Abstract
Introduction
Histoplasma capsulatum is a dimorphic fungus that causes histoplasmosis, a sporadic endemic mycosis with environmental, occupational, and wilderness exposure-related risk factors. The objectives of this review are to describe these risk factors, to alert clinicians to the different presenting manifestations of histoplasmosis, and to recommend effective management and prevention strategies.
Methods
Internet search engines were queried with keywords to select articles for review over the study period, 1950 to 2018. Articles selected for review included case series, epidemiologic analyses of surveillance data, clinical and laboratory updates, immunologic investigations, and observational and longitudinal studies. Articles excluded from review included systemic mycosis reviews and clinicopathologic conference reports.
Results
The principal transmission mechanism of histoplasmosis is by inhalation of spores aerosolized by soil disruption, resulting in pneumonic infections ranging from asymptomatic to disseminated. Although histoplasmosis is more common in endemic regions, nonendemic regions have reported increasing autochthonous and imported cases. Immunocompromised persons are at significantly increased risks of contracting histoplasmosis. Environmental and wilderness-related risk factors for histoplasmosis include bird and bat watching, cave and cave entrance exploration, and bamboo removal and burning. Occupational risk factors for histoplasmosis include road construction, roofing, bridge and water tower work, demolition, and masonry.
Conclusions
Histoplasmosis can result in considerable morbidity. Increased awareness of disease risk factors among the public and the international healthcare community will improve the timely diagnosis and treatment of histoplasmosis and prevent disease progression and dissemination.
Introduction
Histoplasma capsulatum is a dimorphic fungus that causes histoplasmosis, a regionally endemic pulmonary mycosis. Histoplasma exists in nature as a spore-forming mold in soils contaminated with bird or bat droppings. 1 Up to 90% of persons living in the Ohio and Mississippi river valleys of the United States are exposed to the infective spores (microconidia) of Histoplasma during their lifetimes, but only about 1% will develop symptomatic infections.2,3 However, attack rates can exceed 50% during sporadic outbreaks of histoplasmosis.2,3 Once thought to be confined to narrow geographic regions of the Americas, authochthonous and imported cases of histoplasmosis have been reported worldwide.4,5 Although the causes for this expanded disease distribution remain uncertain, climate change has provided a warming ecosystem favoring mold growth, and the HIV pandemic has provided more susceptible human hosts.4,6
