Abstract
The current therapy for myocarditis is immunosuppressive therapy. However, in rare cases in which patients do not respond to intervention, their condition can rapidly deteriorate to myocarditis with shock, which is characterized by extensive and diffuse lymphocyte infiltration in the myocardium. Most cases of myocarditis are caused by virus-mediated damage of cardiomyocytes, and its clinical manifestations are ventricular arrhythmia and hemodynamic disturbances. Extracorporeal membrane oxygenation is an effective intervention, which regulates hemodynamic stability and avoids systemic hypoperfusion. This intervention has been used to sustain hemodynamic stability in patients with myocarditis and shock. We report here early application of extracorporeal membrane oxygenation for successful treatment of a patient with myocarditis and shock.
Introduction
According to a meta-analysis of the Global Burden of Disease Study 2013, the annual incidence of morbidity of acute myocarditis is estimated to be 22/100,000 people. 1 Among all other types of myocardial injury, heart failure accounts for 21.3%. Myocarditis is an inflammatory disease of the myocardium. Potential causes of myocarditis include a variety of viral infections, drug toxicity, and autoimmune diseases. Among these causes, viral infections are the most common cause of myocarditis. The inflammatory response after viral infection appears to be the major driver of myocardial pathophysiology. 2 The most common immune cells that infiltrate the myocardium are lymphocytes. 3 Viruses can trigger inflammatory amplification, and cause myocardial damage through molecular stimulation and other mechanisms. 4 In most cases, myocarditis is a self-limiting disease, but in some circumstances, it can progress to fatal myocarditis with shock, which manifests as hemodynamic disturbance and ventricular arrhythmia. 5 When myocarditis occurs, heart function dramatically declines owing to acute and severe myocardial damage, and blood perfusion of the main organs cannot be well sustained. Therefore, patients with myocarditis and shock need to survive the acute phase. Extracorporeal membrane oxygenation (ECMO) can simultaneously support cardiopulmonary function, provide a protective effect on the damaged myocardium, and allow sufficient time for recovery of the cardiopulmonary system. In ECMO, the blood is drained via veins and passes through the membrane lung where oxygen is added and carbon dioxide is expelled. The oxygenated blood can be infused into the vein or the artery. We report a successful case of early application of ECMO in a patient with myocarditis and shock.
Case presentation
A 20-year-old female patient who had experienced a cold 10 days previously was admitted to the hospital. She was diagnosed with myocarditis with the symptoms of sinus tachycardia and occasional ventricular premature beats. She had intermittent fever for the previous 10 days, heart palpitations for the previous 3 days, and her condition had worsened within 2 hours after being admitted to the hospital. The patient’s body temperature was elevated to 37.6°C. After she was administered cephalosporin (dosage unknown), her condition slightly improved. Paroxysmal palpitations occurred 3 days previously and lasted for approximately 2 minutes, accompanied by mild chest pain and tightness. This sensation lasted for approximately 30 s and then disappeared. No cough, sputum expectoration, hemoptysis, dyspnea, abdominal pain, diarrhea, or bloating was found. The patient described herself as healthy, and declared no history of infectious diseases, allergies, trauma, surgery, or blood transfusion.
When the patient arrived at the hospital, examination results were as follows: heart rate was 118 beats/minute, occasional ventricular premature contractions were identified, and there was no galloping rhythm on auscultation. An electrocardiographic (ECG) examination showed ventricular tachycardia (Figure 1a). At the same time, blood tests were carried out in the laboratory. Myocardial markers were increased during ventricular fibrillation as follows: troponin I concentration, 2.00 ng/mL (normal range: 0–0.04 ng/mL); myoglobin concentration, >900 ng/mL (normal range: 0–120.0 ng/mL); creatine kinase isoenzyme concentration, 37 ng/mL (normal range: 0–16.0 ng/mL); D-2 polymer concentration, 885 ng/mL (normal range: 80.0–500.0 ng/mL); and amino-terminal brain natriuretic peptide concentration, 8150 ng/mL (normal range: 300.0–450.0 ng/mL).
