Abstract
Objective
To assess whether previous freezing cold injuries (FCI) would affect digit skin temperatures and rewarming rates during a follow-up cold stress test protocol.
Design
Nonrandomized control trial.
Methods
Twenty elite alpinists participated; alpinists with previous FCI requiring digit amputations (injured, INJ: n = 10 total, n = 8 male) were compared with ability-matched, uninjured alpinists (control, CON: n = 10, all male). Digit skin temperature was measured using infrared thermography as an index of peripheral digit perfusion after a cold stress test, which consisted of 30 minutes of immersion in 8°C water.
Results
The INJ alpinists’ injured toes were warmer (approximately 6%) than their uninjured toes immediately after cold immersion (95% CI, 0.01°C to 1.00°C; P = .05); there were no differences between the rates of rewarming of injured and uninjured toes (INJ, 0.5° ± 0.1°C/min; CON, 0.7° ± 0.3°C/min; P = .16). Although the INJ alpinists had colder injured fingers immediately after the 35°C warm bath compared with their own uninjured fingers (32.2° ± 2.0°C vs 34.5° ± 0.5°C; P = .02), there were no differences observed between the rates of rewarming of injured and uninjured fingers after cold exposure (INJ, 1.1° ± 0.2°C/min; CON, 1.3° ± 0.5°C/min; P = .22).
Conclusions
Even after FCI that requires digit amputation, there is no evidence of different tissue rates of rewarming between the injured and uninjured fingers or toes of elite alpinists.
Introduction
Of all body regions, fingers and toes are at a greater risk of freezing cold injuries (FCI, ie, frostbite), especially in alpine, subzero environments. 1 Freezing cold injuries of the lower extremities are more frequent than those of the upper extremities.2,3 Tissue that has sustained frostbite often exhibits immediate functional impairment and sequelae (chronic pain, cold sensitivity, localized osteoporosis) long after the initial incident has occurred. Such affected tissues are therefore anecdotally thought to be predisposed to future cold injury, 4 although evidence-based studies, including follow-up assessments of frostbite, are rare. Indeed, with frostbite that requires amputation, the nonviable tissue is removed, and thus, despite obvious functional impairments, it is not known whether the remaining tissues could be at greater risk for future cold injury, or whether the tissues may remain asymptomatic over time.
Because frostbite is accidental in nature, accurate incidence rates are difficult to determine with certainty, 3 although individuals with previous cold injuries to upper and lower extremities and those with cold intolerance as a result of hand fractures 5 or after nerve injury 6 represent a sizable clinical population that may be predisposed to repeated, future cold injuries. Reported incidence rates of cold injury can be very high in alpinists, reportedly 37% in that population per year, 1 compared with the cumulative lifetime incidence of frostbite in young Finnish men, also reported to be as high as 44% to 68%. 7 Often, immediate or timely medical attention of frostbite injuries in alpinists is impossible; a 10-year epidemiological study of frostbite in the Karakoram mountain range revealed that only 2% of patients could be evacuated to the nearest hospital within 6 hours of the incident. 3 Indeed, few randomized, controlled studies have been conducted to quantify the incidence, epidemiology, and predisposing factors of frostbite in high altitude alpinist populations. Between 1951 and 2009 there were 6571 mountaineering accidents involving 11,979 people reported to the American Alpine Club within the United States alone, 8 thus constituting a relatively large population that is routinely at risk for future cold injuries.
Rapid hand or foot cooling can induce general sympathetic stimulation, and is often used to investigate subsequent rewarming rates of the fingers or toes.5,6 Digit skin temperature is well correlated to skin blood flow; 9 it can be quickly and noninvasively measured in conjunction with a cold stress test, although these techniques are rarely used in follow-up cases of clinical frostbite. When exposed to a cold environment (<15°C), skin temperature can rapidly increase in a cyclical manner (hunting response). Indeed, it has been shown that mean skin temperatures and rates of rewarming can be altered after acclimatization to altitude 10 and improved baseline and recovery temperatures after 15 months of military training in the cold, 11 although the “trainability” of fingers and toes to cold exposure per se in laboratory studies has not clearly demonstrated enhanced adaptations to cold.12,13 These studies have suggested, conversely, that there is an increased injury risk with repeated cold exposure. Therefore, although the literature in cold adaptation is equivocal, cold intolerance is assumed to be related to vascular dysfunction, 14 and measuring temperature responses after cold exposure as a means of characterizing (or indexing) future cold injury is not well established in the clinical literature to date.
