Abstract
Objective
To assess the effect of acetazolamide (Az) on exercise performance during early acclimatization to altitude.
Methods
Az (250 mg twice daily) or placebo was administered for 3 days in a double-blind, randomized manner followed by a rapid ascent to 3459 m in the Italian Alps. Twenty healthy adults (age range, 18–67 years) were tested at 60% of sea-level peak power output for 15 minutes on a bicycle ergometer after 16 to 27 hours of altitude exposure. Exercise performance was measured in relation to peripheral oxygen saturations measured from pulse oximetry (Sp
Results
At altitude, resting Sp
Conclusions
In this study group, and despite higher resting Sp
Keywords
Introduction
The use of acetazolamide (Az) for the prevention of acute mountain sickness (AMS) has been debated for 50 years, but 2 recent meta-analyses have strongly supported its effectiveness. In 2012, Kayser and colleagues reviewed 24 placebo-controlled trials comparing 1011 Az-treated individuals with 854 placebo-treated individuals and showed convincing evidence of its value. 1 Escalating doses from 250 mg, to 500 mg and 750 mg per day revealed increasing reduction in AMS symptoms by 45%, 50% and 55% respectively, compared with placebo. Another systematic review and meta-analysis again scrutinized the published studies and confirmed the overall positive benefits of Az. 2 This study was supported by a linked editorial suggesting a more personalized approach was indicated. 3
In spite of this overwhelming evidence, many are unwilling to take medication that may have unpleasant side effects. Paresthesias and the altered taste of carbonated drinks can be most irritating, although they are less apparent with lower doses. In addition, there are concerns that exercise capacity might be reduced at altitude because this has been observed when taking Az at sea level.4,5 Moreover, exercise tests using hypoxic gas mixtures to simulate altitude in the laboratory have not been consistent. Some have demonstrated impaired exercise performance,6,7 whereas others showed the opposite, that Az improved arterial oxygenation during exercise.4,8 However, the total numbers of individuals investigated have been small, typically 6 per study. In addition, the participants have been young, whereas trekkers to altitude often tend to be older. For example, in one Japanese report 70% of the subjects were older than 50 years. 9 When Az is commenced at high altitude in acclimatized individuals, there is again uncertainty about its effect on exercise. One early study showed 2 of 4 people had reduced exercise capacity at 6300 m, 10 whereas a study of 15 subjects showed no adverse effects at 4700 m after 10 days of acclimatization. 11 Unfortunately, none of these studies addressed the use of Az in practice. Normally, Az is taken prophylactically for 1 to 2 days before altitude exposure. 1 Typically, individuals then ascend to between 3500 m and 5500 m over the following week or so. Such altitude exposure is much longer than that undertaken in laboratory experiments, and there is usually some degree of AMS. Only 1 study has evaluated this treatment schedule. 12 Twenty individuals prophylactically took 500 mg of Az or placebo daily on an ascent to 4,846 m. At 10 days, the Az group performed better with 20% less reduction in exercise performance than the placebo group, and this was associated with retention of muscle mass. Clearly, Az was having a quite different effect from its use in short-term treatment studies.
These differing data and limited long-term studies do not provide a consensus regarding the altitude or the stage in acclimatization when Az is beneficial, neutral, or possibly detrimental to exercise. The purpose of this investigation was to assess whether Az affected exercise performance at modest altitude in the early phase of altitude acclimatization. A height of 3459 m was chosen as this was sufficient to induce a modest degree of AMS, and the availability of a comfortable, easily accessible refuge allowed well-controlled exercise testing.
Methods
Subjects And Drug Trial Of Az Vs Placebo
Twenty healthy individuals were recruited. Ages ranged from 18 to 67 years (mean age, 43 ± 16.5 years). There were 6 women, 4 between 20 to 23 years and 2 older than 50 years. Menstrual cycle phase was not recorded. Sixteen individuals had previous experience of altitude and were classified into low, medium, or high AMS susceptibility, but none had suffered from pulmonary or cerebral edema. In 4 individuals, AMS susceptibility was unknown. No participant had resided above 1500 m in the previous 2 months. Apart from 1 subject with mild hypertension who was taking the angiotensin-converting enzyme inhibitor, ramipril (5 mg/d), none was taking any relevant medication and all had refrained from intense physical activity on the days before testing. All were kept naive regarding the medication and expected outcomes of the study. Because of obvious side effects from Az administration such as paresthesias, individuals were requested not to discuss any aspects of their medication with others, including the investigators.
