Abstract
Loxoscelism is a systemic inflammatory reaction in response to a brown recluse spider bite (BRSB). In this case we describe a patient with a heightened inflammatory response to a presumed BRSB, with Coomb’s positive hemolysis, cytoplasmic antineutrophil cytoplasmic antibody (cANCA) vasculitis, and features of hemophagocytic lymphohistiocytosis (HLH). A 24-y-old female presented with sudden pain and swelling to her lower back, nausea, fever, and tachycardia after a presumed BRSB. Hemolysis began on hospital day 3 (15.9 g·dL-1) with a nadir on hospital day 5 (6.3 g·dL-1). She had an lactate dehydrogenase of 1415 U·L-1, ferritin of 15534 ng·mL-1, persistent fever, and signs of bone marrow suppression despite hemolysis, with thrombocytopenia (100,000 μL-1) and an inadequate reticulocyte response (1.7%) suggestive of HLH. The patient’s blood was Coomb’s and cANCA/antiproteinase 3 positive. She had signs of toxin-induced vasculitis, with respiratory failure requiring bilevel positive airway pressure, radiographs with bilateral pulmonary infiltrates, and a desquamating rash. She received 6 U of packed red blood cells, furosemide for pleural and pericardial effusions, antibiotics, and symptomatic treatment during the acute phase of her illness. Hemolysis improved without glucocorticoids by hospital day 6. The patient demonstrated a dysregulated immunologic and complement-mediated response to the presumed BRSB. The triad of Coomb’s positive hemolysis, cANCA vasculitis, and HLH-like reaction associated with a presumed BRSB is described for the first time in the literature and brings up questions for future research regarding the role of immune modulators and complement inhibitors in the treatment of severe loxoscelism as well as the genetic factors that predispose certain individuals to such reactions.
Case report
A 24-y-old African-American female presented with sudden pain and swelling to her left lower back that woke her from sleep, along with nausea, a tingling sensation in her hands, fever, and tachycardia. The wound on the patient’s left lower back began initially as an area of erythema and swelling with a central vesicle and ecchymotic appearance with minimal fluid collection on bedside