One of the simplest and most convenient methods of noninvasively evaluating dynamic changes in cutaneous temperature is via infrared (IR) thermography. IR thermography has been used to clinically assess skin temperatures after a cold test in outdoor manual labourers, 15 circulatory compromise of non-FCI in soldiers 16 and other such varied conditions as diagnosing type 2 diabetes, 17 burn depths, 18 vascular responses after abstinence from smoking, 19 and detecting compressive neuropathies such as carpal tunnel syndrome, 20 traumatic nerve injury, 6 and hand fractures. 5 Indeed, using IR thermography to measure the spontaneous rewarming rate of the hand after cold immersion has been suggested as a useful tool to prospectively identify individuals who are at higher risk of experiencing an FCI. 11 However, no study has attempted to evaluate the functional capabilities of injured alpinists’ digit perfusion many years after surgery. Therefore, the purpose of this study was to determine whether elite alpinists with previous frostbite injury that required amputation had significantly different rates of rewarming after a cold stress test when compared with their ability-matched, elite alpinist counterparts. It was hypothesized that 1) digits within the injured alpinist population would be colder than matched controls; 2) the injured alpinists’ injured digits would be colder than the digits of their own contralateral, uninjured side; and 3) rates of rewarming would be slower in the injured cohort than in the matched controls for both fingers and toes.
Methods
The protocol of this study was approved by the National Committee for Medical Ethics at the Ministry of Health of the Republic of Slovenia, and it conformed to the Declaration of Helsinki guidelines. Written, informed consent was obtained from each participant before participation in the study. Any identifiable participants have provided their signed consent to publication. Participants were requested to refrain from consuming alcohol and caffeine, and from engaging in exertional activity on the day of testing. All testing was performed in a climate-controlled laboratory environment (between 21° and 23°C, 52% relative humidity) during the northern hemisphere summer (June and July) at the Olympic Sport Centre Planica (Rateče, Slovenia).
Study Population
Twenty elite Slovenian alpinists volunteered for this nonrandomized control study. The alpinists were all high altitude climbers with experience on peaks higher than 8000 m. The alpinists were divided into 2 groups: those who had suffered previous FCI that required amputation on either their fingers or toes (injured: INJ), and those who had not sustained any known freezing or non-FCI (control: CON). All alpinists at the time of testing were still actively climbing in high altitude (>4000 m) environments and participating in seasonal expeditions.
Cold Stress Test
A cold stress test, defined here as a 30-minute cold-water immersion of the hand or foot, was used to determine rates of tissue temperature changes before, during, and after the cold immersion. Each participant arrived approximately 15 minutes before the testing session began. Participants were dressed in a T-shirt and trousers, with shoes and socks removed, and were seated on a semirecumbent chair for the duration of testing. This particular cold stress test protocol follows methods described elsewhere.21,22 Briefly, after instrumentation and 5 minutes of baseline recording, participants placed their hand or foot in a thin, plastic bag to avoid their skin getting wet during water immersion. In the uninjured alpinists we tested the foot on the same side as their specified dominant hand to minimize any chance of slight differences or variations in response between sides of the body, in terms of skin temperature, vasomotor function, etc. The alpinist’s hand or foot was then immersed in a circulated water bath, first for 5 minutes at 35°C to normalize digit temperature, and then for 30 minutes at 8°C in a circulated water bath. Water temperature was maintained via an industrial cooling system that had a small impeller continuously stirring the tank water (Haake, Karlsruhe, Germany). After 30 minutes had elapsed, the hand or foot was removed immediately from the water, taken out of the plastic bag, lightly patted dry if necessary, and allowed to passively warm in room air (approximately 21°C) for 10 minutes. In the case of injury, both the injured and uninjured hand or foot were tested in random sequence. Additionally, an index of core temperature (tympanic temperature) was taken before and after the cold stress test using an IR thermometer (ThermoScan IRT 3020, Braun, Kronberg, Germany).