Several environmental, occupational, and wilderness exposure-related risk factors can predispose residents and visitors to histoplasmosis both in endemic and nonendemic regions. The objectives of this review are to describe these risk factors, to alert clinicians to the different presenting manifestations of histoplasmosis, and to recommend effective management and prevention strategies.
Methods
To meet the objectives of this review, Internet search engines, including PubMed, Medline, Ovid, Google, Google Scholar, and Cochrane, were queried with keywords to select articles for review over the study period, 1950 to 2018. The keywords included “Histoplasma capsulatum,” “Histoplasma capsulatum var capsulatum,” “Histoplasma capsulatum var duboisii,” and “Histoplasmosis: incidence, distribution, geography, travel, treatment, and prevention.” The articles selected for review included case series, epidemiologic analyses of surveillance data, clinical and laboratory updates, immunologic investigations, and observational and longitudinal studies. Articles excluded from review included systemic mycosis reviews and clinicopathologic conference reports. These selected methodologies met all recommended criteria for narrative reviews, including several keywords, use of 2 or more Internet search engines, defined study period, and article inclusion and exclusion criteria. 7 Institutional review board approval was not required for this review of published scientific articles.
Results
DISEASE ECOLOGY OF H CAPSULATUM
H capsulatum requires several special ecologic conditions to support its dimorphic life cycle as a spore-forming mold in nature and as a budding yeast form in human infection. Figure 1 is a photomicrograph (original magnification × 800) of the hyphal filaments with breakaway mold spores (microconidia) of the environmental form of H capsulatum.
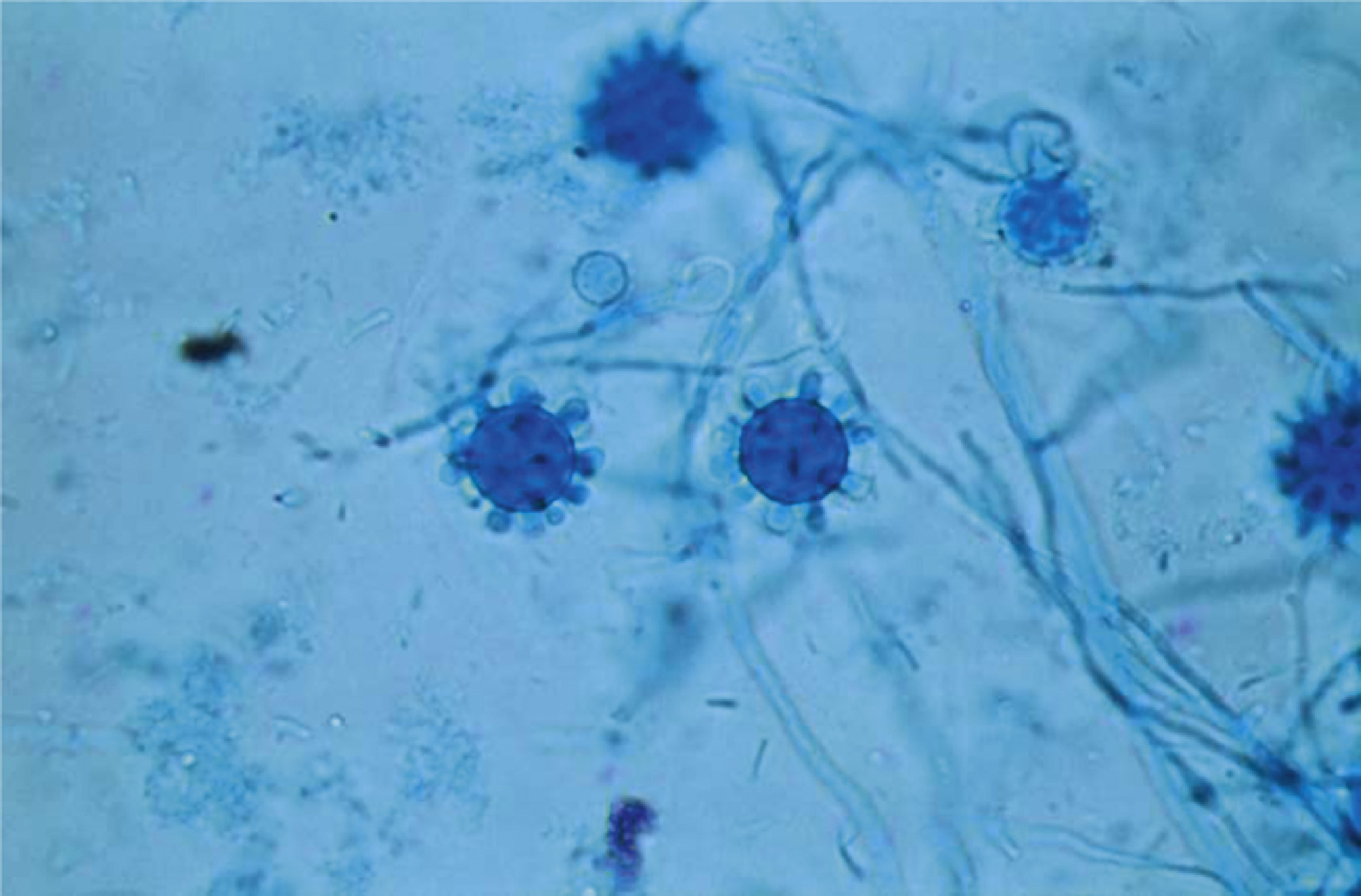
A photomicrograph (original magnification × 800) showing the hyphal filaments with breakaway mold spores (microconidia) of the environmental form of Histoplasma capsulatum. (From US Centers for Disease Control and Prevention [CDC]. Public Health Image Library [PHIL]. PHIL ID #15364. CDC Provider: Dr. Libero Ajello. Available at
In the United States, Histoplasma molds thrive in riverine soils of the Ohio and Mississippi river valleys. 8 These endemic areas for H capsulatum report most of the US cases of histoplasmosis. 8 Figure 2 is a map showing the locations of histoplasmosis outbreaks and the number of outbreak-related cases by state or territory in the continental United States and Puerto Rico over the period 1938 to 2013. 8
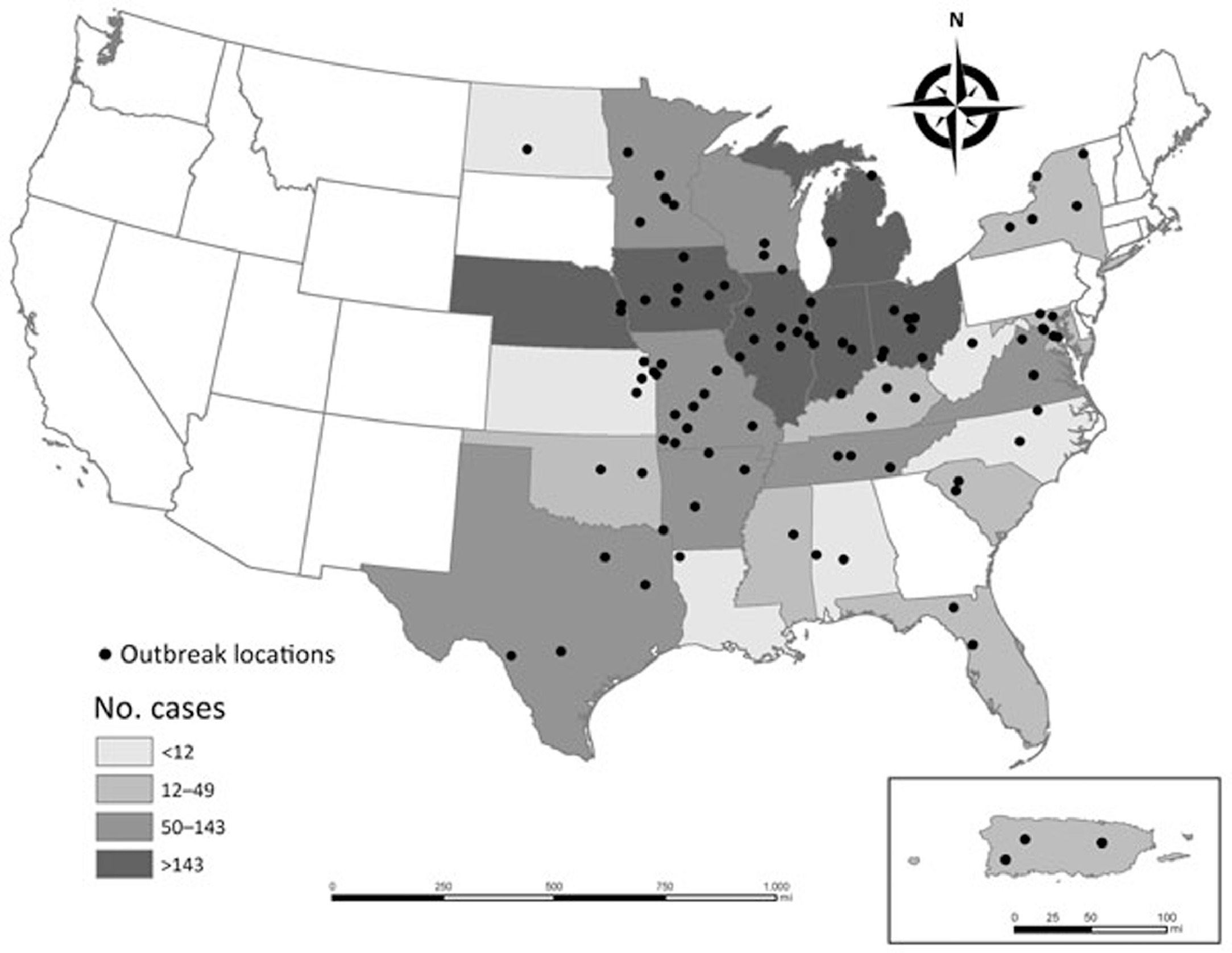
The locations of histoplasmosis outbreaks and the number of outbreak-related cases by state or territory (inset, Puerto Rico), United States, 1938-2013. (From Benedict and Mody.
