Abstract
Discovered in 1947 in a monkey in the Zika forest of Uganda, Zika virus was dismissed as a cause of a mild illness that was confined to Africa and Southeast Asia and transmitted by
Four unexpected mosquito-borne arboviruses immigrated to the Americas from the tropics within 3 decades. 1 First, dengue slipped into the Americas from Southeast Asia (1980s) and is now established on the Mexican border. Then West Nile virus arrived from Africa and the Middle East (1999) and quickly crossed the continental United States. Recently, chikungunya arrived from the Caribbean via East Africa and India (2013), and Zika virus invaded the Americas from the South Pacific (2015). 1 Although all of the mechanisms responsible for these viral migrations cannot be explained, the global movements of these tropical viruses have been facilitated by international air travel capable of transporting virus-infected humans from endemic regions to anywhere in a warming world within 24 hours.
The Epidemiology of Zikavirus Disease
First discovered in 1947 in a rhesus monkey in the Zika forest of Uganda, Zika virus was dismissed as a cause of a periodic, mild febrile illness with rash and conjunctivitis confined to Africa and Southeast Asia. 2 Decades later, Zika erupted outside of its endemic borders on the South Pacific island of Yap in 2007. 3 The Yap outbreak was once again characterized by uncomplicated, short-term febrile illnesses. 3 However, by 2013, Zika virus was first associated with a major neurological complication, Guillain-Barré syndrome (GBS), an ascending flaccid paralysis, with over 40 cases reported in a larger outbreak in the French Polynesian Islands. 4 From the French Polynesian Islands, Zika moved to Easter Island and then on to the Americas, invading Brazil in 2015. 5
Clinical and neuroimaging studies have demonstrated that Zika’s neurological complications represent a spectrum of central nervous system disorders with GBS linked to myelitis and meningoencephalitis, and microcephaly associated with severe brain, optic nerve, and chorioretinal damage. The Zika virus has now caused over 4000 cases of congenital microcephaly, a tragic birth defect characterized by a small, misshapen head with severe brain and ocular malformations, in Brazil. 5 Zika virus has been detected in the amniotic fluid and placentas of infected mothers and in the brains of microcephalic stillbirths and neonates. 5 In just a short period of time (2007–2013), the Zika virus has gained the capability to cause not only asymptomatic (80%) or mild (20%) illnesses in most people, but also severe neurological complications in adults and infants.
In an unprecedented method of arthropod-borne disease transmission, the sexual transmission of the Zika virus from males and females to their sex partners was confirmed serologically by rising immunoglobulin M Zika antibody titers and molecularly by viral RNA detection by reverse transcriptase-polymerase chain reaction.6,7 In addition to local mosquito-transmitted infections, the spread of Zika virus disease will be accelerated by sexual transmission, blood and body fluid contact, and organ transplantation. Such combined mechanisms of infectious disease transmission could result in catastrophic incidences of severe neurological diseases in adults and children.
Since 2015, over 3000 Americans have contracted Zika virus disease while travelling in Zika-endemic regions. Today, the mosquito-borne, local transmission of Zika virus disease occurs as close to the US mainland as its Caribbean territory, Puerto Rico, which is experiencing a $70 billion debt crisis and a weakened public health infrastructure. Puerto Rico has reported over 17,000 mosquito-transmitted cases of Zika virus disease, with over 800 in pregnant women, 1 fatal case of GBS, and 1 neonatal fatality with microcephaly.
Although travel-related or imported Zika virus disease is still the predominant mode of disease transmission in the United States, local mosquito transmission is the predominant mode of disease transmission in the Caribbean and throughout Latin America. The local mosquito transmission of Zika virus disease has now been reported in Florida and is anticipated to occur in additional states shortly as more infected people arrive in the United States from hyperendemic nations in the Caribbean and Latin America.
