Abstract
Amatoxin poisoning from the genus Lepiota may have a deadly outcome, although this is not seen as often as it is from the genus Amanita. In this report, we present a patient who was poisoned by a sublethal dose of Lepiota brunneoincarnata mushrooms. The patient was hospitalized 12 hours after eating the mushrooms. The patient’s transaminase levels increased dramatically starting on day 4. Aspartate transaminase peaked at 78 hours. Starting at 1265 IU/L, alanine transaminase peaked at 90 hours at 5124 IU/L. The patient was discharged on day 8 to outpatient care, and his transaminase levels returned to normal ranges in the subsequent days. A toxin analysis was carried out on the mushrooms that the patient claimed to have eaten. Using reversed-phase high-performance liquid chromatography analysis, an uptake of approximately 19.9 mg of amatoxin from nearly 30 g of mushrooms was calculated. This consisted of 10.59 mg of α-amanitin, 9.18 mg of β-amanitin, and 0.16 mg of γ-amanitin. In conclusion, we present a patient from Turkey who was poisoned by L. brunneoincarnata mushrooms.
Keywords
Introduction
Amatoxins’ responsibility for nearly all fatal mushroom poisonings increases their importance. Amatoxins inhibit nuclear RNA polymerase II, and this inhibition results in impaired protein synthesis and cell death. 1 Because the liver is an organ in which protein synthesis and cell turnover are high, it suffers the most distinct damage in amatoxin poisonings, as happened to our patient. 2 This damage is directly proportional to the dose of toxin received. 3 Liver damage is characterized by massive centrilobular necrosis and in many cases by acute hepatic failure with subsequent complications, including hepatic comas, coagulation disorders, and secondary renal failure.4,5
Amatoxins are contained in some Amanita, Galerina, and Lepiota species. From the genus Amanita, particularly Amanita phalloides, 6 Amanita virosa, and Amanita verna, 7 major causes of poisonings are attributable to their high amatoxin content. Some Lepiota species (Lepiota brunneoincarnata, Lepiota brunneolilacina, Lepiota helveola, Lepiota josserandii) are also very toxic, but are less frequently involved in human poisoning than the Amanita species of current interest.1,8 Amatoxins are very toxic and primarily consist of α-amanitin (AA), β-amanitin (BA), and γ-amanitin (GA).6,7 L. brunneoincarnata mushrooms also contains amanitin, but intoxication from them is very rarely seen in Turkey. 9 It is not fully known which amatoxins in what amounts L. brunneoincarnata mushrooms contain or what amount of consumption of this mushroom may be lethal in humans. In this report, we present a case known to be the first in Turkey. The patient was poisoned in a sublethal dose by this mushroom and was treated successfully.
Case Report
A 39-year-old male patient weighing 72 kg was admitted to the emergency room of a private hospital with diarrhea, nausea, vomiting, abdominal pain, and dehydration. In his anamnesis, he stated that on October 18, 2013, he collected 2 types of natural mushrooms, mixed all of these mushrooms, cooked them, and ate them; no one else ate this meal. When he came to the emergency room, almost 12 hours had passed since he had eaten the mushrooms. This patient’s initial vital signs and physical examination were normal, other than dehydration. The patient’s aspartate aminotransferase (AST) was 30 IU/L, his alanine aminotransferase (ALT) was 34 IU/L, his total bilirubin was 0.5 mg/dL, his direct bilirubin was 0.2 mg/dL, his total protein was 8.1 g/dL, his albumin was 4.3 g/dL, his prothrombin time (PT) was 11.9 seconds, and his international normalized ratio (INR) was 1.03. His hepatitis B surface antigen, hepatitis B surface antibody, hepatitis B core antibody, immunoglobulin M, and antihepatitis C virus antibody were found to be nonreactive. His lactate level, complete blood cell count, electrocardiograph, posteroanterior lung radiography, and complete urine test were normal.
