Abstract
Objective
The purpose of this investigation was to test a field-expedient, cost-effective method to decontaminate, sterilize, and package surgical instruments in an operational (combat) or austere environment using chlorhexidine sponges, ultraviolet C (UVC) light, and commercially available vacuum sealing.
Methods
This was a bench study of 4 experimental groups and 1 control group of 120 surgical instruments. Experimental groups were inoculated with a 106 concentration of common wound bacteria. The control group was vacuum sealed without inoculum. Groups 1, 2, and 3 were first scrubbed with a chlorhexidine sponge, rinsed, and dried. Group 1 was then packaged; group 2 was irradiated with UVC light, then packaged; group 3 was packaged, then irradiated with UVC light through the bag; and group 4 was packaged without chlorhexidine scrubbing or UVC irradiation. The UVC was not tested by itself, as it does not grossly clean. The instruments were stored overnight and tested for remaining colony forming units (CFU).
Results
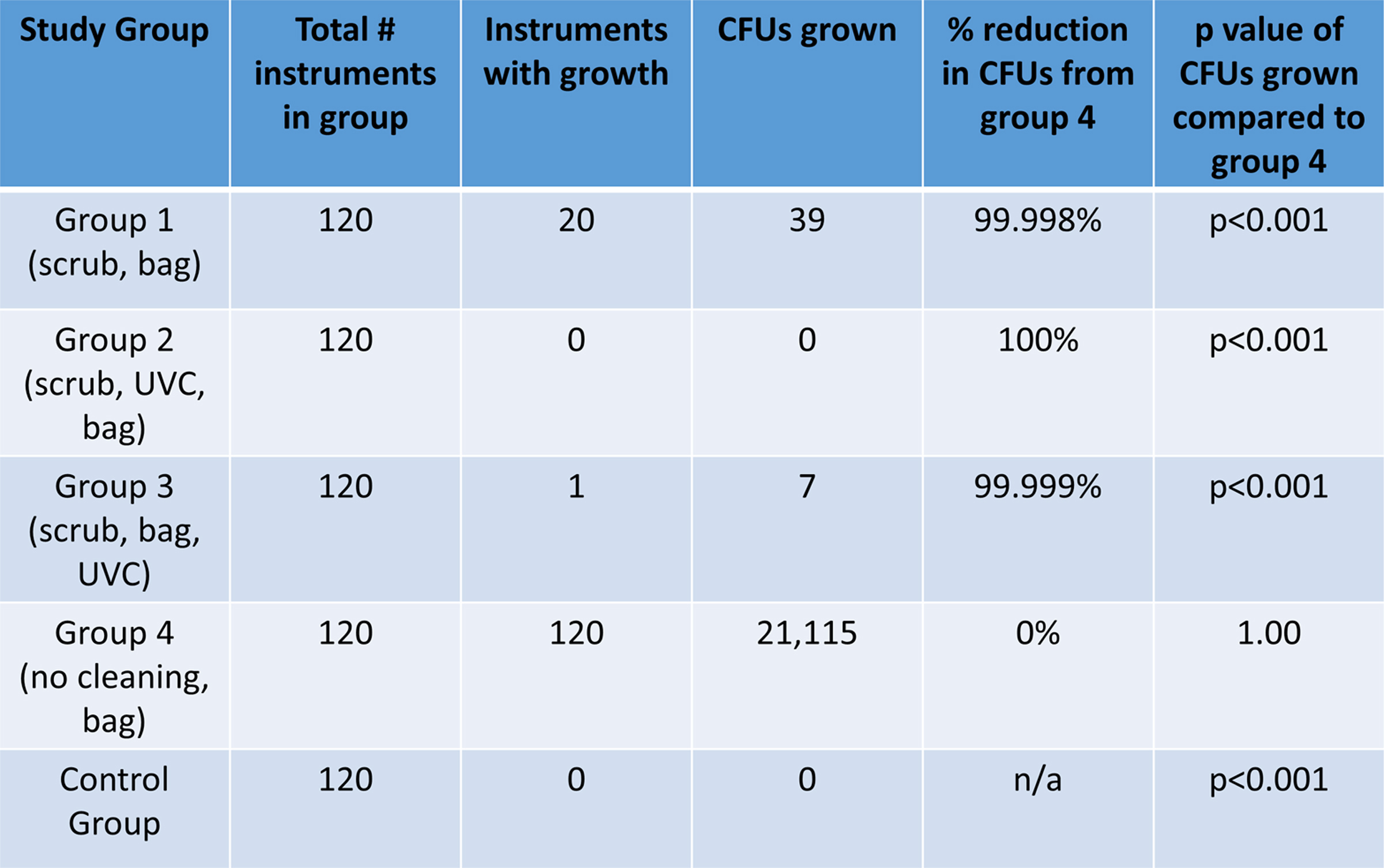
Data analysis was conducted using analysis of variance and group comparisons using the Tukey method. Group 4 CFU was statistically greater (P < .001) than the control group and groups 1 through 3. There was no statistically significant difference between the control group and groups 1 through 3.
