Abstract
Objective
The purposes of this experiment were to determine the repeatability of acute mountain sickness (AMS), AMS symptoms, and physiological responses across 2 identical hypoxic exposures.
Methods
Subjects (n = 25) spent 3 nights at simulated altitude in a normobaric hypoxia chamber: twice at a partial pressure of inspired oxygen (PIO2) of 90 mmHg (4000 m equivalent; “hypoxia”) and once at a PIO2 of 132 mmHg (1000 m equivalent; “sham”) with 14 or more days between exposures. The following variables were measured at hours 0 and 12 of each exposure: AMS severity (ie, Lake Louise score [LLS]), AMS incidence (LLS ≥3), heart rate, oxygen saturation, blood pressure, and the fraction of exhaled nitric oxide. Oxygen saturation and heart rate were also measured while subjects slept.
Results
The incidence of AMS was not statistically different between the 2 exposures (84% vs 56%, P > .05), but the severity of AMS (ie, LLS) was significantly lower on the second hypoxic exposure (mean [SD], 3.1 [1.8]) relative to the first hypoxic exposure (4.8 [2.3]; P < .001). Headache was the only AMS symptom to have a significantly greater severity on both hypoxic exposures (relative to the sham exposure, P < .05). Physiological variables were moderately to strongly repeatable (intraclass correlation range 0.39 to 0.86) but were not associated with AMS susceptibility (P > .05).
Conclusions
The LLS was not repeatable across 2 identical hypoxic exposures. Increased familiarity with the environment (not acclimation) could explain the reduced AMS severity on the second hypoxic exposure. Headache was the most reliable AMS symptom.
Introduction
Acute mountain sickness (AMS) is a relatively common form of altitude illness that can occur after rapid ascents to altitudes above 2500 m or during exposures to (normobaric or hypobaric) hypoxia in a laboratory. 1 Humans vary significantly in their abilities to acclimatize to hypoxia, and researchers often use AMS as a marker of inadequate acclimatization or acclimation. 1 Despite much research, the etiology of hypoxia intolerance is not well understood, 2 and identifying persons who are susceptible to AMS before hypoxia exposure is difficult (eg, Barry and Pollard 3 ).
Repeatability is an assessment of consistency within persons over a series of measurements. 4 Although a previous history of AMS is frequently stated to be a strong risk factor for the recurrence of AMS,3,5 evidence for the repeatability of AMS is not conclusive. Multiple studies reported associations between AMS history and AMS recurrence; 6 –8 however, these studies also reported moderate numbers of false positives (positive AMS history, negative AMS diagnosis) and false negatives (negative AMS history, positive AMS diagnosis), thus questioning the extent to which AMS is repeatable. Three prospective studies reported that AMS was repeatable, 9 –11 but hypoxic exposures were not necessarily comparable in 2 of the studies because of vasopressin use on 1 exposure 12 and a high likelihood of acclimatization on 1 exposure. 10 Furthermore, the sample sizes were small (<20) in 2 of the studies,9,10 and all 3 lacked sham conditions to blind subjects to the conditions.
The physiological processes responsible for individual differences in AMS susceptibility have yet to be determined, and a reliable physiological predictor of AMS remains elusive.
13
Currently, results are inconsistent for associations between AMS and physiological variables such as blood oxygen saturation (Sp
This experiment was designed to determine the repeatability of AMS, AMS symptoms, and objective physiological variables across 2 identical normobaric hypoxia exposures. To prevent bias in self-reported AMS symptoms, a sham exposure was included in the experimental design and subjects were blinded to the experimental conditions. We hypothesized that individual physiological responses to hypoxia would be repeatable across the 2 identical hypoxic exposures and that each of the physiological variables would be associated with AMS.
