Abstract
Only limited clinical scenarios are grounds for induction of therapeutic hypothermia. Its use in traumatic cardiac arrests, including those from lightning strikes, is not well studied. Nonshockable cardiac arrest rhythms have only recently been included in resuscitation guidelines. We report a case of full neurological recovery with therapeutic hypothermia after a lightning-induced pulseless electrical activity cardiac arrest in an 18-year-old woman. We also review the important pathophysiology of lightning-induced cardiac arrest and neurologic sequelae, elaborate upon the mechanism of therapeutic hypothermia, and add case-based evidence in favor of the use of targeted temperature management in lightning-induced cardiac arrest.
Introduction
Therapeutic hypothermia was endorsed only in the past decade as a legitimate treatment for unresponsive cardiac arrest patients with return of spontaneous circulation(ROSC). Definitive research has detailed neurologic benefits, and usage has expanded in recent years. These narrowly defined studies have not attempted to provide insight into its use in many clinical scenarios, including cardiac arrest after lightning strike. This lack of substantive clinical evidence to either support or refute the benefits of therapeutic hypothermia in lighting-induced cardiac arrest has resulted in a lack of guidance in the literature. To this end, we report a fourth unique instance of successful use of therapeutic hypothermia in the setting of lightning-induced pulseless electrical activity (PEA) cardiac arrest.
Case Presentation
An 18-year-old woman sustained an indirect lightning strike and subsequent cardiac arrest while in contact with a tree during a thunderstorm. A bystander provided immediate cardiopulmonary resuscitation (CPR) for approximately 5 minutes. Paramedics then arrived, followed advanced cardiac life support (ACLS) protocols, intubated, and achieved ROSC after a PEA of sinus tachycardia. At the local hospital, the patient experienced a second PEA arrest that again resolved with ACLS efforts. Therapeutic hypothermia was initiated using cooling blankets and ice packs, and the patient was transferred to our level 1 trauma facility.
Upon arrival, her Glasgow Coma Scale (GCS) was 3 without medication. Vital signs on arrival were a blood pressure of 133/103 mm Hg, heart rate of 99 beats/min, mechanically ventilated respiratory rate of 24 breaths/min, pulse oximetry of 100%, and end-tidal carbon dioxide of 28 mm Hg. Pupils were 3 mm and sluggishly reactive bilaterally. A Lichtenberg figure was noted across the rib cage and soot was seen overlying the sternum and posterior neck (Figures 1 and 2). Burns conferred by a necklace were also present around her neck (Figure 2). A blanchable area of erythema was discovered overlying the thoracolumbar junction. No further significant injuries were noted.
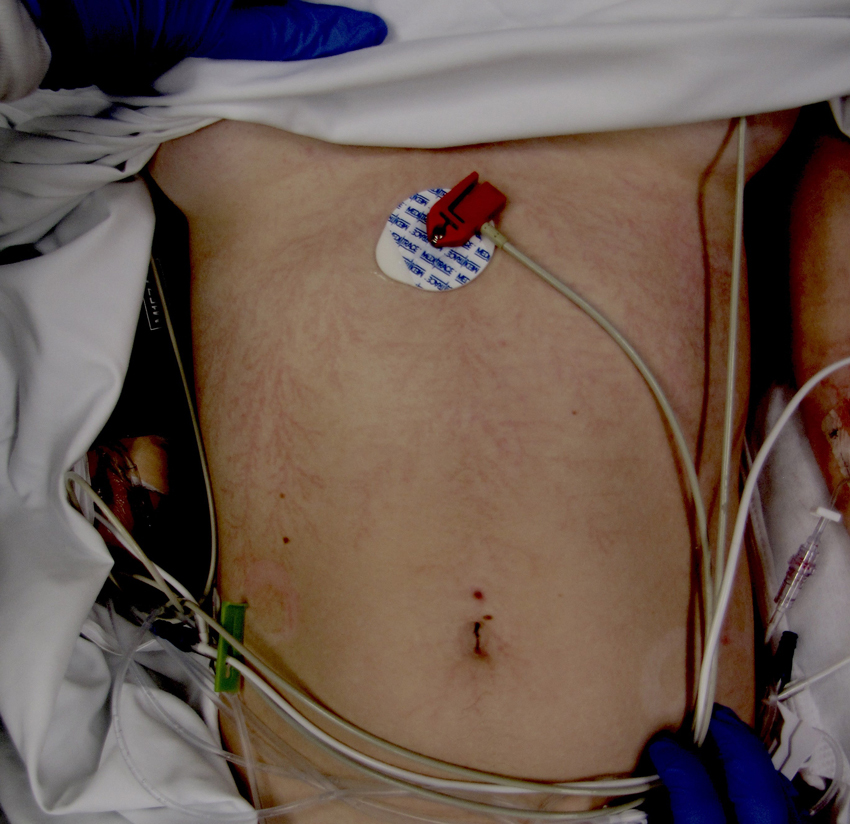
Lichtenberg figure conferred by lightning strike. Photo credit: Forensic Nurse Examiners Team at Christiana Care Health System.