Digit skin temperature was measured on the dorsal surface of each hand or foot using IR thermography (FLIR B335, FLIR Systems, Inc, Wilsonville, OR, USA). This camera has an internal calibration function at the beginning of each use, and a thermal sensitivity of ±0.05°C. Because skin emissivity is 0.98, the measured IR thermography temperatures can be taken as skin temperature. To facilitate capturing a well-contrasted image, we placed a cool towel beneath the hands or feet of the patient immediately before image capture. The camera was placed approximately 30 cm above the dorsal side of the hands or feet. Skin temperature was measured at baseline, immediately after the warm bath, immediately after the cold bath, and at 5 and 10 minutes after immersion. Digit temperatures at each time were measured at the point of the nail bed, or proximal to the amputation site using the company’s in-house spot-tool software (FLIR QuickReport 1.2, SP2, FLIR Systems, Inc). An additional, corresponding reading was recorded at the same location on the uninjured, contralateral digit.
Statistical Analyses
We compared descriptive statistics using the statistical package SPSS version 17.0 (Chicago, IL, USA). On all data sets, a mixed model, 2-way repeated-measures analysis of variance was conducted using 2 within-factors (time and digit) and 1 between-factor (injury status: INJ vs CON). Analysis of finger and toe responses was conducted separately. Although there were n = 10 alpinists in each cohort, data analysis comparing digit injury status constituted a comparison of n = 200 digits (n = 20 alpinists × 10 fingers or toes, respectively). Paired t tests were used to compare differences between specific injured digits and their uninjured counterparts on the contralateral limb for the INJ alpinists only. Bivariate correlations were run on the rate of recovery of pooled digit temperatures (between individuals) immediately after cold immersion until the end of the recovery period, and the current age of the alpinist, or the injury. Patient sample size was based on clinical medical literature, which has reported significant skin temperature differences of less than 1°C in samples of between 7 and 12 persons. 6 ,12,23 All data are presented as mean ± SD with a significance level set at P < .05.
Results
All participants were healthy nonsmokers, with no history of any known cardiorespiratory, musculoskeletal, or circulatory disease. On average, 13.5 years had passed since the FCI event in the INJ alpinist group (n = 10, male = 8, female = 2, all right-handed). Seven alpinists had FCI on their toes, 2 alpinists had amputations on their fingers, and 1 had amputations to both their fingers and toes (Table 1). Alpinists in both groups had experience climbing in extremely cold conditions (temperature ranging from –10° to –50°C) with strong winds (Table 2). There were no significant differences between alpinist groups in terms of age (INJ, 44 ± 10 years; CON, 39 ± 12 years; P = .28) or height (INJ, 1.77 ± 0.09 m; CON, 1.79 ± 0.05 m; P = .44); the control group was heavier than their injured counterparts (INJ, 71 ± 10 kg; CON, 80 ± 7 kg; P = .03). Laboratory ambient temperature and relative humidity remained stable throughout each testing session (21° ± 1°C; 52% ± 8%), as were participants’ tympanic core temperatures measured before and immediately after the test (36.5° ± 0.5°C vs 36.4° ± 0.5°C; P = .88).
Cold injury details for the injured alpinist group
LH, left hand; RH, right hand; LF, left foot; RF, right foot; (↓), partial-length amputations; (↕), full-length amputations; F, finger; T, toe; 1–5 (number of digits affected).
Circumstances in which the injured alpinists obtained (suffered) frostbite, and climbing conditions experienced by the uninjured group
Digit Skin Temperature Differences between alpinist groups
Digit skin temperature assessment via IR thermography (Figure 1) demonstrated no significant differences in baseline finger (INJ, 29.1° ± 3.6°C vs CON, 27.3° ± 3.7°C; P = .33) or toe (INJ, 25.3° ± 2.7°C vs CON, 24.0° ± 2.0°C; P = .57) temperatures between alpinist groups (Figure 2). There were also no significant differences between groups for toe temperatures at any time during the recovery period (P = .12). Toe temperatures recovered up to 53% ± 4% (INJ, 51% ± 3%; CON, 54% ± 4%) of baseline after 5 minutes of recovery, and up to 61% ± 7% (INJ, 60% ± 9%; CON, 62% ± 3%) of baseline after 10 minutes of recovery, regardless of injury status (P = .25).