8
Available at
Other endemic regions for histoplasmosis include Central and South America, Asia, Australia, and sub-Saharan Africa.5,9-12 In Africa, H capsulatum var capsulatum coexists in similar ecosystems with its only other subspecies, H capsulatum var duboisii, which causes similar clinical manifestations. 10 -13 Figure 3 is a world map showing the global regions of greatest endemicity of histoplasmosis. In all endemic regions, Histoplasma species prefer soils containing bird droppings or bat guano.1,13-18 Although birds are immune to infection, bats contract histoplasmosis and spread the fungus in their secretions and droppings, especially within caves and at cave entrances.1,18 The environmental disruption of these preferred habitats is often a precipitator of histoplasmosis outbreaks.
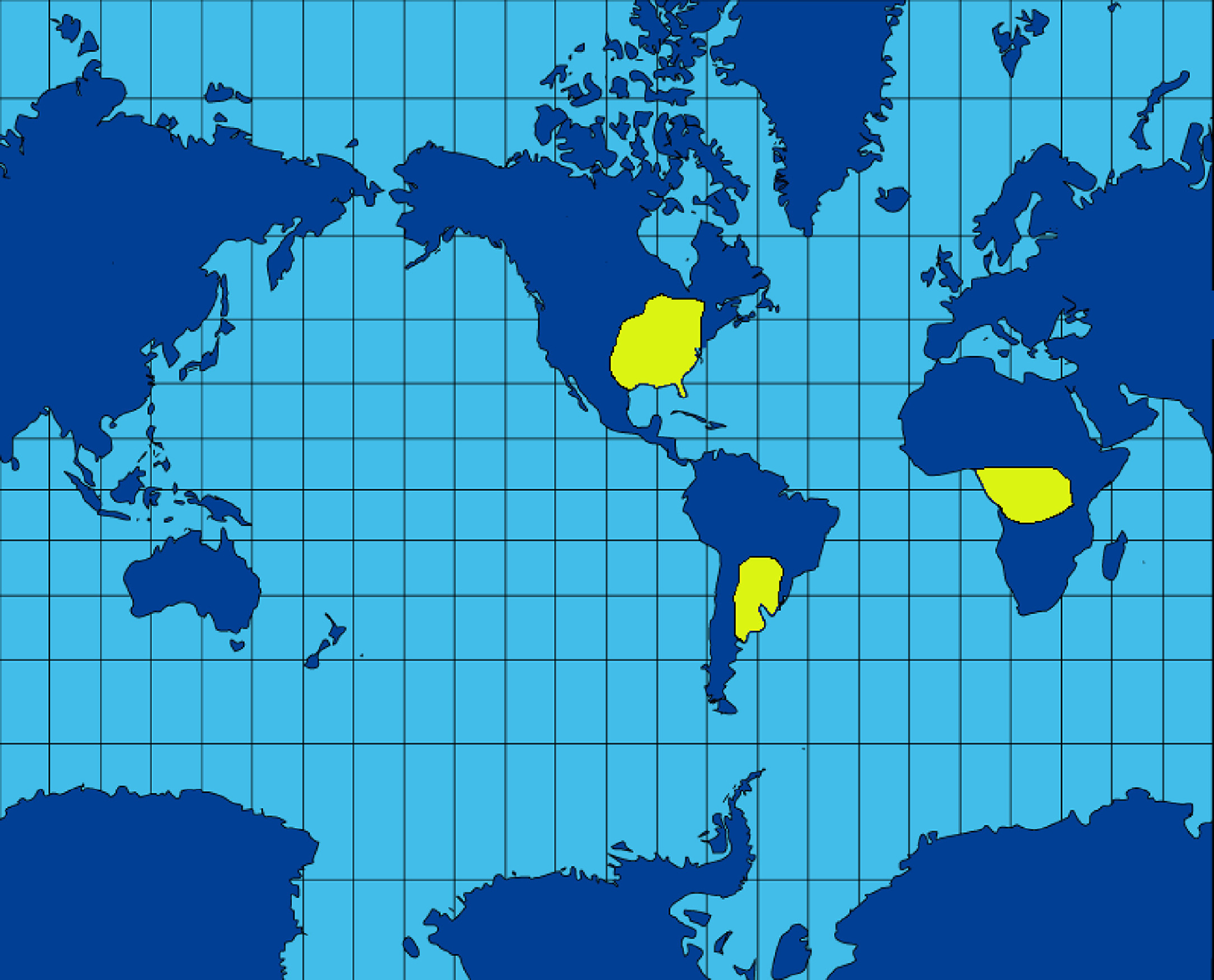
A world map showing the regions reporting the greatest numbers of confirmed cases of histoplasmosis. Compared with the US distribution of histoplasmosis cases, the endemic regions for histoplasmosis cases are probably more widespread in Africa and South America because of underreporting, especially in Africa with 2 causative strains of Histoplasma. (From Trabajo Propio. Wikimedia Commons. Available at
EPIDEMIOLOGY OF HISTOPLASMOSIS
The histoplasmosis-endemic regions of the United States were first mapped during the 1940s and 1950s using a histoplasmin antigen skin test. 2 Later, Baddley et al 12 calculated the annual incidence of histoplasmosis in adults aged 65 y and older to be 3.4 cases per 100,000 persons per y, with an incidence in the Midwest almost twice as high at 6.1 cases per 100,000 persons per y.