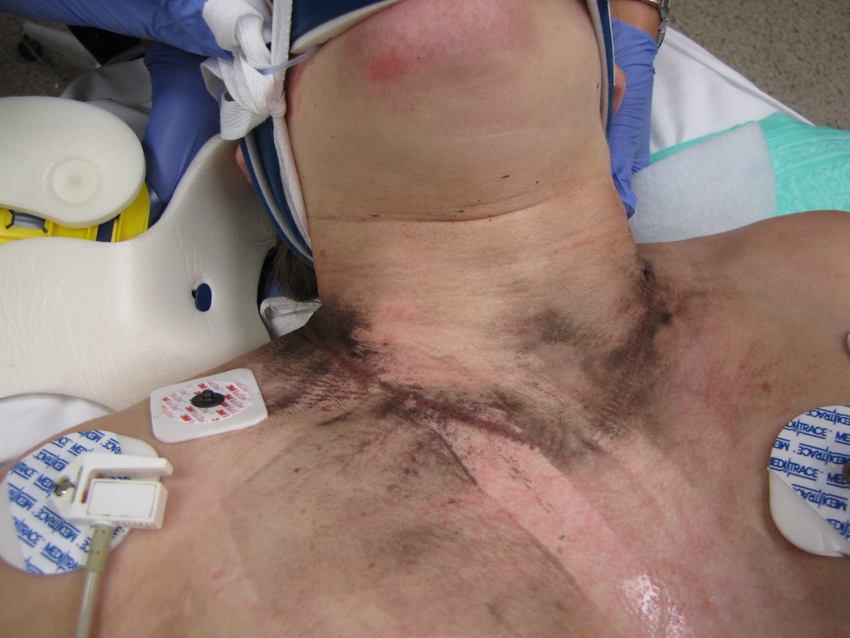
Imprint and soot from lightning strike and presence of metallic necklace. Photo credit: Forensic Nurse Examiners Team at Christiana Care Health System.
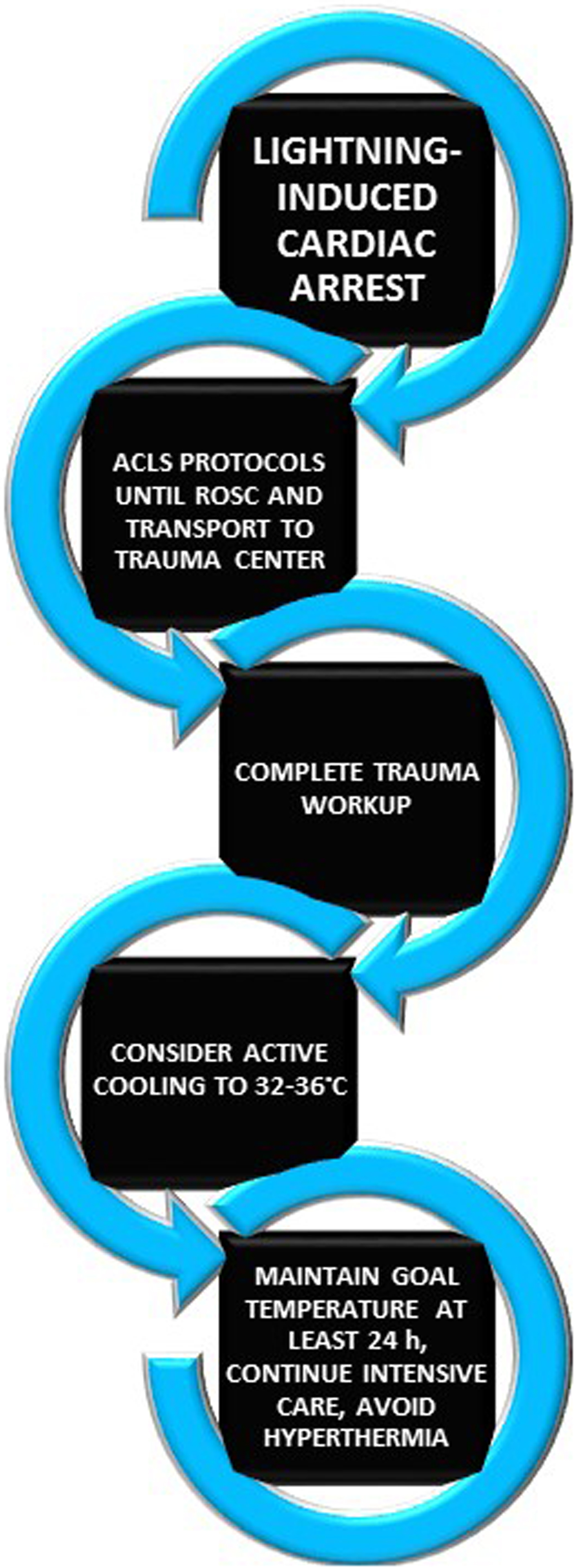
Sample simplified protocol for utilizing therapeutic hypothermia.
