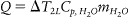Abstract
Objective
Heat illness is a common ailment that, if left untreated, is associated with high morbidity and mortality. Chemical cold packs (CCPs) and ice packs are widely used in the pre-hospital setting and by those with limited resources, yet no controlled studies have compared the cooling of ice to that of CCPs. This study determined the theoretical cooling of CCPs on a benchtop model, comparing the results to similarly sized ice packs, and is the first known comparison of these hyperthermia treatments.
Methods
The CCPs used in Stanford University's Emergency Department were activated in an insulated volume of water (2 L), and temperature was recorded at 1-second intervals in a controlled environment (41°C at 20% humidity). The procedure was repeated with 1-quart ice packs.
Results
The CCPs resulted in a 5.25°C degree temperature drop, with a time constant (time to 63% of initial temperature—a common engineering characterization metric) of 1.72 minutes for the test volume. Ice packs resulted on average in a 19.8°C temperature change, with a time constant of 26.8 minutes. The CCPs provide less overall temperature change and were shorter lived. Application of 6 CCPs on a 50th percentile male (weight 86.6 kg, height 1.7 m), assuming ideal heat transfer, would result in less than 0.5°C temperature change. Similarly configured ice packs would result in a 2.5°C change.
Conclusions
Experiments demonstrate that CCPs are inferior to similarly sized ice packs for thermal regulation, and lose their effectiveness more quickly. These findings support the consideration of ice packs as an alternative to chemical cold packs when cooling hyperthermic patients.
Introduction
Hyperthermia is a common condition in which core body temperature is raised above physiologically normal conditions (≥38°C). Heat stroke is characterized as temperature above 40°C with multiorgan dysfunction. If effective cooling is not rapidly implemented, morbidity and mortality occurs in 30% to 80% of victims. 1 As outcomes are directly attributed to both the magnitude and duration of hyperthermia, successful treatment relies on reducing core body temperature as rapidly as possible. Disagreements continue over which cooling modality is most effective. Furthermore, current literature is limited in describing the efficacy of exertional heat stroke management in resource scarce environments. 2
A commonly used cooling method in pre-hospital care is the application of chemical cold packs (CCP) or ice packs to large arteries near the surface, such as the carotid, axillary, and the femoral, to maximize heat transference and minimize reactive vasoconstriction. This method, despite its wide clinical use, has not been well studied in randomized controlled trials. Instant CCPs use an endothermic reaction to reduce temperature. The hydration of ammonium nitrate (NH4NO3) is most typically used, providing a system enthalpy change of +25.69 kJ/mol at 25°C. 3 Ice packs use the solid to liquid phase change of water to absorb heat from the environment, providing an enthalpy change of +6.0 kJ/mol. 3 Typical cold packs contain approximately 91g ammonium nitrate of unknown purity, and 147 mL water. Cooling rates for CCP have been shown in small studies to be marginally effective (0.028°C/min and 0.027°C/min), 4 whereas ice packs have slightly better cooling rates (0.082°C/min and 0.089°C/min). 5 However, CCPs have not been directly compared with ice packs in the treatment of exertional heat stroke victims. We attempted to determine the cooling efficacy of ice packs and CCP in a thermal model testing heat lost from the system as the result of treatment.
Methods
An activated CCP (MediChoice product #1480069904, Owens & Minor, Richmond, VA) was placed in a 3-L Styrofoam container filled with 2 L water and outfitted with a data-logging thermometer (Figure 1). The container was sealed with a Styrofoam (Dow Chemical, Midland, MI) lid. Owing to the superior insulating properties of Styrofoam, the internal environment of the container can be assumed to be thermally isolated from its surroundings (an adiabatic system). The Styrofoam container was gently agitated throughout the test duration to ensure water temperature homogeneity. Data were obtained at 1-second intervals using a thermocouple probe connected to a data acquisition personal computer. The test was also repeated with a 1-quart Ziploc bag filled with crushed ice (570 mL water content) for comparison. This volume was chosen as it approximated the physical size of the CCP. Immersion of packs in the water volume was approximately equal. Testing was performed in a controlled environment (41°C at 20% humidity). Ambient temperature control helped to standardize the minor heat flux through the Styrofoam wall of the container.

