Abstract
Objective
The effect of altitude on brain function is not yet well understood, nor is the influence of height and speed of ascent. Additionally, the relationship between acute mountain sickness (AMS) symptoms and brain function at altitude is unclear. We hypothesized that a deterioration from baseline measures of brain function occurs after rapid, mechanical ascent to 3459 m and would be less pronounced in persons taking acetazolamide.
Methods
In this double blind, randomized, placebo-controlled study, 20 healthy volunteers (14 men, 6 women; mean age [±SD] 43 ± 16 years) were alternately allocated to acetazolamide 250 mg or to placebo, taken every 12 hours commencing 3 days before ascent. Prosaccadic and antisaccadic eye movements, heart rate, arterial saturation, and Lake Louise AMS scores were assessed at sea level and 15 to 22 hours after ascent to 3459 m.
Results
Arterial oxygen saturation was significantly lower in the placebo group compared to the acetazolamide group at altitude (Wilcoxon signed-rank test, median [interquartile range]: acetazolamide vs placebo: 92% [5] vs 85% [5];
Conclusions
Brain function, measured by saccadic eye movements, appears to be unimpaired after rapid ascent to 3459 m. Although acetazolamide improves oxygen saturations, it may worsen prosaccades, possibly indicating adverse effects of acetazolamide on brain function at moderate altitude.
Introduction
Descriptions of climbing at extreme altitude are rife with stories of poor decision making. Such judgment errors may be caused by tissue hypoxia impairing brain function. Levels of hypoxia impairing brain function may cause reduced performance in a wide range of responses, from simple reflexes to more complex brain functions. 1 Yet, the measurement of altitude effects upon brain function in a reproducible and clinically useful way has remained elusive. A variety of memory and decision-making tasks have been tested previously, but are confounded by learning effects 2 and poor test-retest reliability. Therefore, accurately and reliably testing these deficits is still a major hurdle within altitude research.
A novel way to measure brain function is with the use of saccadometry, which is measurement of saccadic eye movements, or the rapid eye response, toward (prosaccades) or away from (antisaccades) a visual target. Saccadic latencies (the time delay between the appearance of a target to the beginning of a saccade in response to that target) provide a quantitative and sensitive measure of brain function and are considered more robust and reproducible than neuropsychological testing, with considerably weaker learning effects. 3 Prosaccade changes are observed in a range of conditions affecting brain function, including alcohol, 4 antipyschotics, 5 benzodiazepines, 6 and Parkinson’s disease. 7 Antisaccade changes involve more complex cognitive processes and may be more sensitive to brain (dys)function. 3
A small number of studies has looked into measuring saccadic eye movements with high altitude. Early work using saccades (a conference abstract) indicated that saccadic latency does not appear to be affected by 10 minutes of mild hypoxia; 8 however, it is difficult to determine the relevance of such a short hypoxic exposure. More recently, it was demonstrated that both prosaccades and antisaccades are not affected in well-acclimatized persons up to 7500 m. 9 In addition to saccadic latencies, Merz et al 9 considered the rate of antisaccade errors (error rate) as an alternative measure of brain function, but also found no changes in acclimatized persons up to 7500 m. However, the effects of acute ascent to altitude on saccadic parameters have yet to be explored.
Acetazolamide is an established prophylactic for acute mountain sickness (AMS) that is often taken at altitudes above 3000 m. Acetazolamide increases arterial oxygenation by stimulating respiration. 10 Although acetazolamide has been reported to increase brain blood flow at sea level, that was not coupled to any change in cognitive performance. 11 The effect of acetazolamide on brain function at altitude has produced mixed results, however, using traditional measures of neuropsychological testing, despite reduced AMS scores.12,13 We hypothesized that acute ascent to an altitude of 3459 m would result in decreased performance in tests of saccadic latency, and that this effect would be lessened in persons taking acetazolamide, in conjunction with higher arterial oxygen saturations and improved Lake Louise AMS score. If proved useful, measurements of saccades could provide a robust and straightforward measure of subtle impairments in cognitive performance at high altitude and could lead to new ways to identify which mountaineers may be at risk of significant deterioration.
