Abstract
Venomous snakes with hematotoxin—Russell’s viper (
Introduction
Russell’s viper (
We report the case of a boy who had ptosis, exotropia, respiratory failure, rhabdomyolysis, and transaminitis after being bitten by a darkish-colored snake, which was not captured. He did not respond to antivenom for cobra, Malayan pit viper, or Russell’s viper. His neurological and respiratory abnormalities, coagulopathy, and hepatocellular injury finally recovered after receiving antivenom for green pit viper.
Case Presentation
A 12-year-old hill tribe boy, previously healthy, presented with swelling and pain in his left leg after being bitten on his left ankle by a snake in the forest while he was playing with a friend 2 days before he came to Maharaj Nakorn Chiang Mai University Hospital (MNCMUH). The patient described the snake as a 1-foot-long, darkish-colored snake. After the snake bite, the patient had gradual and continuous oozing from the bite wound without bleeding from other sites. One day later, he had more swelling and pain in his left leg. He was taken to the local hospital 26 hours after the snake bite. His vital signs were assessed as follows: temperature 36.2°C, pulse rate 94 beats/min, respiratory rate 20 breaths/min, and blood pressure 140/90 mm Hg. His oxygen saturation was 90% at room air, which was corrected by oxygen supplementation. The physical examination showed blood oozing from the wound and swelling of the left leg. Mild ptosis of the left eye was also noted. Neurological examination was otherwise normal. The initial evaluation of blood tests revealed hemoglobin of 15 g/dL, white blood cell count of 16.05 × 103/mm3, platelet count of 224 × 103/mm3, creatinine 0.57 mg/dL, prothrombin time (PT) greater than 180 seconds, activated partial thromboplastin time (APTT) more than 180 seconds, and venous clotting time (VCT) more than 30 minutes. Because of the desaturation and prolonged coagulation tests, he was treated with 3 doses of antivenom for cobra and Malayan pit viper (Queen Saovabha Memorial Institute, Thai Red Cross Society, Bangkok, Thailand) and 1 dose of tetanus toxoid 0.5 mL and tetanus antitoxin 30,000 units. Despite the treatment, desaturation and prolonged coagulation time were not corrected. After receiving antivenoms, vitamin K, and 6 units fresh frozen plasma, he still had bleeding symptoms and prolonged VCT.
At 51 hours after the snake bite, his left leg became more swollen and inflamed. Ptosis, diplopia, respiratory distress, and red urine developed. He was intubated and then transferred to MNCMUH. Ceftriaxone and amoxicillin/clavulanic acid were given. The physical examination at MNCMUH showed 2 fang marks 0.1 cm in diameter, each with a fang mark distance of 0.8 cm on the left lateral malleolus (Figure 1). He had mild tenderness and swelling of the left ankle without hemorrhagic bleb or ecchymosis. There was no more oozing from the bite wound and no other bleeding from other sites. He had normal consciousness but ptosis of the left eye with palpable fissure distance 0.9 cm of the left eye while his right eye had the palpebral fissure distance of 1.5 cm. He also had exotropia of the left eye (Figure 2). In addition to ophthalmoplegia, he had paradoxical breathing when he was accidentally extubated despite having normal breath sound on both sides on examination. The blood tests on day 2 after the snake bite were as follows: complete blood count with hemoglobin 10.8 g/dL, white blood cell count 10.2 × 103/mm3, and platelets 153 × 103/mm3; blood chemistry with creatinine 0.6 mg/dL, total creatine kinase 135,846 U/L, total protein 6.7 g/dL, albumin 3.4 g/L, alkaline phosphatase 209 U/L, aspartate aminotransferase 3,897 U/L, alanine aminotransferase 1,268 U/L, total bilirubin 0.57 mg/dL, and direct bilirubin 0.07 mg/dL; arterial blood gas showed pH 7.47, pO2 157 mm Hg and pCO2 39.8 mm Hg with FiO2 of 0.4 on ventilator setting; urine analysis showed heme 3+, red blood cell count 2 to 3/high-power field, white blood cell count 20 to 30/high-power field; coagulation tests found PT greater than 200 seconds, APTT more than 200 seconds, VCT greater than 30 minutes, D-dimer 8.5 mg/L, factor X clotting assay 71%, and factor V clotting assay 105%; his chest radiograph was unremarkable. The boy was diagnosed with snake bite and coagulopathy, respiratory failure, and myoglobinuria. He was treated with 5 doses of antivenom for Russell’s viper and 1 dose of fresh frozen plasma in 48 hours without any improvement of the clinical symptoms and coagulopathy. Moreover, his platelet count was gradually decreasing to the lowest number, at 125 × 103/mm3 on day 5 after the snake bite. His blood chemistry gradually improved, however.
