Abstract
Objective
This study aims to examine blood hemostatic responses to completing a 164-km road cycling event in a hot environment.
Methods
Thirty-seven subjects (28 men and 9 women; 51.8±9.5 [mean±SD] y) completed the ride in 6.6±1.1 hours. Anthropometrics (height, body mass [taken also during morning of the ride], percent body fat [%]) were collected the day before the ride. Blood samples were collected on the morning of the ride (PRE) and immediately after (IP) the subject completed the ride. Concentrations of platelet, platelet activation, coagulation, and fibrinolytic markers (platelet factor 4, β-thromboglobulin, von Willebrand factor antigen, thrombin-antithrombin complex, thrombomodulin, and D-Dimer) were measured. Associations between changes from PRE- to IP-ride were examined as a function of event completion time and subject characteristics (demographics and anthropometrics).
Results
All blood hemostatic markers increased significantly (P < .001) from PRE to IP. After controlling for PRE values, finishing time was negatively correlated with platelet factor 4 (r = 0.40; P = .017), while percent body fat (%BF) was negatively correlated with thrombin-antithrombin complex (r = –0.35; P = .038) and to thrombomodulin (r = –0.36; P = .036). In addition, male subjects had greater concentrations of thrombin-antithrombin complex (d = 0.63; P < .05) and natural logarithm thrombomodulin (d = 6.42; P < .05) than female subjects.
Conclusion
Completing the 164-km road cycling event in hot conditions resulted in increased concentrations of platelet, platelet activation, coagulation, and fibrinolytic markers in both men and women. Although platelet activation and coagulation occurred, the fibrinolytic system markers also increased, which appears to balance blood hemostasis and may prevent clot formation during exercise in a hot environment.
Introduction
Ultraendurance events represent an unusual opportunity to study human physiology under uncommon levels of exercise and environmental stress. Moreover, with the rising popularity of ultraendurance running and cycling over the last decade 1 it has become imperative to better understand the effects of these events on human physiology. Ultraendurance events (running, cycling, and triathlon) have been shown to induce changes in blood homeostasis, including platelet activation, coagulation, and fibrinolysis. 2 –5 We followed subjects in the Hotter ’N Hell Hundred (HHH), which typically generates great physiological stress, seen by cyclists losing 2% of their body mass despite intaking fluids approaching 1 L/h, 6 as it consists of a 164-km bicycle ride in northern Texas during summer heat that annually exceeds 37°C.
Activation of blood coagulation results from increased platelet activity, formation of thrombin and, ultimately, the formation of fibrin. Fibrinolysis increases due to a rise in tissue-type plasminogen activator and decrease in plasminogen activator inhibitor. If these 2 systems become unbalanced, risk of stroke and deep vein thrombosis significantly rises. The effects of such imbalance can be seen in complications from several medical conditions, including cancer and trauma. Other risk factors include older age, surgery, immobilization (as with prolonged bed rest, orthopedic casts, and sitting on long flights), combined oral contraceptives, pregnancy, the postnatal period, and genetic factors. 7 –9
Heat stress has been shown to have the potential to affect blood homeostasis, and has been shown to be regulated within a narrow range of temperatures. 10 In response to hyperthermic environmental conditions, subjects in several studies have exhibited increased levels in platelet counts (PC)11,12 and stimulation of platelet activity, made evident by changes in the levels of platelet factor 4 (PF4) and β-thromboglobulin (β-TG). 13 Furthermore, exposure to hyperthermic conditions can increase the concentration of thrombin-antithrombin (TAT) complexes, 11 which indicates potential for coagulation. However, it is also apparent that the human body tries to protect blood homeostasis by activating the fibrinolytic system, as evident in increases in D-Dimers. 11 ,14,15 Thus, prolonged strenuous exercise and heat stress may independently affect blood homeostasis to increase the risk of clot formation, including risk of stroke or deep vein thrombosis.
The benefits of habitual endurance exercise have been shown to include reduced occurrence of ischemic heart disease and lower cardiovascular mortality. However, acute exercise stresses the cardiovascular system, resulting in increased platelet activation, coagulation, and fibrinolysis, among other physiological effects. The activation of the coagulation and fibrinolytic systems are dependent on the duration and intensity of the exercise, which can either accelerate clot formation or lead to the dissolution of clot. 16 The hematologic effects of this systematic stress are especially marked in individuals participating in ultraendurance events. Specifically, PC increase during marathon runs,17,18 ultramarathon runs, 19 and prolonged bicycle rides, 5 while markers of platelet activation (β-TG and PF4) have been found to either increase20,21 or not change22,23 during endurance exercise. Additionally, prolonged endurance exercise produces both a hypercoagulable state (increased TAT), 4 ,5,22,23 which is usually balanced by fibrinolytic activation (increased tissue-type plasminogen activator and D-Dimers). 4 ,5,22,23
Many studies have examined the effect of prolonged exercise on blood homeostasis, 4 ,5,22,23 but to our knowledge no published investigation has examined the combination of heat stress and prolonged exercise on platelet activation. The purpose of this study was therefore to examine blood hemostatic responses to completing a 164-km road cycling event performed in a hot environment. In addition, this investigation, compared with our previous study, examines a larger sample size and includes women. 5 We hypothesized that platelet activation, coagulation, and fibrinolysis would increase during the event.