Individuals were paired for similar characteristics by hierarchy of importance for 1) previous AMS susceptibility, 2) male/female, and 3) age. There were no significant differences between the groups for these 3 variables, nor weight or resting pulse rate. Each pair received 250-mg capsules of Az or placebo (starch) randomly allocated by independent observers, with blinding to subjects and investigators alike. Capsules were taken twice daily starting 3 days before the altitude exposure for a total of 9 doses.
Exercise Test
This was performed on a specially constructed, light-weight (25 kg), horizontal bicycle designed for altitude studies (Alti Cycle). 13 In use, the individual lay supine, constrained by a shoulder harness, with feet strapped into the pedals. Multistage gearing provided high inertia from a 2-kg flywheel rotating at 2900 rpm at 60 rpm pedaling speed, thereby mimicking the sensation of cycling on a normal bicycle. A remotely controlled brake acted on the flywheel to provide controllable resistance, and power output and cadence were measured via a strain-gauged crank-set (Schoberer Rad MeBtechnik, Julich, Germany) linked to a laptop computer. This mode of exercise was chosen because it allowed other studies.
A submaximal exercise test was selected to induce considerable pulse oxygen saturation (Sp
The exercise test at sea level was undertaken on days 6 and 7 before ascent by each individual in the following manner. After a warm-up for 2 to 3 minutes at 50 to 80 W and 60 rpm, the brake resistance was increased by 10- to 20-W intervals every 60 seconds until power could not be sustained. The last minute of constant power was selected as the PPO. Peak pulse rate was recorded and compared with the theoretical peak pulse rate of 220 minus age to provide some assurance that individuals were exercising at or near their maximum. On completion of each test at altitude, the individual was asked to indicate whether the perceived exertion was easy, medium-hard, or hard, to which was added an involuntary category of fail.
Ascent Profile And Altitude Studies
Individuals travelled from sea level to Cervinia in Italy (2050 m) or Zermatt in Switzerland (1608 m) during a 12-hour period. Final ascent to Refugio Testa Grigia (3459 m), in Italy, was by cable car over a 1-hour period to arrive at 4:00
Altitude exercise tests were commenced at 8:00
Self-assessed questionnaires of the Lake Louise Scoring System for AMS
14
were recorded on the evening of arrival, the following morning, and immediately before and after the exercise tests. Because subjects were tested at different times of the day, a mean AMS score was obtained as a measure of accumulated AMS symptoms in relation to hypoxia exposure. This was the mean value of the morning Lake Louise score and that immediately before the exercise test. Sp
Statistical Analyses
Statistical analyses were performed using SPSS version 21 (IBM Corp, Portsmouth, UK). Box and whisker plots for medians and ranges, Mann-Whitney test for differences of means, and Spearman’s correlation for comparison of groups were used.
Ethics
The study was approved by Chichester University Research Ethics Committee (protocol number: 121339) and was performed according to the Declaration of Helsinki. All individuals gave signed informed consent.
Results
The 2 treatment groups were well matched for susceptibility to AMS, male/female ratio, and age (Az, 41 years; placebo, 44 years; >50 years: Az, 4; placebo, 5). Median PPO achieved at sea level was not significantly higher in the placebo group (mean ±1 SD: Az, 210 ± 29 vs placebo, 250 ± 47). The trial capsules were taken by all individuals successfully. A questionnaire recorded immediately before the exercise test (but analyzed later) indicated that 15 had correctly guessed their medication.