In 2016, Benedict and Mody 8 reported a total of 105 outbreaks (defined as >1 case) involving 2850 cases in 26 continental states and Puerto Rico over the reporting period 1938 to 2013. The most common exposure settings were chicken coops and buildings undergoing demolition or renovation. 8 In most outbreak settings (77%), birds, bats, or their droppings were observed. Most outbreaks occurred in regions of highest endemicity with fewer outbreaks reported from states with presumed low levels of endemicity, including Florida, Minnesota, New York, North Dakota, and South Carolina. The authors estimated that 15 to 27% of persons infected in outbreaks required hospitalization for acute respiratory distress, with a case fatality rate of about 1% for acute histoplasmosis pneumonia. 8
In 2000, Limaye et al 19 reported H capsulatum infection in 2 kidney transplant patients from nonendemic areas who received their kidneys from the same organ donor who lived in an endemic area. 19 In 2013, 4 immunosuppressed patients with histoplasmosis were reported from Montana, a state reporting no prior histoplasmosis cases. 20 Of the 4 reported cases, all patients had preexisting immunocompromising conditions, including cancer, infectious mononucleosis, and diabetes. 20 Two patients reported high-risk exposure behaviors for histoplasmosis, including exploring caves and pot planting using guano-containing soils. 20
In 2008 the first authochthonous case of histoplasmosis was reported from eastern Europe in Turkey. 21 In 2018, Elias et al 22 reported the first autochthonous case of histoplasmosis from the Middle East in Israel. In this case a 53-year-old previously healthy woman with chronic lymphopenia presented with weight loss, hoarseness, and a laryngeal mucosal mass that contained multiple intrahistiocytic yeast cells consistent with H capsulatum on biopsy. 22 The authors reported that the patient lived near ancient caves in Galilee where Histoplasma species had been isolated previously. 22
In endemic countries, including the United States, histoplasmosis occurs in 2 to 25% of HIV-infected patients and is the first manifestation of AIDS in 50 to 75% of patients. 23 At the AIDS stage, histoplasmosis-related mortality is about 10% even with highly active antiretroviral therapy in the developed world and up to 40% in the developing world without universal access to this therapy. 23 Histoplasmosis is among the most common opportunistic infections in patients living with AIDS in Latin America, and about 30% of patients with AIDS who contract histoplasmosis will die of it. 9 In several Central American countries, 15% of patients with AIDS and histoplasmosis will have tuberculosis. 24
RISK FACTORS FOR HISTOPLASMOSIS
The significant risk factors for histoplasmosis may be stratified into environmental, occupational, and wilderness-related exposures. The major environmentally related risk factors for histoplasmosis include residency or travel in endemic regions, plant matter or soil disruption, and inhalation exposures in high-risk settings.4,8 High-risk settings for contracting histoplasmosis are mostly confined to endemic regions with empty buildings, chicken coops, farms, stream banks, golf courses, amusement parks, and tennis complexes. 8 These structures and settings all tend to attract flocks of resting or roosting birds, such as seagulls temporarily sheltering from rough seas on the grounds of coastal and lakeside amusement parks, golf courses, and tennis courts. Earlier histoplasmosis outbreaks were more commonly associated with farm and chicken coop exposures. 8 Current outbreaks are more common in natural and workplace settings where bird or bat droppings are present and the environment is disrupted by excavation or construction. 8
In the study by Benedict and Mody, 8 35 outbreaks (33%) were classified as work related, with 26 outbreaks affecting workers alone and 9 affecting both workers and nonworkers, usually subcontractors, over the period 1938 to 2013. 8 The presence of birds, bats, or their droppings were reported in 30 (86%) of the work-related outbreaks. Most work-related outbreaks occurred in buildings (n=15, 43%), but 6 outbreaks occurred at outdoor structures—4 at bridges and 2 at water towers contaminated with bird droppings. 8
The major wilderness-related risk factors for histoplasmosis include disturbance of nesting birds or bats or their droppings, exploring caves or their entrances, and bamboo removal and burning. Although birds, unlike bats, are immune to Histoplasma infection, they support fungal growth by contributing nitrogen and phosphorous to soils with their wastes. Benedict and Mody 8 described the presence of birds or bird droppings in 56% of 105 histoplasmosis outbreaks and bats or their droppings in 23%.