Methods
Subjects
Subjects were recruited through the HHH website, via emails sent to all HHH entrants, and in person at the Multi-Purpose Event Center (Wichita Falls, TX) during the HHH Exposition held on the days before the event (August 27–28, 2015). To be included in the study, subjects had to be healthy, aged 21–65 years, and have previously completed an organized bicycle ride of at least 164 km. Exclusion criteria consisted of the following: 1) inability to speak or understand English; 2) current tobacco use; 3) use of cholesterol-lowering, vasoactive, and/or anticoagulant medications (eg, Coumadin, aspirin); 4) current musculoskeletal injury; 5) diagnosis of cardiovascular, liver, kidney, blood, or gastrointestinal disease or severe metabolic or endocrine disorders (eg, type 2 diabetes); 6) presence of a known medical condition or currently taking medication that could alter fluid balance; 7) history of exertional heatstroke or exercise-heat intolerance; and 8) self-reported use of anabolic or catabolic hormonal substances including testosterone, anabolic steroids, growth hormone, or cortisone.
The methods and procedures of this study were reviewed and approved by the institutional review board of the Uniformed Services University before recruitment of subjects. Potential subjects attended a mandatory information session where the study’s risks and benefits were carefully explained and questions were answered by the investigators. All subjects provided written informed consent after completing the information session. Each subject also completed a medical history form that was reviewed by the event’s medical director.
Anthropometrics, Monitoring Gastrointestinal Temperature, and Environmental Conditions
Anthropometric measurements were completed 1 to 2 days before the event at the Wichita Falls Convention Center, using procedures previously described. 24 Briefly, hip and waist circumference were measured in triplicate using a Guilck tape measure (Perform Better, Cranston, RI). Body fat percentage (%BF) was determined from duplicate skinfolds, calibrated skinfold calipers (Bodycare Harpenden caliper, Southam, United Kingdom), and an appropriate 3-site skinfold equation. 25 A trained investigator performed all measurements. Height was measured with the subject standing against a tape measure attached to the wall, and body mass (Health o meter model 349KLX; Pelstar LLC, Bridgeview, IL) was measured using a floor scale on a level platform, accurate to ±0.1 kg, while subjects wore only bike shirt and bike shorts. To ensure residual sweat was limited in IP measurement, participants were asked to towel down after the event. Body mass index was calculated as body mass (kg) / height (m2). Additionally, body mass was measured in the medical tent at PRE and IP.
An ingestible thermistor (CorTemp; HQ Inc, Palmetto, FL) was used to measure gastrointestinal temperature (TGI) before starting and after completing the event. The thermistor was swallowed by each subject around 2100 hours on the night before the event. Ingestion was verified by a telephone call from an investigator. The TGI was measured with a CorTemp handheld digital thermometer-receiver positioned near the posterior lumbar curve at PRE and IP. TGI was obtained from only 24 subjects because some ingestible thermistors could not be detected.
Ride Characteristics
The 2015 164-km HHH course covers a largely flat terrain that starts and ends in Wichita Falls, Texas. 5 The event began on August 29, 2015 at 0700 hours. Environmental conditions were monitored near the start/finish lines of the event using a Kestrel 4400 Heat Meter (Nielson-Kellerman Co, Boothwyn, PA). Dry bulb temperature, wet-bulb globe temperature, humidity, and wind speed were monitored hourly throughout the event, as previously described. 24
Urine and Blood Collections
Whole blood (approximately 20 mL) and urine samples were obtained before the start of the ride (PRE; 0430–0630 h; August 29, 2015) and immediately postride (IP; within 10 min of subject finishing the ride). Hydration biomarkers included urine-specific gravity by urine refractometry (Model Reichert TS400, Buffalo, NY; urine-specific gravity of <1.025 g·mL−1 was considered euhydrated) and urine color with an 8-point color chart (greater than or equal to 4 was considered dehydrated). 26
Blood was collected from an antecubital vein without stasis (21-gauge needle into evacuated ethylenediaminetetraacetic acid, citrate-theophylline-adenosine-dipyridamole, and sodium citrate vacutainers), while subjects were in a seated position. Subjects were not fasting for the blood draw. Filled vacutainers were placed on ice or in a 4°C refrigerator. Citrate-theophylline-adenosine-dipyridamole and sodium citrate vacutainers were centrifuged at 2000g for 30 minutes, within 40 minutes of blood collection, and the resultant platelet-poor plasma was separated into several individual cryovials and flash-frozen in liquid nitrogen. The frozen cryovials were stored on dry ice and shipped to the Uniformed Services University/Consortium for Health and Military Performance. Upon arrival, the specimens were stored at −80°C in an ultra-low temperature freezer until biochemical analysis. Ethylenediaminetetraacetic acid-whole blood was kept on ice or in a 4°C refrigerator until analysis was conducted at the University of North Texas.
Biochemical Analyses
PCs were measured from ethylenediaminetetraacetic acid-whole blood using an automated clinical analyzer (BC-3200 Auto Hematology Analyzer, Mahwah, NJ). Citrate-theophylline-adenosine-dipyridamole plasma samples were analyzed in duplicate for values PF4 (ThermoFisher Scientific; Frederick, MD) and β-TG (Abbexa Ltd; Cambridge, United Kingdom) via Enzyme Linked Immunosorbent Assays (ELISA). The intra-assay coefficients of variation (CVs) were 7.6 and 4.2%; interassay CVs were 9.1 and 12.8%, respectively. Platelet-poor sodium citrate plasma samples were used to analyze TAT (Assaypro LLC, St. Charles, MO) and thrombomodulin (TM) (Boster Biological Technology, Pleasanton, CA) in duplicate by (ELISA), yielding intra-assay CVs of 2.8 and 2.7%, and interassay CVs of 12.6 and 8.7%, respectively. D-Dimer and von Wildebrand antigen (vWF:Ag) were determined from platelet-poor sodium citrate plasma (Diagnostica Stago Inc, Mount Olive, NJ) by ELISA, with intra-assay CVs of 3.0 and 3.4% and interassay CVs of 4.1 and 8.9%, respectively. For all ELISAs, absorbance was measured using a BIO-RAD iMark microplate reader (Hercules, CA) at the appropriate wavelength for that particular assay.
