Abstract
Objective
This study was designed to evaluate the effectiveness of a new protocol implemented to standardize snakebite management in Iran.
Methods
In this study, 27 patients treated according to the new protocol in 2012 (P+) were compared with 22 patients treated according to the previous modality in the year before implementation of the protocol (P–) in Mashhad Medical Toxicology Centre (MTC). Demographic characteristics and treatment details of all patients were recorded prospectively. Envenomation severity of each victim was assessed according to snakebite severity score (SSS).
Results
After implementation of the protocol, a smaller percentage of patients received antivenom (AV) therapy (78% vs 95%; P = .079). In spite of no significant difference in baseline severity of envenomation between the 2 groups (SSS [mean ± SD], 34.8 ± 18.1 vs 35.5 ± 17.4; P = .801), the P+ group received significantly fewer AV vials (8.4 ± 6.8 vs 12.1 ± 5.6 vials; P = .042) and had a significantly shorter length of hospital stay (2.2 ± 1.5 vs 3.2 ± 1.8 days; P = .027). Moreover, smaller proportion of P+ patients experienced recurrence of venom-induced effects; however, the difference was not significant (18.5% vs 36%; P = .159). The reduction in use of antiallergy treatments to prevent or treat acute hypersensitivity reactions approached statistical significance (41% vs 68%; P = .051). These findings denote a reduction in AV use of approximately 4 vials and a reduction in hospital stay of 1 day for each patient, which translates to approximately $196/patient in healthcare cost savings.
Conclusions
Implementation of a snakebite management protocol at MTC reduced overall antivenom usage, use of antiallergy interventions, and length of hospital stay.
Introduction
Snakebite is a toxicologic emergency that was primarily treated according to anecdotal evidences, local experiences, and empirical basis until recent decades. 1 –3 Thus, considerable attention has been devoted to development of unified integrated protocols for standardizing management of snakebite in different parts of the world. 3 –6 In Iran, a comprehensive protocol has been recently introduced that conjoined antivenom (AV) dosage plan and recommendations for supportive treatments. 7 This is the first Iranian protocol that has been designed to standardize the AV dosage and indications to overcome shortcomings of previous guidelines and to clarify indications of supportive treatments.
Annually, snakebite involves 4500 to 6500 victims in Iran, causing 3 to 9 deaths.2,8 The most prevalent medically important snakes in this country are Echis sochureki, Macrovipera lebetina obtuse, Pseudocerastes persicus persicus, and Naja oxiana: the first 3 belong to the Viperidae family, and the fourth is an Elpidae snake.9,10 The current commercially available AV in Iran (Polyvalent Snake Antivenin, Razi Serum and Vaccine Research Institute, Iran) that has been manufactured during the past 5 decades is capable of counteracting the venoms of these snakes. 11
Mashhad Medical Toxicology Centre (MTC) in Imam Reza Hospital is a referral medical toxicology department in northeast Iran that admits 25 to 60 snakebite victims each year with less than 1% mortality rate. 2 Snakebite events in this part of the country mostly occur during April to late October. 12 During the past 3 decades, a local treatment modality for snakebites mainly designed according to western references has been practiced in MTC.2,8 Although the treatment modality has been effective in terms of saving many lives, its cost-benefit has remained obscure. Moreover, its effectiveness has been affected by differing interpretations of the grading of envenomation severity, undefined therapeutic response (or initial control), and unspecified indications for supportive treatments. 7 Hence, through elaborating the modality, rectifying its defects, and enhancing neglected aspects, an integrated protocol has been developed in MTC by a focus group of medical toxicologists and expert physicians in other areas of medicine. 7 The new protocol includes: 1) a grading scale that enabled a more objective assessment of victims (snakebite envenomation severity scale, SESS); 2) an algorithm consisting of planned course of action and AV dosing (Figure); and 3) a table of instructions for supportive treatments.
