Abstract
After a trip to Belize, a 25-year-old man noticed an erythematous papule on his upper right chest that enlarged over a 6-week period and formed a central aperture. The patient reported feeling movement and intermittent lancinating pains under the skin. The history and examination were consistent with cutaneous myiasis, likely secondary to the human botfly, Dermatobia hominis. The objective of reporting this case is to present a simple method of extraction of a botfly larva using a commercial venom extractor. The patient's upper chest was prepared, and an occlusive dressing was placed over the lesion for 30 minutes. The Extractor Pump (Sawyer Products, Safety Harbor, FL) was applied and activated, and the larva was rapidly extracted completely intact with no significant discomfort to the patient. The wound fully healed without complication. D hominis is a common etiology of cutaneous myiasis endemic to Belize. The larva burrows under the skin of mammals where it develops for a period of weeks before erupting and falling to the soil to pupate. The diagnosis and treatment of botfly infestation is pertinent to doctors in the United States as Central and South America are common travel destinations for North Americans. In this case, a commercially available venom extractor was demonstrated to be a safe, noninvasive, and painless method for botfly extraction in the field without use of hospital resources.
Introduction
The human botfly, Dermatobia hominis, is a member of the Diptera order, the family Oestridae, and the subfamily Cuterebridae. These 2-winged flies are the culprits behind the majority of cases of cutaneous myiasis in the Americas, 1 where they are endemic in the Neotropics spanning from Southern Mexico to Northern Argentina. 2 These flies are active year round in hot, humid, lowland forests below 3000 m in altitude. 3 Cutaneous myiasis describes infestation of the skin of the host (human in this case, although the main host of D hominis is livestock and other mammals) by dipterous larva. These larva feed on the host tissue or bodily fluids as they develop. The life cycle of D hominis is particularly fascinating. After fertilization, the female captures a blood-feeding vector, such as a mosquito or tick, and deposits as many as 50 eggs on the vector's body for use as mechanical transport, a process termed phoresis. The eggs require approximately 6 days of incubation, 2 after which the vector lands on a human to feed, and the warmth from the host's body results in