recommendation, low-quality evidence.
Diuretics
Although their use is documented in older reports, 136 diuretics have no role in HAPE treatment, particularly because many HAPE patients have intravascular volume depletion.
Recommendation
We recommend that diuretics not be used for treatment of HAPE. Strong recommendation, low-quality evidence.
Acetazolamide
Although clinical reports document use of acetazolamide for treatment of HAPE,133,134 there are no systematic studies examining its use for this purpose. The diuretic effect might provoke hypotension in the intravascularly depleted patient, while the added stimulus to ventilation might worsen dyspnea.
Recommendation
We recommend that acetazolamide not be used for treatment of HAPE. Strong recommendation, low-quality evidence.
Dexamethasone
Considering its potential role in HAPE prevention noted above and studies demonstrating effects on maximum exercise capacity, 137 pulmonary inflammation, and ion transporter function in hypoxia 138 , theoretically, dexamethasone may have a role in HAPE treatment. However, no study has established whether it is effective for this purpose.
Recommendation
No recommendation can be made regarding dexamethasone for HAPE treatment due to insufficient evidence.
Suggested Approach to HAPE Treatment
If HAPE is suspected or diagnosed, oxygen should be started, if available, and descent initiated to lower elevation. If descent is not feasible or must be delayed, supplemental oxygen should be continued or the individual should be placed in a portable hyperbaric chamber. Patients who have access to supplemental oxygen and can be adequately monitored in a medical setting (eg, urgent care clinic or emergency department) may not need to descend to lower elevation and can be treated with oxygen alone at the current elevation. Descent should be initiated, however, if oxygenation fails to improve with supplemental oxygen and/or CPAP, if the patient's condition deteriorates despite achieving an oxygen saturation of >90%, or if the patient fails to show signs of improvement with appropriate interventions for HAPE. In more remote settings, early descent should be considered, as the logistics of evacuation become much more complicated if the affected individual cannot descend under their own power.
The addition of nifedipine may not yield additional benefits in well-monitored settings.129,132 In the field setting, where resources are limited, nifedipine can be used as an adjunct to descent, supplemental oxygen, or portable hyperbaric therapy. It should only be used as primary therapy if none of these other measures is available. A phosphodiesterase inhibitor may be used if nifedipine is not available, but concurrent use of multiple pulmonary vasodilators is not recommended. In the hospital setting, CPAP can be considered as an adjunct to supplemental oxygen, and nifedipine can be added if the patient fails to respond to oxygen therapy alone. There is no established role for beta-agonists, diuretics, acetazolamide, or dexamethasone in the treatment of HAPE, although, as noted below, dexamethasone should be considered when concern exists for concurrent HACE.
In selected patients, specifically those who achieve an oxygen saturation of >90% and have adequate support from family or friends and appropriate lodging arrangements, it is reasonable to discharge them from direct medical care with supplemental oxygen, rather than admit them to a healthcare facility. Individuals treated in this manner should be admitted to the hospital if they develop worsening symptoms and/or oxygen saturation while on supplemental oxygen. Descent to lower elevation should be pursued if oxygenation or other aspects of their condition worsen despite appropriate interventions for HAPE, as this suggests that they may have an alternative pathology that requires further evaluation and management.
Individuals who develop HAPE may consider further ascent to higher altitude or reascent only when symptoms of HAPE have completely resolved and they maintain stable oxygenation at rest and with mild exercise while off supplemental oxygen and/or vasodilator therapy. Consideration may be given to using nifedipine or another pulmonary vasodilator upon resuming ascent.
Suggested Approach for Patients With Concurrent HAPE and HACE
Dexamethasone should be added to the treatment regimen of patients with concurrent HAPE and HACE at the doses described above for patients with HACE. Some patients with HAPE may have neurologic dysfunction due to hypoxic encephalopathy rather than HACE, but making this distinction in the field can be difficult. Therefore, dexamethasone should be added to the treatment regimen for HAPE patients with neurologic dysfunction that does not resolve rapidly with administration of supplemental oxygen and improvement in the patient's oxygen saturation. If supplemental oxygen is not available, dexamethasone should be started in addition to the medications for HAPE in patients with altered mental status and/or suspected concurrent HACE. Pulmonary vasodilators may be used in patients with concurrent HAPE and HACE with care to avoid lowering blood pressure as this may decrease cerebral perfusion pressure and provoke cerebral ischemia. For this reason, the phosphodiesterase inhibitors sildenafil and tadalafil may be preferred over nifedipine as they cause less hypotension, although no studies have directly compared the relative blood pressure effects of these medications in this clinical situation.