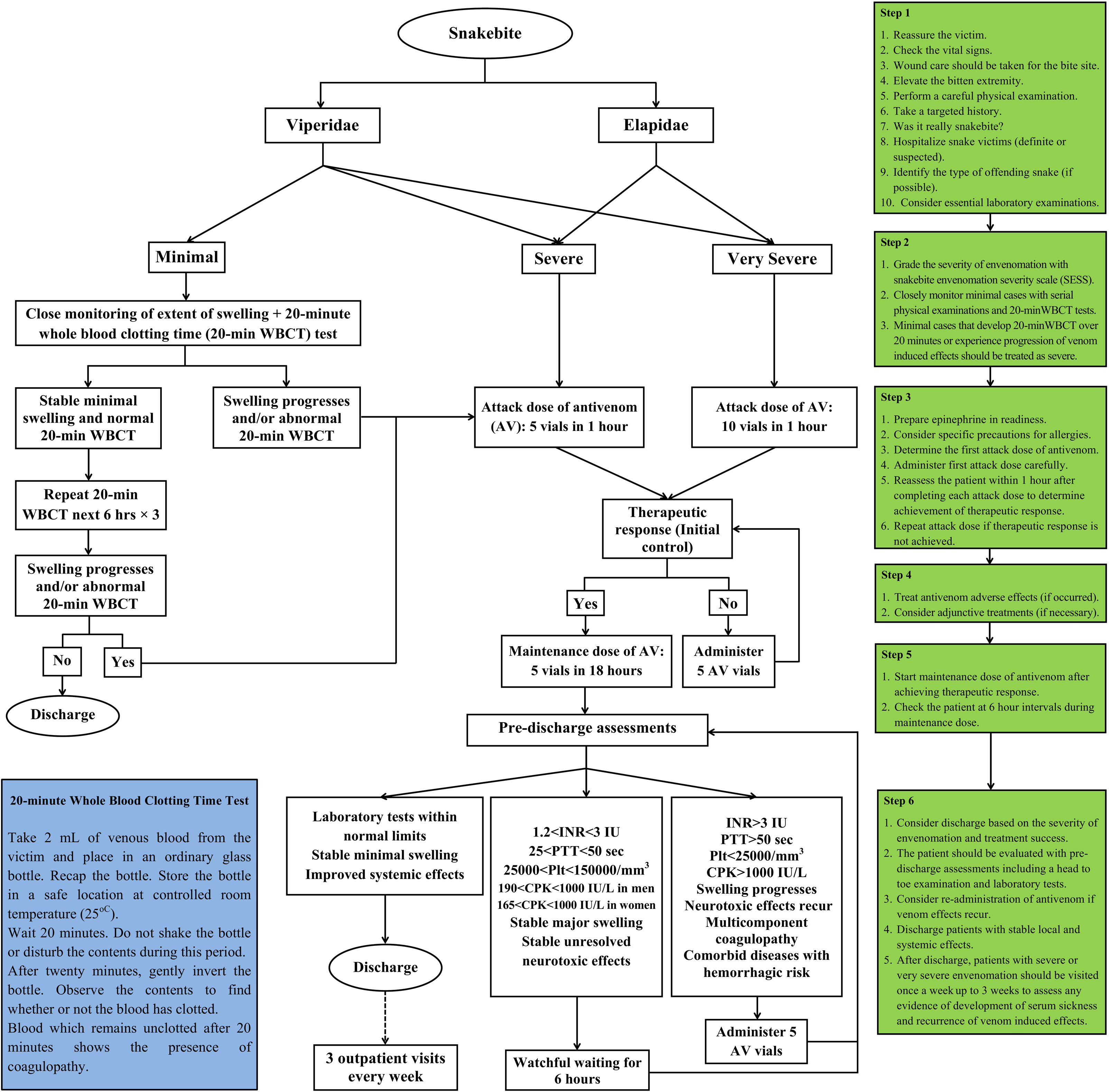
Algorithm for management of snakebite envenomation in the new protocol (adapted from Monzavi et al 7 with permission). AV, antivenom; CPK, creatine phosphokinase; INR, international normalized ratio; Plt, platelet count; PTT, partial thromboplastin time; SESS, snakebite envenomation severity scale; WBCT, whole-blood clotting test.