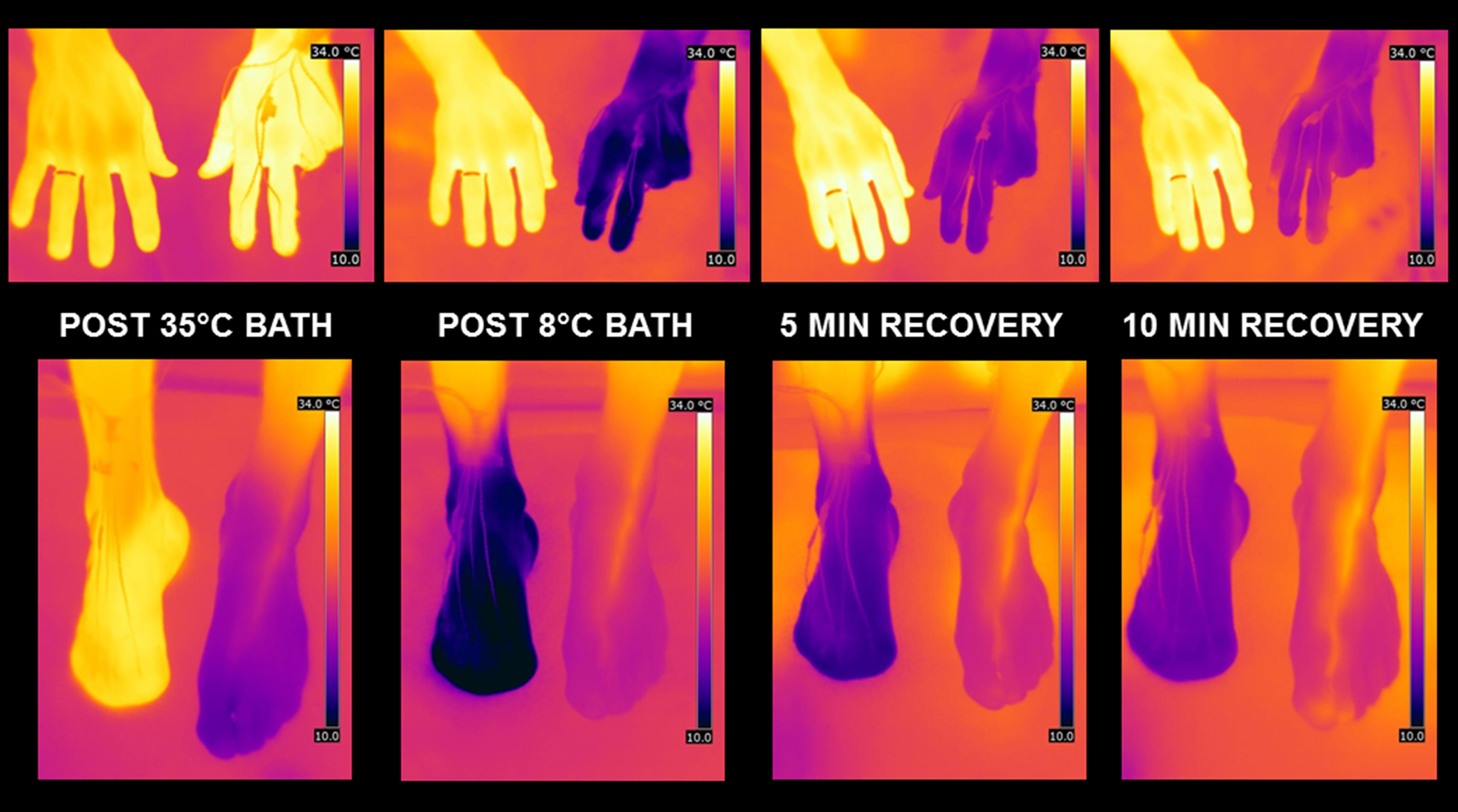
Representative infrared thermographs for the fingers and toes of 2 different injured alpinists. The top hand series shows images of one alpinist’s injured left hand, and the bottom foot series shows images of the other alpinist’s right foot through its heating and recovery time. The temperature scale for each thermograph is uniformly depicted from 10° to 34°C.
