Abstract
Introduction
Skin and subcutaneous infections are dangerous sequelae of soft tissue injuries, especially in austere situations where medical technology is not available. Numerous plant species endemic to North America have been described as having antibacterial properties. Of these, St. John’s wort (Hypericum perforatum), chamomile (Matricaria chamomilla), and white oak (Quercus alba) were selected for testing against Staphylococcus aureus. Our objective was to assess the suitability of all 3 plants as potential antiseptic agents using methods easily replicated in a resource-scarce environment.
Methods
Water-soluble natural products were extracted from different concentrations of each plant part using either mechanical agitation at ambient temperature or boiling in unsterilized tap water. Antibacterial activity of each extract against S aureus was assessed using a conventional agar well diffusion bioassay. Zones of inhibition were measured using electronic calipers and were compared to tap water as the negative control.
Results
Aqueous extracts of St. John’s wort and white oak bark displayed antibacterial effects against S aureus, with St. John’s wort being more potent. Chamomile displayed no inhibitory properties at the concentrations examined.
Conclusions
These data suggest that both St. John’s wort and white oak are potential candidates for infection prophylaxis and therapy in austere wilderness scenarios, with St. John’s wort being the more potent agent. White oak may be more logistically feasible because the larger surface area of a white oak tree allows for harvesting a larger quantity of bark compared to the smaller surface area of the St. John’s wort plant.
Introduction
Lacerations and minor skin wounds are among the most common types of soft tissue injuries sustained in wilderness settings. 1 Left untreated, many such injuries can develop bacterial infections. A study published in 2003 found that, of the 603 injuries in the wilderness documented by the National Outdoor Leadership School’s (NOLS) staff from 1999 to 2002, 186 (approximately 31%) were due to soft tissue injury and 18 soft tissue injuries (3%) were due to wound infections. 2 A separate retrospective study described how 17% of total recorded injuries in the wilderness from 2002 to 2005 were due to soft tissue injury and 4% were attributable to skin infections. 3 This highlights how soft tissue damage in the wilderness can be complicated by infection which is exacerbated by limited access to antiseptics and antibiotics.
Natural products may provide a feasible solution to this problem because plants and trees are present in many outdoor settings. Likewise, herbal remedies have long been used for a variety of ailments. Topical St. John’s wort tea, for example, has been used for burns and wounds in European countries. 4 Another example is honey, which has been used on wounds since antiquity and has even displayed antimicrobial activity against methicillin-resistant Staphylococcus aureus. 5 There is even some evidence that honey may shorten healing time in partial thickness burns. 6 Given the large volume of scientific literature investigating antimicrobial applications of natural products, the use of plants to prevent and treat infections warrants further investigation because it could potentially provide an additional second- or third-line modality in the arsenal of wilderness physicians.
Three plants were chosen for evaluation as potential wound antiseptic agents in the present study: St. John’s wort (Hypericum perforatum), chamomile (Matricaria chamomilla), and white oak (Quercus alba). Aqueous extracts of St. John’s wort teas have demonstrated marked activity against S aureus and other gram-positive organisms using a broth microdilution method. 4 The antimicrobial activity of M chamomilla is less established. For example, M chamomilla acetone extract has displayed antimicrobial activity against S aureus in both the agar well diffusion method and in vivo infected mice wounds. 7 In contrast, another study using microplate wells showed that an extract of M chamomilla powder that had been bathed in 40°C deionized water for 24 h displayed no antimicrobial activity against S aureus. 8 Finally, studies involving the antimicrobial properties of aqueous extracts of Q alba are rare; however, 1 study involving an aqueous extract of Q alba catkins demonstrated activity against Staphylococcus epidermis. 9 Additionally, each of the tested plants grows in North America, thereby increasing the probability of encountering any one plant under a resource-scarce scenario in this region. 10 –12
The aim of our study was to assess these 3 plants as potential alternatives to pharmaceutical-grade, topical antiseptics for use in wound infection prophylaxis and therapy under conditions of resource scarcity. We hypothesized that, using extraction methods feasible in a resource-scarce environment, these plants would demonstrate antibacterial properties against Staphylococcus aureus.
Methods
Plant Selection
H perforatum, M chamomilla, and Q alba were selected based on their availability in North America. 10 –12 Aerial parts from H perforatum and whole flowers from M chamomilla were obtained from Mountain Rose Herbs (Eugene, OR), and Q alba bark was obtained from Starwest Botanicals (Sacramento, CA; Figures 1, 2, and 3). All samples were stored unopened at room temperature until needed.