The aim of this study was to evaluate the effectiveness of the new protocol according to the overall AV volume administered to patients, morbidities, and duration of admission after its institution in 2012.
Methods
Design and Setting
This was a prospective comparative study of snakebite victims admitted to MTC during 2011 and 2012. The study was started prospectively in 2011 with precise data collection of patients who were treated according to previous modality and continued with careful observation of the patients treated according to the new protocol in 2012.
Patients
The patients treated according to the new protocol between April 1, 2012, and October 30, 2012 (P+), were compared with patients who received treatment according to the previous modality in the same period of the year before implementation of the protocol (P–). Demographic characteristics and clinical manifestations of all patients as well as treatment details were recorded in a predesigned checklist by 2 of us (S.M.M. and B.D.). At presentation, the envenomation severity of each victim was also assessed by these researchers according to the snakebite severity score (SSS). 13
Treatments
To start attacking the venom and promptly reversing the venom effects, a specified amount of the AV called the attack dose (or initial dose in other internationally recognized protocols) should be given to the patient at a relatively rapid rate. The attack dose may be repeated according to the patient’s condition (subsequent attack dose). For maintaining a steady AV level in the blood to prevent venom–AV mismatch and recurrence of venom effects, the maintenance dose of AV should be infused at a slow rate. Differences between the 2 treatment plans rely on the definition of these doses, the rate of AV administration, the time to repeat antivenom, indication of supportive treatments, etc. The characteristics of the new protocol and the previous modality are summarized in Table 1. As shown, in the new protocol compared with the previous treatment modality:7,8 The grading system (SESS) has been defined with more details and has been simplified into 3 grades. The extent of <2.5 cm has been defined as the threshold of edema that does not need to be treated. However, for bites on digits, any level of edema has been determined as an indication for AV administration. The systemic venom effects have been clarified with more detailed definitions in the grading scale. The highest AV volume at each attack dose has been limited to 10 vials, half of the amount recommended in the previous treatment modality. The infusion rate of the attack dose has been increased. Therapeutic response has been defined and its assessment has become mandatory. Administration of subsequent attack doses depended on the failure to achieve the therapeutic response. The maintenance dose and the dose for recurrence of venom-induced effects have been fixed at 5 vials. The maximum of total attack doses (upper limit of AV attack dosage) has been set at 20 vials except for uncontrollable bleeding and critically ill patients. The skin test has been discontinued. Pretreatment for immediate allergic reactions (IARs) has been limited to severe atopic syndrome and previous sensitization to biological products. The time to discharge has been defined as 18 hours after admission after 4 normal 20-minute whole-blood clotting tests (20-minute WBCT) for minimal cases and 24 hours after stabilization of severe and very severe cases.
Comparison of the new protocol with the previous treatment modality
ECG, electrocardiograph; INR, international normalized ratio; PT, prothrombin time; PTT, partial thromboplastin time; SBP, systolic blood pressure; WBCT, whole-blood clotting test.
Ethics
The study was approved by the ethics committee of the Mashhad University of Medical Sciences. The protocol was designed according to previous regional studies, local experience, and internationally recognized textbooks, and if controversy existed, the focus group selected the most appropriate and conservative treatment. With a feedback from the focus group, the new protocol was implemented in MTC based on a staffing decision. All patients received meticulous care under the supervision of the researchers. It had also been planned to stop AV administration if the patient showed any sign of IARs.
Statistical Analysis
To compare the means of normally distributed dependent variables between P+ and P– patients, we performed independent samples Student’s t tests, and for nonnormally distributed dependent variables, the Mann-Whitney U test was used. For comparing proportions between the 2 groups, the χ2 test was used. Analyses were done using SPSS for Windows (SPSS Inc, Chicago, IL). Probability values of less than .05 were considered statistically significant.