High Altitude Travel Following COVID-19
With the advent of effective vaccines and evolution in the overall burden of COVID-19 and its effects on society, large numbers of people are again engaging in high altitude travel. On average, most people infected with SARS-CoV-2 recover with no lasting effects on gas exchange, pulmonary mechanics, cardiovascular function, or exercise capacity, although a subset of individuals, particularly those who had severe disease in the early waves of the pandemic, have had persistent functional deficits for many months following infection. Evaluation before high altitude travel is warranted in those individuals with persistent symptoms at least 2 wk after a positive COVID-19 test or hospital discharge as well as in those who required care in an intensive care unit or experienced myocarditis or thromboembolic disease due to COVID-19. Testing should include pulse oximetry at rest and with activity, pulmonary function testing, plain chest radiography, electrocardiography, B-type natriuretic peptide test, high-sensitivity troponin test, and echocardiography. Computed tomography scanning may be useful in those with hypoxemia and abnormal findings on plain chest radiography, while cardiac MRI should be pursued in those with elevated high-sensitivity troponin or abnormal echocardiography. Cardiopulmonary exercise testing should be considered in individuals with marked limitation in exercise capacity or those who are planning heavy exertion at high altitude. Depending on the results of these tests, individuals may need to modify or defer their high altitude travel plans. 139
Conclusions
We have provided evidence-based guidelines for prevention, diagnosis, and treatment of acute altitude illnesses, including the main prophylactic and therapeutic modalities for AMS, HACE and HAPE, and recommendations regarding their role in disease management. While these guidelines cover many of the important issues related to prevention and treatment of altitude illness, several important questions remain to be addressed and should serve as a focus for future research. Such research might include identifying objective standards for diagnosing AMS, direct comparisons of commonly used medications, optimal dosing regimens for prevention and treatment of altitude illness in the pediatric population, and the role of simulated altitude in altitude illness prevention and climbing performance.
Supplemental Material
sj-pdf-2-wem-10.1016_j.wem.2023.05.013 - Supplemental material for Wilderness Medical Society Clinical Practice Guidelines for the Prevention, Diagnosis, and Treatment of Acute Altitude Illness: 2024 Update
Supplemental material, sj-pdf-2-wem-10.1016_j.wem.2023.05.013 for Wilderness Medical Society Clinical Practice Guidelines for the Prevention, Diagnosis, and Treatment of Acute Altitude Illness: 2024 Update by Andrew M. Luks, MD, Beth A. Beidleman, ScD, Luanne Freer, MD, Colin K. Grissom, MD, Linda E. Keyes, MD, Scott E. McIntosh, MD, MPH, George W. Rodway, PhD, APRN, Robert B. Schoene, MD, Ken Zafren, MD, and Peter H. Hackett, MD in Wilderness & Environmental Medicine
Footnotes
Acknowledgment
The authors acknowledge the past contributions of their friend, the late Dr Paul S. Auerbach, to previous versions of these Clinical Practice Guidelines.
Author Contribution(s)
Drafting of the manuscript (AML); participation in all phases of the editing process (AML); participation in working groups to review and revise recommendations and content and then participation in the editing process (AML, BAB, LF, CKG, LEK, SEM, GWR, RBS, KZ, PHH). All authors approved the final manuscript.
Declaration of Conflicting Interests
AML reports book royalties for authorship from Taylor and Francis, Wolters Kluwer, and Springer. PHH reports payments from Up to Date, is a speaker for Wilderness and Travel Medicine and International Society for Travel Medicine (ISTM), and reports support for attending meetings and/or travel from ISTM and Eurac Research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.