Methods
In this double blind, randomized, placebo-controlled study, 20 healthy volunteers (14 men, 6 women; mean age [±SD] 43 ± 16 years) were studied. Minimization was used to reduce group differences in AMS susceptibility, age, and sex. Subjects were randomly allocated to receive either 250 mg acetazolamide or identically matching placebo, resulting in 2 groups of 10 subjects balanced for AMS susceptibility, age, and sex. Acetazolamide and placebo was self-administered every 12 hours for 3 days before ascent and continued until after completion of testing.
All participants resided at elevations between 50 m and 150 m; they had no recent (within 2 months) exposure to high altitudes. The Research and Ethics Committee of the University of Chichester granted approval for these studies, and participants gave their written and informed consent. Participants were studied on 2 occasions, at sea level approximately 4 weeks before departure and on the second the day after an acute ascent by cable car to 3459 m (Refugio Guide del Cervino, Aosta, Italy). All saccadic latencies and physiological data were preprocessed by a blinded investigator before group analysis.
Visually guided horizontal saccades were recorded using a saccadometer (portable miniaturized infrared 1 kHz saccadometer, low-pass filtered at 250 Hz with 12-bit resolution). 7 The participants wore the oculometer on their head, secured by an elastic strap and resting on the bridge of the nose (Figure 1); 3 built-in low-power lasers projected red 13 cd m−2 spots subtending some 0.1 degrees in a horizontal line in the midline at ±10 degrees. 10 Each trial started with a central fixation light, and then after a random delay, a second target light appeared randomly either to left or to right, to which the participants were instructed to make a saccade. Prosaccadic and antisaccadic eye movements were measured at baseline (2 weeks preceding ascent) and after 15 to 22 hours at altitude (3459 m). As the testing protocol required approximately 20 minutes per subject, to minimize group differences in altitude exposure, the order of testing was determined by an independent investigator so that experimental subjects alternated between placebo and active drug and paired according to randomization minimization, as above. Prosaccadic latency was taken as the time for the eyes to move from a central fixation point to an unexpected new target either to the right or left (10 degrees), and during antisaccades, the latency was calculated as the time to move their eyes away to the opposite direction from the new fixation. Five saccadic blocks were recorded at each timepoint, following an internationally standardized antisaccade protocol. 3
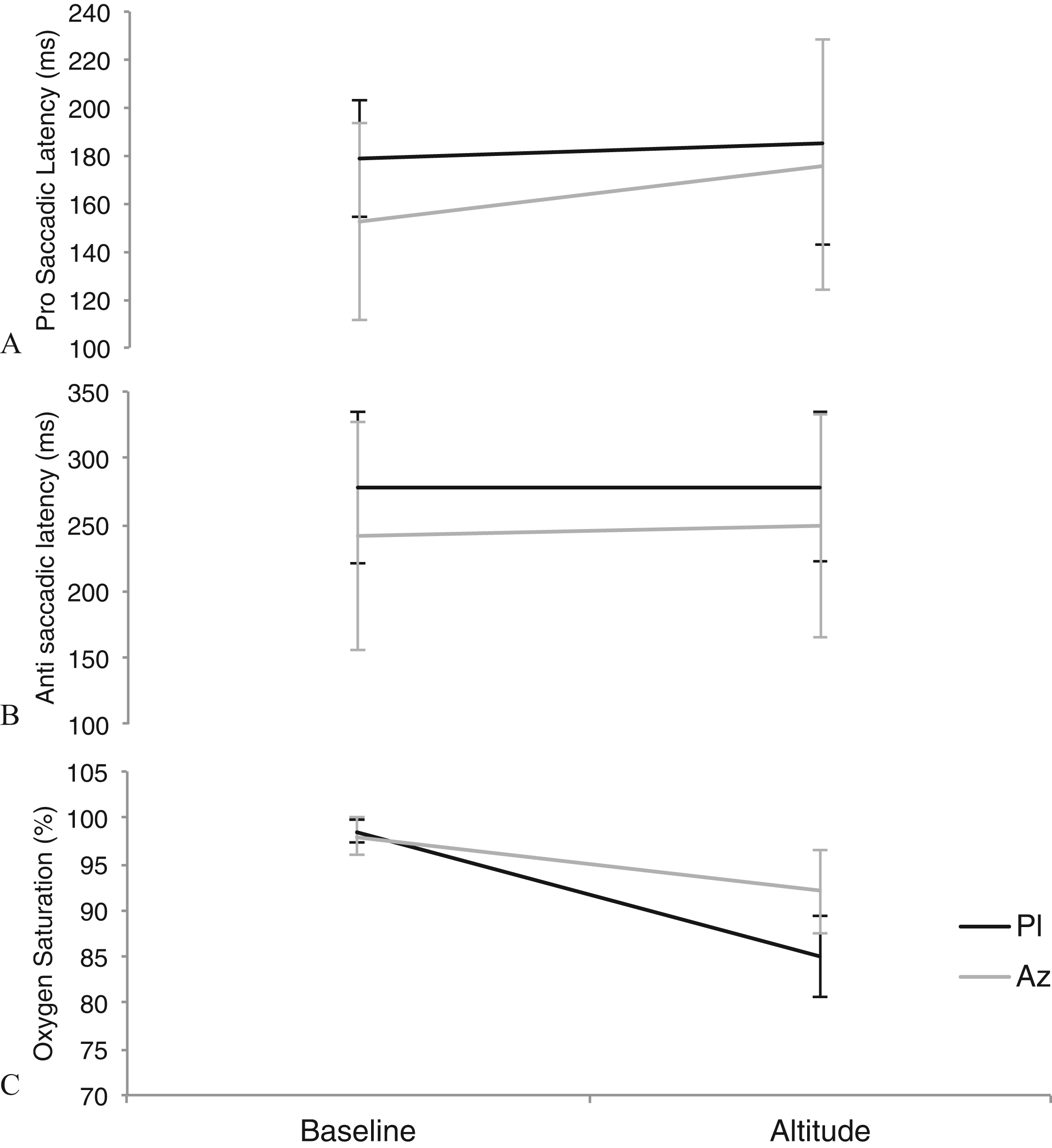
