Abstract
We report the case of a patient who presented with respiratory failure, recurrent ventricular fibrillation, ventricular arrhythmias, and hypotension after an intentional ingestion of aconite flowers. Significant ingestion of this plant can produce life-threatening cardio- and neurotoxicity that may require evacuation from the wilderness to a medical facility capable of advanced treatment and intensive care monitoring.
Introduction
Aconitum is a genus of over 300 species of flowering plants belonging to the family Ranunculaceae. In China alone, 1 there over 200 species of this plant. Toxicity of the plant has been acknowledged since ancient times. 2 All of its parts are toxic, especially the roots and tubers, which contain a higher concentration of C19-diterpenoid alkaloids (aconitine, mesaconitine, hypaconitine) than its flowers and leaves. 1 In Europe, the most widespread Aconitum species are monkshood (Aconitum variegatum and napellus) and wolfsbane (Aconitum lycoctonum). Monkshood is a toxic perennial plant native and endemic in the Alps and in the mountainous areas of western and central Europe. It bears beautiful blue/purple flowers during the flowering period (June to September). Routes of entry of these toxins are oral (accidental or intentional ingestion) and dermal.
Case report
A healthy 63-year-old male trekker voluntarily ingested, for unspecified reasons, approximately 30 monkshood flowers (Figure 1) during a hike in the Brescia Prealps area. Within 2 h of ingestion, the subject had profuse salivation, leading him to call for help. An airborne rescue team, dispatched from the helicopter emergency medical service base of Brescia, arrived on the scene 18 min after the emergency call and found the patient to be conscious and alert, with marked hypersalivation, blood pressure 120/70 mm Hg, heart rate 70 beats·min-1, and eupneic breathing. The patient reported paresthesia and numbness in all 4 limbs with no motor deficits, and both pupils were equal, round, and reactive to light. An initial electrocardiogram performed on the helicopter on the way to the hospital showed sinus rhythm with premature ventricular contractions (PVCs). As part of supportive treatment in the field, a peripheral venous line was placed and fluids (500 mL bolus of a lactated Ringer’s solution) were administered. The national poison center confirmed monkshood poisoning from photographs taken by the patient himself and sent digitally by the rescue team.

Aconitum napellus in the Italian Prealps. Aconitum napellus is an herbaceous perennial plant of the family Ranunculaceae. These plants are chiefly native to the mountainous parts of the Northern Hemisphere, growing in the moisture-retentive but well-draining soils of mountain meadows. The hood-like shapes of his flowers give the plant its common name of monk’s hood. Used with permission.
Upon arriving at the emergency department, approximately 45 min after placing the initial emergency call, the patient was awake, alert, and fully oriented. He was nauseous and experiencing hypersalivation and generalized malaise. His vital signs and rhythm strip were similar to what were found in the field (PVCs and nonspecific T waves in the inferior leads). An orogastric tube (36 French gauge) was inserted, and gastric lavage (7 L of warm water) was performed until clear. An “unquantified” amount of presumed plant and flower-like material was recovered. The orogastric tube was removed, and activated charcoal (60 g) was administered by mouth. The intravenous (IV) fluids were continued and osmotic laxatives (Macrogol) were administrated.
The patient was transferred to the intensive care unit (ICU). After he was admitted into the ICU, the patient reported worsening paresthesia in the perioral area and worsening of his general malaise. His condition quickly began to deteriorate, with active vomiting, profuse diaphoresis, and generalized pallor and cold extremities. In the ICU, the patient developed respiratory arrest due to muscle paralysis. He was sedated with midazolam (0.3 mg·kg-1) and fentanyl (2 mcg·kg-1), intubated, and mechanically ventilated (Table 1). He became hypotensive (90/60 mm Hg) and tachycardic (130 beats·min-1) with frequent ectopy (primarily PVCs/ventricular ectopy). The ICU staff started therapy with amiodarone (ie, 150 mg bolus+600 mg continuous infusion) and aggressive IV fluid therapy. Approximately 1 h after admission to the ICU, the patient experienced a sudden loss of consciousness with generalized, tonic-clonic seizure-like activity and accompanying ventricular tachycardia. Lidocaine (150 mg initially, and a second dose of 50 mg) was administered and the patient was defibrillated 4 times (at 360 joules each) until sinus rhythm/sinus tachycardia was again achieved.