Methods
Subjects
Twenty-six healthy nonsmoking subjects (17 male; 9 female) were recruited, all of whom resided at low altitude (ie, <200 m above sea level) and had not ascended above 2500 m (excluding commercial flights in pressurized airliners) in the 2 months preceding each exposure. Subjects were asked about their caffeine use, and those who acknowledged withdrawal symptoms from abstention were excluded because caffeine intake was restricted and symptoms of caffeine withdrawal can mimic symptoms of AMS. 20 On their first visits to the laboratory, subjects were familiarized with the procedures and the testing environment. The Clinical Research Ethics Board of the University of British Columbia approved this study, and each subject provided written informed consent before participating.
Experimental Design
This experiment utilized a single-blind, sham-controlled design. Subjects slept 3 nights in a normobaric hypoxia chamber (Colorado Altitude Training, Louisville, CO) located approximately 100 m above sea level at the University of British Columbia’s Vancouver Campus. The chamber (approximate volume of 15.6 m3) was a transparent box housed in a large room with natural lighting. The temperature was controlled at 22°C ± 3°C, but humidity was not controlled. Subjects were exposed to hypoxia on 2 occasions (H1 and H2) with partial pressure of oxygen (PIO2) of 90 mmHg (4000 m equivalent)
21
and to a sham condition on 1 occasion (SH) with PIO2 of 132 mmHg (1000 m equivalent),
21
with a minimum of 14 days between each exposure. The PIO2 was measured with the chamber’s built-in sensors, and airflow into the chamber was modified as necessary by the chamber to maintain the desired hypoxic dose. An exhaust fan vented the chamber to limit CO
Subjects entered the chamber in the evening and remained in the chamber for 12 hours before exiting the next morning. Two subjects occupied the chamber simultaneously for most exposures, but a single subject occupied the chamber for 6 exposures owing to scheduling conflicts. Subjects were randomly divided into 3 groups, with each group experiencing SH on the first, second, or third exposure. Making the chamber slightly hypoxic for the SH exposure was necessary to mimic the sound of the hypoxic exposures. The SH PIO2 did not lower the subjects’ Sp
To limit confounding effects on various measurements, subjects were asked to refrain from the intake of food and drink for 2 hours, caffeine for 12 hours, alcohol for 24 hours, and food rich in nitrates for 48 hours before entering the chamber. 22 Subjects ingested water ad libitum in the chamber and were offered a standard meal after 1 hour. While in the chamber, subjects rested (ie, performed no physical activity).
Physiological Measurements
All variables were measured in room air before subjects entered the chamber (hour 0) and inside the chamber before subjects exited (hour 12). Subjects were awoken 30 minutes before exiting the chamber to allow for data collection.
Hypoxia tolerance was assessed using the Lake Louise score (LLS) questionnaire, 23 which required subjects to rate 5 symptoms of AMS (headache, gastrointestinal symptoms, fatigue, dizziness, and sleep difficulty) on a scale of 0 (not present) to 3 (severe). A LLS of 3 or greater with a headache score of 1 or greater was considered a positive diagnosis (AMS+), and a LLS not meeting these criteria was considered a negative diagnosis (AMS−). 23
While subjects were supine, HR and Sp
Nocturnal HR and Sp
Data Analysis
Data analysis was performed using SPSS version 21.0 (IBM, Armonk, NY), and alpha was set to 0.05 for all statistical tests.
The severity of AMS and the severity of each individual AMS symptom during each condition (at hour 12) were compared with a one-way analysis of variance (ANOVA) and Friedman tests, respectively. Post hoc analysis was performed with Bonferroni-corrected paired samples t tests and Wilcoxon signed rank tests, respectively. The exact McNemar’s test was used to compare the incidence of AMS and its symptoms across exposures. The percent agreement 26 was calculated as a measure of repeatability. To determine if familiarity with the chamber affected the LLS, tests for linear trends were performed (independent variable, condition; dependent variable, LLS).