The reported species of birds in 59 bird-related outbreaks included chickens in 41% (n=24), unspecified blackbirds in 32% (n=19), pigeons in 15% (n=9), and seagulls in 2% (n=1). 8 The species of black-colored birds identified in bird-related outbreaks included crows, grackles, starlings, and redwing blackbirds. 8 The types of birds were not reported in 14% (n=8) of the bird-related outbreaks.1,13,16,17
Bats not only support fungal growth in caves with their wastes, they also inoculate guano-covered cave surfaces with infective spores. During the period 1938 to 2013, 9 cave-exploration associated outbreaks of histoplasmosis were reported from the southernmost and traditionally nonendemic locations of the United States and its territories, including Florida, Texas, and Puerto Rico. 8 Four of the 5 outbreaks in Puerto Rico were associated with bat guano–filled cave exposures. 8 In addition to Puerto Rico, Costa Rica, Cuba, and Trinidad have now been confirmed as high-risk regions for histoplasmosis exposures in caves and at cave entrances as a result of both Histoplasma-infected bat and contaminated guano exposures.8,18,25-28
After a 2011 family cookout near a bamboo grove in rural Arkansas, all 19 family members reported fever, cough, chest pain, or shortness of breath 2 to 9 d after bamboo exposures. 29 All 19 attendees participated in a bamboo bonfire and hot dog roast, 3 cut bamboo, 3 built a bamboo fort, 1 raked bamboo leaf litter, and 8 raked bamboo ash. Although no bats were reported in the area, the bamboo grove was described as a longstanding redwing blackbird (Agelaius phoeniceus) roost. Chest radiograph results were abnormal in 11 of the 14 attendees who had chest radiographs. Seven persons had laboratory confirmed histoplasmosis, and 11 persons were considered probable cases. Among the 18 attendees with confirmed or probable histoplasmosis, 16 sought medical care and 7 were hospitalized. Seven patients with confirmed histoplasmosis were treated with oral itraconazole, and all patients recovered.
A similar outbreak of histoplasmosis was reported from northern Louisiana in 1980 that raised the possibility that heating Histoplasma microconidia in conjunction with fire-generated air currents could create ideal conditions for transmission by infectious aerosols. 30 In this case, 6 workers cleared a 260 m2 (2800 sq ft) field of bamboo that was laden with blackbird feces. 30 The bamboo was then bulldozed to midfield and burned. All 6 workers developed serologically confirmed acute histoplasmosis pneumonia.