Hb, hemoglobin; WBC, white blood count; PLT, platelets; AST, aspartate aminotransferase; ALT, alanine aminotransferase; PCO2, carbon dioxide partial pressure; PO2, oxygen partial pressure; SatO2, oxygen saturation; HCO3, bicarbonate; BEecf, base excess in the extracellular fluid compartment; Lact, lactate; P/F, arterial oxygen partial pressure/fractional inspired oxygen.
Approximately 2 h and 15 min after admission to intensive care, the patient developed ventricular tachycardia alternating atrial fibrillation with a rapid ventricular response (ie, 130–140 beats·min-1) and another drop in systemic blood pressure to 90/50 mm Hg. IV fluid boluses were administered, resulting in transient reversal of hypotension. After approximately 1 h of mechanical ventilation, the patient again experienced pulseless ventricular fibrillation. Once again, lidocaine (150 mg) was administered, and the patient was defibrillated 5 times (360 joules each). Asystole ensued, and cardiopulmonary resuscitation was performed. Epinephrine (1 mg 1:10,000 0.1 mg·mL-1 IV push) was administered with almost immediate return of spontaneous circulation and normal sinus rhythm, resulting in the cessation of cardiopulmonary resuscitation. Pharmacological therapy with catecholamine support was initiated for ersistent hypotension: norepinephrine (0.03 microgram·kg-1·min-1) was begun, followed by the addition of dopamine (3 microgram·kg-1·min -1), and magnesium sulfate (1 g IV infusion) (Figure 2).
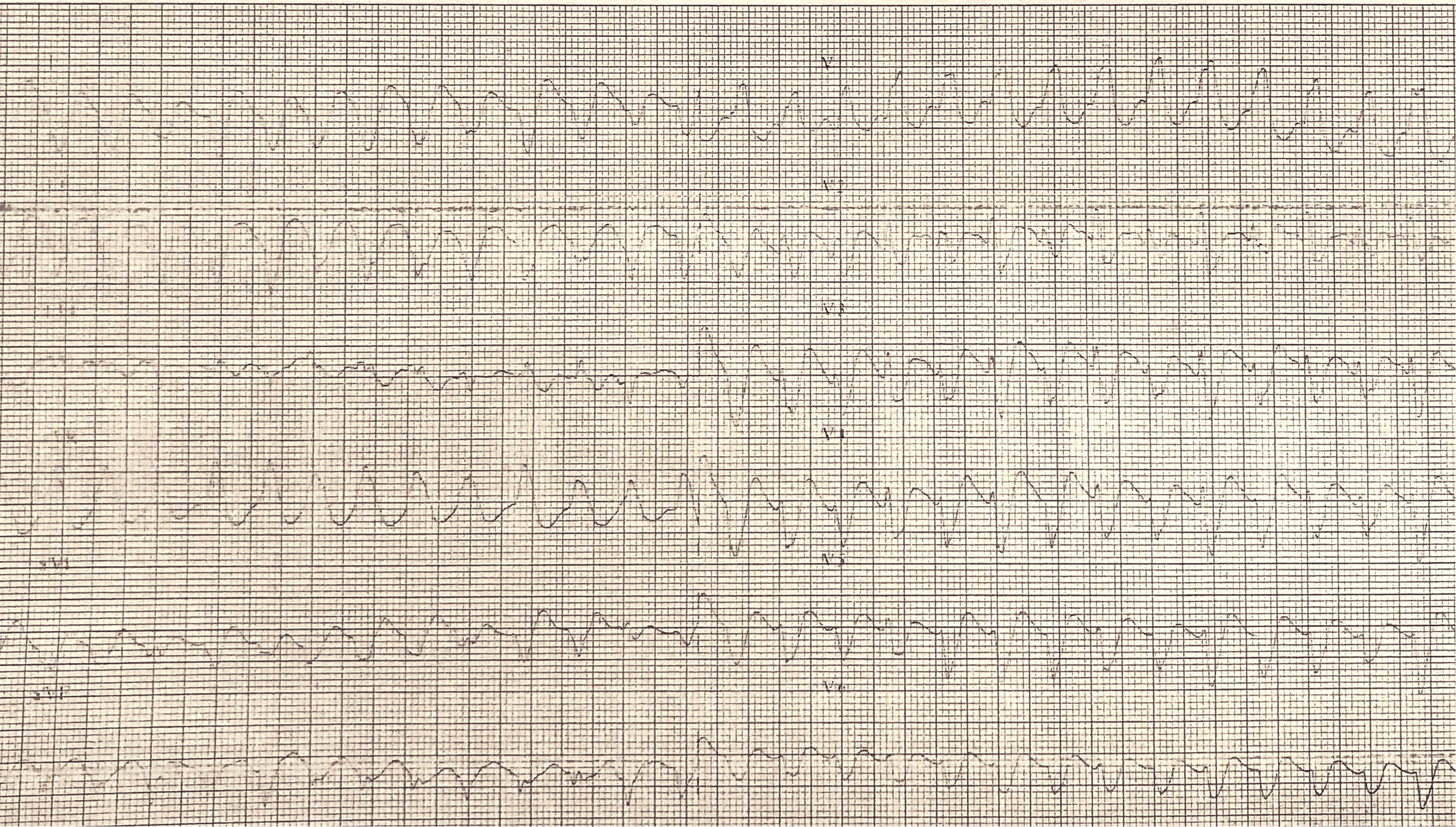
Electrocardiogram performed in intensive care unit. The tracing shows the first episode of ventricular tachycardia.