the eggs rapidly hatching and the first instar larva entering the host's skin through a follicle, the mosquito bite site, or direct invasion. Entry into the skin usually takes less than an hour, but the larva can survive on the skin surface for as long as 20 days. 4 The larva will develop in the hypodermal layer for 5 to 12 weeks, growing to a size of 18 mm to 24 mm in length 3 before erupting from the host's skin and falling into the soil to pupate for 20 to 30 days, becoming an adult fly. 1 The adult fly lives only 9 to 12 days, with the sole purpose of mating and finding another vector, as its mouth parts are nonfunctional, and it cannot feed. 2
While growing in the skin, the larva goes through 3 instar stages (delineated by molts). The stage II larva is flask shaped, whereas the stage III larva is more fusiform, as in this case. Owing to a requirement for contact with the air for respiration, botfly skin lesions are characterized by a central punctum with an opening, which is maintained throughout larval development and allows for larval respiration (Figure 1). The larva is oriented in its subcutaneous pocket with its posterior surface toward the breathing aperture, superficially, and its head buried deep in the cavity. There are 2 hooklike maxillae at the cephalic end that help to burrow and feed on the host tissue. 5 Caudally directed, concentric parallel rows of small hooks or spines along the anterior body segments help to lock the stage III larva in place in the subcutaneous tissue (Figure 2). 6 Spiracles are also noted on the posterior end of the larva through which air is exchanged through the breathing aperture in the skin (Figure 1). Waste is excreted through the posteriorly directed and superficially oriented anus. 7
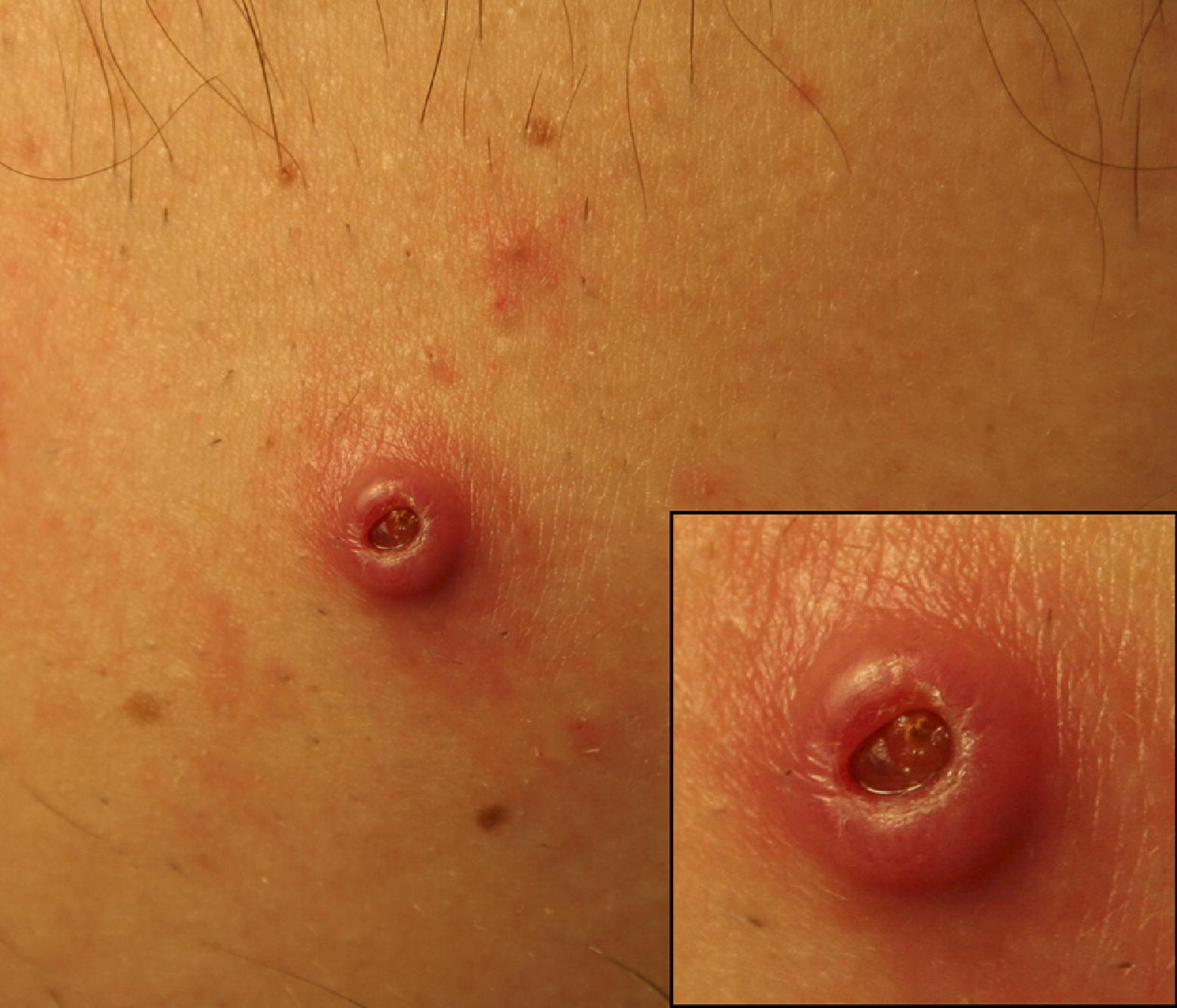
Skin lesion of botfly myiasis third-instar larva on the upper right chest of the patient, with magnified inset. Note the central punctum or orifice through which the larva breathes. Respiratory spiracles at the posterior end of the larva can be seen in the center of the lesion. Motion could be visualized through the breathing aperture.

Dermatobia hominis larva after extraction. The larva measures 2 cm. The anterior portion of the larva with noticeable dark-colored maxillae is at the right aspect of the image. The spiracles on the posterior aspect of the larva can be seen through the breathing aperture in Figure 1. Notice concentric parallel rows of spines in the anterior segments, which lock the larva in place.