Experimental test setup.
It was hypothesized that optimal cold treatments will absorb heat from the human body through conduction and can be predicted with the solution to the heat flux equation: T = ΔT (e−t/tc +1). The thermally insulated container would allow direct detection of the enthalpy change associated with treatment (hydration of ammonium nitrate or phase change of ice to liquid). Statistical analysis was performed in Matlab.
Results
The cooling curves followed an exponential decay, predicted by the solution to the heat flux equation. Such trend facilitates analysis and an exponential equation could be written for the 2 cases. Both experimental and model data are presented in Figure 2. To offset minor initial temperature fluctuations, temperature data were plotted as temperature difference from initial (41°C). For the test with CCP shown here, the experimental data fit the exponential decay curve until t = 13 minutes, whereupon heating from the exterior environment caused a water volume temperature increase. Experimental ice pack data depicted in Figure 2 showed an initial drop deviating from predicted temperature curve due to a temporary cessation of mixing. Experimental results realigned with predicted exponential decay upon resumption of mixing. Time constant (time the temperature takes to reach 63% of original value) comparison showed 2 orders of magnitude difference between the 2 different treatments. Results showed that CCPs remain effective for roughly 5 minutes, with an average temperature rate of 0.6°C per minute whereas ice packs could continue to cool well past 25 minutes. These results are presented in Table 2. The ice pack could provide much more enthalpy change as compared with a similarly sized CCP. This model noted the thermal energy absorbed by a 2-L volume of water using the following equation (see Table 1 for definition of symbols):
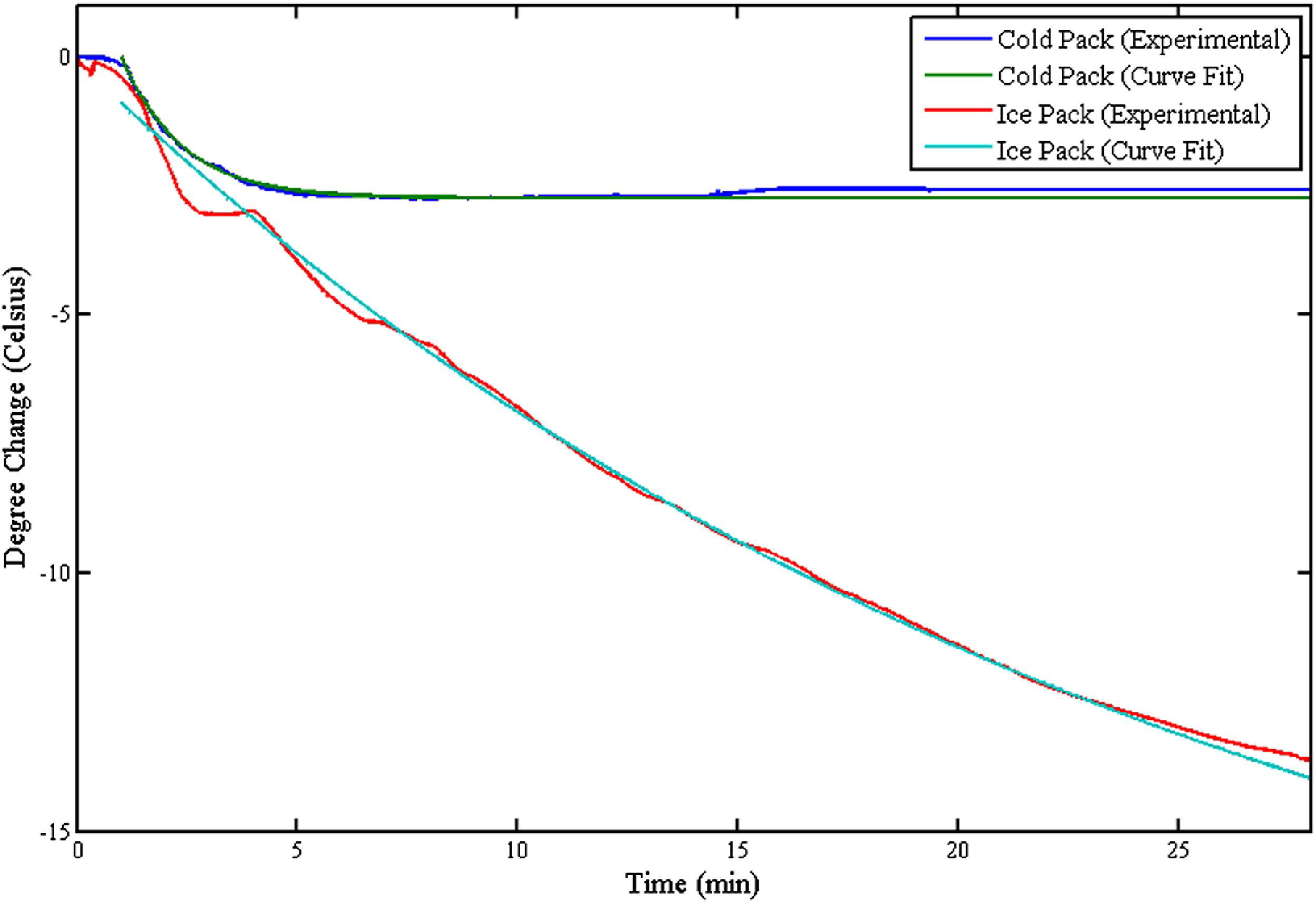
Comparison of ice pack versus chemical cold pack treatment.
List of symbols
Characteristics of experimental and curve-fitting results
The overall change in temperature for a human body can be determined by: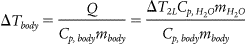
The thermal mass of a 50th percentile male can be determined by the mass (86.6 kg) multiplied by its specific heat capacity (3490 J/kg°C). 6 The temperature change induced on an average male from 6 cold packs placed near the large arteries, as per standard practice, is less than 0.2°C per minute over the lifetime of a cold pack (approximately 5 minutes). A single treatment would not reduce a hyperthermic patient's core temperature (41°C) to physiologically normal (37°C). With cold packs being exchanged every 5 minutes, it would require 18 cold packs overall to achieve sub-38°C core temperature within 15 minutes. Alternatively, ice packs of roughly the same size can induce a much larger temperature change (3.3°C) in 4 minutes. Although the higher enthalpy change per mole associated with the dissolution of ammonium nitrate into water would suggest that it would be the superior cold source, the amount of ammonium nitrate used in CCPs is too small to effect a therapeutically significant temperature change.