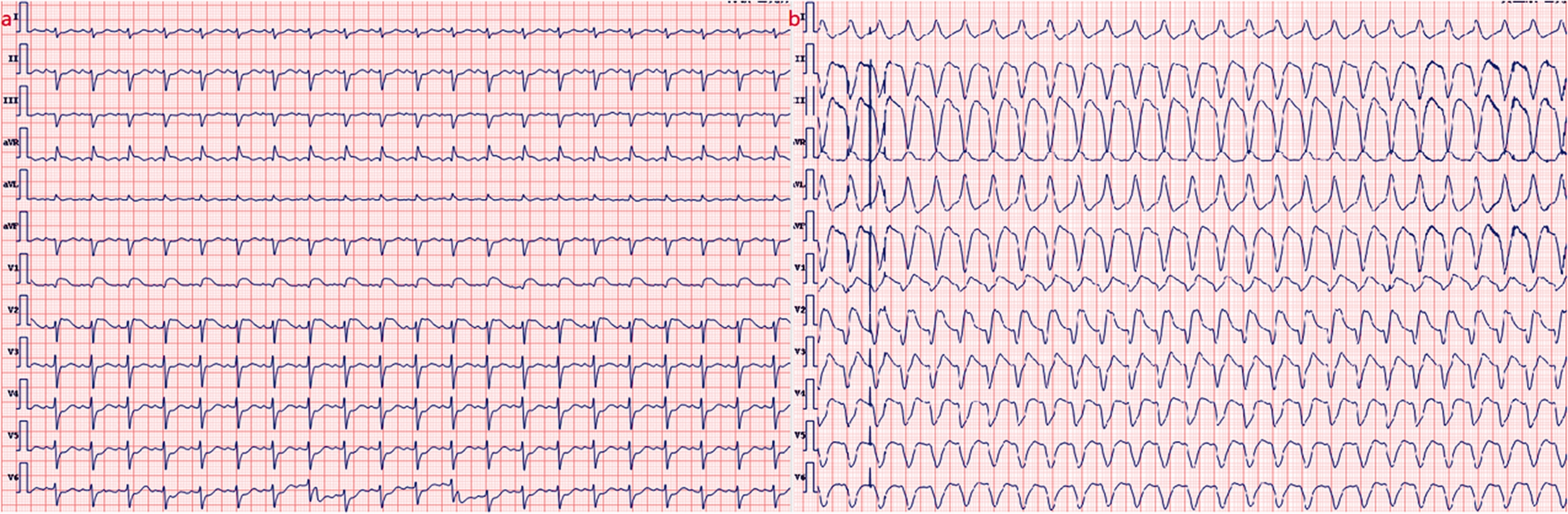
Electrocardiographic results before and after myocarditis with shock. (a) Electrocardiogram of the patient showing sinus tachycardia and occasional ventricular premature beats at the time of admission. (b) Electrocardiogram of the patient showing ventricular tachycardia at the time of ventricular fibrillation shock.
Three hours after admission, the patient reported a headache and tinnitus during the initial consultation process. She suddenly showed a slow response to our questions, and no blood was able to be withdrawn from her vein. In the next minute, a sequence of ventricular fibrillation occurred simultaneously. She was immediately provided with bedside cardiac compression and single-channel 270 J non-synchronous electrical defibrillation. An ECG examination was performed after defibrillation (Figure 1b). An intravenous bolus of 270 mL of 5% glucose + 0.3 g amiodarone was also administered. After electrical defibrillation, the patient had a pupil response. During the time of the coma, her blood pressure was 65/40 mmHg, her blood oxygen saturation was 50 mmHg, and there was shallow and spontaneous breathing. Laboratory examination results showed an elevation in several myocardial markers, including troponin I, myoglobin, creatine kinase isoenzyme, and amino-terminal brain natriuretic peptide (the patient’s family refused a myocardial biopsy). She was then diagnosed with myocarditis and shock on the basis of a repeated hemodynamic decline, arrhythmia, and an elevation in myocardial markers.