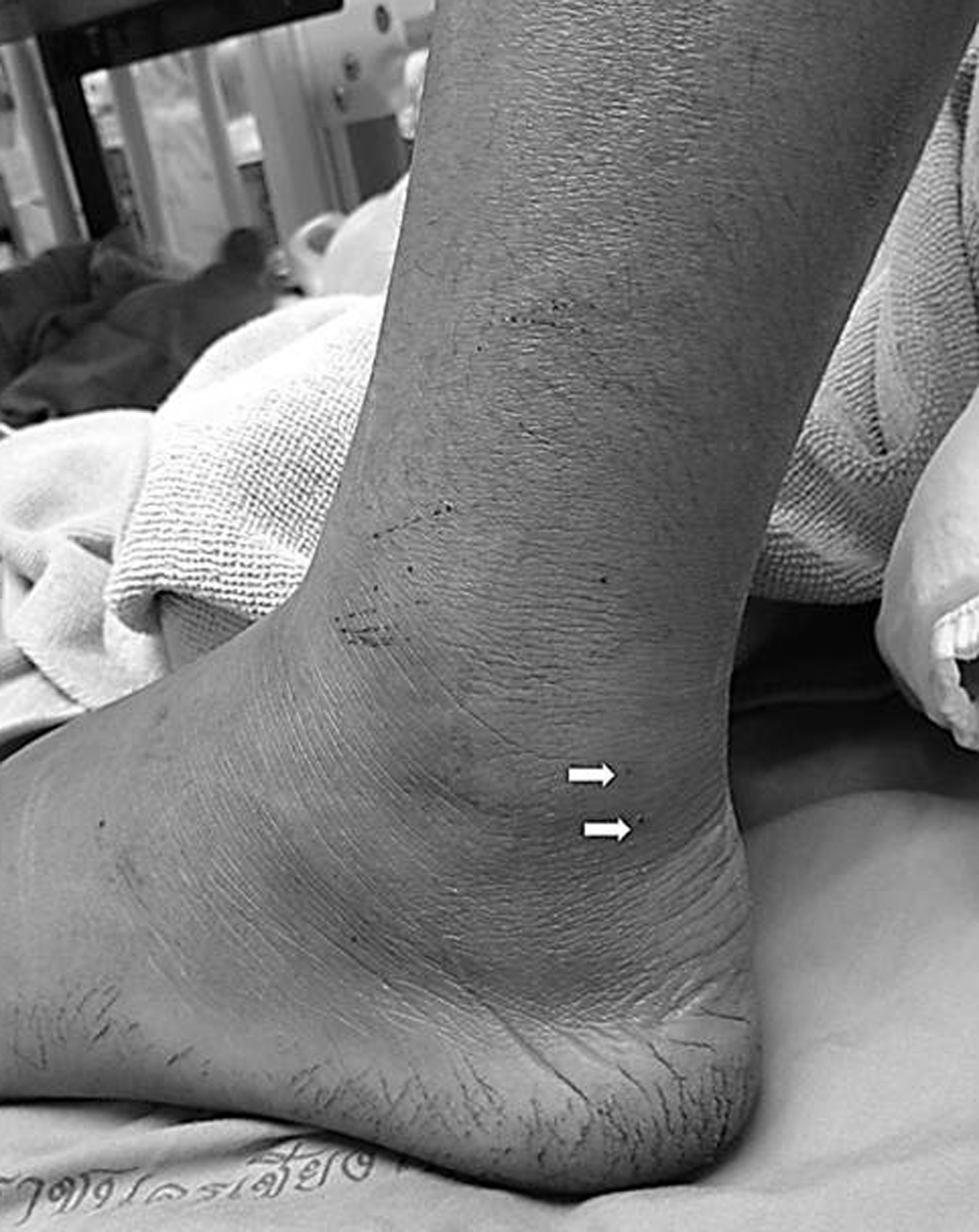
Fang mark on left ankle, below to left lateral malleolus (arrows).

Exotropia and ptosis of the left eye on day 1 of admission.
After consultation with the institutional toxicologist and Ramathibodi Poison Center at Mahidol University in Bangkok, the patient was treated with antivenom for green pit viper on day 5 after the snake bite. He subsequently had clinical improvement of left ptosis, exotropia, and respiratory weakness within 12 hours after the first dose of antivenom. The coagulation tests also became normal within 6 hours and the platelet count within 24 hours after antivenom treatment. On day 10 of the snake bite, however, his VCT, PT, and APTT were prolonged again. He was given a second dose of antivenom for green pit viper, and again, his coagulopathy was rapidly corrected, and blood testing remained in the normal range until he was discharged from the hospital. He was admitted to the hospital for 12 days and was discharged uneventfully on day 14 after the snake bite.
Discussion
The presentation of this patient was similar to the syndrome caused by the bite of a snake with hematotoxin because he had pain and swelling of the affected part and abnormal coagulation tests, including prolonged VCT, PT, and APTT, and low fibrinogen and platelet count. These findings were compatible with Russell’s, Malayan, or green pit viper toxicity. 1 ,5–14 However, he also had ptosis, exotropia, and respiratory distress, which were compatible with the syndrome caused by the bite of a snake with neurotoxin, such as a cobra or king cobra. Therefore, he had indications for antivenoms for both hematotoxin and neurotoxin. 1 Because of his description of the snake, the geographic range, and the severity of his clinical manifestations, he was treated with antivenoms for Malayan pit viper and cobra, 2 of the most likely snakes, although the chance of 2 snake bites at the same time was low. After 3 doses of antivenoms, however, he did not have any clinical and laboratory improvement but, rather, worsening respiratory symptoms.