Example of plant parts used for H perforatum.

Example of plant parts used for M chamomilla.

Example of plant parts used for Q alba.
Pathogen Selection
Although most wounds in wilderness settings are likely contaminated by polymicrobial populations, Staphylococcus is listed by the Wilderness Medical Society clinical practice guidelines for basic wound management in the austere environment to be among the relevant bacterial pathogens to consider when selecting appropriate treatment in such scenarios. 13 S aureus ATCC 29213 was purchased from the American Type Culture Collection (Manassas, VA) because it was isolated from a wound, likely making it similar to an organism one might encounter in the event of a wound sustained in the wilderness.
Plant Product Extraction
Weighed plant parts were placed in 30 mL of tap water contained in 125-mL Erlenmeyer flasks and were either heated to a boil and immediately removed for cooling or aerated with wrist-action agitation on a mechanical shaker (Wrist Action Shaker Model 75; Burrell Scientific, Pittsburgh, PA) at 21°C for 2 h. This time limit was chosen arbitrarily as a reasonable time period for an average human to hike uninterrupted over uneven terrain. Organic debris was removed by vacuum filtration through Whatman filter paper (Whatman No.1; Fisher Scientific, Pittsburg, PA). Cleared liquid extract was stored in polypropylene centrifuge tubes (50 mL; Corning, NY) at 4°C. Heated extracts were allowed to sit for 2 h to cool before filtration. Shaken extracts were immediately filtered. The methods were designed to simulate resource-scarce methods of extraction such as heating over a basic heat source (eg, campfire) or storing the plant part in a canteen with the agitating motion of hiking potentiating extraction of water-soluble natural products.
Bacterial Cultivation and Bioassay Conditions
All extracts were tested against S aureus using a conventional agar-well diffusion bioassay using a modification of the disk agar diffusion bioassay protocol outlined in a previous study. 14 Cells from S aureus working cultures were inoculated into 20 mL of Mueller-Hinton broth contained in 125-mL Erlenmeyer flasks and incubated for 15 to 18 h at 37°C with rotary aeration (Excella E24 benchtop incubator shaker; New Brunswick Scientific Co., Inc, Edison, NJ). Bacteria from these broth cultures were then used to inoculate culture tubes (18 × 150 mm, Kimax) of Mueller-Hinton broth to an optical density of 0.025 at 620 nm (OD620 = 0.025) using a Spectronic 20 optical spectrophotometer (Thermo Electron Corp., Madison, WI). These cultures were incubated at 37°C with aeration as before until an OD620 of 0.05 was reached. One hundred microliters of each cell suspension was pipetted onto the center of Fisherbrand 100 mm × 15 mm polystyrene petri dishes containing Mueller-Hinton agar and was uniformly spread to obtain an even cell lawn. Sterile glass pipettes were used to bore wells into the agar (approximately 6 mm). Seventy-five microliters of each plant extract was delivered into each well. Wells containing 75 μL of nonsterilized tap water served as negative controls for each plate. Bioassay plates were incubated for 15 to 18 h at 37°C, and zones of inhibition (ZOIs) were measured with electronic calipers. The diameter of the agar well was measured and subtracted from the total zone diameter, and the remainder was expressed in millimeters (Figure 4 A and B). The edges of the ZOIs were determined as the area where bacterial growth began. Only clear zones indicating complete inhibition were measured. Any ZOIs containing attenuated bacterial growth without uniform areas of complete inhibition around the wells were measured as ZOI = 0 mm during data collection (Figure 4 E). Attenuated bacterial growth was assessed visually as a lower density of bacterial colonies on the agar compared with the control. Asymmetry was handled by taking the shortest distance from the center of the well to the start of bacterial growth. This value was then multiplied by 2 and the well diameter was subtracted to give the final measurement (Figure 4 C and D).