Within the next 3 hours, we identified six events of ventricular fibrillation and performed a sequence of defibrillation as described above. The patient was immediately administered a mixture of epinephrine 1 mg/mL, dopamine 10 mg/mL, lidocaine 20 mg/mL, and esmolol 10 mg/mL. Additionally, a 20-mL 0.9% sodium chloride injection with potassium chloride 2 g and vitamin C 2 g was provided. Methylprednisolone 40 mg was also administered by an intravenous bolus injection. An intravenous drip containing 250 mL of 0.9% sodium chloride, dopamine 200 mg, 10 mL of potassium magnesium aspartate, magnesium sulfate 1 g, sodium bicarbonate 12.5 g, and 2 meta-hydroxylamine 0.1 mg/mL was started. Suddenly, the patient’s spontaneous breathing disappeared, and we provided tracheal intubation with a ventilator to assist breathing. At the seventh event of ventricular fibrillation, an ECG showed continuous ventricular tachycardia and the patient was unconscious. Her heart rate was 150 beats/minute, and her blood pressure was 70/55 mmHg. We decided to proceed with ECMO intervention.
Six hours after the onset of the patient’s complications, ECMO was started by following the standard operating procedure. Initially, the patient’s bilateral inguinal area was disinfected, and left femoral artery access and right femoral vein access were established under ultrasound guidance. The skin was then gradually exposed to reveal the artery, and an 18 Fr arterial cannula was placed in the left femoral artery to a depth of 15 cm. This was followed by insertion of a 22 Fr venous cannula into the right femoral vein to a depth of 45 cm. Forty minutes after the preparation, the ECMO was successfully connected, and the machine ran smoothly at 2675 rpm (1200 × g) with a flow rate of 3.5 L/minute. The machine was functioning well, all cannulated vessels were fixed, and the vital signs of the patient were stable. A color Doppler ultrasound examination of the heart was performed after ECMO (Figure 2a). Most of the cardiac function parameters, including the large vessels, atrium, left and right ventricles, bicuspid valve, tricuspid valve, aortic valve, pulmonary artery, and pericardium, were normal, except for the ejection fraction, which was slightly lower than the normal range. Six days later, the patient was weaned off from ECMO 6 and was discharged from the hospital 14 days later with a promising cardiac magnetic resonance imaging (MRI) result. After ECMO and upon discharge from the hospital, the troponin I concentration had decreased to 0.08 ng/mL, myoglobin to 84.4 ng/mL, and creatine kinase isoenzyme to 2.7 U/L. The aspartate aminotransferase concentration was 25.04 U/L (normal range: 14.00–36.00U/L) and the creatine kinase concentration was 101.67 U/L (normal range: 30.00–135.00 U/L). A color Doppler ultrasound examination of the heart was performed when the patient was discharged (Figure 2b) and showed that the ejection fraction had increased from 44% just after ECMO to 72%. Cardiac MRI was also performed at the time of discharge (Figure 3). MRI demonstrated normal left ventricular morphology, no obvious abnormal ventricular wall motion signal, and a regular trace of pericardial effusion. The patient was administered antiarrhythmic drugs, including a β-receptor blocker.

Reports of echocardiographic examinations after extracorporeal membrane oxygenation and at discharge. (a) Report of an echocardiogram of the patient after extracorporeal membrane oxygenation. Most of the cardiac function parameters were normal, except for a low ejection fraction. (b) Report of an echocardiogram at discharge shows that the ejection fraction is greatly improved.

Results of a cardiac magnetic resonance imaging (MRI) examination. (a) Cardiac MRI shows a four-chamber sequence of micro-pericardial effusion. (b) Cardiac MRI shows a two-chamber sequence of micro-pericardial effusion. In panels a and b, the arrows indicate the position of a small amount of pericardial effusion c: Cardiac MRI shows a four-chamber sequence of a plain scan. d: Cardiac MRI shows a four-chamber sequence of plain scan enhancement. In panels c and d, the cardiac structure was normal without any indication of fibrous hyperplasia.