At altitude (Figure 1), resting Sp
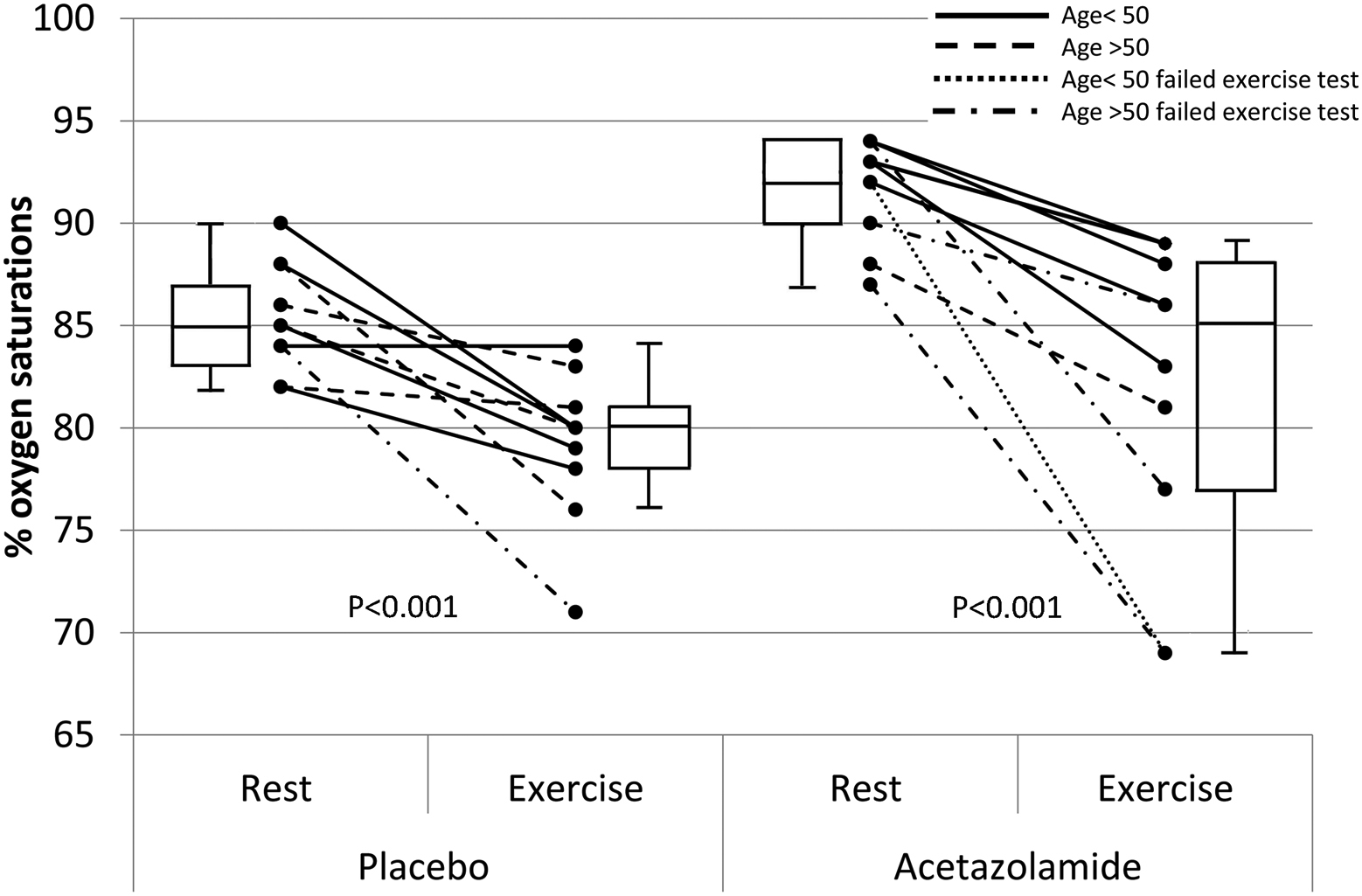
Peripheral O2 saturations (Sp
The exercise test was technically satisfactory in all subjects. Sp
Five subjects failed to complete the exercise test, with 4 of the 5 having the largest fall in percentage Sp
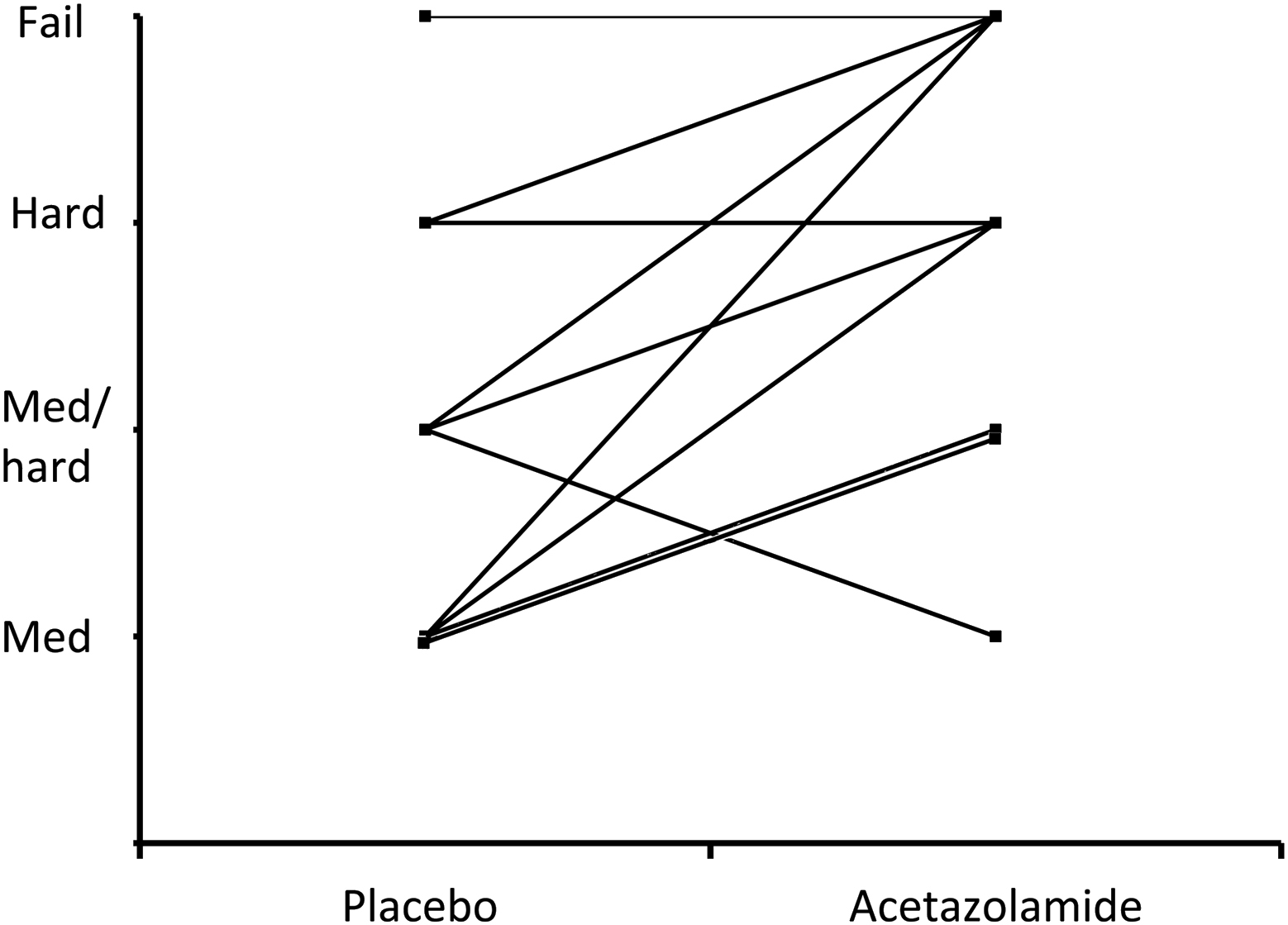
Perceived exercise difficulty for placebo individuals at 3459 m compared with their matched acetazolamide partners (P < .01).
Discussion
The results show that exercise performance at 3459 m was reduced in many subjects taking Az. The reduction was in terms of its perceived difficulty and the failure to complete the test in 4 subjects of whom 3 had large reduction in Sp
There have been several detailed publications of the effect of Az on exercise at reduced oxygen concentrations during early exposure to hypoxia. All were for subjects exposed to hypoxic gas mixtures commencing immediately before the exercise tests. Schoene and colleagues investigated 6 men (age, 23–35 years) in a crossover study of 3 250-mg doses of Az over 24 hours and using an F
Our study differed from these reports in 3 main aspects. First, subjects were exposed to altitude for 18 to 27 hours before the exercise test and, importantly, during a period of sleep with its attendant hypoxia. This caused a measureable amount of AMS symptoms, which were slightly less in the Az group (although not significant). Second, the altitude of our study at 3459 m (F