The effect of the condition on each continuous physiological variable was determined using a two-way repeated measures ANOVA (time [0 and 12 hours] vs condition [H1, H2, SH]). Post hoc analysis was performed with Bonferroni-corrected paired samples t tests within each exposure and across exposures at hour 12. The effect of condition on mean nocturnal HR and Sp
Results
Subject Characteristics
Twenty-five subjects (9 female and 16 male) completed this experiment, and 1 male subject withdrew from the study after completing 2 exposures. The mean (SD) age of subjects was 24.6 years (6.2 years), mean height was 175 cm (8.4 cm), and mean weight was 72 kg (12 kg). One subject’s FENO data were excluded from analysis because she exhaled more than 100 parts per billion (ppb), approximately 5 times larger than the mean, and high FENO can indicate the presence of asthma, 27 which was an exclusion criterion for the FENO component of the study. After 8 subjects had begun the experiment, BP measurements were added as outcome variables (n = 17 for these variables). Consecutive exposures were separated by a median of 21 days (14–108 days), and H1 and H2 were separated by a median of 28 days (14–138 days).
Normobaric Chamber Conditions
A mean PIO2 of 90 mmHg (87–93 mmHg) was maintained for all hypoxic exposures, and a mean PIO2 of 132 mmHg (129–135 mmHg) was maintained for sham exposures. The partial pressure of carbon dioxide (P
Repeatability of Ams and Its Symptoms
The incidence and severity of AMS were much greater in both hypoxic exposures (H1 and H2) compared to the SH exposure (Table 1, Figure 1). The severity of AMS was significantly lower (−35%) in H2 relative to H1 (P < .001; Table 1), but the difference in the incidence of AMS from H1 to H2 (also −35%) was not statistically significant (P = .065). Agreements for the incidence and severity of AMS on H1 and H2 were low (Table 1).