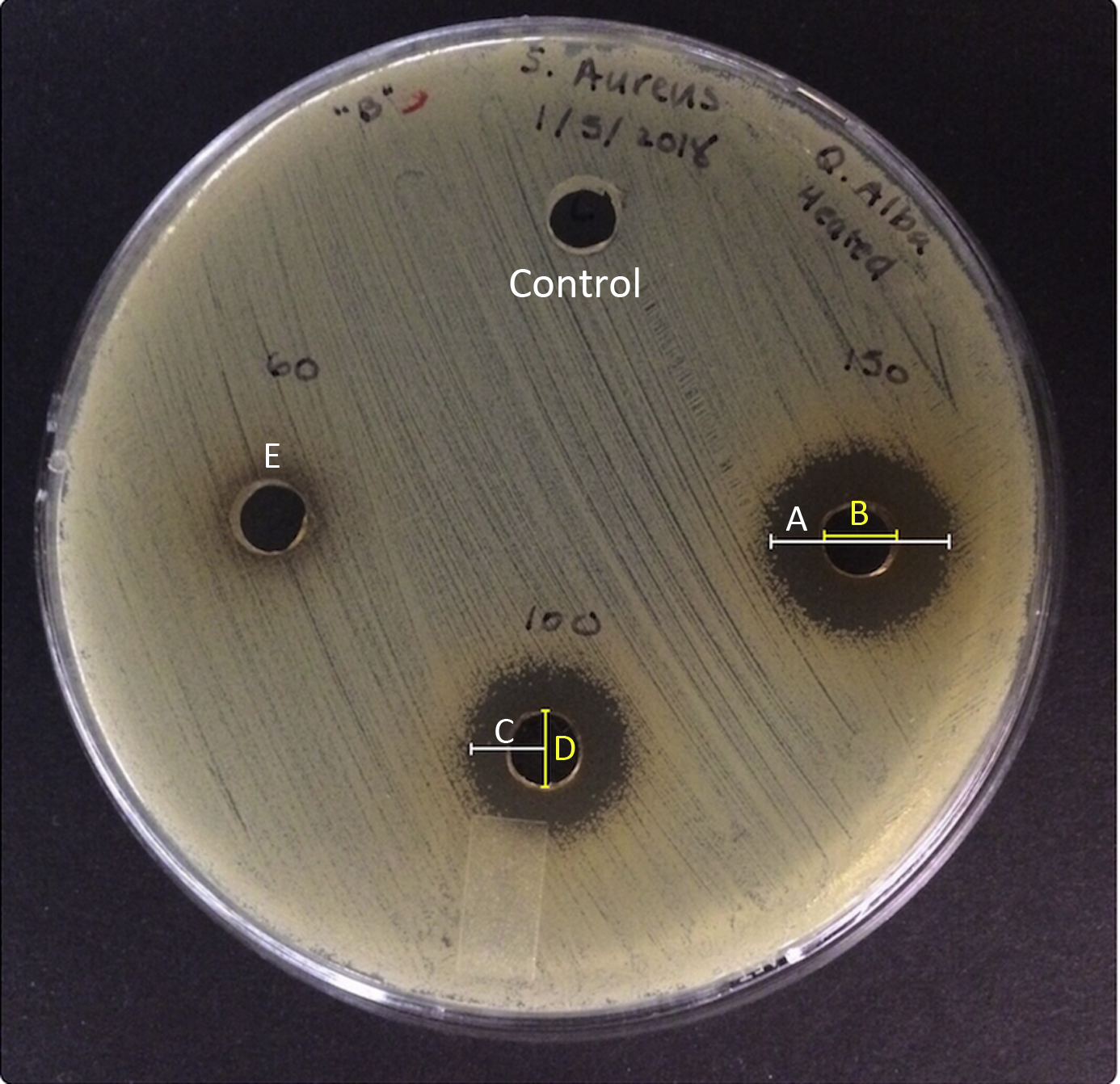
Representative agar diffusion plate illustrating the effect of heated Q alba extract on growth of S aureus. A, Zone of inhibition (ZOI) diameter. B, Well diameter. C, Measurement for asymmetric zones of inhibition. D, Well diameter subtracted from 2 × C to achieve final value for asymmetric ZOIs. E, Example of a zone with attenuated bacterial growth.
Each plant product was extracted 3 times at concentrations of 7.5, 15, 30, 60, 100, and 150 mg·mL-1 for heated extracts and 7.5, 15, and 30 mg·mL-1 for ambient extracts. Each extract was tested 3 separate times. This yielded 9 total data points per concentration per extraction method. These data points were then entered into SAS version 9.4 to calculate the mean and standard deviation. The average ZOI of extract, when appropriate, was used in the factorial analysis of variance (ANOVA). Higher concentrations were not tested for ambient extracts because it was deemed improbable to find sufficient plant parts in the wilderness to fill the volume of most canteens at higher concentrations without expending significant time and energy. Lower quantities of water could be used in a canteen to compensate for fewer plant parts, but the high amount of empty volume and agitating motion would likely lead to inefficient extraction because wet plant parts would adhere to the walls of the canteen and would not consistently be in contact with the solvent. Heated extracts were tested at higher concentrations because the static nature of heating a container allows for easier manipulation of the water to plant part ratio.