Median (± interquartile range) measurements at baseline (sea level) and altitude (3459 m) for the placebo (Pl) group (
Heart rate, arterial oxygen saturation (Go2 fingertip pulse oximeter; Nonin Medical, Plymouth, MN) and Lake Louise AMS scores were assessed at baseline and altitude (3459 m). Lake Louise scores consisted of a cumulative score of a 0 to 4 point scale for each of the following: headache, gastrointestinal symptoms, fatigue or weakness, dizziness/lightheadedness, and difficulty sleeping (Figure 2).

Saccadometer headpiece (oculometer), worn across the bridge of the nose and against the forehead. The headpiece contains 3 lasers to project either a central fixation point or a visual target to the left or right of the central fixation, to which the participants were instructed to make a saccade.
Statistics
Although normality tests (Shapiro-Wilk) were above significance (
In the acetazolamide group, 1 subject was removed from the analysis as an outlier, as the baseline saccadic latencies were greater than 2 standard deviations of the mean.
Results
Sea Level
There were no differences in both baseline physiology and saccadic latency between the acetazolamide and placebo groups, and no learning effect across baseline trials was observed (Table).
Saccadic and physiological measures from baseline (150 m) to altitude (3459 m) for groups taking acetazolamide 250 mg every 12 hours or matched placebo
All paired comparisons within groups were carried out using a Wilcoxon signed-rank test, whereas all independent between-group comparisons were carried out using a Wilcoxon rank-sum test. Significant values were taken as
Pl, placebo; Az, acetazolamide.
High Altitude
Arterial oxygen saturations were significantly lower in the placebo group compared with the acetazolamide group at altitude (acetazolamide vs placebo: median [interquartile range] 92% [5] vs 85% [5];
In the placebo group, there was no significant effect of altitude on prosaccadic or antisaccadic latency. A reduction in error rate was observed from baseline to altitude in the placebo group (
Discussion
Rapid mechanized ascent to 3459 m in the placebo group had no effect on prosaccadic or antisaccadic latencies, whereas we observed a significant increase in prosaccadic latencies on ascent to altitude with acetazolamide. This increase in prosaccades was seen despite higher oxygen saturations than in the placebo group; and when the groups were directly compared at altitude, there was no difference in Lake Louise scores, heart rate, or saccadic latencies. Therefore, although the moderate altitude was not severe enough to observe a measurable effect of acetazolamide on AMS symptoms, it is possible that acetazolamide may negatively impact brain function.