Incidence and severity of acute mountain sickness and its individual symptoms in 1 sham and 2 normobaric hypoxia exposures
The number (percentage) of subjects who had each symptom and acute mountain sickness (AMS).
The number (percentage) of subjects who had the same response on first hypoxia exposure (H1) and second hypoxia exposure (H2).
For individual symptoms, the median (range) is reported; for AMS, the mean (SD) is reported.
Significant difference from the sham exposure (SH).
Significant difference between the 2 hypoxic conditions.
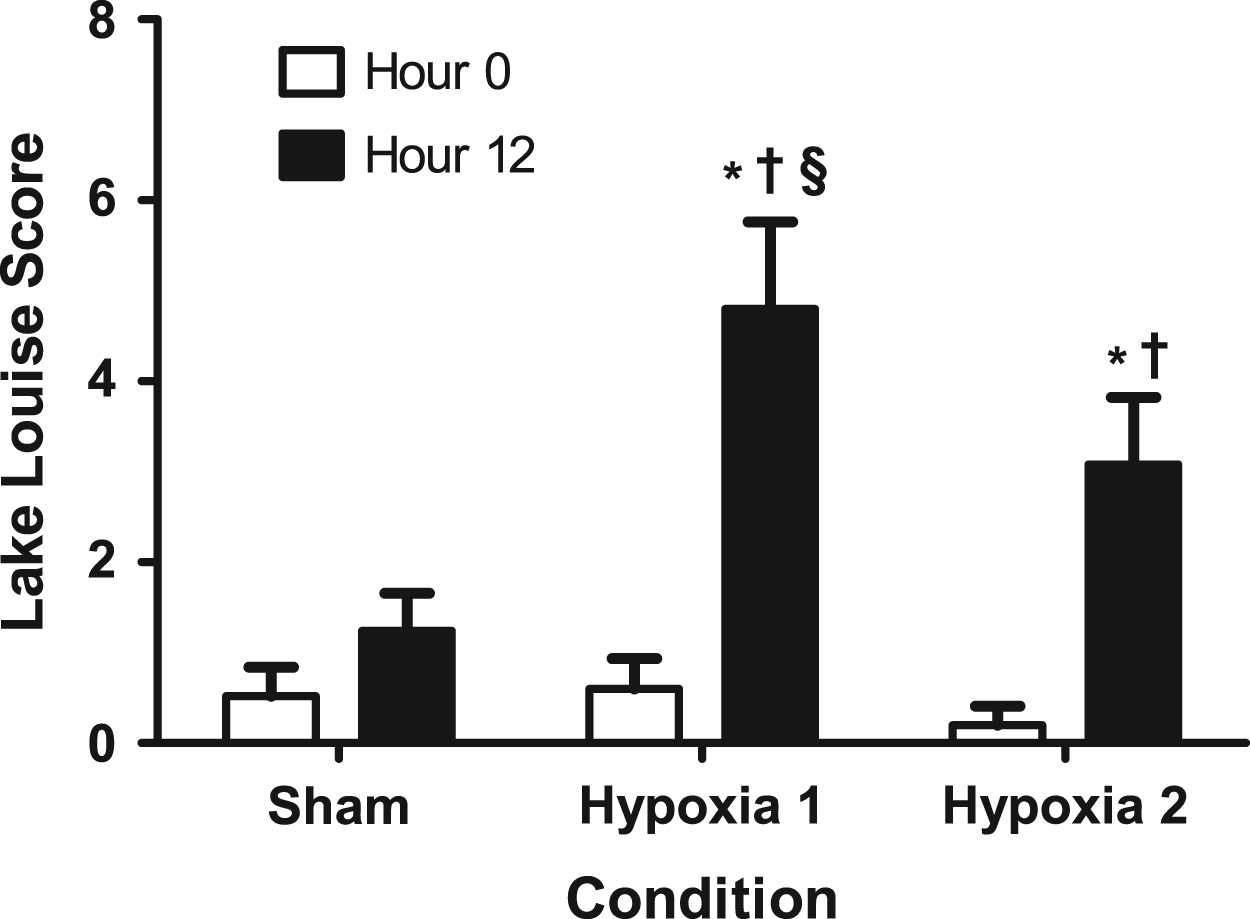
The Lake Louise score at hour 0 (open bars) and hour 12 (solid bars) of the sham, first hypoxic, and second hypoxic exposures (PIO2 was 132 mmHg, 90 mmHg, and 90 mmHg, respectively). *Denotes a 12-hour mean that was significantly greater than the 0-hour mean of the same condition (P < .05). †Denotes a 12-hour mean that was significantly greater than the 12-hour sham mean (P < .05). §Denotes a 12-hour mean that was significantly greater than the 12-hour mean of the second hypoxia exposure (P < .05). Error bars represent 1 SD of the mean, and n = 25.
The incidence and severity of all 5 individual symptoms comprising the LLS were significantly greater in H1 relative to SH, but only the incidence and severity of headache symptoms were significantly greater in H2 relative to SH (Table 1). Agreements for the incidence and severity of AMS symptoms were variable (Table 1). The incidence and severity of headache were not statistically different between H1 and H2 (Table 1).
The decrease in LLS across SH exposures was not statistically significant (P = .066), but the linear trend for a decrease in sleep difficulty scores was statistically significant (P = .025; Figure 2).