Results
Epidemiological Profile of Snakebite Patients in Both Years
Forty-nine victims (65% men) were enrolled during the 2-year period of the study. Most of the victims (39%) were young adults (20–39 years old). The majority of bites (90%) occurred in rural areas (Table 2). Echis sochureki was responsible for most bites (39%), followed by Macrovipera lebetina obtuse (16%). The identity of offending snakes in the remaining cases could not be established (45%). In the greater number of events, upper extremities (51%) were affected followed by lower extremities (45%). No or fewer than 10 AV vials in total were administered for a higher proportion of patients (61%). Most of the patients were discharged in less than 3 days of hospital stay (75%). None of the patients required intensive care unit admission, and no deaths occurred.
Demographic features of patients, circumstances of the bites, treatments, and outcomes (n = 49)
ICU, intensive care unit.
Attack dose + maintenance dose.
Analysis of Effectiveness
Twenty-two patients were treated according to the previous modality (P–) and 27 patients after implementation of the new protocol (P+). The 2 groups showed no significant difference in age (P = .894) and sex distribution (P =.703). No significant difference in the extent of edema (P = .085), frequency of bites on digits (P = .449), and the development of coagulopathy (P = .336) was noted between the 2 groups (Table 3). In addition, in both years, no posttreatment complications including renal failure, bleeding complications, and neurologic sequelae were seen except a case with dermal necrosis that was treated with previous modality.
Comparative analysis of patients treated with the new protocol and previous modality
AV, antivenom; IARs, immediate allergic reactions.
Thrombocytopenia and/or increased coagulation tests.
Remanifestation of venom effects after achievement of therapeutic response.
Administration of corticosteroid/chlorpheniramine (± epinephrine) either as pretreatment or treatment for allergic reactions.
Pretreatment for IARs (administration of corticosteroids or chlorpheniramine before AV therapy) was significantly reduced by adherence to the new protocol (33% vs 64%; P = .035). Nevertheless, the occurrence of IARs was not significantly different between the 2 groups (P = .678). Moreover, the reduction in overall anti-IAR care delivered to the patients (either as pretreatment or treatment) approached statistical significance (41% vs 68%; P = .051).
After implementation of the protocol, a smaller percentage of patients received AV therapy, although the difference was only close to statistical significance (78% vs 95%; P = .079). In spite of no significant difference in baseline severity of envenomation between the 2 groups (SSS [mean ± SD], 34.8 ± 18.1 vs 35.5 ± 17.4; P = .894), the P+ group received significantly fewer AV vials (8.4 ± 6.8 vs 12.1 ± 5.6 vials; P = .042) and had a significantly shorter length of hospital stay (2.2 ± 1.5 vs 3.2 ± 1.8 days; P = .027). In addition, smaller proportion of P+ patients experienced recurrence of venom-induced effects; however, the difference was not significant (18.5% vs 36%; P = .159).
These findings denote approximately 4 vials reduction in AV use and 1 day reduction in hospital stay for each patient. As the production cost of each AV vial (Razi antivenin) is $28 US and charges for 1-day stay in public hospitals are about $84 US in Iran, 14 the implementation of the protocol resulted in approximately $196 US/patient savings in healthcare cost.
Discussion
Snakebite as a medical toxicologic emergency has often been associated with excessive fears and concerns for both the patient and the treating physician. In the past, some physicians were inclined to administer high doses of AV for all victims to not miss any seemingly minimal case with underlying severe envenomation. In addition, in some old-fashioned treatment modalities and guidelines that were generally based on unreliable subjective experiences and evidences from countries with higher incomes or more dangerous snakes, high doses of AV have been recommended. Moreover, in some developing countries, the cost of manufacturing and distribution of AV products have been covered by the governments. These have resulted in overtreatment of the victims and waste of medical resources. 6 ,8,15,16 In this study, we showed that implementation of an updated evidence-based protocol can reduce unnecessary treatments, morbidities, and overall healthcare costs.