Within minutes after arrival to the trauma bay, she had a GCS of 8 with spontaneous eye opening, flexor activity, and decorticate posturing with no movement in the lower extremities. She was sedated with fentanyl, propofol, and cisatracurium to facilitate further imaging studies. An electrocardiogram revealed sinus tachycardia without ectopy. Chest radiograph, focused ultrasonography in trauma (FAST), and computed tomography of the head, cervical spine, chest, abdomen, and pelvis found no signs of traumatic injury.
Relevant laboratory findings included an arterial blood gas indicating a metabolic acidosis with a pH of 7.26, partial pressure of carbon dioxide of 40 mm Hg, bicarbonate level of 18.8 mEq/L, lactate of 3.9 mEq/L, creatinine kinase of 1019 IU/L, troponin of 0.17 ng/mL, and white blood cell count of 19.6 103/mm3.
The patient was admitted to the surgical intensive care unit and a triple lumen femoral Icy Catheter (ZOLL Medical Corporation, Chelmsford, MA) was placed to achieve a temperature goal of 34°C (Figure 3).
By the third hospital day, gradual 24-hour rewarming had been completed. Subsequently, she was following commands but experiencing episodes of sinus tachycardia up to 160 beats/min when stimulated. This was palliated with fentanyl and midazolam, and she was extubated without incident the same day. Shortly after she was found to be oriented to self only and failed a dysphagia screen.
On hospital day 4, she exhibited a GCS of 14 with disorientation to place and delayed word finding. She followed all commands and was able to ambulate with assistance. She was cleared for a regular diet and transferred to the pediatric general medical unit in keeping with hospital age protocol.
On hospital day 7, she was transferred to a specialty inpatient rehabilitation center. At the time of discharge, she was stable, ambulating well, and had only signs of mildly impaired balance and coordination.
Upon discharge from rehabilitation 1 month later, the patient maintained a diagnosis of mild cognitive impairment characterized by decreased attention and short-term recall of auditory and written information. At 2-year follow-up, she reported a full return to her baseline.
Discussion
ACLS recommendations were recently updated substantially in 2015. 1 These current ACLS guidelines endorse the use of targeted temperature management (TTM) with active cooling from 32°C to 36°C for at least 24 hours in comatose patients with ROSC after cardiac arrest 1 (Figure 3). This reflects both an increase in the acceptable temperature range and the inclusion of nonventricular tachycardia and ventricular fibrillation arrests. Routine use of prehospital cooling remains a subject of study but is not recommended. 1
The success of therapeutic hypothermia in cardiac arrest is based upon an ability to be neuroprotective in the setting of hypoxia. 2 Traditionally, a lack of study has prevented widespread expansion of inclusion criteria. No substantive evidence exists that additional patients do not benefit. Causes of cardiac arrest that remain a relative rarity are therefore likely to remain absent from clinical use guidelines without a concerted effort to study them. Lightning-induced cardiac arrest lends itself to this phenomenon. Only case reports detailing therapeutic hypothermia after lightning strikes exist, and they include 10- and 14-year-old boys resuscitated with surface cooling and a 12-year-old boy treated with chilled fluid and surface cooling. 3 –5 All had promising neurologic outcomes. Although our resuscitation followed 2012 ACLS protocols, no substantive differences exist for treatment of a PEA arrest as cardiopulmonary resuscitation, and epinephrine remains the cornerstone of treatment.