ultrasound and evolved into the classic dermonecrotic wound characteristic of a brown recluse spider bite (BRSB) (Figure 1). The characteristic dermonecrotic lesion coupled with the overall clinical picture (and widespread presence of Loxosceles reclusa in Missouri, where this case took place) suggested the diagnosis of Loxosceles envenomation by our medical toxicologist. The patient was initially admitted to the observation unit due to persistent tachycardia, nausea, and ongoing pain with a significant area of redness and edema surrounding the bite site (livedeoid plaque). Overnight the patient developed worsening tachycardia, hypotension, and increased work of breathing and was admitted to the intensive care unit, where she progressed to respiratory distress and respiratory failure requiring noninvasive positive pressure ventilation (performed with bilevel positive airway pressure). Computed tomography scan showed bilateral moderate-sized pleural effusion, and echocardiogram showed preserved ejection fraction with a moderate-sized pericardial effusion.
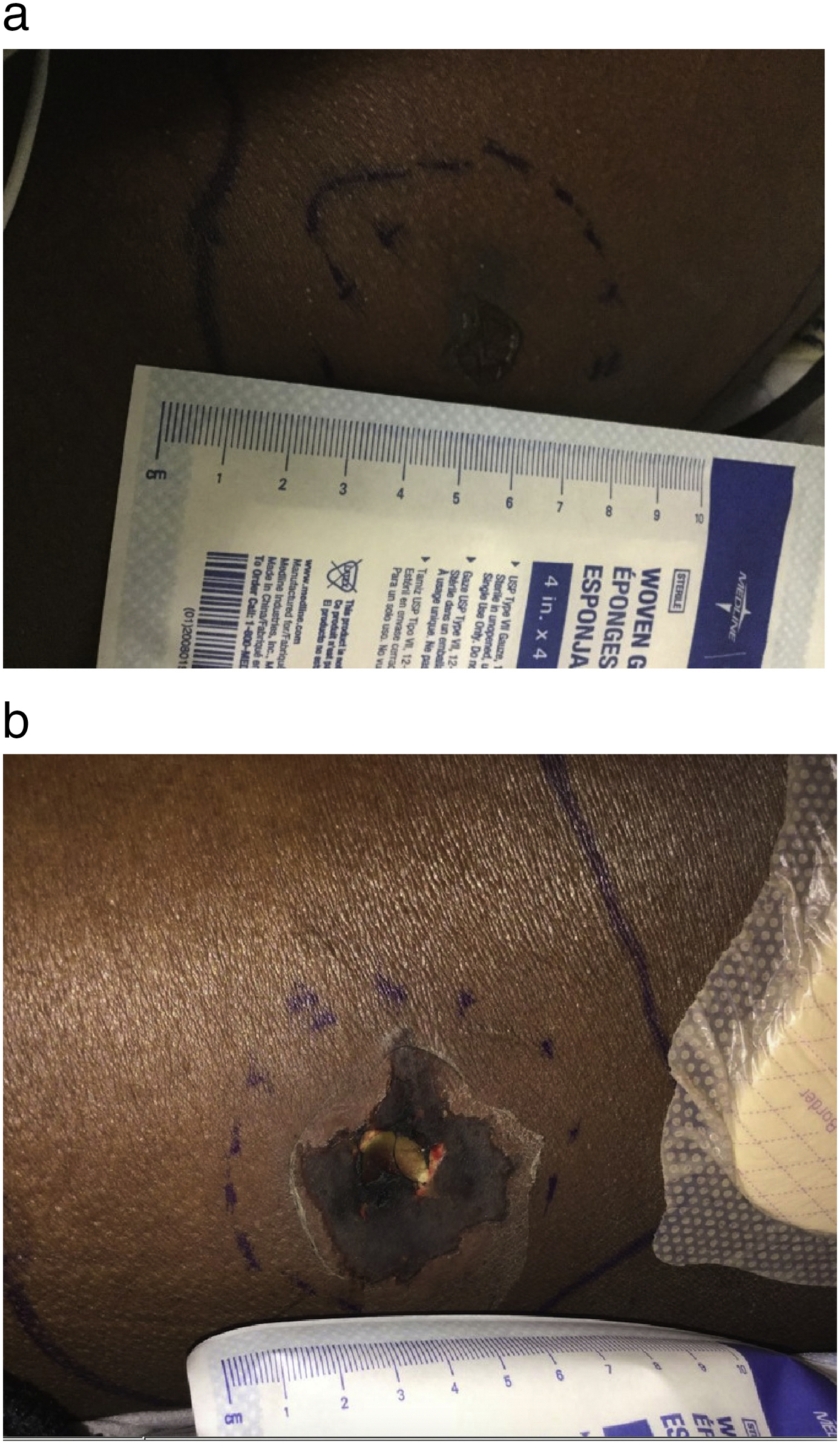
Evolution of dermonecrotic lesion.
Pronounced hemolysis began on day 3 of hospitalization with a decrease in hemoglobin from 15.9 g·dL-1 (normal 12.0–15.5 g·dL-1) at admission to 6.3 g·dL-1 on hospital day 5. Hemolysis work-up revealed the patient’s blood to be Coomb’s positive, monospecific C3 to be 2 +, and cytoplasmic antineutrophil cytoplasmic antibody (cANCA) and antiproteinase 3 positive. Lactic dehydrogenase reached a maximum of 1415 U·L-1. The patient’s laboratory values showed a heightened inflammatory response, with a white blood cell count of 33,600 μL-1 and ferritin peaking at 15 534 ng·mL-1 (normal range 20 to 200 ng·mL-1 in women). She showed signs of bone marrow suppression in addition to hemolysis with thrombocytopenia (platelet minimum 100,000 μL-1) and an inadequate reticulocyte response (1.7% during hemolytic phase). The patient began showing clinical signs of toxin-induced vasculitis, with increasing oxygen requirement and repeat chest radiographs showing bilateral pulmonary infiltrates secondary to toxin-induced capillary leak phenomenon. She also developed a desquamating rash (Figure 2).