A nasogastric tube was put in place, and gastric lavage was performed on the patient. Activated charcoal was initiated and continued at a dose of 50 g every 6 hours for 3 days. The patient was rehydrated via intravenous administration of 0.9% sodium chloride and 5% dextrose to guard against the risk of hypoglycemia. The patient was given acetyl cysteine (150 mg/kg for 1 hour, followed by 50 mg/kg for 4 hours, followed by 150 mg/kg for 16 hours), ranitidine (50 mg, 4 times a day), vitamin B (250 mg/3 mL, once a day), vitamin C (500 mg/5 mL, once a day), corticosteroid (40 mg, once a day), fresh frozen plasma (15 mL/kg, once a day), and vitamin K (1 mg, once a day).
The patient, who had been monitored and treated in a medium-sized hospital, was referred to a university hospital on the fourth day for more comprehensive treatment because his liver enzymes had increased. During the period in which the patient was being monitored in the emergency internal diseases intensive care unit of the university hospital, his relatives contacted our Clinical Pharmacology and Toxicology Unit through social media after they had searched on mushroom poisonings on the internet. The next day, we went with the patient’s relatives to the place where he had collected the mushrooms (Turkey, Sakarya, Kaynarca district, on soil, in pasture, 41°1′53.17″N-30°18′34.91″E, 60 m, October 25, 2013, Akata 5992), and we collected some of these described mushrooms (Agaricus bisporus and L. brunneoincarnata). The patient and his relatives confirmed that these were the suspicious mushrooms he had eaten. Five L. brunneoincarnata mushrooms in the sizes and amounts he said he had eaten were set aside to be analyzed. These mushrooms were identified macroscopically as L. brunneoincarnata, and their identification was verified microscopically as well (Figure 1). 