The table included in this study was constructed using Microsoft Word version 14.7.1. A factorial ANOVA was applied using Statistica version 13.3. This was performed to compare the main effects of plant type, extract concentration, and the interaction effect of plant type and extract concentration on ZOI diameter. Plant type had 2 levels (H perforatum and Q alba). Extract concentration had 2 levels (100 mg·mL-1 and 150 mg·mL-1). Statistical significance was defined as P < 0.05.
Results
H Perforatum
The 7.5, 15, and 30 mg·mL-1 concentrations of H perforatum inhibited S aureus growth in an apparent concentration-dependent manner when extracted by mechanical agitation at ambient room temperature (21°C). Even larger ZOIs were obtained when extracted by heating, and concentration dependence continued for the additional concentrations of 60, 100, and 150 mg·mL-1 (Table 1).
Susceptibility of S aureus ATCC 29213 to H perforatum, Q alba, and M chamomilla aqueous extracts at varying concentrations and temperatures measured by zone of inhibition
ND, not determined.
M Chamomilla
All ambient and heated extracts of M chamomilla failed to inhibit S aureus growth at all concentrations examined. Heated extracts of 100 and 150 mg·mL-1 could not be tested to completion because at these concentrations the chamomile flowers absorbed most of the solvent and failed to produce sufficient quantity of testable extract.
Q Alba
Only heated extracts at high concentrations (100 and 150 mg·mL-1) produced ZOIs. Heated extracts at low concentrations (7.5, 15, and 30 mg·mL-1), as well as ambient temperature extracts at any concentration, failed to produce ZOIs (Table 1).
Statistical Analysis
The main effect for plant type produced an F-ratio of F(1,8)=30.20 (P < 0.001), denoting a statistically significant difference in ZOI production among plant types. The main effect for extract concentration was not significant with an F-ratio of F(1,8)=2.08 (P = 0.19). The interaction effect of plant type and extract concentration was also not significant F(1,8)=0.79 (P = 0.40).
Discussion
In this study H perforatum produced the largest ZOIs at each concentration compared with the other plants used in this study. This suggests that H perforatum is a promising candidate for use in wound cleaning or prophylaxis in a resource-scarce environment.
M chamomilla extract did not exhibit antibacterial properties in this investigation, a result that is inconsistent with 2 previous studies that have assessed its antistaphylococcal properties in vivo and in vitro.7,15 This disparity is likely because other studies included organic solvents such as acetone or methanol extracts or essential oils. No other published studies have assessed the antibacterial properties of aqueous M chamomilla flower extracts against S aureus under simulated resource scarcity. Historically, M chamomilla has displayed inconsistent antistaphylococcal activity. 12 Therefore, the inability of M chamomilla to produce ZOIs in the present study supports the notion that it is an unsuitable agent for use under resource-scarce conditions.
The antistaphylococcal results produced by Q alba in this investigation are supported by previously published research. One study found that aqueous extracts of Q alba catkins displayed antibacterial activity against Staphylococcus epidermidis. 9 Another study demonstrated antibacterial properties of Q alba extract using wood water from white oak wood. 16 Based on our review of the current literature, this is the first study to assess the antibacterial effects of Q alba bark extract against S aureus under simulated resource scarcity. The results of the present study and prior studies together suggest that Q alba bark is a potentially viable agent for wound therapy under resource-scarce environments.
A factorial ANOVA comparing 100 and 150 mg·mL-1 of heated Q alba bark extract with the same concentrations of heated H perforatum extract revealed a significantly greater diameter of the ZOIs of H perforatum (P < 0.001). This suggests that, when controlling for concentration, H perforatum produced more potent antistaphylococcal activity compared to Q alba and is a better candidate for wound treatment. However, the larger surface area of a white oak tree (Q alba) compared to a smaller herb (H perforatum) means that Q alba could be a more logistically feasible option. Additionally, the main effect of extract concentration on ZOI diameter was not significant (P = 0.19), indicating the antistaphylococcal effects remained similar as the concentrations increased between 100 to 150 mg·mL-1 for both plant extracts. This may be due to maximum extract being obtained for the given quantity of solvent.