The epidemiological profile of patients in the present study generally resembles the previous reports from Iran and other countries. 6 ,8,12,15,17–20 Snakebite mostly affects a young male population. 12 ,17–20 It is an environmental threat especially in rural areas. 12 ,18,21 The extremities are the most affected parts of the body. 6 ,8,12,15,17–20 However, in some studies including the present one, the upper extremities were more involved compared with the lower extremities. 8 ,12,17,18 Conversely, in reports from India, Bangladesh, and Afghanistan, the lower extremities were mostly affected. 6 ,19,20 This discrepancy is probably related to clothing cultures in these countries, as in the Indian subcontinent, the lower extremities are less covered with clothes and are easily exposed to bites. In this study, Echis sochureki was the most common offending snake among identifiable cases. Echis sochureki is highly distributed and is responsible for most snake attacks to humans in the region. 2 ,9,10,15,18 To reverse the effects of its venom, Razi Antivenin has shown to be a potent product. 7 ,8,11,18 It is a F(ab′)2 polyvalent AV derived from equine serum through a costly manufacturing process. 11 Hence, it is potentially a hypersensitivity-inducing agent, especially in high volumes. 21 These facts emphasize the necessity of rationalizing the AV usage and maximizing healthcare resources. In recent years, great efforts have been focused on developing national protocols for snakebite management in different countries to achieve these goals. 4 –7
In the United States, Crotalidae Polyvalent Immune Fab (CroFab) has replaced the Wyeth Antivenin (Crotalidae) Polyvalent (ACP) since 2000. 22 Premarketing studies recommended 4 to 6 vials as the initial dose. 23 In a postmarketing study by Dart et al, 22 6 vials (2 vials every 6 hours) were recommended as the maintenance dose to prevent recurrence of venom-induced effects. In light of these studies, Weant et al 4 proposed a multiaspect protocol to ensure appropriate patient evaluation, CroFab use, and follow-up in 2010. The protocol led to a decrease of 2.2 vials in AV use and 0.8 day shorter hospital stay, which meant $2000 US/patient healthcare cost savings. 24 Lavonas et al 5 reassessed and augmented the protocol with other practical aspects in 2011.
In India also, many efforts have been made toward a rational AV usage. In 1999, Tariang et al 25 proposed a low-dose AV regimen instead of a high-dose conventional regimen. They showed a savings of 5 AV vials (1000 rupees or $23 US per patient) with no difference in morbidities and mortalities by applying their new dose regimen. In 2004, Srimannarayana et al 26 also showed that lower-dose AV regimens compared with conventional regimens can significantly reduce AV use and recurrence of venom effects with similar mortalities. Considering this information, Ghosh et al6,27 reassessed available guidelines and devised a clinical protocol for standardizing snakebite management in West Bengal in 2008. Their protocol resulted in a 66% reduction in AV use, equivalent to $150,000 US annual cost saving, and a 24% reduction in mortality rate.
In Iran, it was shown that 5 vials of AV as an attack dose is effective and adequate to control venom effects in most cases.8,28 Nevertheless, in the previous accepted modality, very high volumes of AV was proposed.2,8 Therefore, the new protocol has been devised to reduce variations in practice and normalize AV usage. 7 In the present study, we showed that the protocol can lower the AV use and duration of admission. These have been achieved with clear definitions of AV therapy indications and end points, the indications of repeating AV, uniform attack and maintenance AV doses, and applicable severity grading scale. According to the new protocol, patients without systemic manifestations and less than 2.5 cm of local edema will be graded as having minimal severity of envenomation and will not receive AV. 7 Instead, they will receive 18 hours of close observation, and within this time frame their coagulation status will be checked with a 20-minute WBCT every 6 hours. The value of the 20-minute WBCT to guide the successful management of systemic envenomation has been ascertained in earlier studies.29,30 When compared with prior practice in which any minor edema was an indication for AV, the new protocol limits administration to victims with more than 2.5 cm of swelling or systemic findings. Nevertheless, in the new protocol, any level of edema on digits is still an indication for AV administration similar to the previous modality.2,7 Also, in the World Health Organization guideline for the management of snakebites, AV therapy has been recommended for any swelling after bites on the digits (toes and especially fingers). 31 The reason for this exception is that the subtissue compartments of the digits are surrounded with limited space and tight fascial membranes, and subsequent to a swelling, the elastic limit of the skin can be rapidly reached and so the possibility of tissue necrosis and compartment syndrome is higher.32,33
Furthermore, the new protocol delineated the time to discharge to be after 4 normal 20-minute WBCTs within 18 hours after admission for minimal cases and 24 hours after therapeutic response for severe and very severe cases. 7 These new ceilings also reduced the length of hospital stay. Overall, the reduction in hospital stay and AV use was equivalent to $196 US per each Iranian patient. Considering that a brain magnetic resonance imaging study costs about $60 US in Iran 14 and $424 US in the United States (approximately 7 times that in Iran), 34 the cost saving resulting by application of the new protocol can be equal to $1372 US/patient in the United States.