9 The details of the mushrooms are as follows: family, Agaricaceae Chevall; species, L. brunneoincarnata Chodat & C. Martín 1889; cap, 20 to 40 mm, convex to planoconvex; scales of cap, brownish to chestnut brown on whitish pink background; stem, 20 to 40 × 5 to 10 mm, rather short, whitish above the ring zone, pink vinaceous below the ring zone and covered by brownish scales; gills, whitish to cream, free and crowded; flesh; thin and whitish; basidia, 18 to 22 × 6 to 7 µm, clavate; spores, 7 to 9.5 × 4 to 5 µm, ellipsoid to almond shaped and hyaline; and ecology, grows on soil, among grasses, in meadows and pastures, often close to woodland edges, in late summer to autumn.10,11 First the wet weights (29.9 g) of the 5 mushrooms that were set aside were measured, and then their dry weights (2.82 g) were taken after a drying procedure. The toxin amount of the representative mushrooms was analyzed using the method previously described.6,7 It was calculated that the patient may have ingested a total of 19.93 mg of amatoxin; 10.59 mg of this was AA, 9.18 mg was BA, and 0.16 mg was GA.
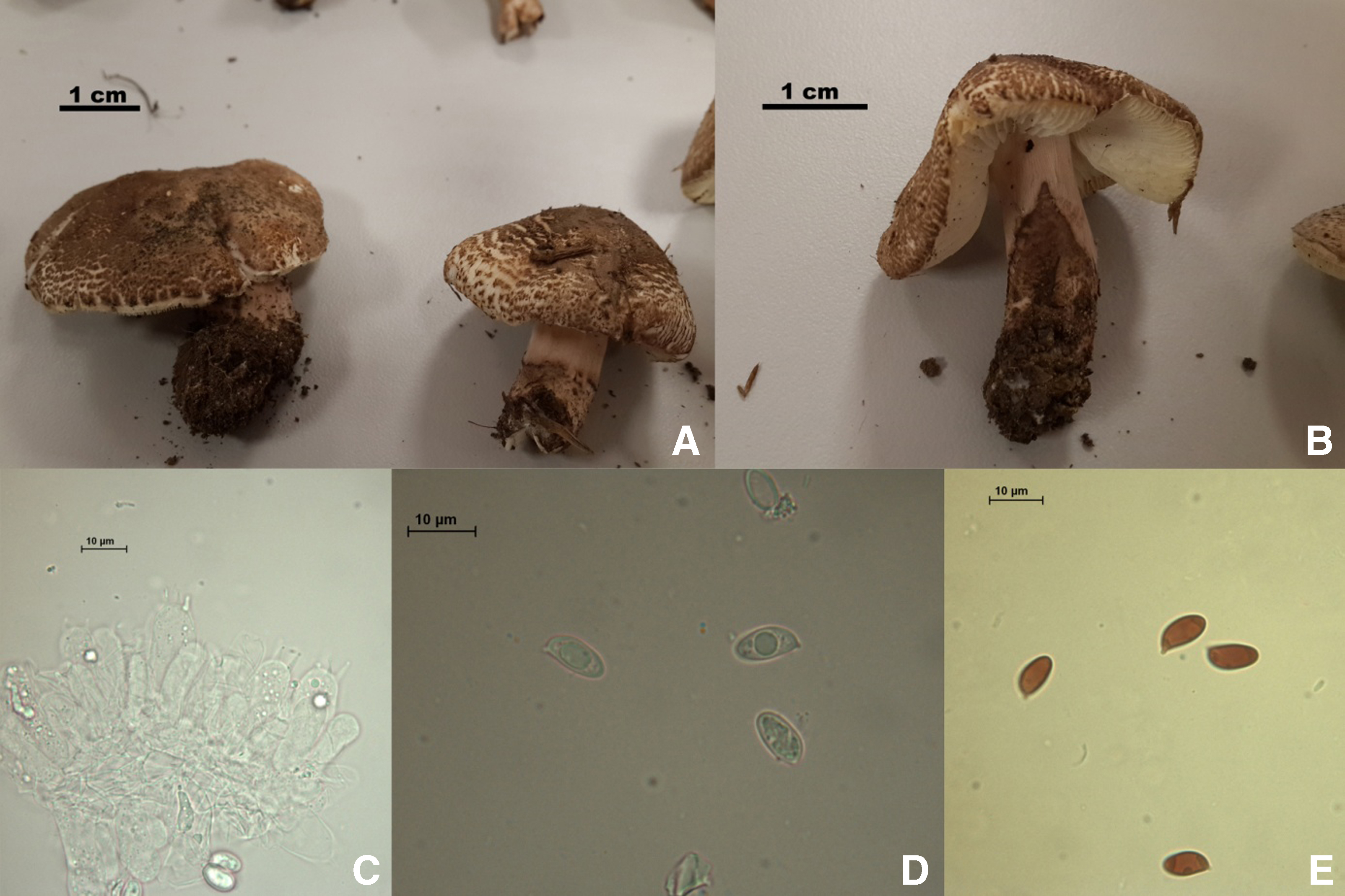
Macroscopic and microscopic features of the Lepiota brunneoincarnata mushrooms collected from the region where the patient picked mushrooms. A, B, Fruiting body. C, Basidia. D, Spores. E, Dextrinoid spores. 9
The AST and ALT values of the patient started to go up dramatically (AST, 1265 U/L; ALT, 1430 U/L) 78 hours after he ate the mushrooms and continued to go up dramatically during day 4 (Figure 2). The ALT level increased during day 4 (1925 IU/L) and reached a peak value at the end of the day at approximately 90 hours (ALT 5124 IU/L). On day 5, the patient’s transaminases levels entered a general declining trend, and on day 8, when he was discharged, they were almost normal (AST, 59 IU/L; ALT, 1261 IU/L). On day 25, when he was invited for a follow-up check, his AST and ALT values were at normal levels.