Statistical Analyses
Tests for normality of distribution (Kolmogorov-Smirnov χ2 test) and homogeneity of variance (Levene’s test) were performed for all data sets (SPSS version 20.0). When data failed this test, appropriate log transformation procedures were applied and data were retested. Dependent-samples t tests were conducted to examine blood hemostatic marker changes from PRE to IP. nQuery Advisor software (Statistical Solutions, Saugus, MA) was used to determine statistical power for the dependent variable (platelets, PF4, β-TG, vWF:Ag, TAT, TM, and D-Dimer), which ranged from 0.78 to 0.84. Cohen’s d was calculated and used to estimate effect size in order to indicate mean change in standard deviation units of the change values. Next, associations between subject characteristics (ie, anthropometrics and event completion time) and IP blood hemostatic concentrations at IP were examined. Since urine color is an ordinal variable, nonparametric analyses were used to examine urine color (Wilcoxon signed-ranks test for changes over time, Spearman’s rank order correlation for relations with blood markers). Partial correlations were analyzed between subject characteristics and IP blood hemostatic marker levels while controlling for PRE blood hemostatic marker concentration. Simple PRE-IP difference scores were not used in order to minimize assumptions required for analysis. 27 Sex differences in subject characteristics and blood hemostatic markers were also explored. The level of significance was set at P < .05. Data are presented as mean±SD unless otherwise indicated.
Results
Thirty-seven subjects (28 men and 9 women) completed the event in less than 9 hours with a mean finishing time of 6.63 hours (fastest: 4.65 h; slowest: 8.89 h). Their personal characteristics are provided in Table 2. Body mass decreased slightly, but significantly from 79.8 kg at PRE to 78.7 kg at IP (P < .001). Compared with male subjects, female subjects were significantly younger (d = 1.0; P < .05; male subjects = 53.9 y, female subjects = 45.6 y) and had a higher %BF (d = 2.5,=; P < .001; male subjects = 19.1%, female subjects = 29.6%) and slower event times (d = 0.8; P < .05; male subjects = 6.42 h, female subjects = 7.29 h). During the event, PRE and IP urine color, USG, and TGI were measured; however, only TGI changed significantly (P = .013) from PRE to IP, which are shown in Table 1. 24 Environmental conditions on August 29, 2015 were recorded throughout the day at the medical tent. The dry bulb temperature ranged from 26.1 to 39.3°C, while the wet bulb globe temperature ranged from 22.3 to 34.6°C (Figure 1). Both dry bulb and wet bulb globe temperature peaked at 1420 hours. Percent relative humidity was 56.5% at 0800 hours and fell to 26.8% by 1300 hours (Figure 1). Wind speed ranged from 0 to 6.1 km/h. During the event, partial cloud cover was observed. 24
Selected characteristics of cyclists
BMI, body mass index; W/H ratio, waist-to-hip ratio. 24
Values for male subjects, female subjects, and total number of participants are mean±SD.
Subject urine and internal temperature characteristics 24
PRE, preride; IP, immediate postride; M, males; F, females; USG urine specific gravity; TGI, gastrointestinal temperature.
Values are mean and SD except for urine color, which is based on 8-point color rating system, with colors that are equal and darker than the number 4 indicating dehydration. 26
Inside parentheses (average of male subjects/female subjects).
n=33 (male subjects = 26; female subjects = 7) for USG and urine color; n=24 (male subjects = 19; females = 5) for gastrointestinal temperature.
Significantly different from PRE (P < .05).