The D hominis larva secretes and excretes many substances, particularly proteases, which have been only partially characterized but contribute to its success as a parasite in relation to tissue invasion, nutrition, and host immune system evasion. 2 In cattle, it was demonstrated that immunoglobulins did not bind directly to the living larva, indicating immunomodulatory properties of the larva's secretions. Additionally, large amounts of soluble antigens were identified along the fistulous breathing aperture, suggesting their involvement in maintaining this opening. 8 It has also been postulated that the larva may secrete an antimicrobial substance, as these lesions do not usually become superinfected despite the open wound. 3
In the United States, D hominis is not a rare cause of myiasis owing to the preponderance of travelers returning from South and Central America. Numerous prior case reports and small case series found in the literature offer a variety of different treatment methods for botfly cutaneous myiasis. The objective of reporting our case is to present a quick, simple, noninvasive method of extraction of a human botfly larva using the Extractor Pump (Sawyer Products, Safety Harbor, FL) to extract the larva out of the skin. This device was originally developed to “vacuum” venom from a snake bite wound. The configuration of the device is such that the operator depresses the syringelike mechanism to activate suction after obtaining an airtight seal on the skin using the appropriately sized plastic cup adapter (Figure 3G). One other case report of this method of larval extraction was found in the literature. 9
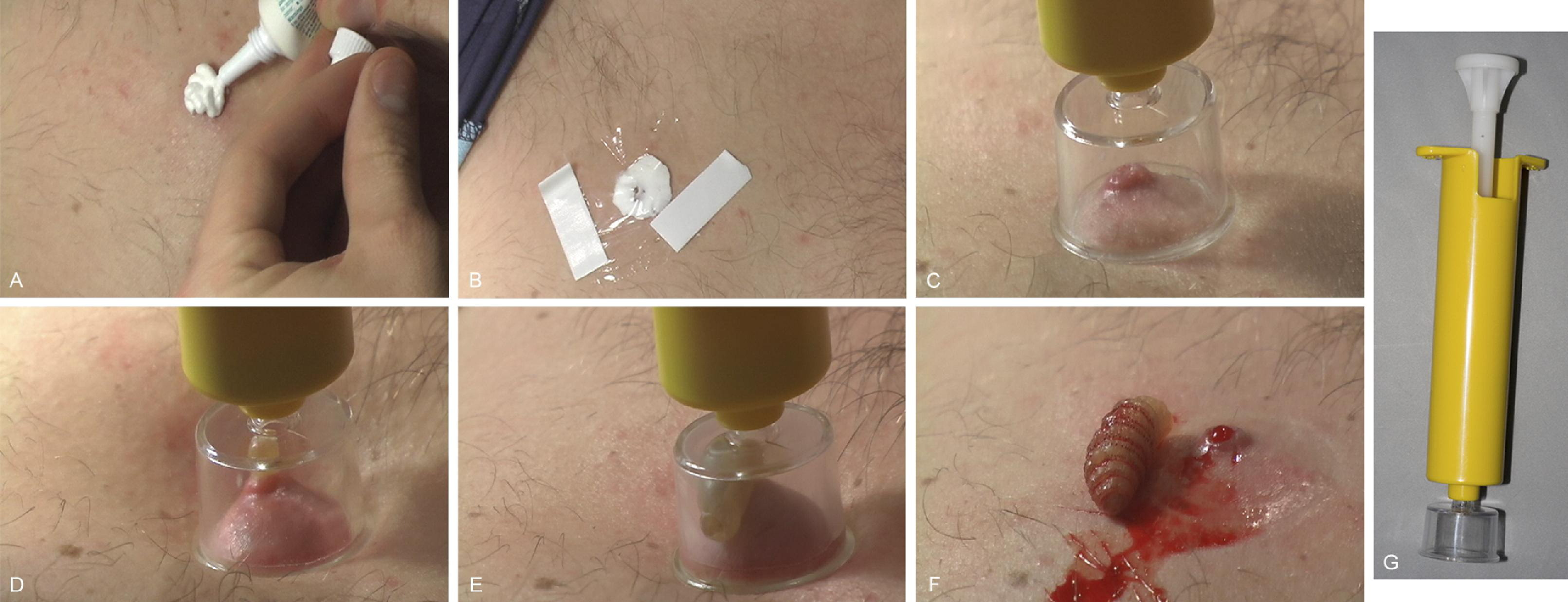
Sequence of images using the venom extractor to remove the botfly larva from the skin. (A) The placement of antibiotic cream over the respiratory aperture. (B) Thin plastic wrap was placed over the cream to create an occlusive dressing and left in place for 30 minutes. The vacuum extractor was (C) applied and (D) activated. In (D) and (E), the action of the extractor is pulling the larva from the subcutaneous cavity. (F) The intact larva and wound immediately after extraction. (G) The venom extractor device. See video in the online version of this article for real-time extraction sequence.