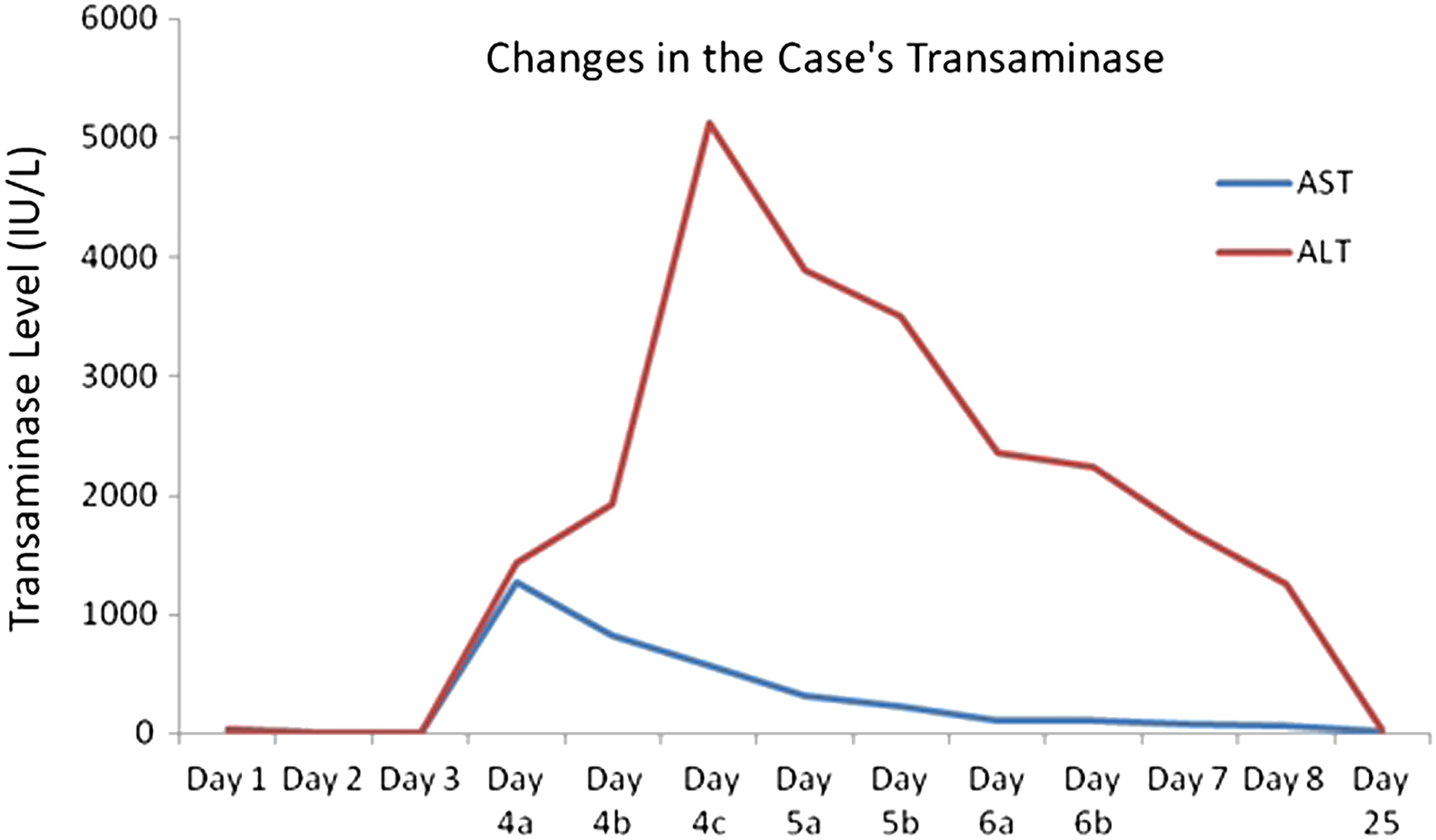
Change in the patient’s transaminase levels. ALT, alanine transaminase; AST, aspartate transaminase.
The patient was icteric on day 3, and his total bilirubin value reached its highest level (17.3 mg/dL) on day 4 and decreased during the following days. Although the patient’s creatinine values remained low during his treatment, no apparent renal problems were observed, and his creatinine returned to normal during the following days. The patient’s PT and INR values on day 4 reached as high as 18.2 seconds and 1.6, respectively, but decreased during the following days. The patient was discharged on his request at the end of day 8, when he was recovered clinically. He was checked in the outpatient clinic 17 days later.
Discussion
A large number of fatal mushroom poisoning incidences occur every year in Europe and America, and mushrooms containing amatoxin are blamed in almost all of them. 12 Unless their macroscopic differences are explored very carefully, as in our case, these mushrooms can be confused with edible mushrooms (Agaricus spp such as Agaricus campestris, Agaricus silvicola, and Agaricus arvensis; Lepiota naucina; and Leucoagaricus leucothites).