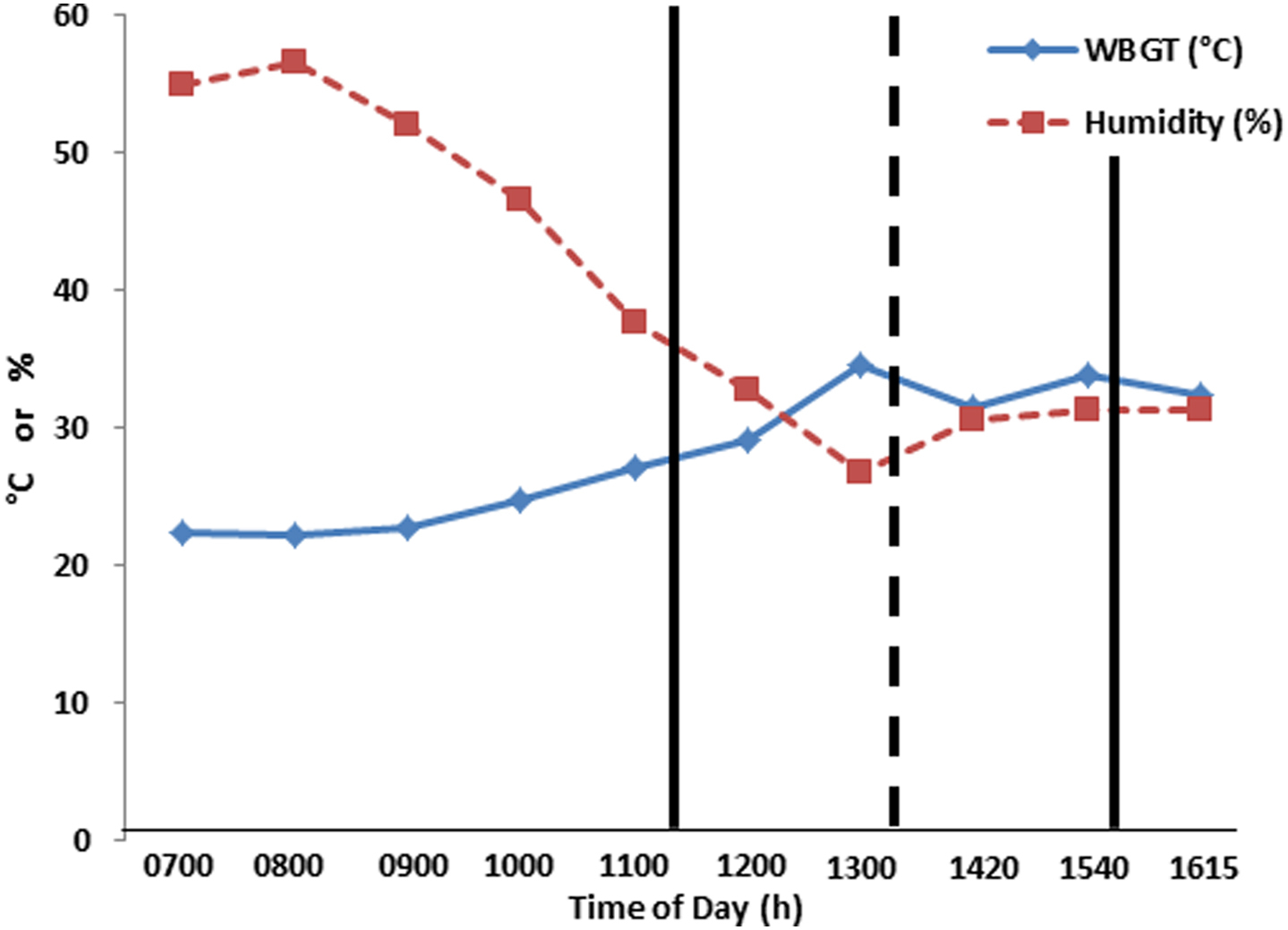
Wet bulb globe temperature (WBGT) and relative humidity measured at the first aid station. Straight bold lines represent fastest and slowest participant. Dashed bold line represents average finishing time of participants.
The concentrations of platelet, platelet activation, coagulation, and fibrinolytic markers all increased significantly (P < .001) from PRE to IP (Table 3). The platelet markers, PC, and vWF:Ag ratio increased by 29.1% and 96.1%, while the platelet activation markers PF4 and β-TG increased by 66.3% and 50.8%, respectively. The individual responses for PF4 and β-TG are presented in Figures 2A and B. Additionally, the coagulation and fibrinolytic markers TAT, TM, and D-Dimer increased by 15.4%, 8.9%, and 37.7%, respectively.
Platelet, coagulation, and fibrinolytic markers pre- and immediate postride