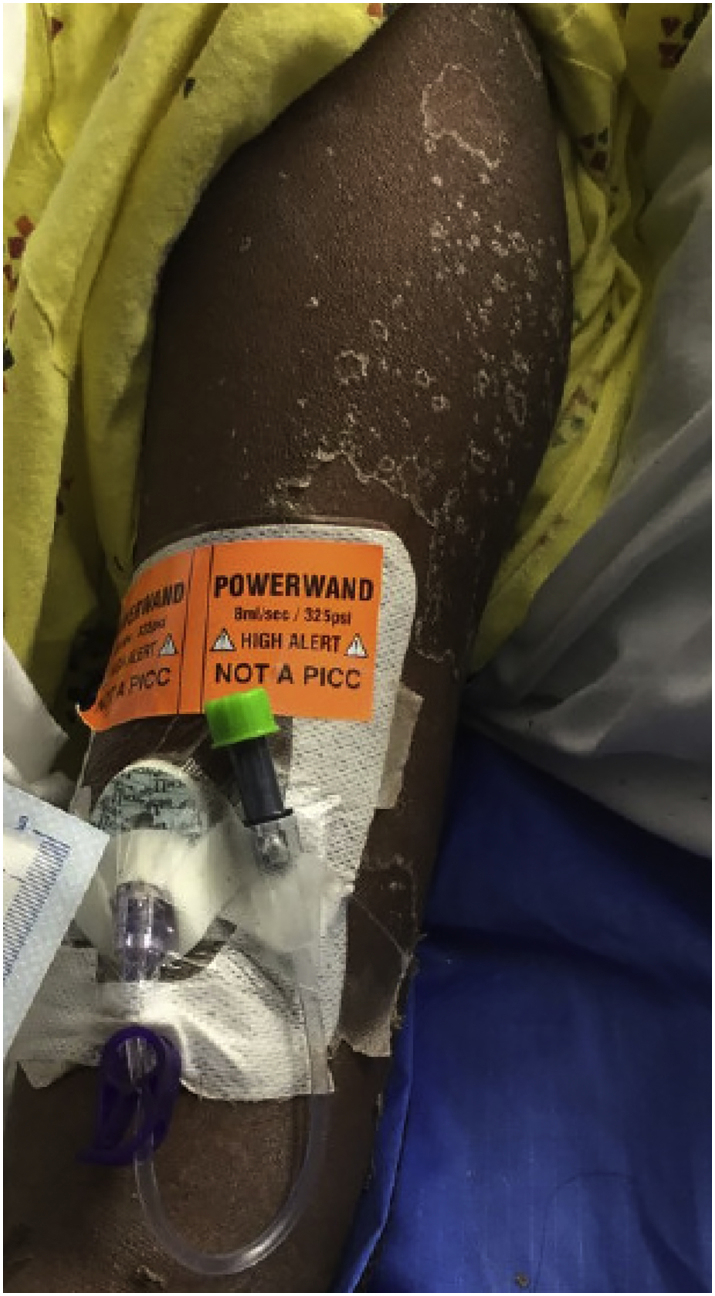
Desquamation rash.
International normalized ratio and partial thromboplastin time were mildly elevated throughout the course of the patient’s illness, with elevated d-dimer as well as fibrinogen. The patient’s renal function remained normal throughout her stay (creatinine 0.5–0.8 mg·dL-1). She continued to have persistent sinus tachycardia with a heart rate of 130 to 160 beats·min-1. Repeat echocardiogram demonstrated showed normal systolic and diastolic function, persistent pericardial effusion without signs of tamponade physiology, and no evidence of pulmonary hypertension (right ventricular systolic pressure 32 mm Hg). Her troponin levels remained normal.
She received a total of 6 U of packed red blood cells, furosemide for pleural and pericardial effusions, empiric broad-spectrum antibiotics, supplemental oxygen, and symptomatic treatment during the acute phase of her illness. Our team opted against treatment with glucocorticoids because we believed patient’s hemolysis to be most likely due to toxin-induced complement activation instead of immunoglobulin-mediated (negative monospecific immunoglobulin G). The patient’s clinical picture and hemolysis improved by hospital day 6, and she was able to be transferred to a less complex care unit. Her wound was treated by the wound care team with debridement and daily dressing changes and did not require skin grafting.
Discussion
This patient’s hospital course illustrates the complex mechanisms by which presumed brown recluse spider envenomation can have devastating inflammatory and immune-mediated effects. L reclusa (Sicariidae) is a venomous spider found in parts of the midwestern United States, adjacent to several southeastern states, and parts of northern Mexico. 1 –3 The clinical presentation of envenomation by several species of recluse spiders (Loxosceles spp) ranges from local skin necrosis (necrotic arachnidism), to mild systemic symptoms such as nausea, vomiting, fever, and arthralgias, to severe systemic loxoscelism that manifests as hemolytic anemia and multiorgan dysfunction.1,3 Severe systemic loxoscelism is a rare entity that has been found to affect children and young adults disproportionately.1,3 There is an antivenom available in Brazil that has not been approved by the Food and Drug Administration for use in the United States and has not been proven to improve outcomes. 4
One of the active toxins in brown recluse (L reclusa) venom is sphingomyelinase D, an enzyme that catalyzes the breakdown of sphingomyelin, a main component of animal cell membranes. The proposed mechanism behind both the skin necrosis of the brown recluse bite and the hemolysis associated with systemic loxoscelism was once thought to be attributed to direct toxicity of sphingomyelinase D on the cell membranes of erythrocytes.5,6 However, the amount of venom released by brown recluse spiders is extremely small, and therefore other mechanisms have been proposed to explain the widespread effect of sphingomyelinase D on erythrocytes. Evidence has shown that the hemolysis associated with brown recluse spider bites is most likely associated with complement activation. 1 ,3,5,6 This patient, and many other patients with hemolysis associated with presumed brown recluse bite, was found to be direct antiglobulin testing (Coomb’s) positive. She was found to be C3 positive, but others have been found to be both C3 and IgG positive. 1 ,5,6
The proposed mechanism of hemolysis involves reduced expression of glycophorin A via activation of an endogenous metalloprotease, which in turn makes the cell membrane more vulnerable to complement-mediated destruction. 1 ,5,6 Newly altered erythrocytes affect the expression of glycophorin A on previously unexposed erythrocytes, thereby propagating the hemolysis swiftly throughout the body. 1 The IgG bound to patients’ erythrocytes in BRSB is thought to be due to nonspecific binding to altered proteins on a cell membrane affected by sphingomyelinase rather than as a specific autoantibody. 1 The difference in individual immune response to brown recluse spider venom could possibly be due to variations in serum complement and immunoglobulin levels. 1