PATHOGENESIS OF HISTOPLASMOSIS
The infective microconidia of H capsulatum are aerosolized by contaminated soil disruption and convert into budding yeast forms when inhaled to the alveolar level of the lungs. Although infected, most immunocompetent persons will remain asymptomatic with positive histoplasmin skin tests because pulmonary macrophages will engulf microconidia and transport them to lymph nodes for destruction. After the inhalation of a large spore inoculum, about 20% of exposed persons will experience temporary acute influenza–like symptoms after an incubation period of 3 to 21 d. A few will develop acute pneumonia that may progress to chronic granulomatous pneumonia lasting months, especially in immunocompromised persons. Acute and chronic infections may progress rapidly to disseminated infections, especially in immunocompromised persons with diabetes, HIV infection, and AIDS. 3
Figure 4 is a methenamine silver–stained bronchoalveolar lavage specimen showing the yeast forms of H capsulatum var duboisii in the lungs undergoing asexual replication by budding. Figure 5 is an axial slice from a computed tomographic scan of the chest at the level of the carina that shows the classic miliary nodular appearance or “snowstorm” pattern of acute histoplasmosis pneumonia.
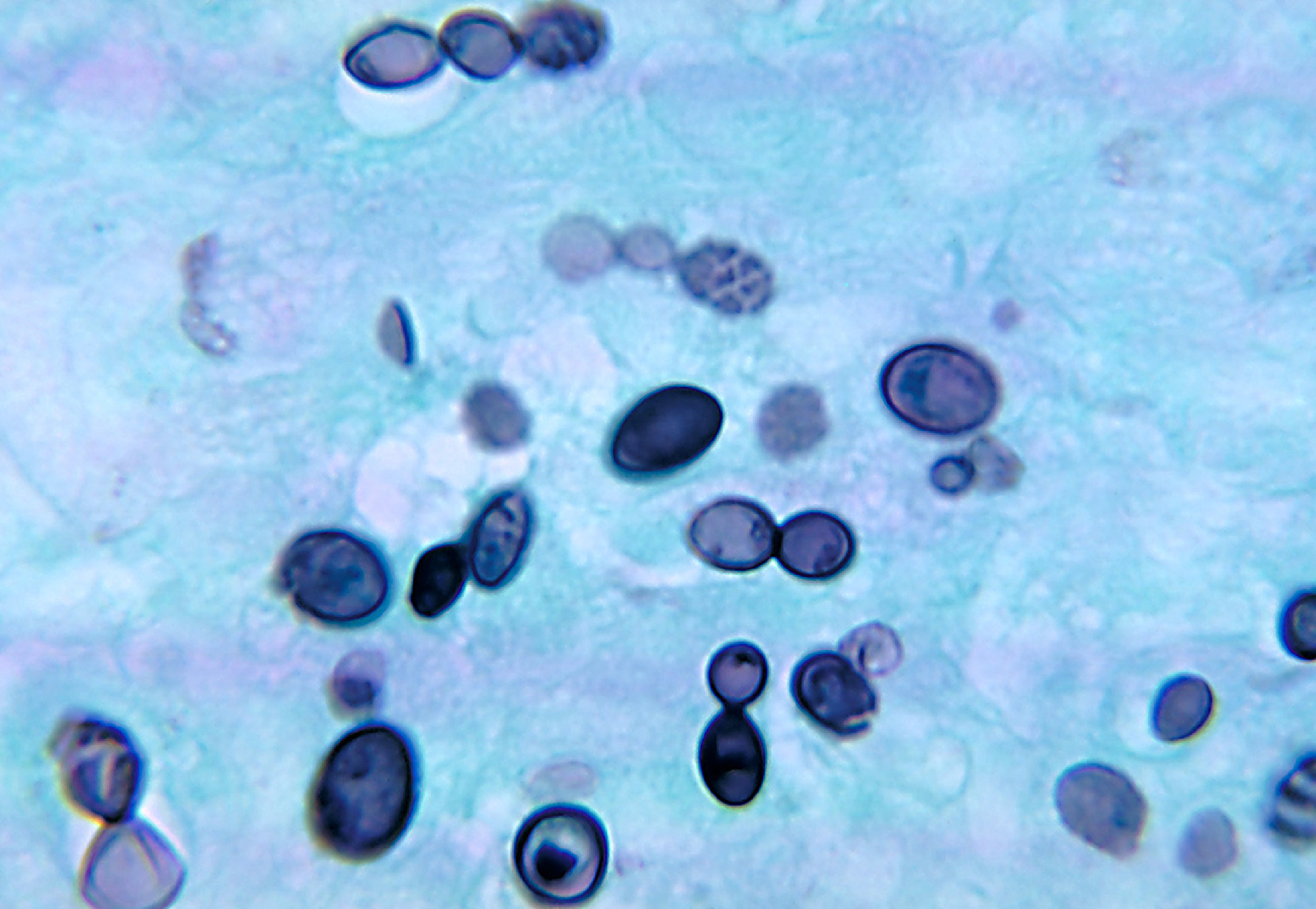
A methenamine silver–stained sputum specimen showing the typical invasive yeast cells of Histoplasma capsulatum var duboisii (African strain) in the lungs undergoing replication by budding. (From US Centers for Disease Control and Prevention [CDC], Public Health Image Library [PHIL]. PHIL ID #4219. CDC Provider: Dr. Libero Ajello. Available at
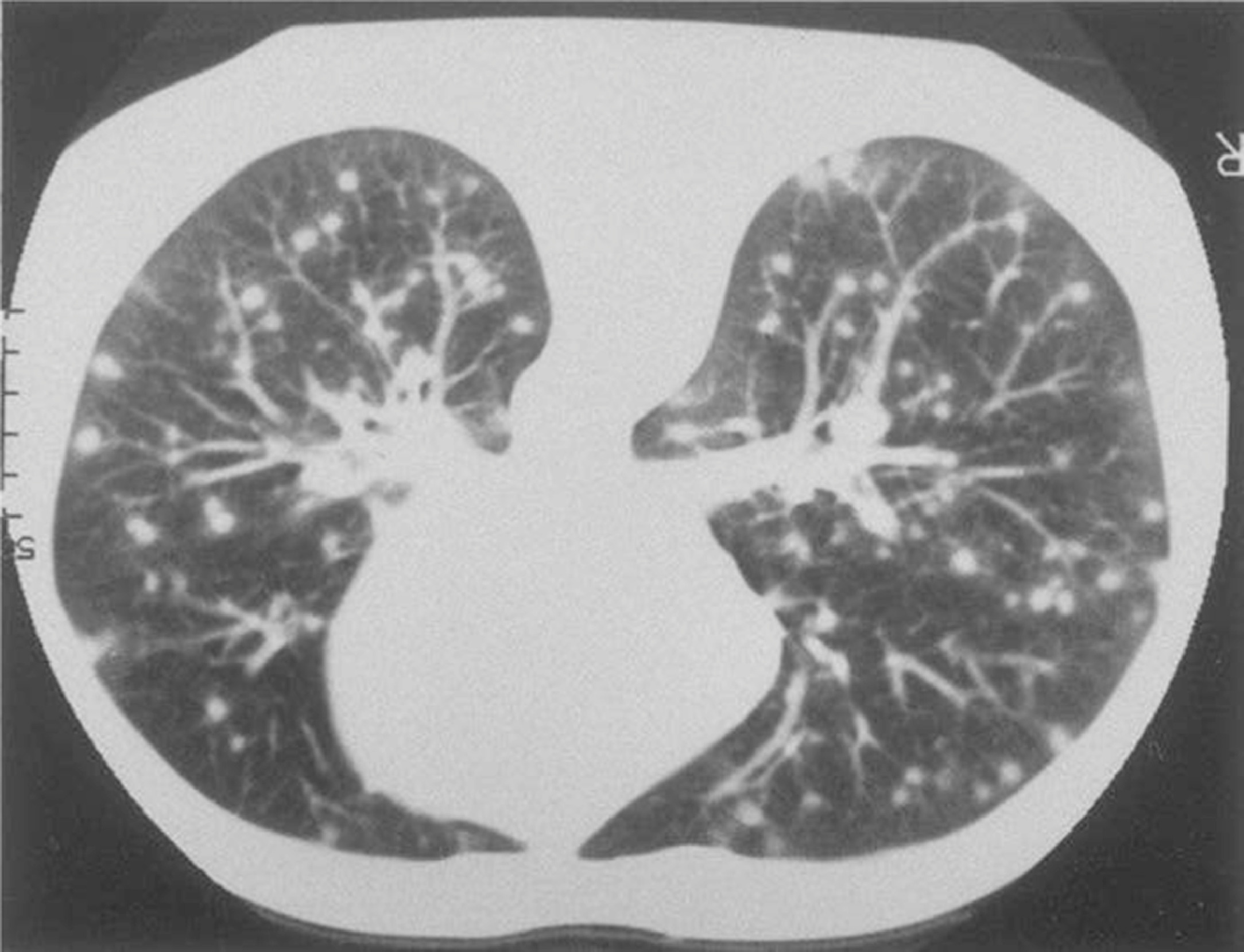
An axial slice from a computed tomography study of the chest at the level of the carina that shows the classic miliary nodular appearance or “snowstorm” pattern of acute histoplasmosis pneumonia. (From US Centers for Disease Control and Prevention [CDC], Public Health Image Library [PHIL]. PHIL ID #472. CDC Provider: Not identified. Available at
In disseminated infections, hematogenous dissemination of yeast forms may occur in rare cases, causing friable, granulomatous lesions on the skin, conjunctiva, or mucosal surfaces; or in lymph nodes, bone, intestines, and liver. Cutaneous, mucosal, and disseminated gastrointestinal histoplasmosis may also occur without significant pneumonia, usually in immunocompromised patients, especially patients with AIDS.