Conclusions
Vacuum sealing of chlorhexidine-scrubbed contaminated instruments with and without handheld UVC irradiation appears to be an acceptable method of field decontamination. Chlorhexidine scrubbing alone achieved a 99.9% reduction in CFU, whereas adding UVC before packaging achieved sterilization or 100% reduction in CFU, and UVC through the bag achieved disinfection.
Introduction
Army physician assistants provide treatment as close as possible to the point of injury, and are the main credentialed provider of Army prehospital care. They are equipped with enough basic supplies and equipment to provide initial emergency medical treatment and resuscitation, acute care, and routine primary care. Examples of these treatments are emergent, invasive life-saving interventions such as surgical cricothyrotomy as well as finger and tube thoracostomy, in addition to urgent procedures such as wound debridement and laceration repair. All these invasive procedures are usually performed with clean or sterile reusable surgical instruments. A current field sterilization practices survey was sent by e-mail to 1500 Army active duty, Reserve, and National Guard physician assistants. Of the respondents, 18% used an autoclave or steam sterilizer, 22% performed cold sterilization, 30% transported their instruments to a higher level facility for sterilization, and 30% used some other method to decontaminate their instruments. Examples of “other” methods reported were use of hydrogen peroxide, betadine, isopropyl alcohol, ethyl alcohol, povidone iodine wipes, Cavicide wipes, and bleach (see the Table 1 for definitions of cleansing methods1,2).
Definitions
Autoclaves are not authorized for most prehospital units, and cold sterilization requires storage of chemicals that are bulky and have short shelf lives when mixed and activated. Even reliable sterile storage before use in this environment is difficult, as instruments are stored in trauma packs or aid bags where the usual cellophane and paper packaging (peel pack) is easily damaged. Currently, there is no standardized prehospital/field method to decontaminate or package surgical instruments for Army prehospital use. This lack highlights the need for the development and testing of a cost-effective, simple, and portable method that requires minimal resources to achieve high-level disinfection or sterilization, as well as durable storage of reusable surgical instruments suitable for the operational (combat) and austere (wilderness) environments.
Methods
This was an experimental laboratory study to develop an alternative method to achieve high-level disinfection or sterilization and packaging of reusable semicritical and critical surgical instruments in austere environments. This is a pilot study as there are no similar studies looking at the effectiveness of prehospital decontamination of surgical instruments and durable packaging that prevents contamination. This study tested a method of scrubbing contaminated surgical instruments with chlorhexidine, then irradiating them with ultraviolet-C (UVC) light, and finally, packaging them in a vacuum sealed bag. In all, 124 surgical instruments were issued from the Madigan Army Medical Center (MAMC) Central Material Services for this experiment. They were autoclaved and issued in double peel packs. Thirteen types of instruments were selected based on a representation of what is used in the field; variety and variability of grooves, teeth, and handles; and Central Material Services availability. All instruments are made of surgical steel (Figure 1). Individually packaged 4% chlorhexidine gluconate sponge scrubs were obtained. A commercially available portable UVC wand, vacuum sealer, and nylon vacuum sealer bags were purchased by the MAMC Department of Clinical Investigation. All bacteria samples were obtained from the American Type Culture Collection.