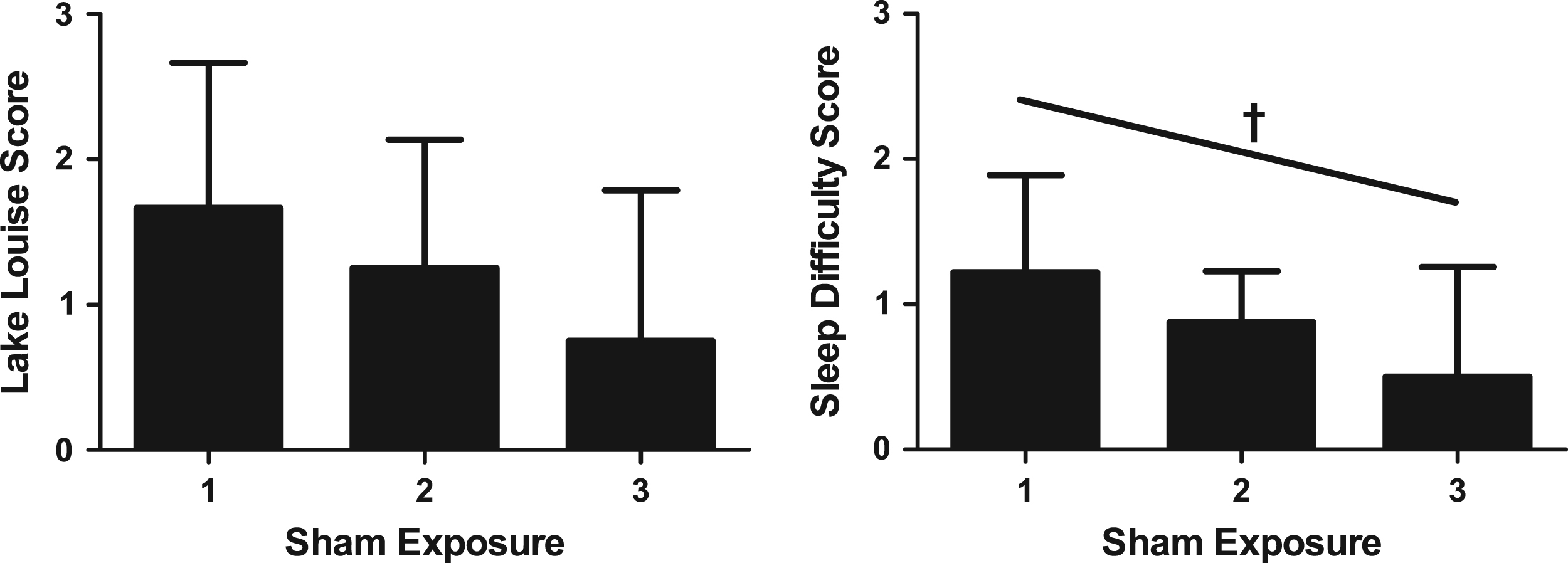
The mean Lake Louise score (left) and sleep difficulty item score (right) from hour 12 of sham conditions that randomly occurred on the first (n = 8), second (n = 8), or third (n = 9) exposure of the study. Error bars represent 1 SD of the mean. †Denotes a statistically significant linear trend for decreasing sleep difficulty scores (P = .025). The linear trend for the Lake Louise Score was not statistically significant (P = .066).
Repeatability of Physiological Responses to Hypoxia
As shown in Table 2, Sp
Physiological responses to one 12-hour sham exposure and two 12-hour hypoxia exposures
Data are presented as mean (SD).
HR, heart rate; Sp
The percent change is calculated for hour 12 relative to hour 0 of the same exposure.
Intraclass correlations (ICC) are calculated for mean values measured at hour 12 of the first hypoxia exposure (H1) and second hypoxia exposure (H2).
Statistically different from hour 0 in the same exposure after Bonferroni correction (P < .008).
Statistically different from hour 12 of sham exposure (SH) after Bonferroni correction (P < .008).
Statistically significant ICC at hour 12 of H1 and H2 (P < .05).
Nocturnal HR was greater on H1 (63.2 [10.6] beats/min) and H2 (60.1 [11.5] beats/min) relative to SH (52.5 [9.1] beats/min; P < .001 for both comparisons); and nocturnal HR was greater on H1 compared to H2 (P = .005). Relative to SH (97.5% [1.5%]), nocturnal Sp
Predicting and Diagnosing Ams
Twelve subjects were AMS+/AMS+, 2 subjects were AMS−/AMS−, 9 subjects were AMS+/AMS−, and 2 subjects were AMS−/AMS+ on H1 and H2, respectively; therefore, the AMS diagnosis on H1 had a sensitivity of 86%, a specificity of 18%, a positive predictive value of 56%, and a negative predictive value of 50% for the AMS status on H2.