CLINICAL MANIFESTATIONS OF HISTOPLASMOSIS
Several factors determine the severity of Histoplasma infection, including the size of the inhaled inoculum of microconidia and the patient's age and immune status. As noted, less than 1% of exposed individuals will develop symptoms; of those, 60% will present with respiratory symptoms of chest pain, cough, and shortness of breath. 13 Additional clinical manifestations of histoplasmosis in immunocompetent patients resemble influenza. These symptoms may include fever, chills, abdominal pain, nausea, and vomiting, especially in children; and malaise, fatigue, weight loss, severe headache, myalgias, and arthralgias in adults.3,8,13
The clinical manifestations of histoplasmosis in the immunocompromised may share fewer presenting features and have a broader range of presenting findings, including skin and mucosal lesions, painful lymphadenopathy, leukopenia, and granulomatous lesions in the airway, colon, eyes, liver, or brain. The shared features in the immunocompromised often include a prolonged course of fever, weight loss, cachexia, and pancytopenia.
In patients with AIDS and disseminated histoplasmosis, the presenting manifestations of disease include severe cachexia, weight loss, and other opportunistic fungal infections, such as thrush, and granulomatous lesions in the liver, spleen, brain, or colon. 31 Norton et al 32 reported 2 cases of gastrointestinal histoplasmosis with colonic perforation and pneumoperitoneum in patients with AIDS who presented with chronic abdominal pain and melena.
African histoplasmosis caused by H capsulatum var duboisii is coendemic with H capsulatum var capsulatum in sub-Saharan central and west Africa and likely underreported. 33 All cases of African histoplasmosis reported from Europe have been imported. 33 Although the primary mode of transmission of African histoplasmosis is by airborne inhalation of contaminated soil, transmission by direct inoculation of microconidia in soil-contaminated wounds has also been reported. 33 In HIV-negative patients, the infection is typically located in the lungs. Patients with HIV will manifest more disseminated disease, with granulomatous lesions in the skin and lymph nodes and osteolytic lesions in the skull, ribs, and vertebrae. 33 As in H capsulatum var capsulatum–caused histoplasmosis in HIV-infected patients, the case fatality rate in disseminated African histoplasmosis is about 20%. 33 Diagnostic and treatment strategies are similar to those recommended for H capsulatum var capsulatum–caused histoplasmosis.
DIFFERENTIAL DIAGNOSIS OF HISTOPLASMOSIS
In immunocompetent persons with symptomatic pulmonary histoplasmosis, the differential diagnosis includes influenza, community-acquired pneumonia, tuberculosis, and psittacosis.34,35 The differential diagnosis of disseminated histoplasmosis in immunocompromised patients is significantly more extensive than the differential diagnosis in the immunocompetent, especially in patients with AIDS and diffuse reticulonodular pneumonic disease. In such cases the differential diagnosis should also include miliary tuberculosis, Pneumocystis pneumonia, Kaposi sarcoma, cytomegalovirus pneumonia, lymphoma, and other opportunistic fungal infections, such as coccidioidomycosis, paracoccidiomycosis, cryptococcosis, and penicilliosis.32,33
LABORATORY DIAGNOSIS OF HISTOPLASMOSIS
Because chest radiographs and thoracic computed tomographic scan findings mimic other diseases, such as pneumonia and tuberculosis, and are not diagnostic of histoplasmosis, the final diagnosis of histoplasmosis is dependent on laboratory tests.36,37 The clinical laboratory tests for histoplasmosis include antigen enzyme immunoassay (EIA) tests, serologic tests, fungal cultures, and tissue histopathologic findings.36,37 The Histoplasma EIA tests, developed in the late 1970s, rapidly detect the presence of H capsulatum antigens in blood and urine and are the recommended initial screening tests for histoplasmosis.36,37 Antigen EIA tests are also useful for rapid diagnosis in patients with immunocompromising conditions, such as AIDS. A positive blood or urine Histoplasma EIA test is usually defined as ≥0.6 ng·mL−1.3,38 The sensitivity of EIA tests, however, increases with the severity of illness, and EIA tests may yield false negative results in patients with less severe disease and lower antigen burdens. 38 Unlike serologic tests designed to detect Histoplasma antibodies, EIA tests detect histoplasmin antigen, do not depend on delayed antibody formation, and can confirm infection immediately in most cases.
Serologic tests, such as complement fixation and immunodiffusion, will confirm EIA-made diagnoses of acute histoplasmosis retrospectively because it takes 6 or more weeks for antibodies to H capsulatum antigens to develop.3,37 These tests will detect antibodies to Histoplasma yeast or mycelia in convalescent compared with acute sera with sensitivities of about 80%.3,39 The serologic tests will be positive in 90% of patients with pulmonary disease and 85% of patients with disseminated disease. 34 Serologic tests for histoplasmosis may be inaccurate in patients with AIDS. 32 In these cases, diagnoses should be confirmed by culture or histopathologic examination.