Discussion
Evidence supports providing the hyperthermic patient with the most rapid cooling therapy available. Pre-hospital providers should consider the theoretical limitations of chemical ice packs placed in the traditional 6 areas of the human body, as prior studies have found that 6 strategically placed CCP are not an improvement over passive cooling. 4 Of note, the prior investigators agitated their CCP at 15 minutes and replaced it at 30 minutes, whereas our findings indicate that more frequent replacement is preferred for optimal heat flux, although it remains to be seen whether CCP therapy would only provide local tissue cooling. Additionally, a single application would not be sufficient for clinically significant core temperature depression. Wilderness medicine providers may consider conserving their limited resources for higher yield cooling techniques. Application of cold packs to locations with arterial venous anastomosis, such as the palm or soles of the feet, may provide superior cooling rates than those traditionally described. 7
Chemical cold packs are only one of many treatments available for hyperthermic patients in a pre-hospital setting. Cooling methods available in the wilderness may include evaporative cooling and cold water immersion. Evaporative cooling can be achieved by loosening or removing clothing, spraying with tepid water to maximize water-vapor skin interface while minimizing shivering, while facilitating convection with air movement by fanning. Studies using cold or warm cooling mist water achieve rates from 0.04°C to 0.08°C per minute. 8 Another evaporative method such as utilizing the downdraft from a helicopter is slightly more effective (0.10°C/min), if more logistically challenging. 9
Immersion studies with ice water cooling has been shown to be twice as rapid to reduce core temperature as the evaporative spray method (0.20°C/min vs 0.11°C/min). 10 Immersion takes advantage of water's high heat conduction, which is 25 times that of the air, as well as the high thermal gradient between ice water and skin, but the practicalities of this technique limit its usefulness in many wilderness scenarios. The ideal temperature of immersion water has been debated, 11 as there is concern that reflexive vasoconstriction could theoretically insulate the body, and cause shivering and paradoxical heat generation.12,13 Current expert consensus is that the colder the temperature of the immersion water, the better.