Case Presentation
Two weeks after a trip to Belize in early April, a 25-year-old man noticed an erythematous papule on his upper right chest. The patient had been on vacation and spent a significant amount of time in the jungle, birdwatching in Cockscombe Jaguar Preserve near Maya Center. Initially, the patient felt this papule likely represented a mosquito bite, until it began to hurt and feel like a bee sting approximately 2 weeks after he first noticed the lesion. It enlarged over the next 2 weeks and formed a noticeable, small, central opening in the skin through which secretions were extruded and occasionally found on the patient's clothing. The papule continued to increase in size, and the patient reported feeling movement and intermittent lancinating pains under the skin. These sharp pains would occur 3 or 4 times a day for 3 to 4 days at a time, with pain-free periods of approximately 3 days between episodes. During the eighth week, the pain became somewhat worse.
On physical examination, a 1.5-cm erythematous nodule was seen, with a 3-mm central opening through which movement could be occasionally seen and the caudal end of the larva could be identified (Figure 1). There was no surrounding erythema or signs of infection. The patient's examination and symptoms, coupled with his travel history, were highly suggestive of human botfly infestation or cutaneous myiasis secondary to D hominis. The patient wanted to undergo an attempt at vacuum extraction of the larva owing to his discomfort and unease with having a larva developing under his skin.
The patient's right upper chest was cleaned with an alcohol scrub, and an occlusive dressing was created by placing a generous amount of triple antibiotic cream over the lesion and taping a small piece of plastic wrap over the cream. This dressing was left in place for 30 minutes. Toward the end of the 30-minute period, there was a clear increase in the amount of secretion coming from the aperture in the skin and a significant increase in larval movement. The occlusive dressing was removed and the area wiped clean with an alcohol scrub. The Extractor Pump was applied with an appropriately sized plastic adapter cup completely encompassing the nodule and activated (ie, syringelike mechanism depressed) according to the manufacturer's instructions. The larva was rapidly extracted in approximately 45 s with no significant discomfort to the patient (Figure 3). The larva measured just over 2 cm and was completely intact and motile after extraction (Figure 2). Morphologic examination by the author showed a fusiform-shaped larva with anterior maxillae, caudal spiracles, and concentric, parallel hooks around the anterior body segments consistent with third instar D Hominis larva. Subsequently, the wound was cleaned on the surface and dressed with triple antibiotic ointment and a simple gauze bandage. After 2 weeks, the wound had healed by secondary intention without any special wound care or complications. No scalpel or suture was required. Two months after the extraction, the patient had minimal to no visible scar.