The AMS+ and AMS− subjects’ data were only compared on H2 because the distribution of subjects was unbalanced on H1 (ie, 21 AMS+, 4 AMS− on H1). None of the physiological variables was significantly associated with AMS (Table 3). The mean proportion of time elapsed below each Sp
Physiological responses to the second 12-hour normobaric hypoxia exposure in subjects with and subjects without acute mountain sickness
Data are presented as mean (SD).
AMS−, without acute mountain sickness; AMS+, with acute mountain sickness; HR, heart rate; Sp
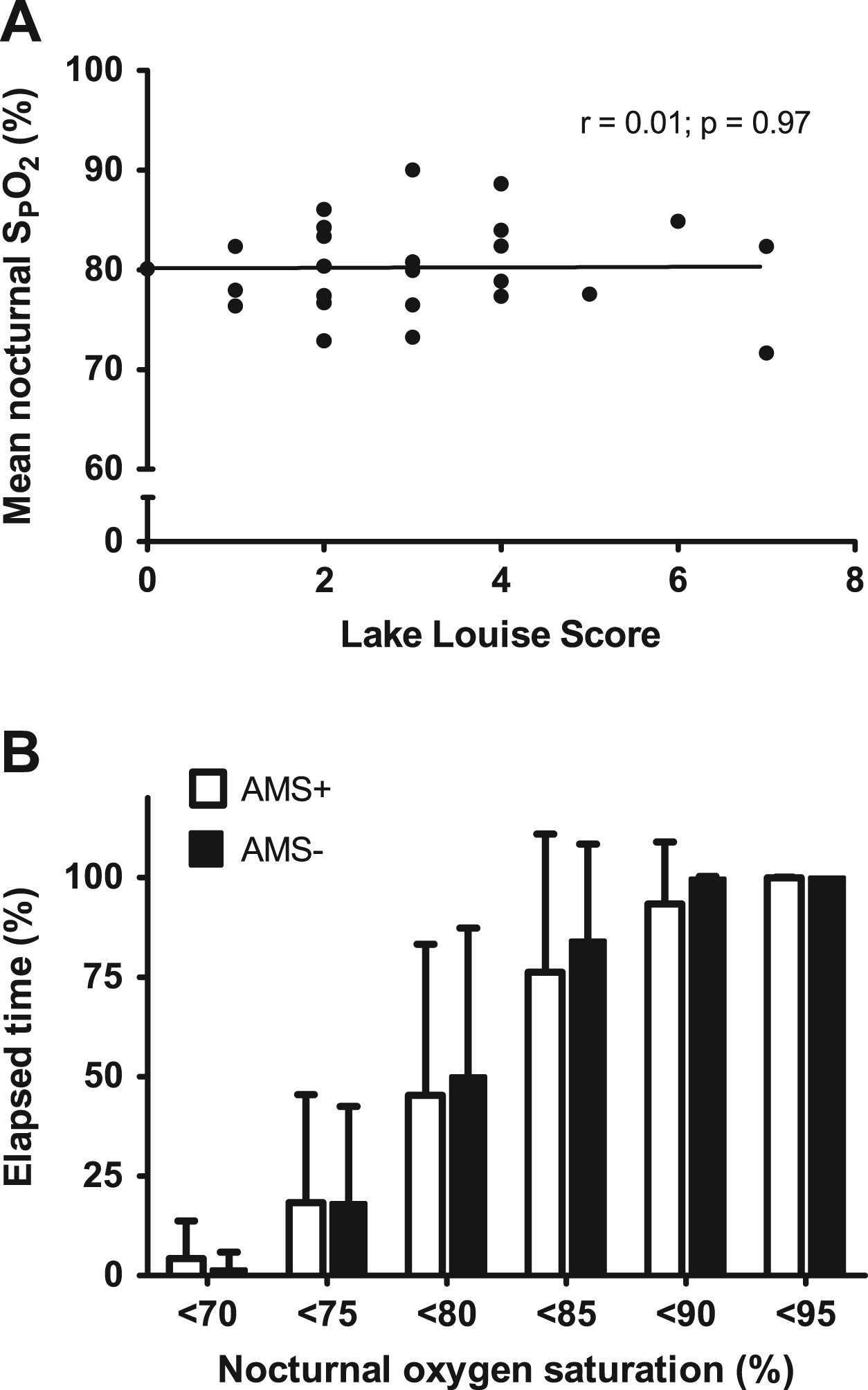
Nocturnal oxygen saturation (Sp
Discussion
This is the first experiment to assess AMS repeatability with a single blind, sham-controlled design. Contrary to our hypotheses, the severity of AMS (measured with the LLS) was lower on the second hypoxic exposure relative to the first, and none of the physiological variables was associated with AMS. As hypothesized, however, physiological variables were moderately to strongly repeatable across H1 and H2. We suggest that familiarization with the environment may be responsible for the decrease in AMS symptom severity across hypoxic exposures.
The difference in the AMS incidence between H1 and H2 was not statistically significant; however, that the severity of AMS was significantly lower in H2 still suggests that AMS was not repeatable in this experiment. The severity of AMS is more sensitive to changes than the incidence of AMS, making it a better index for repeatability: for any decrease in LLS, whether a change in AMS incidence would occur is dependent on 1) the threshold LLS for a positive AMS diagnosis, and 2) the mean severity of AMS induced by the experimental conditions. The lack of repeatability suggests that it may be inappropriate to categorize subjects as AMS susceptible or AMS resistant after 1 hypoxia exposure.