As the numbers of imported and mosquito-borne cases of Zika virus disease are increasing over time in the United States and its territories, interested readers are encouraged to check the updates on Zika cases and complications at publication time and periodically on the US Centers for Disease Control and Prevention website available at
The Mosquito Vectors of Zika Virus and Their Biting Behaviors
Three flaviviruses, yellow fever, dengue, and Zika, and 1 alphavirus, chikungunya, are all transmitted to humans by female
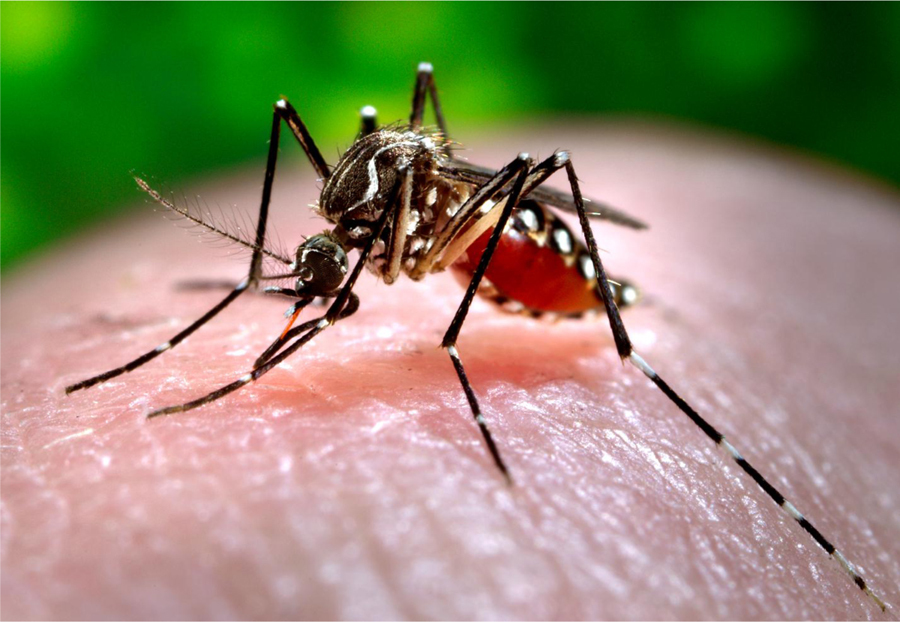
A female

A female
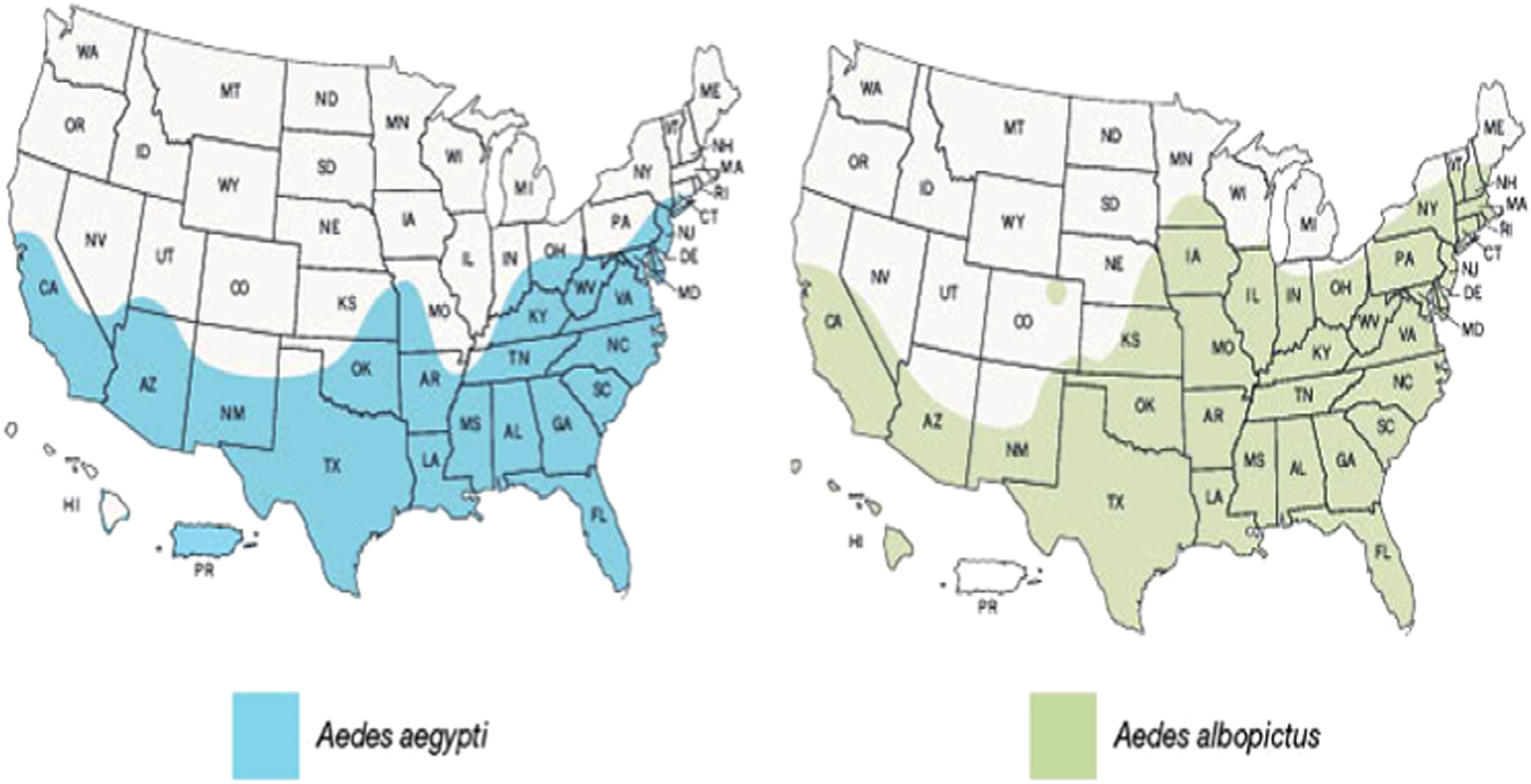
The regional geographic distribution ranges of
Since
The distribution and distinguishing features of
Outdoor enthusiasts are cautioned that
Control and Prevention Strategies for Zika Virus Disease
Although virologists and vaccinologists have learned much more about the immunological mechanisms of flaviviral disease transmission from recent experiences with investigational West Nile and dengue vaccines, a Zika virus vaccine is far away, especially with federal research funding stalled in Congress. In addition, a Zika vaccine that is rushed from production into mass administration before human trial testing could itself precipitate GBS or be inactivated by cross-reacting antibodies from prior flaviviral experiences with dengue, West Nile virus, or even the yellow fever vaccine.