Discussion
Cutaneous myiasis secondary to D hominis is a relatively commonly encountered condition in the United States. Although the human botfly is not endemic to the United States, the clinical manifestations of infestation by this insect can be seen upon the return of travelers from South and Central America who have engaged in adventure activities or ecotourism, such as hiking in the jungle. The tumbu fly of subsarahan Africa, or Cordylobia anthropophaga, is likely the second most commonly encountered cause of cutaneous myiasis in American travelers. Additionally, flies native to the United States can also result in cutaneous myiasis in humans, most notably Cuterebra species. Human infestation from these flies is rare, and the most common hosts are rodents and rabbits. 1
In the current case report, an entomologist was not available for specimen review, but the patient's travel history, symptoms, lesion natural history, and morphology of the larva encountered is classic for D hominis cutaneous myiasis. Most patients present with a single papular lesion, usually on exposed portions of the skin, often the scalp, arms, or upper chest, in areas prone to mosquito bites. Multiple lesions are certainly possible. The lesion enlarges over a period of weeks, and the patient notices an opening in the skin at the center of the lesion. Commonly, intermittent sharp pains are felt at the site, with an accompanying “crawling” feeling under the skin. In equivocal cases, ultrasonography can be used to aid in diagnosis, and will show motility, a hyperechoic segmented fusiform structure (the larva) with a thin hypoechoic rim (the cavity), posterior acoustic shadowing, and internal flow on Doppler sonography. 5 ,10–12 On physical examination, there is a localized papule or nodule with mild erythema and a characteristic breathing aperture at its center. 13 Movement of the larva can be seen through this aperture, especially if the larva is late stage and the lesion is gently prodded to agitate the larva. Typically, there are no systemic symptoms and little surrounding inflammatory reaction, although eosinophilia and elevated white blood cell count have been reported. 5 It seems uncommon that these lesions become superinfected, and that has been hypothesized to be due to antimicrobial effects of the larva's excretory/secretory products. 2 Given this lack of systemic effect, it is generally considered safe to allow the infestation to take its natural course if the patient is able to tolerate the complete cycle. The larva would naturally grow through 3 instars in its subcutaneous pocket for 5 to 12 weeks before exiting the skin through the breathing aperture and dropping into the soil to pupate, with no long-term sequelae to the patient after the wound heals. 5
The majority of people, however, are disturbed knowing that a larva is developing under their skin, and coupled with intermittent pain from the lesion, they usually desire to have it removed as soon as a diagnosis has been made. To this end, a number of remedies and treatments have been previously described. In the hospital setting, the patient will most likely receive a simple surgical incision and debridement. 14 This is a relatively low cost procedure without many complications, but it does require local anesthesia, and there is a risk of incising the larva and spilling contents into the wound, which could lead to inflammation and possible infection. This risk also applies to the method of using manual pressure at the sides of the papule coupled with forceps, as needed, to pull and push the larva out. This pressure method is not consistently successful, 9 owing to the robust anchoring mechanisms of the larva, and does not allow for irrigation of the wound. Moreover, it carries an increased risk of an inflammatory reaction or infection if the larval integument is violated. One paper described the larva being pushed out by injection of lidocaine, negating the need for a surgical incision. 15 There are also reports of myiasis being treated by medical therapy before extraction, using topical ivermectin, an antiparasitic agent used for treating D hominis infestation in cattle. The authors of those reports state that this killed the larva within 4 to 5 hours and made extraction by manual pressure easier. The larva must still be extracted, however, or retention will cause a significant inflammatory granulomatous reaction with increased risk of infection as the larva decays. 16
Many home remedies have been described, centering around coaxing the larva out of the wound by a combination of suffocation and providing a medium for the larva to migrate into. Placing pork fat or other meats over the wound has been described. 17 It is likely more the suffocation than the enticement of meat with this method that results in some limited success at migration of the larva from the wound, as the larva's mouth parts with which it burrows and feeds are buried in the deep aspect of the subcutaneous wound. The most basic form of this treatment is creating an occlusive dressing with petroleum-based products to cover the breathing aperture and suffocate the larva, necessitating its migration toward the surface. This method was employed at the beginning of the procedure in the patient described here to “loosen up” the larva, in the hope of causing the release of some of the anchors as the larva struggled to clear a path through which to breathe. Many previous reports describe leaving occlusive dressings in place for hours to days, with mixed results. Generally, the larva partially backs out of the cavity.9,17 In the current report, the occlusive dressing was in place for 30 minutes. At this time, there was increased larval activity and secretions, as it was clearly attempting to clear the breathing aperture. Although only a subjective observation, the increased secretions during this time do support a role for these secretions in maintaining the breathing aperture. It is thought that this short occlusion did aid in a swift and easier extraction, despite the larva remaining inside the burrow.