Surgical instruments used in the study.
There were 4 intervention groups and 1 control group. The same 120 instruments were used in each group. The instruments in the 4 intervention groups were inoculated with methicillin-resistant Staphylococcus aureus, Streptococcus pyogenes, Pseudomonas aeruginosa, and Acinetobacter baumanni. For inoculation, the functional unit (distal 3 cm) of each instrument was dipped in Luria broth containing a 106 concentration of each type of bacteria for 1 minute to ensure even distribution of bacteria on the instruments. After inoculation, the instruments were placed in a laminar flow hood and allowed to dry for 2 hours. The control group had no inoculum placed on them. Additionally, an internal control group of 4 instruments that did not undergo any intervention were inoculated in the same manner for each intervention group to ensure adequate inoculum. Instruments were resterilized by an autoclave after each intervention to ensure sterility before their reuse.
The 4 intervention groups were as follows: 1) chlorhexidine only in a vacuum-sealed bag; 2) chlorhexidine plus UVC exposure, placed in a vacuum-sealed bag; 3) chlorhexidine, placed in a vacuum-sealed bag plus UVC exposure through the bag; and 4) vacuum-sealed bag only, no chlorhexidine and no UVC irradiation. The instruments in the control group were removed from their sterile double peel packaging under a laminar flow hood to prevent contamination, and individually sealed in vacuum bags. The intervention groups were treated on a laboratory bench and not under a laminar flow hood.
Each instrument in group 1, after inoculation, was scrubbed vigorously with the chlorhexidine sponge for 30 seconds on the functional portion of the instrument (Figure 2A). One scrub sponge was used for 4 instruments, then discarded and replaced. Instruments were rinsed with tepid tap water until all gross chlorhexidine was removed, dried with a sterile 4×4-inch gauze pad, and sealed in an individual vacuum bag. The instruments in group 2 were cleaned and dried in the same manner, and then individually hung by a wire on an apparatus opposite the functional portion of the instrument (Figure 2B); each instrument was exposed to UVC radiation by waving the wand up and down from 4 inches on each side of the instrument for 45 seconds, and then it was sealed in a vacuum bag. The instruments in group 3 were cleaned and dried in the same manner as groups 1 and 2; they were then sealed in individual vacuum bags, hung by wire and clip, and UVC irradiated in the same fashion as group 2, except through the bag (Figure 2C). The instruments in group 4, the “bag only” group, were not cleaned or exposed to UVC light; they were individually sealed in a vacuum bag (Figure 2D).

(A) Chlorhexidine scrubbing; (B) UVC wanding; (C) UVC wanding through bag; (D) commercially available vacuum sealer.
After placement in vacuum-sealed bags, the instruments in all experimental groups were labeled 1 to 120, then stored in a covered container with no direct light for as long as 24 hours. After storage, each instrument was removed from its vacuum-sealed bag, and the functional unit was swabbed with a presoaked sterile cotton tip applicator. The cotton tip applicator was placed in 10 mL phosphate-buffered saline solution, then vortexed for 5 seconds to loosen any bacteria into the solution. A 100-µL sample was taken and streaked out onto Luria broth agar plates. The agar plates were incubated at 30°C for 18 hours. The 4 internal control instruments were tested in the same manner. After incubation, colony forming units (CFU) were counted in each of the intervention groups and compared with the control group and with group 4 to see how effective the respective disinfection and sterilization conditions were, as measured by percent reduction in CFU.
Because this was a novel study with no prior information, the power analysis was done on an estimated percentage of reduction of density (5% to 10%) of bacteria across the 5 groups. The sample of 600 (120 per group) is sufficient to capture that difference at an alpha level of 0.05 at a power of 80%. The statistical method of choice was one-way analysis of variance with post-hoc analysis of comparing groups by the Tukey method.