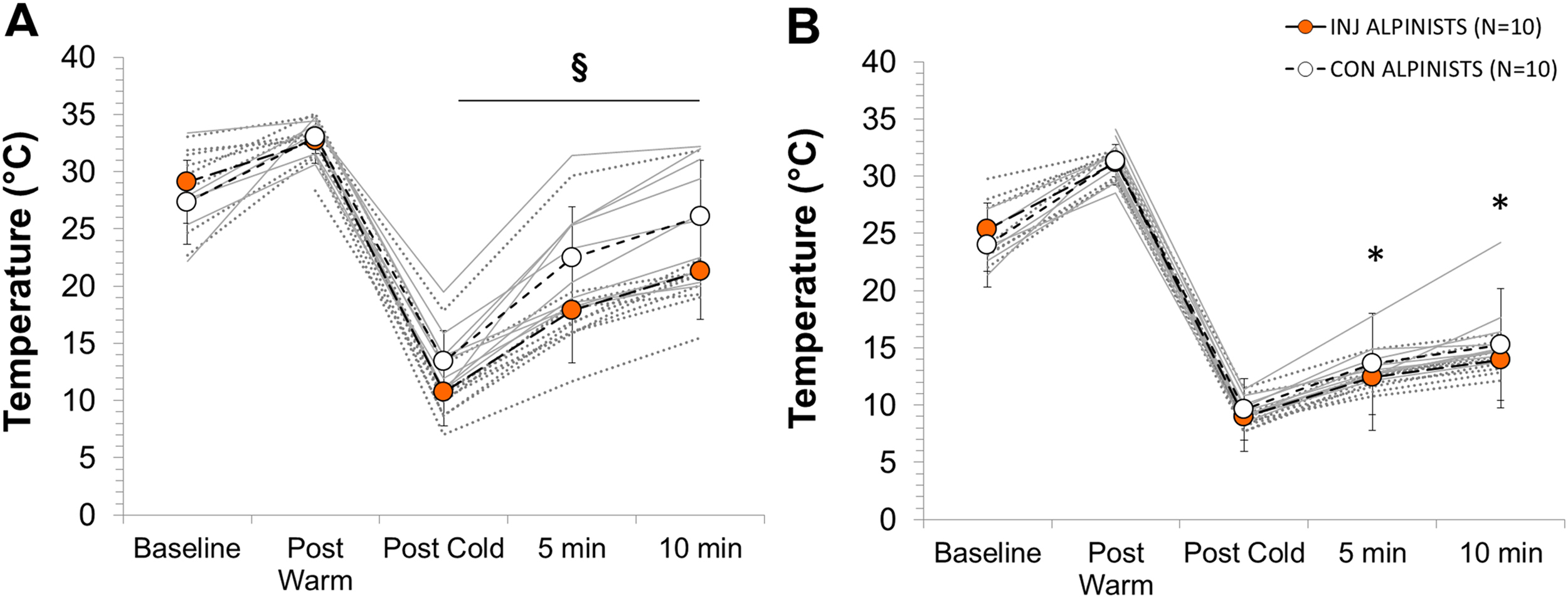
Mean digit temperature changes for (A) fingers and (B) toes separated by alpinist group at baseline, immediately after the 35°C warm bath (post warm), immediately after 30 minutes in the 8°C cold bath (post cold), and during the recovery period 5 and 10 minutes after immersion. Individual alpinist’s data are depicted in gray lines (Injured = dotted gray, Control = solid gray). § indicates a significant interaction effect between alpinist groups during the rewarming recovery phase; * indicates a significant temperature increase compared with post cold time, (P < .05).
The INJ alpinists’ 2nd and 5th finger temperatures were statistically significantly colder by 22% and 20% immediately after the cold bath compared with their CON counterparts (2nd finger 95% CI, –5.7° to –0.09°C; P = .04; 5th finger 95% CI, –5.1° to –0.31°C; P = .03; Figure 3). Mean finger temperature remained 20% (P = .04) and 18% (P = .03) lower for INJ alpinists than for the CON alpinists after 5 and 10 minutes of recovery, respectively. After 5 minutes of recovery there were 2 digits that were significantly cooler in the INJ group compared with the CON group (2nd finger 95% CI, –10.3° to –1.5°C, P = .01; 3rd finger 95% CI, –9.9° to –0.8°C; P = .02). After 10 minutes of recovery, 3 digits were significantly cooler in the INJ group than in the CON group (2nd finger 95% CI, –10.0° to –1.2°C, P = .02; 3rd finger 95% CI, –9.9° to –1.2°C, P = 02; 5th finger 95% CI, –9.1° to –0.2°C, P = .04). However, thumb temperature was never significantly different between alpinist groups at any time (baseline, P = .33; post 35°C bath, P = .30; post 8°C cold bath, P = .09; 5 minutes after test, P = .16; 10 minutes after test, P = .07).