Emergency Departments typically have additional capabilities allowing for more rapid cooling rates. Such capabilities should be used with expediency as degree and duration of elevated body temperature is the best predictor for patient outcome. 8 Intravenous introduction of cooled saline has the additional benefit of rehydration and increasing blood pressure, often a confounding ailment of hyperthermic patients. Hemodialysis and gastric lavage with cooled saline have also been used to remove heat. Cold packs are only one part of an overall cooling strategy, specific to the severity of hyperthermia in the patient and the capabilities of the Emergency Department.
The popularity of CCPs as a source for cold therapy may be due their ease in storage and transport, as well as their relative low cost. Ice may provide superior cooling, but requires greater infrastructure to maintain a readily accessible source. Such infrastructure may be considered for stationary clinics with high incidences of hyperthermic patients.
Future directions of study include direct comparison of treatments in hyperthermic people to provide more clinically applicable results and further elucidation of efficacy. Also, studies investigating alternative locations for applying CCPs could prove more beneficial than the traditional paradigm.
Study Limitations
This benchtop model allows for comparison between 2 different cooling methods and was not an attempt to reproduce cooling rates found in other studies.4,5 The rates of cooling predicted by these experiments for a human body exceed those found in literature describing actual application.4,5 That can be attributed to the idealized heat exchange between the cold source (CCP or ice pack) and the volume of water. During real world application, energy will be absorbed by the cold source from both the human body and the surrounding environment, resulting in reduced effect on body temperature. The rate of heat gained by the cold source may be also reduced, providing cooling beyond 5 minutes in real world applications.
Results from the cooling of the 2-L water volume are extrapolated to represent the human body. This assumes human body temperature homogeneity, namely, the entire body is uniformly cooled down. In clinical applications, cooling may be used to protect vital organs and is concentrated on those areas. Localized cooling may exhibit higher cooling rates than seen in these tests.
Temperature homogeneity of the test volume was achieved by mechanical shaking of the container. Such energy input deviates from the adiabatic assumption. However, such input is assumed to be minuscule in comparison with the change in enthalpy associated with treatment.
The model did not attempt to mimic the basal metabolic heat output of the human body (60 W to 90 W). 6 The addition of a heat source would have reduced the overall temperature change and increased the time constant associated with each treatment. However, such additional complexity to the experimental model would have equally biased both treatments toward smaller temperature changes and shorter efficacy times. Because the relative efficacy of the treatments was desired, this deviation was considered acceptable.
Only 1 type of model of CCPs was used in this study. The amount of ammonium nitrate present in each CCP may differ between manufacturers. Additionally, other manufacturers use ammonium chloride (NH4Cl) as their source for enthalpy change (14.7 kJ/mol). Subsequently, the enthalpy change provided may vary according to manufacturer and model number.
Conclusions
This study is the first to directly compare the effectiveness CCPs and ice packs for hyperthermic cooling. In these experiments, the ice pack provided significantly higher enthalpy change over a longer period than similarly sized CCPs. Care providers may want to consider using ice packs in lieu of CCPs when infrastructure allows for their use. Further investigation is needed to determine the clinical effectiveness of using both CCPS and ice packs in the treatment of hyperthermic patients.
Footnotes
Acknowledgments
Special thanks to Vinh Cao, Dennis Grahn, and Prof Craig Heller for providing technical expertise and laboratory space, as well as to Stanford University Hospital Division of Emergency Medicine for their financial support.