PRE, preride; IP, immediate postride; PF4, platelet factor 4; β-TG, β-thromboglobulin; vWF:Ag, von Wildebrand factor antigen; TAT, thrombin antithrombin complex; TM, thromobomodulin.
Values are mean±SD.
Significantly different from PRE (P < .001).
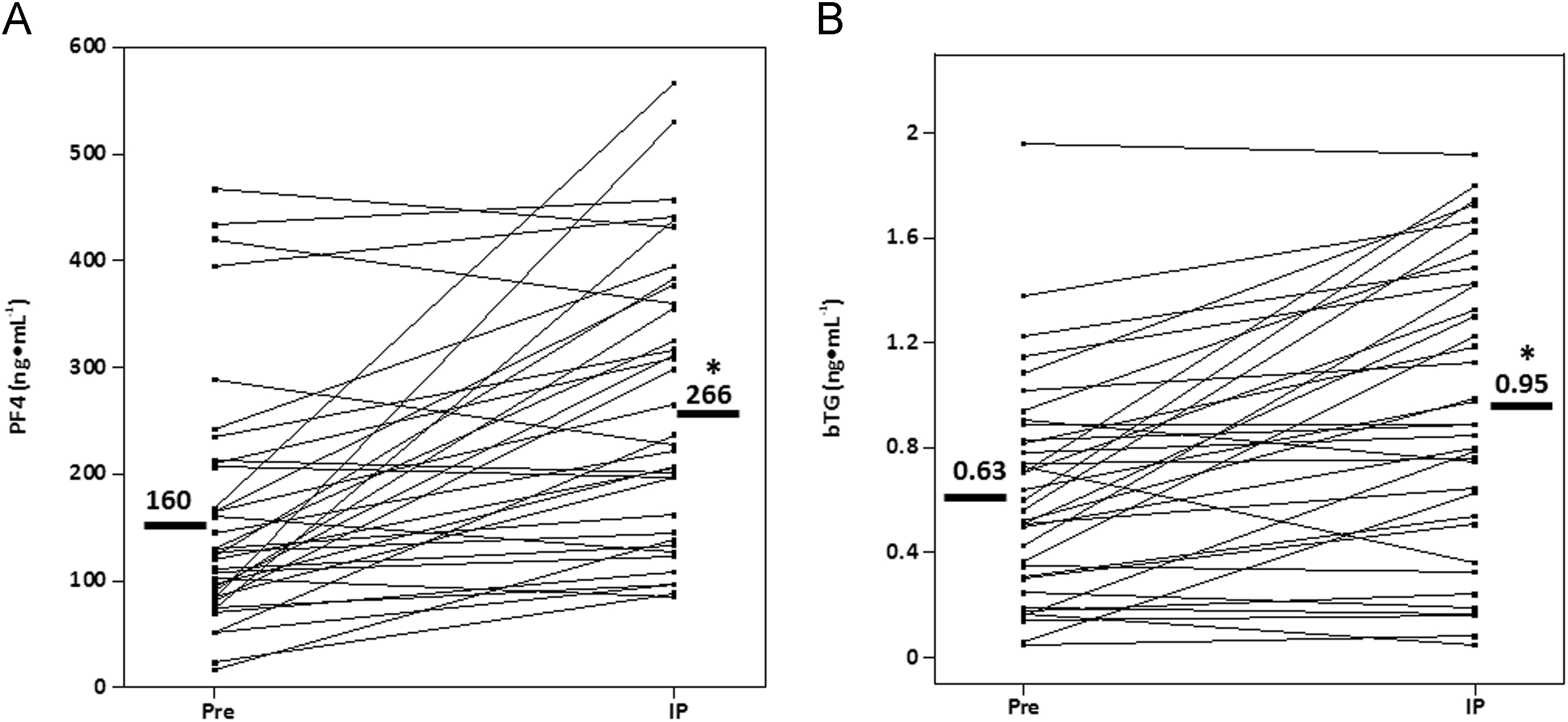
Individual PF4 (A) and β-TG (B) responses to 164-km of cycling. Straight bold line represents participants’ average. PRE, preride; IP, immediate postride; PF4, platelet factor 4; β-TG, β-thromboglobulin. *, significantly different from PRE (P < .001).
Correlations between PRE blood hemostatic marker concentrations and subject characteristics revealed no significant relationships except for the waist to hip (W/H) ratio, which was positively related to PF4 (r = 0.40; P < .05) (Figure 3).
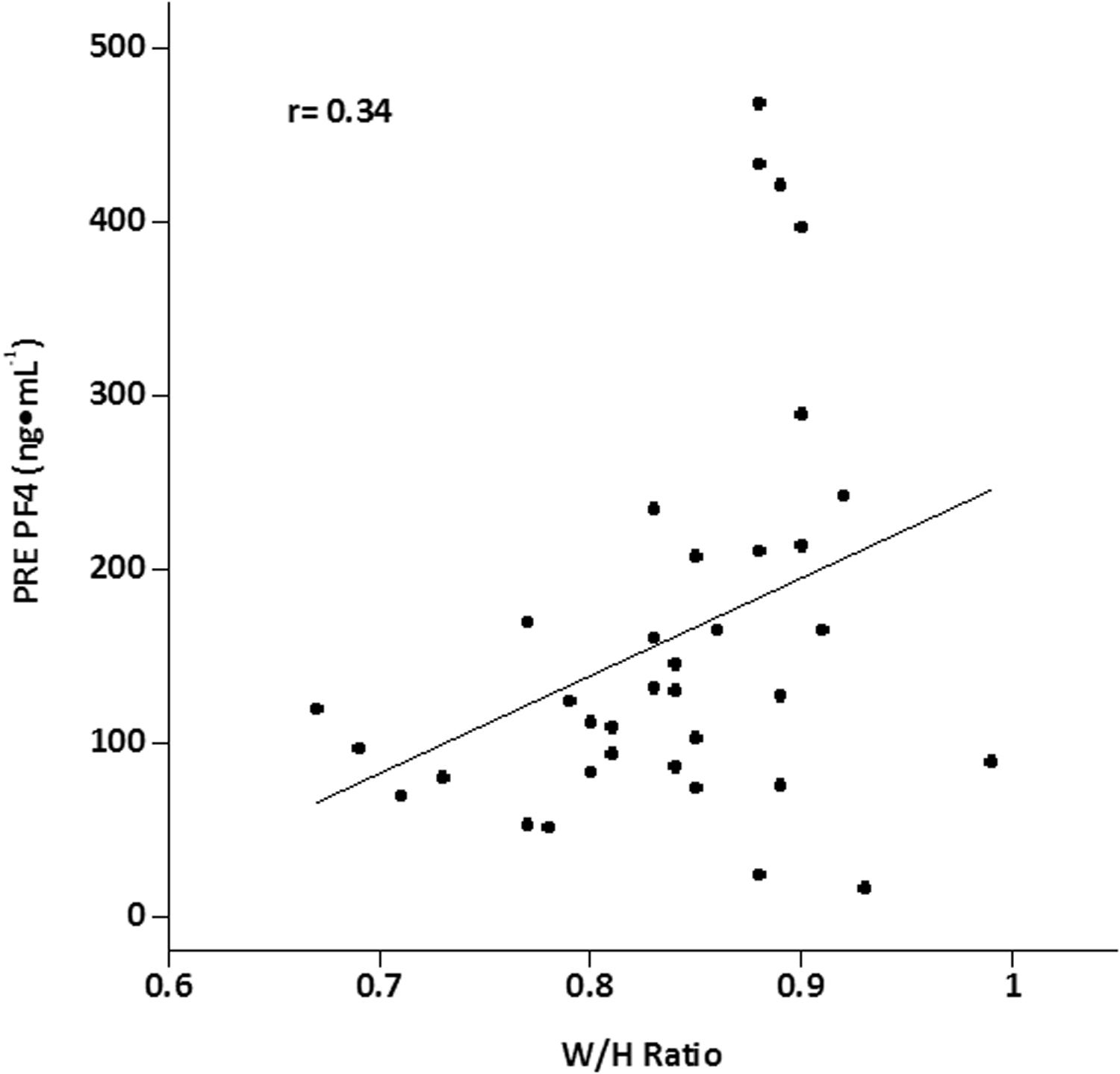
Relationships between baseline PF4 and waist-to-hip ratio.