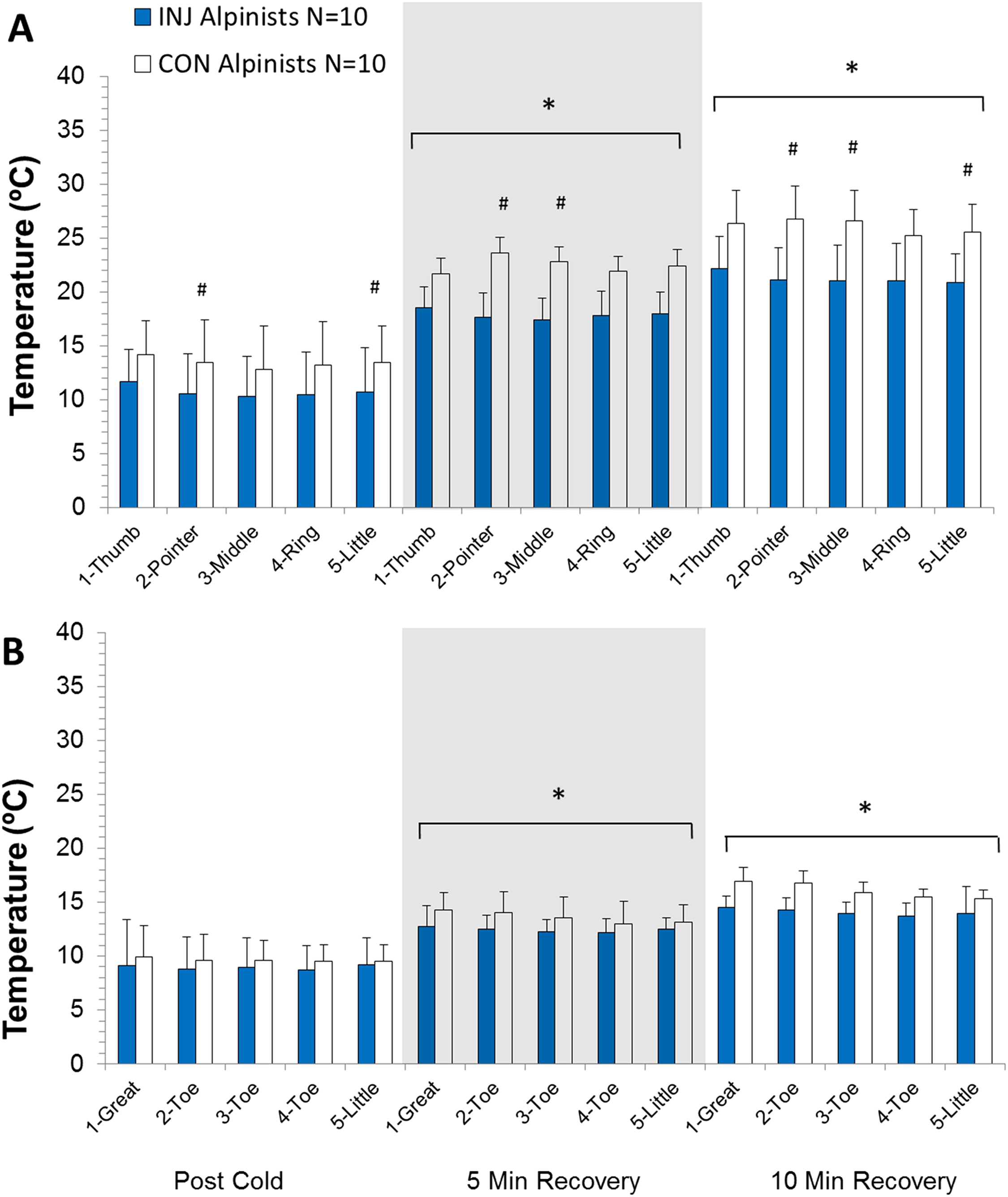
Absolute individual digit temperature changes for (A) fingers and (B) toes during the 10-minute recovery phase of the cold stress test protocol in injured (INJ) and control (CON) alpinists. Shown are changes for 1-thumb, 2-pointer finger, 3-middle finger, 4-ring finger, 5-little finger; the same order for the toes is shown starting with 1-great toe through to 5-little toe. * indicates significant difference in temperature compared with all other times; # indicates significant difference between alpinist groups for that finger (P < .05).