9
Therefore, the only effective national strategies to immediately prepare the United States for the Zika virus invasion are vector control measures and personal protection from mosquito bites. In addition to a basic knowledge of the
Mosquito Vector Control Strategies
Mosquito vector control strategies may be classified as physical, chemical, biological, and genetic. The most effective combinations of vector control strategies should be well matched to the habitat, egg-laying preferences, and the biting behaviors of the targeted mosquito vectors. Although state and local governments typically provide for regional chemical, biological, and genetic vector control programs, every citizen has a personal responsibility for implementing simple physical vector control strategies, such as removing yard debris, unblocking clogged drains and roofing gutters, and emptying or covering all potential freshwater containers. Table 2 describes the 4 types of mosquito control strategies and their most effective applications in vector control programs targeted at
Vector control strategies and their individual utilities in
Personal Protective Strategies to Prevent Mosquito Bites
In addition to some simple physical control strategies, such as window screens and air-conditioned households, personal protective strategies to prevent mosquito bites include wearing light-colored, long-sleeved shirts and long pants and applying effective mosquito repellents throughout the day. Mosquito repellents may be divided into two basic chemical classes: 1) synthetic chemicals, such as DEET, picaridin, and IR3535 (Avon Skin So Soft); and 2) plant-derived oils and synthetics, such as oil of lemon eucalyptus, oil of citronella, and permethrin. 10 Table 3 describes the range of mosquito repellents and insecticides available worldwide as stratified by their active ingredients, formulations, strengths (%), efficacies against mosquitoes, precautions, and adverse effects. 10 Although recommendations to combine clothing impregnated with insecticides, such as permethrin, with mosquito repellents, such as DEET or picaridin, are not supported by randomized controlled trials, the most effective uses of mosquito repellents are to apply a topical repellent on exposed skin and to wear permethrin- or other synthetic pyrethroid-impregnated clothes that act as contact insecticides and provide better and longer lasting protection against mosquito bites. 10
Available mosquito repellents: formulations, efficacy, safety, and toxicity 10
Protective Efficacy Scale: 0, no protection provided; +, minimal level of protection; ++, moderate level of protection; +++, maximal level of protection.
FDA Pregnancy Categories: A, human studies have demonstrated no evidence of risk to the fetus; B, animal studies have demonstrated no evidence of risk to the fetus; C, animal studies have demonstrated adverse effects on the fetus; D, investigational or marketing experiences or human studies have demonstrated adverse effects on the fetus, but potential benefits may warrant use of the drug in pregnancy despite the risks; X, studies in animals or humans have demonstrated fetal abnormalities; N, FDA has not classified the drug.
Conclusions: What can we expect? What can we do? What preventive measures will work or fail?
The local mosquito-borne transmission of Zika virus disease in the United States is inevitable as more viremic persons arrive from Zika-endemic areas during the summer Atlantic coastal mosquito-breeding and tropical storm seasons. Although aerial insecticide spraying of freshwater wetlands can control West Nile virus–transmitting mosquitoes, it is less effective for
Mosquito-borne transmission will be magnified by sexual transmission from asymptomatic males to their sex partners as Zika remains capable of replication for months in semen. Protective measures will include not only physical and chemical vector control (Table 2) and personal protection with pyrethroid-impregnated clothing and topical insect repellents (Table 3), but also proper condom use for prolonged periods during sex (throughout pregnancy and up to 9 months) by males who have had Zika virus disease or visited Zika-endemic areas. The defensive lessons learned now and combined with ongoing research in flaviviral immunology and genetic mosquito vector control will better prepare us for the next arthropod-borne pandemic in our changing world ecosystem.
Financial/Material Support: Support provided by departmental and institutional sources.
Disclosures: None.
Footnotes
Submitted for publication May 2016.
Accepted for publication July 2016.