Results
Using an ANOVA test, there was a significant difference between all the groups when anchored to the control group (P < .001). Therefore, a post-hoc analysis using the Tukey method was performed. The Tukey method compared all groups to one another. This post-hoc analysis did not find a statistically significant difference in groups 1, 2, and 3 when compared to the control. However, it did find a significant difference between group 4 (P < .001) and the control. The post-hoc analysis also found a statistically significant difference (P < .001) with group 4 compared to groups 1 through 3 together. Moreover, the post-hoc analysis with group 4 and the control group found a significant mean difference in CFU of 175.96 (95% CI: 166.3 to 185.61).
Group 1 (chlorhexidine only; n = 120) resulted in 20 instruments with CFU remaining, thus achieving disinfection. The total number of CFU remaining was 39, a 99.9% reduction in CFU as compared with the bag-only group. The other 100 instruments were completely free of bacteria. The instruments in group 2 (chlorhexidine plus UVC, then bag; n = 120) showed no bacterial growth, thus attaining sterilization. Group 3 (chlorhexidine, then bag, then UVC; n = 120) resulted in 1 instrument with 7 CFU remaining and 119 without growth, attaining high-level disinfection. All instruments in group 4 (bag only; n = 120) remained contaminated, with an average of 175.96 CFU per instrument and a total of 21 115 CFU for all 120 instruments. The control group of instruments (n = 120) had no bacterial growth (Table 2). The internal control instruments had an average of 276.3 CFU per group, thus proving an adequate amount of bacteria was inoculated on the surgical instruments in each of the intervention groups.
Results for experimental groups 1 through 4 and control group. CFU(s), colony forming units
Discussion
This study addressed the need for a scientifically researched method of sterilizing and packaging surgical instruments for prehospital aid stations and other austere conditions. It not only tested the ability of UVC irradiation when used with mechanical cleansing to achieve sterilization of grossly decontaminated surgical instruments, but also a recommended method of cleaning, sterilizing, and packaging surgical instruments. Cleaning is reinforced as an important step in the decontamination process: by manual cleaning alone, chlorhexidine sponge scrubs achieved a 99.9% reduction of bacteria. Further studies are needed to test the duration and environmental parameters of sterility maintenance in vacuum-sealed bags.
Study Limitations
We only tested 4 different types of aerobic bacteria; no anaerobic bacteria, bacterial spores, fungi, or viruses were tested. No UVC-alone arm was used owing to previously studied limitations of using surfaces contaminated with organic material. 3 We used irradiance doses based on the known bacteria in this study. Finding a common irradiance dose for eradication of all microbes is a practical approach; however, waving a wand for a long time may be impractical in the field. Some microbes have repair mechanisms known as photoprotection, which might make UVC irradiation less effective. 4 Nevertheless, given the results of this study, if the austere medical practitioner has no other means of sterilization, the application of UVC irradiation after mechanical cleaning may be beneficial.
Conclusion
Irradiation with UVC is effective for sterilizing surgical instruments contaminated with aerobic bacteria after being scrubbed with chlorhexidine sponge scrubs, and immediate vacuum sealing maintains that sterility. After the scrubbing of surgical instruments, UVC radiation through a vacuum-sealed bag was effective at reducing the microbial load by 99.9%. Proper mechanical cleaning remains important in significantly reducing bioburden on surgical instruments, as it sterilized 83% of the instruments and reduced the total microbial load by 99.9%. Additionally, UVC light was able to sterilize a variety of surgical instruments with numerous grooves, teeth, and edges. Prior studies have not used UVC on critical items or have overcome shadowing by grooves through the use of large cabinet (nonportable) size devices. 5 Although further studies are necessary, the simplicity and low resource requirement of this technique appears promising for prehospital and austere applications.
Footnotes
Acknowledgments
Thanks to Dr Raywin Huang, statistician for Madigan Clinical Investigations, for his assistance with data analysis and calculations. This study was conducted by the authors while assigned to the Department of Emergency Medicine and Department of Clinical Investigation, Madigan Army Medical Center, Joint Base Lewis-McCord, WA, with no additional funding sources or conflict of interest to disclose.
This study was conducted by the above authors while assigned to the Department of Emergency Medicine and Department of Clinical Investigation, Madigan Army Medical Center, Joint Base Lewis-McCord, WA, USA with no additional funding sources or conflict of interest to disclose.