Headache, the cardinal symptom of AMS, 28 was the only symptom with a significantly greater incidence and severity in H1 and H2 relative to SH. The incidence and severity of headache were not statistically different in H1 and H2, suggesting that headache symptoms may be influenced less by familiarization than other AMS symptoms. Sleep difficulty was the only other symptom that occurred commonly on H2, but the elevated sleep-difficulty scores in SH suggest that the chamber (independent of hypoxia) was responsible for much of the difficulty sleeping.
The decrease in LLS from H1 to H2 was not likely due to hypoxia acclimation. Although 5 days above 3000 m in the 2 months preceding an ascent to 4559 m decreased the risk of AMS, 7 to our knowledge, sustained acclimation multiple weeks after a single 12-hour exposure to hypoxia has not been demonstrated (in awake or sleeping subjects). A washout period similar to the one employed in this study (ie, 12–14 days) was previously used to prevent carryover effects between 2 longer (22-hour) hypoxic exposures (barometric pressure 446 mmHg; 4300 m equivalent). 29 That objective physiological responses were similar to H1 and H2 also suggests subjects were not acclimated to hypoxia on the second hypoxic exposure.
Our data suggest that familiarization to the environment affects the LLS, and our results reaffirm the subjective nature of the LLS. An unfamiliar sleeping environment and the anticipation of unpleasant symptoms (the potential symptoms of AMS were a required element of the consent forms) may have been responsible for the greater LLS on H1. MacNutt et al 30 suggested that a greater “psychological tolerance” of altitude (acquired from previous ascents) could explain the reduced AMS symptoms observed in subjects reascending to altitude. Although our subjects did not ascend to altitude, they may have been anxious about the experiment. In support of this suggestion, sleep quality improved significantly with the number of previous exposures to the chamber, and mean HR, SBP, DBP, and MAP were slightly lower in H2 compared with H1 (at hour 12), possibly indicating reduced anxiety. Although familiarization with the environment has been postulated to be a potential confounding variable in studies of this sort, 29 this study is the first to demonstrate that familiarization with the testing environment affects self-reported LLS.