Regarding safety, some studies have shown increased risk of irritation and photosensitization with topical application of St. John’s wort. 17 Another valid concern is the systemic interaction between H perforatum and certain types of antidepressant medication. Overall, there is little data regarding the interaction of topical aqueous H perforatum extract with antidepressants, specifically selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors. However, one review article on H perforatum drug interactions states that topical preparations of H perforatum can be generally viewed as benign because serum concentrations of a topical H perforatum preparation would likely be below therapeutic level. 18 Another source warns against topical Q alba bark use in large wounds because of potential reactions of dermatitis; however, the effect on smaller wounds is not specified. Additionally, Q alba does contain salicylates, and caution should be used in humans with aspirin allergy until future studies can more definitively guide its application in this context. 19 Overall, there is little research specifically regarding topical application of Q alba bark extract in wound cleaning. Possible adverse reactions should be considered in the design of studies involving human wound treatment with H perforatum or Q alba.
In several areas of North America and Canada there is an association of Cryptococcus spp with trees. 20 Blastomyces dermatitidis is also associated with rotting wood. 21 This could be a potential hazard for cutaneous inoculation if an extract made with oak bark contained either of these fungal species. The boiling of water to make oak bark extract may in itself reduce the risk of unwanted inoculation; however, research regarding the use of heated water to disinfect organic matter colonized by Cryptococcus or Blastomyces is limited. One study demonstrated that exposure to 45°C for 5 min killed the majority of melanized and nonmelanized Cryptococcus neoformans cells. 22 Another study showed that of 23 isolates of B dermatitidis exposed to 100°C in suspension buffer only 2 displayed growth when incubated for 12 wk afterwards. 23 Future in vivo studies involving locally acquired extracts in areas endemic to C gattii and B dermatitidis would be required to elucidate the clinical significance of the risk of cutaneous inoculation with either Cryptococcus spp or B dermatitidis.
Limitations
Our study was limited by several experimental factors. First, the plants used were industrially grown and harvested, which implies that they were grown under more uniform conditions compared to plants that would be encountered in the wild. This could affect the composition of the plants and make their antibacterial properties more or less potent than the same species of plants encountered in the wild. Second, the plant parts used were dried by the distributor for storage. This could alter the antibacterial strength of the plants by concentrating or inactivating 1 or more of their antibacterial components. Third, only a single organism was used when simulating a skin infection in the wild. In reality, wound infections in the wilderness are likely polymicrobial. 13 Therefore, additional testing against a wider array of pathogenic organisms is required before definitive recommendations for real-world application can be made. Fourth, our study was designed to simulate conditions without access to medical pharmaceuticals. Thus, there were no positive controls used as comparison. Finally, although this study used vacuum filtration to expedite extraction time and ensure sufficient quantity of extract for multiple agar plates, such methods will likely not be available in austere circumstances. Gravity filtration will likely be the most expedient method available in such situations and should be used in future studies.
Conclusions
Wounds in the wilderness can easily be complicated by infection. Two out of 3 of the plant extracts surveyed (H perforatum and Q alba) displayed antistaphylococcal properties, with H perforatum consistently displaying more potent antimicrobial activity compared to Q alba. This suggests that both plants are viable for future evaluation in infection prophylaxis or therapy under these conditions. Future studies should focus on in vivo testing of simulated resource-scarce extracts to evaluate clinical significance and test locally acquired extracts to evaluate the significance and variability of geographic factors in extract strength. Additionally, polymicrobial wounds should be evaluated by examining other organisms and mixed cultures.
Footnotes
Acknowledgments
The authors acknowledge Dr Tom Curtis, associate professor at Oklahoma State University Center for Health Sciences, for assistance with data management.
Author Contributions
Study concept and design (AJW, LSB, FRC); obtaining funding (AJW, LSB, FRC); acquisition of data (AJW, NWN); analysis of data (AJW, LSB); drafting of manuscript (AJW, NWN, LSB); critical revision of manuscript (AJW, FRC, LSB); and approval of final manuscript (AJW, NWN, LSB, FRC).
Financial/Material Support
This study received funding from the Wilderness Medical Society Houston grant, supported by the Academy of Wilderness Medicine®.
Disclosures
None.
☆
Data from this study were presented at the Wilderness Medical Society 2018 Summer Conference on August 5, 2018, in Midway, UT, and at the Oklahoma State University Center for Health Sciences Research Day, February 16, 2018 in Tulsa, OK.