On day 2, ventilatory parameters remained unchanged, and the patient was well adapted to the mechanical ventilator. The ventilator was set in synchronized intermittent mandatory ventilation (volume control)+pressure support mode, and the patient showed good respiratory exchanges, pH 7.43, and lactates in progressive reduction (Table 1). He had mild hypernatremia, blood pressure of 100/50 mm Hg, and heart rate of 52 beats·min-1, rhythmic. Therapy with catecholamines was continued.
On day 3, the patient was hemodynamically stable and rhythmic, with circulation sustained only by low-dose noradrenaline. The patient was ventilated with gas exchanges at the limit of the norm (arterial oxygen partial pressure/fractional inspired oxygen ratio=220 mm Hg). Laboratory examinations including complete blood count and serum chemistries were mostly in the normal range, except for creatine kinase (elevated at 12,277 u·L-1), aspartate aminotransferase (elevated at 258 u·L-1), and alanine aminotransferase (elevated at 107 u·L-1) (Table 1).
On day 4, hemodynamic parameters were normal. Blood pressure was 130/60 mm Hg, and the heart rate 70 beats·min-1. The patient was successfully extubated and connected to a venturi mask at 40%. Creatine kinase was in sharp decline to 6484 u·L-1 and troponin I improved to 0.047 ng·mL-1 (Table 1).
On day 5, the patient was once again awake, cooperative, and hemodynamically stable, in normal sinus rhythm without cardiac ectopy. Intravenous therapy with pressor agents was completely discontinued. The creatine kinase improved to 2350 u·L-1 and troponin I improved to 0.015 ng·mL-1 (Table 1).
On day 6, the patient continued to be hemodynamically stable and eupneic (oxygen saturation 98%, respiratory rate 26) with a venturi mask at 40%. He was transferred out of the ICU and into the internal medicine ward. Inpatient supportive medical and psychiatric care continued for several more days. The patient was discharged from the hospital 11 d after admission with full recovery and a return to normal baseline of all laboratory evaluations.