Although the lethal dose of amatoxins in humans is not very well known, we determined in a recent case report of a patient who was poisoned by A phalloides that an amatoxin intake of more than around 0.32 mg/kg per oral dose could be lethal. 13 An amatoxin exposure a little less than that, 0.28 mg/kg per oral dose, was estimated by patient history in this case. Making the calculations and evaluations based on patient’s history can be considered as limitations of the study. However, it should also be noted that calculations were done after taking the patient’s self-report and family confirmations.
Paydas et al 14 described 27 successive mushroom poisoning cases that occurred in a region called Middle Taurus in southern Turkey. The patients were treated for 14 days after eating a mushroom identified as a species of Lepiota. Another Lepiota case, again from Turkey, was reported by Işiloğlu and Watling. 15 However, both of these studies involved mushroom intoxications from Lepiota species (Lepiota castanea, L helveola) other than L. brunneoincarnata.
A prolongation was seen in the coagulation parameters of our patient, especially starting in the middle of day 4, but they returned to normal during the subsequent days. Similarly, no significant renal problems were seen in our patient. Although 14 of 27 intoxicated patients monitored by Paydas et al 14 lost their lives as a result of liver failure, the publication stressed that there were no deaths associated with renal failure. In the case reported by Kervégant et al 16 relating to a child who was discharged on day 11, the child’s AST and ALT values (1018 IU/L and 3205 IU/L, respectively) increased, and PT time was prolonged (18 seconds). However, the child’s transaminase values were not elevated as high as those of our patient. In a recent study, Eren et al 17 showed that patients who had died had very high ALT (2345–4048 IU/L) and AST (2075–3464 IU/L) levels. It was reported that patients experienced hepatic comas after ALT and AST values increased, demonstrating a significant relationship between mortality and ALT and AST levels. As a result, ALT and AST levels can be used as an important and practical parameter that indicates the degree of mushroom poisoning. When we compare these values, which have been determined based on A phalloides poisonings, with those of our patient, we see that AST increased to just below these levels on day 4. Interestingly, the elevation in the patient’s ALT was even higher than those in fatal poisonings.
When compared with A phalloides poisonings, this dramatic increase in transaminases levels and different prognosis could be related to possible differences in the contents of L. brunneoincarnata mushrooms.18,19 There has been no comprehensive study until the current report about the toxin composition of this mushroom. In the mycological literature on Lepiota mushrooms, 24 species are presumed to be amatoxin-producing. The presence of amatoxin in these species, including L. brunneoincarnata, has been shown using thin-layer chromatography. 4 ,20,21 However, there is no information in these studies as to which amatoxins are present and in what amounts. One of the frequently used methods of amatoxin analysis today is the reversed-phase high-performance liquid chromatography method because it produces precise and specific results in identifying toxin content in mushrooms. With this method, toxins can be analyzed in blood, urine, and various organs in cases of poisoning. 22
The analysis we carried out showed that the patient may have ingested approximately 19.93 mg of amatoxin from approximately 30 g of L. brunneoincarnata mushrooms. Although it is argued in some studies23,24 that the amount of L. brunneoincarnata that must be eaten for fatal hepatotoxicity is at least 100 g, a result that differs from our report, it is obvious that there is a need for more detailed studies on the subject. In amatoxin intoxications, the clinical manifestation is closely associated with the species of the mushroom eaten and the amount eaten as well as the characteristics of the person who is poisoned. In particular, personal tolerance, body weight, and general health influence the clinical course of a poisoning. 22 However, in view of the laboratory findings of ALT levels exceeding 5000 IU/L, an elevated bilirubin level, and prolonged coagulation parameters as well as the observations from clinical monitoring, we can say that the dose of amatoxin consumed by our patient came very close to a lethal dose.
In conclusion, we presented a case from Turkey involving a patient who was poisoned by a sublethal amatoxin dose from L. brunneoincarnata mushrooms and who was successfully treated. Because we could obtain correct and sufficient anamnesis regarding this mushroom, we were able to conduct advanced analyses on the mushroom eaten. This demonstrated once more the importance of having poison centers with teams experienced in mushrooms in regions where many natural mushrooms grow and are often consumed and in which the identification and toxin analysis of mushrooms can be carried out. Such centers will provide benefits to clinicians in treating poisonings.
Footnotes
Acknowledgment
The authors would like to thank Jilber Barutciyan for his valuable support in the preparation of the manuscript.