Discussion
We experienced a 20-year-old female patient with myocarditis and shock. The moderate manifestations of this patient included palpitations and occasional ventricular premature beats. During the consultation process, we observed that her blood could not be withdrawn, the patient described that she had experienced tinnitus, and she started to lack a response while we were asking questions. Our case showed rapid progression of myocarditis with shock. The rapid progression of myocarditis with shock has a high mortality rate of 50%. 7 The early manifestations of patients with myocarditis are often moderate and are easily missed. One major characteristic of myocarditis is dramatic hemodynamic changes. 8 As soon as hemodynamic changes occur, we suggest that diagnosis and a treatment plan be immediately impelemented. 7
Myocarditis has a wide variety of clinical manifestations, ranging from mild symptoms, such as chest pain with mild ventricular dysfunction, to life-threatening arrhythmias and severe heart failure.9,10 Therefore, the prognosis of patients with myocarditis is unpredictable, with partial or complete recovery without medical intervention or progression to advanced heart failure within a few days. 8 The immune cells that are involved in histopathological changes in myocarditis are the three major subtypes of lymphocytes, eosinophils, and giant cells. Among them, lymphocytic myocarditis is the most prominent subtype. 3 Myocarditis with shock can present with sudden onset, and its main manifestations include rapid hemodynamic changes, cardiogenic shock, and fatal arrhythmias.3,11,12
Myocarditis with shock is an inflammatory disease of the myocardium with a high mortality rate. Early application of ECMO increases the prognosis of patients.13–15 ECMO is an efficient technique against hemodynamic instability and systemic hypoperfusion, and can be applied as the first-line mechanical support technique for patients with myocarditis and shock. 16 ECMO can effortlessly prevent the deterioration of hemodynamics in patients with myocarditis and shock.17–19 In fact, consistent with our case report, the registration of extracorporeal life support organizations indicates that early application of ECMO before cardiac arrest may give lead to outcomes. 14
Regarding the advantages and disadvantages of each imaging tool, an ECG and echocardiogram are more convenient and less time-consuming than cardiac MRI. However, an ECG and echocardiogram may still have a strong advantage in early diagnosis. In contrast to an ECG and echocardiogram, cardiac MRI can better evaluate the function of myocardial tissue. Cardiac MRI can be used to effectively determine the prognosis of myocarditis, 20 owing to its resolution for identifying fibrosis, edema, and inflammatory cell infiltration in the myocardium.21,22 Edema can progress to fibrosis, with a reduction in the occurrence of edema indicating less myocardial fibrosis. 23 Patients with myocarditis can recover quickly possibly because there is no problem with their own cardiac function, but there is ventricular fibrillation caused by myocarditis. Once the risk factor of myocarditis is removed, cardiac function can recover. This is different from many patients with poor cardiac function who have ventricular fibrillation. These patients may have basic diseases and a poor prognosis. Patients with giant cell myocarditis may show a small amount of fibrosis, and giant cell myocarditis often occurs in young people. The amount of research related to myocarditis and fibrosis is increasing, and a reduction in fibrosis may be a new research field in the future. 24 In our case, cardiac MRI showed that there was no visible fibrosis in micro-pericardial effusion, which suggested that our early application of ECMO was beneficial to the patient.
Conclusion
The treatment of reversing arrhythmia should be carried out as soon as possible in case of myocarditis with shock. The timing of early application of ECMO is a key intervention to treat patients with myocarditis and shock.
Footnotes
Ethics statement
Informed consent was signed by the patient’s parents. All details have been de-identified such that the identity of the patient may not be ascertained in any manner. The requirement for review board approval was waived because of the nature of this study (case report).
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This research was funded by the Health Commission of Jilin Province in 2018 (Project Number: 2018SCZ008).