Subject characteristics and IP blood hemostatic marker concentrations were analyzed for partial correlations, controlling for PRE blood hemostatic concentrations. Finishing time was negatively related to PF4 (r = 0.34; P = .017) (Figure 4), while %BF was negatively related to TAT (r = –0.35; P = .038) and to TM (r = –0.36; P = .036). No relations were found between blood hemostatic markers and IP USG or urine color. PC was negatively related to TGI (r = –0.50). Furthermore, female subjects had lower IP concentrations of TAT (d = 0.63; P < .05) and natural logarithm thrombomodulin (InTM) (d = 6.42; P < .05) than male subjects.
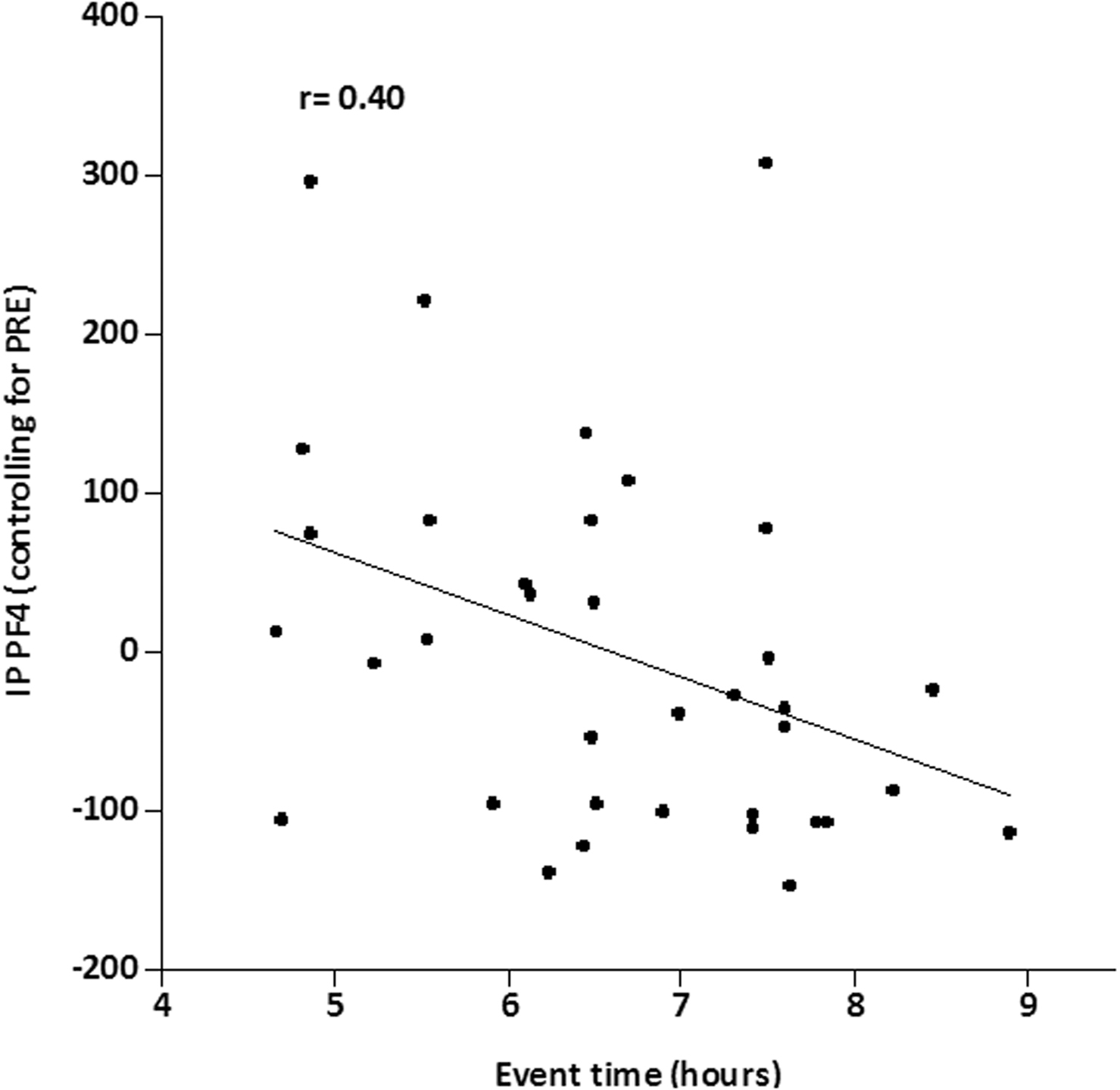
Relationship between IP PF4 (controlling for PRE) and event time.