In addition, with the new protocol, the rate of recurrence of venom effects diminished. This is probably caused by addition of a fixed 5-vial maintenance dose to the new protocol, which is able to prevent redistribution of deposited venom to the blood circulation after clearance of the attack dose of AV. 35 Moreover, the use of antiallergy medications was standardized by using the protocol. The development of IARs after AV therapy is reported to be very low. 1 ,5,8,21 On the other hand, most IARs, when they occur, are minor and satisfactorily responsive to antiallergic treatments. 1 ,5,21 Hence, pretreatment for hypersensitivity reactions is controversial and not advocated in most approaches. 1 ,4–7,21 Ghosh et al, 6 Weant et al, 4 and Lavonas et al 5 also abandoned anti-IAR pretreatment in their protocols.
Excessive use of biologic products predisposes patients to acute and late-onset hypersensitivity reactions. Shortages in national and regional medical stockpiles attributable to ceased or decreased production and inadequate distribution are a potential dilemma in both high- and low-income countries.36,37 Thus, it is necessary to use medical resources more wisely and to keep hospital beds available for patients with more serious conditions. Local and national evidence-based protocols are key elements to minimize both risks and medical resource wastage.
Limitations
There are some limitations to this study. The relatively small sample size of the study may expose the findings of the study to type II errors. Further prospective studies with larger sample sizes may refine the statistical significance of the study. Another limitation of the study was that the development of serum sickness could not be compared between the groups. This is because of the fact that although the patients were asked to come to outpatient clinics for 3 follow-up visits during the first 3 weeks after discharge, 7 the vast majority of them did not adhere and came after more than a month (range, 1–4 months). Despite our ability during the visits after a month to evaluate renal function by measurement of serum creatinine, hematologic status by complete blood count, and neurologic condition by neurologic examinations, the development of serum sickness, which is most probable in the first 3 weeks after AV therapy,3,7 could not be precisely assessed. However, we can assume that when a patient did not complain or give a history of specific signs of serum sickness (such as skin rashes, fever, itching, lymphadenitis, and arthralgia) during the mentioned time, they were either absent or too minor to be noted.
Moreover, the results presented in this study were obtained from a center in northeast Iran and may not be able to show the exact picture of the effectiveness of the protocol and the snakebite epidemiology in all parts of the country. After introduction of the protocol and its utilization in a single referral medical toxicology center, the next phase will be training the protocol to other national centers and evaluating its effectiveness on a larger scale.
Conclusions
A standardized protocol can reduce major variations in snakebite management, diminish unnecessary AV administration, and reduce healthcare costs. The designing of a region-specific multidisciplinary protocol based on medical capacity and infrastructure of each country may be cost-beneficial and is recommended.
Footnotes
Acknowledgments
We would like to thank Dr B. Dormanesh, AJA University of Medical Sciences, for her kind support. We also acknowledge Mr S. M. Ziaie and the staff of Mashhad Medical Toxicology Centre for their unstinting assistance during this study.
☆
Conflict of interest: The authors declare that they have no conflict of interest.