It is important to remember that maximal or submaximal exercise cannot be maintained for long periods, so bears little resemblance to the prolonged periods of lower-intensity exercise that occurs on treks. Brief periods of excessive oxygen desaturation that might occur with Az during exercise are insignificant compared with oxygen saturations that are 15% to 20% higher throughout the day and night. 15 Over many days this could have an important accumulative effect resulting in better sleep, less appetite suppression, and greater retention of muscle mass, any of which could lead to better exercise performance.
Limitations Of This Study
The investigation contained only 20 individuals so was relatively small but nevertheless larger than most laboratory series. In addition, the altitude attained was modest, and there was no crossover trial. The latter is difficult to achieve as a mountain study. A week or more is required to deacclimatize and to wash out Az, so all individuals would need to return to sea level, the United Kingdom in our case. As regards the exercise test, a V
Conclusions
We have shown that during early acclimatization to 3459 m, 500 mg of Az daily reduces the ability to exercise hard. Placebo subjects experienced more AMS than those on Az but were able to exercise to a greater degree, had lower Sp
Footnotes
Acknowledgments
Statistical analysis was kindly undertaken by Dr S. Harding, Binding Site Ltd, Birmingham, UK. The study was supported by the Birmingham Medical Research Expeditionary Society and the JABBS Foundation, which are both registered UK charities. All authors declare no support from any organization for the submitted work, no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years, and any other relationships or activities that could appear to have influenced the submitted work.