In 2002, Boggild et al 9 reported a case of treating botfly myiasis with extraction using a commercial venom extractor after failure of a trial of an 18-hour petroleum jelly occlusive dressing and attempt at extraction with lateral pressure. After these attempts, the posterior end of the larva was seen coming out of the breathing aperture, and the venom extractor was subsequently applied with rapid results—a less than 1-s extraction of a 1.5-cm larva, which was approximately 5 to 7 weeks into its development. 9
This paper presents the second report of extraction of D hominis using the same commercial venom extractor, the Extractor, helping to further validate this technique. There are some important similarities and differences to note between these 2 cases. Both reports are with larvae that are third instar. The larva in the current report was larger (2 cm vs 1.5 cm in the earlier report) and was estimated to be further along in maturation (8 weeks vs 5 to 7 weeks). Importantly, in the current case, only 30 minutes of occlusive dressing time was used, compared with 18 hours in the prior report, with no difference in outcome, suggesting that a short period of occlusion is a time-saving viable alternative for third instar larval extraction. Boggild et al 9 did report a shorter extraction time (1 s vs 45 s in this case). This difference in extraction time could possibly be attributed to the larva being larger in the current case, or it might be related to the longer occlusion time of 18 hours and the attempt at extraction with manual pressure in the prior case report. There was no apparent difference in overall outcome of uncomplicated larval extraction.
This procedure was performed outside of the confines of the hospital. The Extractor is commercially available, lightweight, inexpensive ($15 to $20), and portable for practical field use. However, adding this device to an already cramped first aid kit may not be prudent, as its efficacy in changing the clinical course of a snake bite or other envenomation is not clear, and its use for larval extraction would generally not be required until the patient's return home, as the lesion does not come to the attention of the host for a number of weeks. With this in mind, it may be more practical to stock a small number of these devices in a travel medicine clinic that commonly sees this patient population, educating patients traveling to endemic areas to return to the clinic if skin lesions of this nature develop after their trip. Boggild et al 9 reported rates of cutaneous myiasis as high as 5% to 10% of all skin lesions seen at their institution. This incidence reported from a Tropical Disease Unit (Toronto, Ontario, Canada) is likely higher than for most general practice clinics. With this in mind, it should be considered that other commonly stocked devices such as syringes could possibly be adapted to perform a similar function, but that has not been reported, and although adaptation of such a device may work, it would likely complicate the performance of this procedure.
The advantages of using a venom extractor for removal of botfly larvae include ease of use, noninvasiveness, and rapid, relatively painless extraction without disruption of the larval integument. With the low number of cases reported, there is certainly the possibility for failure of this device in treating other cases. Fortunately, if this method does fail, conversion to surgical excision or use of pressure and forceps should not be made any more complicated by its failure. Consideration should be given to other treatment methods, especially in dealing with first or second instar larva (0 to approximately 3 weeks development 18 ), as both reports of this method are of third instar larva. It is possible that this vacuum method may work well during these early stages, but no data are available regarding its efficacy. The larva will be smaller as a first or second instar, which may be beneficial, but the breathing aperture is also significantly smaller and barely perceptible. At early stages, these lesions are often thought to be mosquito bites by the patient or furuncles, misdiagnosed and treated with antibiotics by physicians, 3 because the characteristic breathing hole is not apparent and symptoms are less specific. No data are available for use of this vacuum method for cutaneous myiasis caused by other larval species (ie, Cordylobia anthropophaga or Cuterebra).
D hominis is a well-known common etiology of cutaneous myiasis endemic to Belize, Central America, and the Northern part of South America. Preventive measures used for mosquitoes, such as insect repellent and netting, should help to prevent botfly infestation when traveling in endemic areas. 19 The diagnosis and treatment of botfly infestation is pertinent to doctors in the United States, as Belize (among other endemic regions in South and Central America) is a common travel destination for North Americans. In this case, the Extractor Pump was demonstrated to be a safe, noninvasive, and painless method for third instar botfly larva extraction in the field without use of hospital resources, helping to validate its widespread use.
Footnotes
Acknowledgments
The author would like to thank Will Gravlee, Robert Wadkins, and Brett Akers for their assistance in treating this patient and documenting the case. The author has no financial interest in the product discussed.
Supplementary data
Supplementary data associated with this article can be found in the online version at