In general, identification of snake bite is based on the examination of the snake by experienced medical personnel. If the snake is not available to be examined, as in our case, the details of the snake and circumstances of its bite obtained from the patient and a witness, combined with a clinical syndrome of symptoms and signs and incidence of common snake bites in each area can be used to infer the species of snake. 1 According to a report by the World Health Organization, the Malayan pit viper (40%), green pit viper (37%), and cobra (16%) are 3 of the most common causes of venomous snake bites in Thailand. 1 These findings were similar to a local report written in Chiang Mai. 2 The epidemiology of snake bite was quite different in Bangkok, where green pit viper was much more prevalent (77% to more than 90%) than the other types of venomous snakes.4,14 Although serologic testing to identify the species of snakes is the reliable method for diagnosing definite type of snake bite,15,16 these tests are not available in most general hospitals, and the interpretation of blood tests of patients who received antivenom is still problematic.
Russell’s viper bite was the most likely diagnosis as it could explain most symptoms of this patient, according to a report by Kularatne.
11
That case series of 336 patients in Sri Lanka who were bitten by Russell’s viper (
The patient in this case had red urine with positive urine heme by urine dipstick, but no significant red blood cells or red blood cell cast in the urine, and markedly elevated total creatine kinase, so he was suspected of having rhabdomyolysis and myoglobinuria rather than hematuria or hemoglobinuria. Rhabdomyolysis has a wide range of prevalence, from 14% to 73% of patients bitten by Russell’s viper. 10 The depletion of platelet count, fibrinogen, and factors V and X were commonly found in patients with Russell’s viper bites. 5 However, the results of factors V and X clotting assay in this patient should be carefully interpreted because he received fresh frozen plasma before blood testing.
Kularatne 11 reported the manifestations of hematotoxicity from Russell’s viper usually improved within 48 hours in most patients (94%) after antivenom administration. 11 Unlike the previous report, our patient did not have any response to antivenom for Russell’s viper, even though he received 5 doses of antivenom within 48 hours. Severe liver injury was found in only 1 report of a patient who was bitten by Russell’s viper. 15 Renal failure, which was a common complication (in 5 of 7 patients) in the case series of Russell’s viper bites in Thailand, was not found in our patient. Therefore, a Russell’s viper bite was less likely in our patient.
Many types of green pit viper are found in all parts of Thailand, but 3 of the most common species are
Rojnuckarin et al 17 reported that, owing to the large molecule of green pit viper venom, the clearance of its venom antigen could be more delayed than cobra or Russell’s viper venom antigen. Moreover, symptoms can recur in patients bitten by vipers because of slow absorption of the venom from the wound, as seen in this patient.3,17 In addition, this patient had severely prolonged coagulation tests while his platelet count was relatively normal. These results were compatible with the kinetic study of venom antigen of green pit viper that showed early prolongation of coagulation test but delayed thrombocytopenia. 17 The proposed mechanism was because of different components in green pit viper venom, with longer half-life that could stimulate platelets and cause thrombocytopenia.
The only report regarding neurological involvement after a green pit viper bite was from Thailand, but that patient’s course was complicated by tetanus. 19 Our patient, however, received tetanus toxoid, tetanus antitoxin, and antibiotics that could cover anaerobic bacteria since admission to the local hospital; thus, the possibility for tetanus for this patient was low. To our knowledge, the mechanism of neurological involvement in this case is still unclear. Also, there is no explanation for the hepatocellular injury. Myoglobinuria might be a result of a local muscle injury from proteolytic enzymes in the snake venom. 1 Although Coca-Cola urine was found in a patient who had acute tubular necrosis after a green pit viper bite, 5 no laboratory test of myoglobinuria was available to confirm the etiology of the brown-colored urine.
Study Limitations
There are several limitations to this report. Firstly, no blood testing confirmed the exact type of snake. Serological testing for confirmation of green pit viper bite is still not available for routine service in Thailand, and interpretation of the test in a patient who received antivenom is still doubtful. Secondly, although no bleeding was detected on a computed tomography brain scan of this patient, magnetic resonance imaging would have been more sensitive to detect brain parenchyma lesion, especially infarction, than computed tomography. Thirdly, all symptoms might have resolved spontaneously, not by the effect of antivenom, because this patient had been admitted for a week when he recovered. Fourthly, the explanation for liver injury and rhabdomyolysis in this patient was unclear.
Even though the specific species of snake could not be identified, this patient was likely bitten by a snake with hematotoxin, as supported by the markedly abnormal coagulation tests.
Footnotes
Acknowledgments
We would like to thank Ramathibodi Poison Center at Mahidol University, Bangkok, for the excellent consulting service. We also would like to thank Mr Ken Dolen and Dr Pimlak Charoenkwan for English language editing of the manuscript.