Discussion
Aconitum toxins affect several excitable tissues, such as the myocardium, nerves, and muscles. This case presented not only major life-threatening conditions in Aconitum poisoning, such as respiratory depression and ventricular arrhythmias,3,4 but also other important conditions, including neurotoxicity (paresthesia in all 4 extremities, circumoral and extremity numbness, and generalized muscular tonic-clonic/seizure-like activity),1,5 and gastrointestinal features (nausea, vomiting and abdominal pain).1,5 The neurologic and cardiologic phenomena are due to the actions of C19-diterpenoid alkaloids (aconitine, mesaconitine, hypaconitine) on the voltage-gated sodium channels 6 of the cell membranes of excitable tissues. This can be explained by the high affinity of C19-diterpenoid alkaloids to the voltage-sensitive sodium channels at site 2 in the cell membrane. 7 More precisely, site 2 is located on the S6 transmembrane segments of all 4 pore-forming α-subunits in the cell membrane. 8 This interaction between toxin and voltage-sensitive sodium channels causes a persistent activation of the sodium channels by increasing late sodium current (INa), which brings about sustained depolarization of the cells. 7 The sodium channels of the depolarized cells become refractory to excitation and are able to generate afterdepolarizations (early or delayed afterdepolarizations). The afterdepolarizations are responsible for the triggered activity of the heart, one of the mechanisms responsible for arrhythmias. However, recent research also indicates an inhibitory action of the C19-diterpenoid alkaloids on potassium channels 8 (IKr) and calcium channels 9 (ICa-L). These actions seem to further contribute to the generation of early or delayed afterdepolarizations.
The deadliest complication of aconite poisoning would seem to be lethal ventricular tachyarrhythmia (polymorphic ventricular tachycardia, ventricular fibrillation, and the rarer bidirectional ventricular tachycardia). Clinically, this was the challenge in our patient: trying to control recurrent, ventricular arrhythmias in the face of hypotension and respiratory failure. In this particular case, recurrent ventricular fibrillation had shown sensitivity to electrical cardioversion and standard antiarrhythmics. It has been generally reported that aconite-induced ventricular arrhythmias are often refractory to direct current cardioversion and antiarrhythmic drugs. 10 A recent review 3 and multiple case reports 4 ,5,11 in the medical literature seem to suggest that amiodarone and flecainide are reasonable first-line treatments. Our experience with this individual case demonstrates that cardioversion and antiarrhythmics were, in fact, very useful. In our case, lidocaine seemed to be a bit more helpful at times; however, its effects seemed somewhat transient as compared with amiodarone. In addition, amiodarone-dosing limits prevented using it as frequently as the arrhythmias were occurring in the first 48 h of care. Lidocaine was also available for administering more rapidly than amiodarone, and by that time our patient had reached the recommended maximum dose of amiodarone. Based on the evidence of this clinical case, we hypothesize a broader toxicity of the compounds of C19-diterpenoid alkaloids on the human heart, which needs further investigations.
In this case, given that the amount of poison ingested was potentially life-threatening, the gastric lavage was performed. Although no complications were recorded, this practice of decontamination has high risk of developing major morbidities (aspiration pneumonitis, esophageal or gastric perforation, fluid and electrolyte imbalances, arrhythmias). Activated charcoal 11 should instead be considered in patients who present within 1 h of ingestion and have an intact or protected airway.
Aconitum poisoning in a wilderness setting presents unique challenges. Aconitum species are found in circumboreal-arctic zones, and poisoning occasionally occurs in remote mountain settings with the accidental ingestion or use of plants as an herbal remedy by the local population and mountain travelers. Management of such intoxication involves a wide spectrum of possibilities. Interviewing the victim and witnesses and having a photograph of the plant may reveal useful identification information. The first responder should obtain expert advice and begin rescue logistics as early as possible by contacting the most appropriate local emergency dispatch center, emergency department, or poison center. Any victim poisoned by an aconite should be immediately evacuated from the field and administered definitive medical care. Once this has been achieved, the victim must be admitted to a medical facility for at least 24 to 48 h, even if asymptomatic, given the onset of life-threatening arrhythmias. There is no antidote for aconitine poisoning; treatment directed toward inhibiting further absorption of toxins and carefully managed supportive therapies, especially intensive cardiopulmonary monitoring, 5 should be considered as an integral part of good clinical care in Aconitum poisoning.
Acknowledgments: We thank Mr. Amos Da Lozzo, photographer of “Destinazione Montagna,” for providing photos of the plant of Aconitum napellus present on the rescue scene.
Author Contributions: Report production and literature review (FA, PP); patient care and report creation oversight (PB, GP, CC, GB).
Financial/Material Support: None.
Disclosures: None.