That AMS severity was not repeatable is in disagreement with previous studies, 9 –11 but between-study comparisons are difficult for a number of reasons. Firstly, only 1 previous study 11 used the LLS questionnaire, and methods of assessing AMS (eg, Hackett’s score 31 ) used in the other studies will likely have different psychometric properties compared to the LLS that will affect repeatability. Secondly, the methods used to calculate or confirm AMS repeatability differed substantially across studies, preventing a direct comparison of quantitative repeatability statistics. Thirdly, methodological differences make direct comparisons problematic because 2 studies occurred at high altitude,10,11 1 study utilized a hypobaric chamber, 9 and our study was conducted in a normobaric chamber. It is possible that there would a greater effect of familiarization in a chamber experiment than in a high-altitude experiment because sleeping in a chamber would be a novel experience for the subjects, whereas sleeping in a high-altitude setting could be similar to sleeping in a low-altitude setting in many ways. Finally, we included the SH exposure to reduce subject bias, which was not done in any of the previous studies.
Pulse oximetry is frequently used in field studies of AMS, but pulse oximetry has not been conclusively demonstrated to be a reliable diagnostic tool for AMS.
32
Mean HR (wakeful) was highly repeatable in this study, but similar to previous studies (eg, Wagner et al
33
), HR was not associated with AMS. The mean Sp
In addition to pulse oximetry, we tested BP and FENO for associations with AMS. Blood pressure was not associated with AMS, but our sample size was relatively small (n = 17) for these comparisons. The ICCs of SBP, DBP, and MAP were lower than those measured in normoxia, 38 possibly because of the small range of each variable in our data. 39 The FENO was unaffected by normobaric hypoxia, a finding that supports previous studies, 19 ,25,40 and the high repeatability of FENO agrees with data collected in normoxia. 41 The AMS+ subjects had a lower mean FENO than the AMS− subjects, which, although not statistically significant (P = .065), is consistent with previous studies.19,42
Study Limitations
There are several limitations to this experiment that should be considered when interpreting our results. Firstly, we cannot rule out the possibility that a consistent history of AMS symptoms across multiple (>2) independent exposures to hypoxia would be useful for predicting AMS on future exposures. Secondly, as with any study with a small to moderate sample size, we also cannot rule out the possibility of type I errors. Thirdly, the duration of the exposures were a compromise between feasibility and the typical time course of AMS, and increasing the exposure length may affect the repeatability of AMS. Finally, it is unclear whether our results can be generalized to high altitude settings. Subjects were exposed to normobaric hypoxia instead of hypobaric hypoxia, and the 2 modes of hypoxia may differ in terms of their capacities to induce AMS.43,44 Exposure to normobaric hypoxia also requires subjects to be confined to a small space that is unfamiliar and potentially uncomfortable, possibly making this mode of hypoxia different from a true altitude exposure.
45
Similarly, the normobaric hypoxia chamber used in this study was not able to maintain a CO
Conclusions
We demonstrated that AMS severity (measured with the LLS questionnaire) was not repeatable in response to 2 identical 12-hour normobaric hypoxia exposures, and that the AMS status on H1 was not a reliable predictor of the AMS status on H2. The measured physiological responses to normobaric hypoxia were moderately to strongly repeatable, but none was associated with AMS status. Finally, a greater focus on headache may be warranted in future AMS studies: headache was repeatable across hypoxic conditions, and headache was the only AMS symptom with an elevated severity (relative to sham) on the second hypoxic exposure.
Footnotes
Acknowledgments
The Natural Sciences and Engineering Research Council of Canada (NSERC) provided funding for this project, and the Canadian Foundation of Innovation (CFI) provided funding for the normobaric hypoxia chamber. Martin J. MacInnis is the recipient of an NSERC Canada Graduate Scholarship.
☆
Presented in part at the 18th International Hypoxia Symposium, February 26–March 3, 2013, Lake Louise, Alberta, Canada.