Our patient also developed cANCA/ antiprotease-3 (PR-3) vasculitis, a rare entity previously described only in case reports. 7 The proposed mechanism involves an inflammatory response and cytokine release triggered by the brown recluse toxin. 2 These cytokines cause PR-3 or myeloperoxidase found in neutrophil vesicles to be expressed on the cell surface of neutrophils, which are then bound by ANCAs, inciting neutrophil-mediated damage of nearby vascular endothelium. 8 –10 Binding of ANCA to PR-3 and myeloperoxidase also causes activation of the complement system (via factors C3 and C5), which further activates neutrophils, causing a positive feedback loop leading to self-perpetuating inflammation and tissue damage. 9 The patient’s ANCA vasculitis manifested in respiratory distress from presumed pulmonary capillary leak phenomenon or diffuse alveolar hemorrhage. 7 Our team treated the patient with supportive care for her respiratory failure with noninvasive positive pressure ventilation at the beginning of her disease course, eventually weaning to supplemental oxygen via nasal cannula and finally to room air and via judicious management of her volume status. The immune- and complement-mediated components of ANCA vasculitis raise the question of a role of glucocorticoids, complement inhibitors such as eculizumab, or other immune modulators such as cyclophosphamide or azathioprine in treating patients who appear to have an uncontrolled activation of the immune and complement systems secondary to presumed or confirmed BRSB.
The third unique aspect of the patient’s apparent pathologic immune response to presumed brown recluse spider venom was that she displayed certain clinical features of secondary hemophagocytic lymphohistiocytosis (HLH). 2 HLH is a clinical syndrome caused by high production and release of cytokines, causing accumulation and dysregulation of natural killer cells and cytotoxic T cells that presents with a wide spectrum of clinical manifestations with rapid deterioration and considerable morbidity and mortality.2,11 Some clinicians have proposed that for adults, HLH should be considered when there is evidence of a predisposing disease, fever, organomegaly (from lymphocytic infiltration), cytopenias of at least 2 cell lines, elevated ferritin, elevated lactate dehydrogenase, and hemophagocytosis on bone marrow aspirate. Treatment of HLH includes a prolonged course of high-dose steroids to dampen the pathologic immune response. 2 The patient’s predisposing factor was the presumed BRSB; she had persistent fever throughout her stay, a maximum lactate dehydrogenase of 1415 U-1, a maximum ferritin of 15,534 ng·mL-1, thrombocytopenia, and hemolysis with inadequate reticulocyte response. Bone marrow aspirate, soluble CD25 level, and natural killer cell activity assays were not performed due to patient improvement over the course of her stay without high-dose steroid treatment.
The patient’s course of illness after a presumed BRSB illustrates the numerous mechanisms by which sphingomyelinase can cause a pathologic reaction in certain individuals who may have a genetic or age-related predisposition to dysregulated immunologic or complement-mediated response to the toxin. The presence of the triad of Coomb’s positive hemolysis, cANCA/PR-3 vasculitis, and secondary HLH-like reaction associated with presumed BRSB is described for the first time in the literature and raises questions for future research regarding the role of immune modulators other than glucocorticoids and the possible role of complement inhibitors in the treatment of severe systemic loxoscelism with hemolysis and multisystem organ failure.
Limitations
This case must be termed a presumed case of BRSB. Without the capture and identification of the arachnid by an entomologist, our team accepted the most likely diagnosis based on the collaboration with our medical toxicologist with extensive experience with BRSBs. Although other Loxosceles species within the Sicariidae family have sphingomyelinase D as an active toxin, Loxosceles reclusa was the most likely species, given the geographic area in which this case took place. Other less likely considerations would be methicillin-resistant Staphylococcus aureus or clostridial infection, or envenomation by another member of the Sicariidae family.
Author Contributions
All authors (KS, AS, CZ, CB, MT) were involved in the following: study concept and design; drafting of the manuscript; critical revision of the manuscript; and approval of final manuscript.
Financial/Material Support
This project did not receive any financial or material support.
Disclosure
None.