Use of therapeutic hypothermia after PEA cardiac arrest and after lightning-induced arrest is a fairly new phenomenon. A recent retrospective study of 100 patients with nonshockable cardiac arrest showed a nearly threefold increase in neurologic improvement when intravascular therapeutic hypothermia was utilized and patients had strict time limits between cardiac collapse and resuscitation efforts. 6
The recent change in nomenclature from therapeutic hypothermia to targeted temperature management is reflective of the fact that temperature management after cardiac arrest goes beyond cooling. Fevers of 38°C or greater, a common occurrence within 48 hours of ROSC, are associated with increased rates of death. 7 Therapeutic hypothermia is effective at mitigating this, but the effect is present in noncooled patients as well. 8
Although uncommon, lightning and floods are the leading weather-induced causes of death in the United States. 9 An average of 53 Americans have been killed each year by lightning strikes and hundreds more are injured annually. 10 Injury patterns are unpredictable and range from mild symptomatology to death. 11 However, lightning-induced cardiac arrest may be more survivable than other causes while still leaving substantial neurologic deficits in survivors. 11
Lightning is a complex cause of cardiac arrest as it acts to suddenly depolarize the entire myocardium, suppress ventilation through spasm of the thoracic muscles, induce catecholamine release and autonomic nervous system stimulation, and cause brain hemorrhage, edema, and direct injury to any and all affected tissue. 11 It additionally may confer major trauma, such as burns and blast injuries. Central nervous system injury is of particular interest as it is both a target of protection in TTM and focal point of both acute and delayed injury from lightning strike, although the frequency of long-term neurologic sequelae is not well studied.
Potentially permanent neurologic complications can occur from damage to vulnerable regions of cortex in hypoxic settings. 9 Short-term memory loss, personality changes, and impaired judgment may result in addition to loss of motor or sensory function. 9 Cerebral infarction and myelopathy are rare complications of direct current on vulnerable tissue. 8 Autonomic damage may occur as well, but damage to the peripheral nervous system is unlikely. 9
Even if reperfusion is achieved within 30 minutes after cardiac arrest, global ischemia will have induced selective neuronal death, and a poor neurologic outcome can be anticipated. 11 Injured cells that do not die immediately may become necrotic, recover, or undergo later apoptosis. 11 If cooling is initiated rapidly, animal experiments have shown clear neuroprotective effects for cells that are not immediately irreversibly damaged. 7
A decrease of core temperature by just 1°C can diminish metabolic needs of neuronal tissue by 6% to 7%. 12 Other proposed mechanisms of therapeutic hypothermia include stabilization of cellular membranes and minimizing toxic free radical production after injury. 13 This is of particular interest in the setting of lightning. Free radical release from electrical injury is believed to be both derived from the lipids found in myelin and directly damaging to them. 14
Hypothermia may also limit cerebral edema, a possible outcome after lightning-induced neural death and vascular necrosis, and impairment of the blood brain barrier.14,15 Furthermore, it is believed that therapeutic hypothermia combats impaired glutamate reuptake and stymies the release of the inflammatory cytokines interleukins 1 and 6 and tumor necrosis factor alpha.16,17
The diversity of beneficial effects discovered in the research of TTM lends itself to improving patient outcomes in a variety of settings. The increasing use of TTM is a testament to this, and promising outcomes may potentially be had in improving the daily life of survivors of cardiac arrest. At this point, the use of prehospital cooling has not shown reliable benefit. 1 Prehospital prevention of fever when extrication would be prolonged has not been evaluated.
Our clinical result, and those cases presented before it, lends credence to the idea that the many lightning strike survivors who experience and survive a cardiac arrest each year may stand to benefit from TTM if adequate resources are available to provide it. We hope for further research on TTM to evaluate its role in more clinical scenarios.
Footnotes
Acknowledgment:
We would like to thank the Forensic Nurse Examiners Team at Christiana Care Health System for material support.
Author Contributions: Study Concept and Design: (DS), (BF), (SM). Acquisition of Data: (DS), (BF), (SM), (MP). Analysis of Data: (DS), (BF), (SM), (MP). Drafting of Manuscript: (DS), (BF), (SM), (MP). Critical Revision and Approval of Manuscript: (DS), (BF), (SM), (MP)
Financial/Material Support: None.
Disclosures: None.
Submitted for publication January 2016
Accepted for publication May 2016