Digit Skin Temperature Differences within the injured alpinist group
A second rationale for this study was to compare the FCI digits of the INJ alpinists with their own, uninjured fingers or toes to determine any intraparticipant differences. Indeed, when comparing the skin temperature of an injured alpinist’s toe (TInj) with that of his or her own corresponding toe on the uninjured side (TCOR), the injured digit was, conversely, slightly warmer immediately after the warm bath by approximately 3% (95% CI, 0.1° to 2.9°C; P = .04) and immediately after the cold bath by approximately 6% (95% CI, 0.004° to 1.000°C; P = .05) than the uninjured toe. For the toes, there were no significant differences between TInj and TCOR after 5 (P = .23) and 10 minutes (P = .32) of recovery. The injured fingers (FInj, n = 6 digits) were approximately 8% cooler than their corresponding fingers (FCOR) at baseline (95% CI, –3.1° to –0.1°C; P = .01) and 7% cooler immediately after the warm bath (95% CI, −4.0° to –0.5°C; P = .02). There were no other significant differences between injured vs corresponding digits, or between the uninjured digits on the same side of the injury vs the corresponding digits on the contralateral side.
Digit Skin Temperature Rates of recovery and correlation to injury age
The hands warmed up (all, 1.2° ± 0.4°C/min; INJ, 1.1° ± 0.2°C/min; CON, 1.3° ± 0.5°C/min; P = .22) at double the rate of the feet (all, 0.6° ± 0.2°C/min; INJ, 0.5° ± 0.1°C/min; CON, 0.7° ± 0.3°C/min; P = .16) irrespective of group, during our 10-minute passive rewarming protocol. Both locations showed no correlational relationship to either how old the alpinist was at the time of testing (fingers R2, .001; P = .96; toes R2, .001; P = .94), or the age of the injury site itself (fingers R2, .112; P = .35; toes R2, .008; P = .81).
Discussion
The present study demonstrates that there were no statistically significant differences in finger or toe skin temperature recovery rates between INJ and CON elite alpinists in response to a follow-up cold stress test. There also were not any differences between injured and uninjured toe digit temperatures within the INJ cohort alone, either immediately after the cold stress test or throughout the 10-minute recovery period. The INJ alpinists’ finger temperatures for the 2nd, 3rd, 4th, and 5th digits were cooler than those of the CON group at various times throughout the cold stress test protocol. Whether these temperature differences at baseline and during the postwarming period constitute clinically significant differences that could predispose these digits to future cold injury is unlikely, given the nonuniform nature of the data observed.
The variable responses observed between fingers and toes and between digits within a given hand or foot highlight the necessity to individually assess each digit’s response to a cold stress stimulus, especially if one is conducting follow-up clinical assessments of previous frostbite or other tissue damage. Vasodilation and vasoconstriction responses are not generalizable between uninjured hands and feet, 24 and the present study builds on this research to suggest that even after FCI that requires amputation, tissue perfusion is also variable between digits. 22 Elite alpinists are generally very physically active, highly trained athletes. Intermittent exposure to high altitude has been shown to increase endothelium-dependent vasodilation, 25 possibly enhancing vascular responses to the cold. However, it is not clear whether peripheral cold adaptation per se occurs at high altitude, although some studies report enhanced finger vasodilation responses after 45 days at an altitude of 5100 m, 26 and after a 3-week Himalayan expedition (3985–6828 m), 10 whereas others report no changes after repeated finger exposure to cold immersion at elevations of 4350 m. 26 Given that in the present study the intergroup differences in finger temperature rewarming were small and not uniform, the clinical and practical relevance of these findings is not clearly demonstrated.