Previous literature is mixed on the effects of acetazolamide on brain function at altitude. Earlier work at similar altitudes has found conflicting results, demonstrating worse 13 or better 12 neurocognitive performance despite consistently reduced incidence of AMS. These discrepancies may partly be explained by differences in study design: subjects who performed poorly rapidly ascended to the study altitude by airplane, 13 whereas subjects who demonstrated improvement walked to altitude. 12 Therefore, while acetazolamide reduces AMS, it may worsen brain function after rapid ascent to moderate altitude. Consequently, the apparent increase in saccadic latency in the current study from baseline to altitude in the acetazolamide group may be indicative of a decline in brain function. Although consistent with previous research involving rapid ascent, 13 without concurrent decreases in antisaccade latencies and traditional neuropsychological tests, this interesting finding warrants further investigation.
In the absence of acetazolamide, deterioration in brain function appears to be determined by height and speed of ascent. A previous study that used both neuropsychological tests and prosaccades and antisaccades during slow, acclimatized ascent (191 to 201 m daily) to 6265 m showed no deterioration in cognitive performance or alterations in saccadic parameters. 11 That observation appears to demonstrate that well-acclimatized persons do not have cognitive impairment measurable by neurocognitive tests or saccadometry, despite the sensitivity of saccades in detecting even subtle brain dysfunction seen in several other conditions. 4 –6 Conversely, rapid ascent to only 4383 m has been demonstrated to impair improvement of cognitive tasks using only neuropsychological tests, compared to a similarly repeatedly tested control group, 12 and rapid simulated ascent to altitudes of 5334 m and 7620 m showed marked declines in cognitive performance aspects. 14 Therefore, although it appears clear that rapid ascent to altitudes above 4483 m may indeed induce deficits in cognitive performance, the results of our study show that the danger of this impairment at moderate altitudes (3459 m) is much less apparent. Further research using rapid ascent to altitudes above 4000 m may reveal whether measures of saccadic eye movements correspond with neuropsychological tests, and whether acetazolamide plays a role in brain dysfunction at altitude.
Study Limitations
This study was limited by a number of factors, which could be improved for future research into brain function at altitude. A moderate altitude of 3459 m appears to be insufficient to induce differences in AMS scores between acetazolamide and placebo in 20 subjects; therefore, the study was lacking sufficient power to compare AMS and saccadic latencies. Without concurrent neuropsychological measures, it is difficult to compare the sensitivity of saccades in assessing brain function, and future studies will need to include a range of brain function measures to fully assess the affect of altitude and acetazolamide. Additionally, the effect of acetazolamide on cognition was not tested at sea level in these subjects, and thus the isolated effect of acetazolamide is not known. Therefore, the finding in the current study of increased prosaccadic latency at altitude from baseline for the acetazolamide group, without any corresponding differences for antisaccadic latencies/errors, or acetazolamide alone, needs careful interpretation. With antisaccades likely to show more subtle changes in global brain dysfunction, 3 this observed difference in prosaccades is unlikely to be related to dramatic cognitive impairment in subjects. We hypothesize that this result was influenced by interindividual variability in our relatively small group of subjects, and future testing would likely require both subjects and concurrent neurophysiological tests to establish the validity of this finding.
Conclusion
This study has shown that there was no difference in saccadic latencies from baseline to moderate altitude in a group receiving placebo, and no difference in saccadic latencies or AMS between acetazolamide and placebo at altitude. However, this study illustrates the practicality of examining saccadic latencies at altitude, and the absence of a learning effect, suggesting this method may be preferable (although not yet comparable) to paper-based brain function testing. Although brain function as measured by saccadic eye movements at moderate altitude (3459 m) appears to be unimpaired by hypobaric hypoxia, saccadic latencies may nevertheless be a useful tool for investigating brain dysfunction during the early stages of hypoxic exposure, before acclimatization, when brain dysfunction has been reported to occur.14,15
Footnotes
Acknowledgments
The study was supported by the Birmingham Medical Research Expeditionary Society and the Jabbs Foundation, which are registered UK charities. Olivia K. Faull is a Commonwealth Scholar funded by the UK government, and Chrystalina A Antoniades is supported by a grant from the Wellcome Trust (073735), by the National Institute of Health Research (NIHR), by the Oxford Biomedical Research Centre (BRC), and by the Dementias and Neurodegenerative Diseases Research Network (DENDRON). Kyle T. S. Pattinson is supported by the National Institute for Health Research of Oxford Biomedical Research Centre based at Oxford University Hospitals NHS Trust and University of Oxford.
☆
Presented in part at the High Altitude Research Conference, November 22, 2013, Birmingham, UK.