Discussion
Over the past decade, numerous studies have investigated the effects of acute exercise on blood homeostasis in both laboratory and field settings. To our knowledge, no investigation has examined markers of platelet activation in response to long-duration endurance exercise in a hot environment, and only 1 study examined coagulation and fibrinolytic markers in response to such conditions. 5 However, that study had a small sample size (n=16) and did not include women. Additionally, unlike previous events the present study investigated platelet activation, coagulation, and fibrinolytic responses to a 164-km recreational road cycling event held during the hot Texas summer utilizing a larger sample of men and women. Our results yield novel physiological insights with potential health implications.
Subjects who completed the cycling event exhibited increased concentrations of platelets, platelet activity (PF4 and β-TG), coagulation (TAT), and fibrinolytic markers (D-Dimer). Additionally, a positive relationship between PF4 concentrations and W/H ratio was observed, indicating a greater predisposition for platelet activity in those with greater W/H ratio during prolonged cycling in the heat.
Even though the risk of cardiovascular events is significantly decreased in individuals who partake in habitual physical activity compared with sedentary individuals, the majority of venous thrombotic embolisms due to exercise occur in individuals over 35 years of age as a result of platelet-rich thrombi.28,29 Thus, prolonged endurance exercise increases the risk of sudden cardiac arrest in older individuals. 30 Nonetheless, we did not find a relationship between age and any markers of blood homeostasis, even though 34 out of the 37 subjects were over the age of 35 years (range, 26–65 y). This finding may be explained as follows: a) completing this event requires significant prior endurance training, and b) our inclusion criteria required that study subjects had previously completed a prolonged endurance ride and be apparently healthy. Thus, although the cardiovascular health and fitness of our subjects were not measured in the current study, both are likely to be much greater in individuals who complete the HHH than the age-matched general population. Moreover, moderate-intensity exercise training reduces platelet reactivity and enhances fibrinolysis during rest. In addition, this type of training enhances platelet reactivity and augments hyperfibrinolytic activity during strenuous exercise as seen during this event. 31 Prolonged strenuous exercise in mild ambient conditions has been documented to increase PC by 12 to 24%. 16 ,21,32 This phenomenon occurs as platelets are released from the spleen, bone marrow, and lungs.33,34 Heat stress has been documented to increase PC independently of exercise by 7.5 to 34%. 11 ,12,15 In the current study (2015 HHH), PC increased by 29%. This large increase is similar to, although slightly smaller than, our findings of a 39% increase in PC for the 2013 HHH event, which was held under similar environmental conditions as the current study. 5 In combination, these results indicate that prolonged road cycling in the heat induces a substantial increase of PC.
Platelet activity was measured by examining the platelet-specific proteins PF4 and β-TG, which are released from α-granules during platelet activation.35,36 Even though these proteins are similar in structure, they exhibit different functional properties, 35 and compared with β-TG, PF4 has a higher affinity to heparin and is rapidly cleared from the circulation. Previous research has provided conflicting results for platelet function; this discrepancy in findings might, in part, be due to the difficulty of accurately measuring these proteins, since artificial elevation can occur in vitro during sample collection, handling, and processing. 36 Therefore, in order to distinguish between in vitro and in vivo secretion, we measured PF4 and β-TG in tandem. 37
In general, studies have produced conflicting results regarding the effects of endurance exercise on concentrations of PF4 and β-TG, with both increases 20 ,21,38,39 and no changes reported for these proteins. 40 –42 However, this divergence in findings could be the due to the type, intensity, and/or duration of exercise used. 43 PF4 response to aerobic exercise increases in older individuals and individuals with cardiovascular disease (CVD).44,45 In our study, we also determined that age was significantly correlated with baseline PF4, which suggests that our subject sample of cyclists may be at risk for heart attack, stroke, or venous thrombosis.
Even though heat stress has been shown to independently affect blood homeostasis, 11 ,13,14 only a few studies have examined the effect of heat stress on markers of platelet activation. In those studies, passive hyperthermic conditions resulted in an increase of both PF4 (55–150%) and β-TG (23–52%).13,46 In the present study, we also measured an increase of PF4 and β-TG (66% and 51%, respectively), which is comparable to previous studies that involved heat stress in the absence of exercise. However, we found no relationship between TGI and PF4 or β-TG (change from baseline), suggesting that the combination of prolonged endurance exercise and heat stress did not have an additive effect on platelet activation markers in our study. Additionally, we determined that finishing time was negatively related to PF4, thus riding intensity, not ride duration, of the event may be increasing PF4 expression.
We also examined vWF:Ag, a glycoprotein released from endothelium cells and platelets that has a role in platelet adhesion. 47 Previous research has demonstrated that rigorous exercise48,49 and heat stress10,50 independently increased vWF:Ag concentrations in the circulation, thereby increasing the risk of clotting. In the present study, vWF:Ag increased 96% during the event; this increase is slightly higher than the response found during a previous (2013) HHH event (65%). 5 Mechanistically, the release of vWF:Ag can occur due to any of several different stimuli, including sheer stress, epinephrine, thrombin, and fibrin.51,52
Because TAT is a sensitive marker of coagulation and is formed by the neutralization of thrombin by antithrombin III (AT), it has been used as a marker of thrombin generation. TAT reportedly forms during prolonged endurance exercise4,53 and during hyperthermia. 11 Mechanistically, thrombin formation occurs with strenuous exercise via platelet activation, aerobic metabolism with lactate formation, and/or muscle damage. 53 –55 Since cycling is a low-impact activity, cycling exercise theoretically produces less thrombin formation than a running event. 4
In an investigation of a prior (2013) HHH event, we observed an increased formation of thrombin (+86% from pre- to postevent), as measured by thrombin-antithrombin complexes. 5 The current study subjects (2015 HHH) likewise experienced a significant but smaller increase of thrombin-antithrombin complexes (+15.4%). This difference might in part result from 2 factors. First, baseline concentrations of TAT were greater for the 2015 HHH (7.65 μg.mL) than for the 2013 HHH (2.89 μg.mL). 5 Second, the current study (2015 HHH) examined subjects who completed the event in less than 9 hours, whereas the previous study (2013 HHH) examined only subjects who completed the event in less than 6 hours; thus, the greater intensity of the ride in the previous study could have induced a greater increase in TAT.