The most specific, confirmatory tests for histoplasmosis are fungal cultures and the histopathologic identification of H capsulatum in tissues. Blood, bone marrow, sputum, lesion aspirates, or tissue biopsies may yield positive cultures of Histoplasma. Histoplasma yeast forms may be identified histologically in sputum, needle biopsy specimens of lesions in the lungs or liver, or surgically excised specimens, such as lymph nodes, liver biopsy specimens, and lesion biopsy specimens (Figure 4).
In addition to microscopic techniques, fungal cultures, antigen and antibody detection tests, and molecular tests (polymerase chain reaction), a new generation of rapid point-of-care tests have now been reported to have increased specificities and shorter turnarounds in diagnosing invasive fungal infections of the lungs. 38 These latest diagnostic tests include the Aspergillus-specific lateral flow assay for pulmonary aspergillosis and the Histoplasma-specific loop-mediated isothermic amplification assay for pulmonary histoplasmosis. 38 Such rapid point-of-care tests will certainly augment the older, traditional laboratory tests in diagnosing histoplasmosis.
TREATMENT OF HISTOPLASMOSIS
Immunocompetent patients with asymptomatic histoplasmosis will require no treatment. 39 In most cases, acute histoplasmosis pneumonia in immunocompetent patients will also require no antifungal therapy unless symptoms, especially fever and malaise, persist for more than a month. In these cases, treatment with oral itraconazole, 100 mg twice a d for 3 to 6 mo, should be instituted to prevent progression to chronic pulmonary disease. 39 Chronic pulmonary disease should be treated in both immunocompetent and immunocompromised patients with oral itraconazole, 200 mg twice a d for 12 to 24 mo. 39 Disseminated histoplasmosis should be treated aggressively beginning with intravenous induction therapy with itraconazole, 200 mg intravenously twice a d for 3 d, followed by itraconazole, 200 mg intravenously every d for 14 d. 39 After intravenous therapy, patients with disseminated histoplasmosis should be continued on oral itraconazole, 200 mg twice a d for 12 to 24 mo. 39
Relapsing disease will occur in 10 to 20% of immunocompetent patients with disseminated histoplasmosis and in 80% of patients with disseminated histoplasmosis and AIDS. 39 In these cases, retreatment with itraconazole is indicated. Without long-term treatment with itraconazole, the mortality from disseminated histoplasmosis is 80%. 39 In cases of relapsing disease and severe acute pneumonic disease, intravenous amphotericin B remains a treatment option. 39 The regimen in these cases should include the intravenous administration of the deoxycholate formulation of amphotericin B, 0.7 to 1.0 mg·kg−1 daily for 1 to 2 wk, followed by intravenous itraconazole, 200 mg 3 times a d for 3 d, and then oral itraconazole 200 mg twice a day for 12 wk. 16
Recently, isavuconazole has offered a new option for the management of invasive fungal infections of the lungs, specifically pulmonary aspergillosis, and may provide advantages over the other currently available antifungals, such as amphotericin and other azoles, in treating histoplasmosis, with improved pharmacokinetic and safety profiles. 40
PREVENTION AND CONTROL OF HISTOPLASMOSIS
Environmental testing of soil samples for Histoplasma using fungal culture–based methods is resource intensive and not recommended without an inexpensive commercially available test. Molecular methods to detect H capsulatum in soil samples are in development and confined to research studies. 41 The US National Institute of Occupational Safety and Health (NIOSH) no longer recommends formalin decontamination of soil or other environmental materials because of the health hazards of formalin and the impracticality of treating large areas or other settings, such as caves. 41 NIOSH provides the following recommendations for workers who disrupt contaminated soils or accumulations of bird or bat droppings: 1) exclude birds or bats from buildings; 2) post warnings about health risks to workers; 3) control dust by wetting surfaces during construction, demolition, or excavation in endemic areas; 4) properly dispose of all contaminated wastes; and 5) select and wear appropriate personal protective equipment. 41
Persons who visit caves or any other areas with high concentrations of bird or bat droppings should also consider using personal protective equipment, including gloves, protective clothing, and respirators. 41 Bamboo groves near residences and schools that serve as bird roosts for more than a year should be removed by workers wearing protective gear and disposed of properly. 29 Lastly, sporadic clusters of urban histoplasmosis may result from an increasing popularity of keeping backyard chicken coops and racing pigeon flocks. 42 Public health officers should anticipate these rediscovered hobbies as potential risk factors for histoplasmosis, especially in the immunocompromised, and inspect, license, and regulate amateur bird breeders. 42
Because the poultry industry is an agricultural mainstay today, several vaccines for the primary prevention of avian infectious diseases are in use or development. 43 Vaccines for the primary prevention of endemic human mycoses are also in development and would benefit immunocompromised patients at risk of fungal pneumonias, such as coccidiodomycosis and histoplasmosis. 44 In animal models, experimental vaccines have induced the production of T-helper immune cells active against Histoplasma antigens.45,46 Endemic mycoses, including histoplasmosis, can activate heat shock proteins, which are similar to cytokines and increase pathogen virulence and ability to evade host immune responses. 47 Immunization against heat shock proteins offers another avenue for protection against disseminated fungal infections, especially in the immunocompromised. 47
Conclusions
Histoplasmosis is more common in endemic and nonendemic areas than previously reported and can result in considerable morbidity, especially in the immunocompromised. Histoplasmosis is increasingly imported from endemic areas to nonendemic areas worldwide. Healthcare providers, public health officials, occupational health and safety officers, and construction managers should exercise an increased awareness of the risks of histoplasmosis in order to institute effective measures to reduce human exposures to Histoplasma microconidia in endemic areas and during the disruption of bird or bat droppings or contaminated soils.
Footnotes
Acknowledgment
Support for Dr. Diaz was provided by departmental and institutional sources.