It is also not possible to discern whether adaptation responses would act to modify the interindividual variability regarding susceptibility to FCI, or regarding specific finger-temperature responses during the cold-water immersion. We measured all alpinists while they were not participating in expedition training, so the acute effects of high altitude exposure per se should be a less significant factor in the present study. The alpinists in the cohort group may have had augmented behavioral responses to cold through their high altitude mountain experiences (learning, tradition, expedition experience). However, because these alpinists were not participants with known comorbidities (eg, diabetes, atherosclerosis, vascular disorders) or existing sequelae attributed to the digit amputation (increased cold sensitivity, chronic pain, localized osteoporosis), they may represent the best-case scenario for relatively young, active people who suffer an FCI sustained through leisure pursuits, and not with other predisposing factors of frostbite (alcohol consumption, psychiatric illness, or vehicular trauma). 27
Quantifying dynamic vascular responses in the peripheral circulation to cold exposure is critical when conducting follow-up testing of participants who have had previous cold injuries 22 because these digits may display different rewarming profiles compared with the uninjured counterparts. By determining individual digit responses, this technique has produced results contrary to our initial hypotheses. We believe this communicates a significant message to clinicians and medical personnel: even after considerable tissue trauma after frostbite and amputation, tissue perfusion can be generally well-maintained after recovery, even after significant time has passed since the initial injury was sustained. Historically, cold stress testing has been occasionally used to quantify rewarming patterns in alpinists returning from high altitude sojourns, 10 after military training in cold climates,11,16 outdoor manual labor exercises, 15 and after traumatic ulnar nerve surgery or hand fractures.5,6 Unfortunately, there are little, if any, standardized guidelines to using cold stress testing as a follow-up tool in clinical practice. The variations in hand or foot submersion time, testing 1 finger or all digits, ambient temperature of the testing environment, hydration status of the individual, and even the amount of clothing worn by the patient can each affect test results. This variability in testing methodology has been criticized in the literature of late.28,29 What is clear from the present study is that tissue responses can vary significantly depending on either the tissue’s vasodilation or vasoconstriction response to a warm and cold stimulus; thus, we maintain that any cold stress test must include a warming phase to standardize tissue baseline temperatures and a cooling phase of at least 10 minutes to observe sufficient differences between digit sites, as well as a passive rewarming phase, preferably at least 10 minutes after the cold stimulus has been completed. In general, rewarming responses seen in a single digit are not generalizable to other digits, either in the hand or feet, 24 which is a significant limitation in the cold physiology literature, in which general practice has been to immerse only 1 finger (usually the middle finger) up to the distal phalanx. 29 –31 It has been suggested that a useful application of cold stress testing could be to preemptively assess one’s risk to cold injuries on an individual basis. 29 Whether current resistance indices of frostbite 30 are accurate predictors of future cold injury remain to be proven.
The INJ alpinists acted as their own control for a portion of this research, and thus it could be argued that the digits matched to their opposite, uninjured counterparts may inherently have different vasomotor reactions to the cold stress test by virtue of simply being on the contralateral side of the body. Indeed, there has been literature investigating whether IR thermography of participants with peripheral nerve injury had uniformly equivalent responses to skin temperature changes on both sides of the body. 32 The researchers found that in normal persons, differences in skin temperature were 0.24° ± 0.07°C between sides, whereas in patients with peripheral nerve injury, this difference deviated by approximately 1.55°C, ie, by approximately the same factor as we found between the TInj and TCOR in our INJ alpinists’ fingers. Participants also completed a 5-minute immersion in warm 35°C water for the purposes of standardizing all digit temperatures before cold immersion began. Although it is unlikely this protocol affects subsequent digit vasoconstrictor responses per se, it should be noted that differences in cold stress test protocols do exist within the literature.
We found small, but statistically significant temperature differences in some fingers, although there were only 6 injured finger digits within the entire INJ alpinist population. This study was based on literature in the clinical and physiological fields, which reported differences testing populations of between 7 and 12 persons, which are considerably fewer than large, placebo-controlled clinical trials. However, it is not uncommon in thermal physiology research to have fewer participants if uniform directional changes in a dependent variable are anticipated. Notably, for obvious ethical reasons, it is not possible to induce a cold injury in healthy volunteers to directly test whether an FCI may affect blood flow responses; hence, the design and unique population studied here. Finally, this study investigated a target group of healthy, unmedicated, elite athletes, and conclusions drawn from this study are not meant to be generalized to clinical patients or military personnel who have suffered non-FCI, clinical compressive neuropathies, Raynaud’s disease, or postoperative care, and who continue to experience sequelae during their recovery period.
Conclusions
There were no significant differences in toe or finger temperature recovery rates between previously injured and uninjured frostbite digits after a follow-up cold stress test. Although the injured alpinists exhibited modestly cooler fingers at baseline and after warming than the uninjured alpinists, the practical relevance of this difference remains to be established. A prior FCI that requires amputation may not necessarily predispose that digit to altered vascular function to a repeat cold exposure.
Footnotes
Acknowledgments
The authors would like to thank the participants for altruistically giving their time and experience to this project. The research was supported, in part, by a grant from the Swedish Armed Forces (No. 922: 0905). Gratitude is expressed to Dr. Metka Milčinski for providing valuable feedback on manuscript preparation and to Mr. Adam McDonnell for his assistance with data collection.