TM is a cell membrane integral glycoprotein expressed by endothelium cells that has dual roles in the coagulation and fibrinolytic systems. It acts as a procoagulant by activating thrombin-activatable fibrinolysis inhibitor, 56 and it acts in the fibrinolytic system by converting thrombin from a procoagulant enzyme to an anticoagulant enzyme, which in turn elevates the activity of protein C. 57 TM is stimulated by endothelium cell damage through hydrogen peroxides and stimulated leukocytes. 58 Although elevated levels of TM have been observed after running events,59,60 swimming failed to elicit a TM response. 59 Thus, elevation of plasma TM is thought to occur via mechanical stress from ground impact forces and weight bearing. The effect of passive heat stress after bathing in hot spring baths 61 resulted in no increase of circulating TM concentration. In the current study, we found a slight but significant increase of TM (9%) during cycling in the heat. Although cycling involves no ground impact forces, our results suggest that long-duration cycling exercise in the heat is sufficient to cause an elevated release of TM.
Fibrinolytic activity is typically increased during exercise, counteracting a hypercoagulable state. We investigated in vivo fibrinolytic activation by measuring D-Dimer concentrations, which are split fibrin products. D-Dimer concentrations are increased both during intense endurance training4,12 and in resistance training. 62 D-Dimers can also be formed in response to external heat stress, 14 , or by wearing an insulated suit that raises the body’s internal temperature. 15 In a previous field study, 5 we found a 38% increase in D-Dimer concentration during the 2015 event, compared with a 54% increase during the 2013 event. Since the 2015 study included women and a greater number of subjects overall, its results may be more representative of the D-Dimer response to the HHH event.
The baseline expression of PF4 from platelets in our study was positively correlated with the W/H ratio. Therefore, individuals with greater waist circumference who participate in prolonged endurance cycling events in extreme conditions may be at greater risk of clot formation, deep-vein thrombosis, or pulmonary embolism. However, in our study, concentrations of TAT and lnTM were negatively correlated with %BF. This suggests that the cyclists in our study who exhibited lower %BF, as a measure of overall obesity, are more sensitive to forming thrombin and releasing TM than those who have higher levels of body fat. 63
The present study had several limitations. First, the study’s participant size is relatively small, which can mean that statistically significant differences were not detected (type 2 statistical error) and presents the usual difficulties in comparing small studies. Second, we examined only IP results. Future research should track the same measures through 1 day postride to follow the effects of participation through the end of the normal recovery period. Third, the changes over the event clearly demonstrate that this was a recreational ride in that core temperature was minimally increased. Last, since we did not collect data on perceived or actual intensity of the event (power, heart rate, rate of perceived exertion, etc.), we did not determine how hard each participant actually cycled. However, the average rider rode at 21 km/h, which represents a relatively low exercise intensity given the nearly flat course, but in relatively hot, dry conditions (range: wet bulb globe temperature, 22.3–33.9°C; humidity, 26.8–56.5°C).
In summary, completing a 164-km road cycling HHH event in a hot environment altered blood homeostasis in both men and women, as evidenced by increased postexercise concentrations in markers of platelet activation, coagulation, and fibrinolysis. Furthermore, we found a relationship between PF4 (controlling for prerace levels) and event completion time. However, female subjects in our study demonstrated lower IP concentrations of TAT and InTM than men; this suggests that men participating in the HHH may be at a greater risk for having thrombolytic event. In conclusion, the HHH event produced a hypercoaguable state; however, the fibrinolytic system also was upregulated, appearing to balance blood hemostasis and may have prevented clot formation during exercise in a hot environment.
Acknowledgments: The authors gratefully acknowledge the HHH Expo committee for the use of facilities in the Wichita Falls Multi-Purpose Event Center. Additionally, we would ike to thank Tufts University, Jean Mayer USDA Human Nutrition Research Center on Aging for help in the analyses of the biomarkers.
Author Contributions: Study concept and design (BRK, JLV, ECL, and LEA); acquisition of the data (BRK, DEL, JLV, ECL); analysis of the data (BRK, JBK, JLV, and DEL); drafting of the manuscript (BRK, JLV, ECL, LEA, JBK); critical revision of the manuscript (BRK, JBK, JLV, ECL, LEA, and PAD); medical monitoring of the participants (KHW). All authors read and approved the final manuscript.
Financial/Material Support: External funds were provided by the Center Alliance for Nutrition and Dietary Supplementation. Internal funds were also provided by the University of North Texas and University of Connecticut.
Disclosures: None. The opinions and assertions expressed herein are those of the authors and should not be construed as reflecting those of the Uniformed Services University, Department of the Army, Department of the Air Force, Department of the Navy or the United States Department of Defense.
