Abstract
Objective
Prolapse of mitral valve leaflets is a frequent disorder and the most common cause of severe mitral regurgitation in western countries. However, little is known about the effects of altitude on mitral valve prolapse. We studied the prevalence and echocardiographic characteristics of mitral valve prolapse at moderately high altitude and sea level.
Methods
A total of 936 consecutive subjects who were admitted to 2 study institutions at Kars, Turkey (1750 m) and Istanbul, Turkey (7 m) were enrolled in this study to determine prevalence of mitral valve prolapse. Demographic and 2-dimensional echocardiographic characteristics of participants were recorded.
Results
Prevalance of mitral valve prolapse was found to be significantly higher in people living at moderate altitude compared with those living at sea level (6.2% vs 2.0%; P = .007). Overall echocardiographic features regarding valve thickness (4.1 ± 0.80 mm vs 3.6 ± 0.66 mm; P = 0.169), maximal valve prolapse (4.6 ± 2.08 mm vs 3.9 ± 0.91 mm; P = .093), and frequency of mitral regurgitation (89% vs 73%; P = .65) were similar between groups, although anterior valve prolapse was seen more frequently at moderate altitude (50% vs 11%; P = .056) and posterior leaflet prolapse was significantly more frequent at sea level (66% vs 10%; P = .002).
Conclusions
Mitral valve prolapse is more frequently observed at moderately high altitudes. Further studies are needed to determine clinical importance of our findings.
Introduction
Mitral valve prolapse (MVP) is the most common nonischemic cause of severe regurgitation that necessitates surgical intervention in developed countries. 1 An increased propensity for arrhythmias, sudden death, and infective endocarditis is observed with this condition. Mitral valve prolapse is a valvular abnormality that can be caused by histologic abnormalities of valvular and subvalvular tissue, including chordae tendinea and mitral annuli, a disproportion between the left ventricle and mitral valve, or various collagen vascular disorders. 2 Echocardiographically, MVP is defined as an abnormal protrusion of one or both mitral leaflets beneath the annular plane. 2 Primary MVP is a result of progressive myxomatous degeneration, in which redundancy and thickness of leaflets are increased and excess leaflet tissue is present along with a dilated mitral annulus and elongated chords (Figure 1). Mitral valve prolapse is linked to congenital connective tissue disorders such as Marfan or Ehlers-Danlos syndromes, and sporadic forms are found to be associated with genes regulating connective tissue strength or remodeling. 3 Secondary MVP is caused by a wide variety of conditions, including rheumatoid heart disease, ischemic heart disease, or aortic regurgitation with jet impingement on the mitral valve.2,3
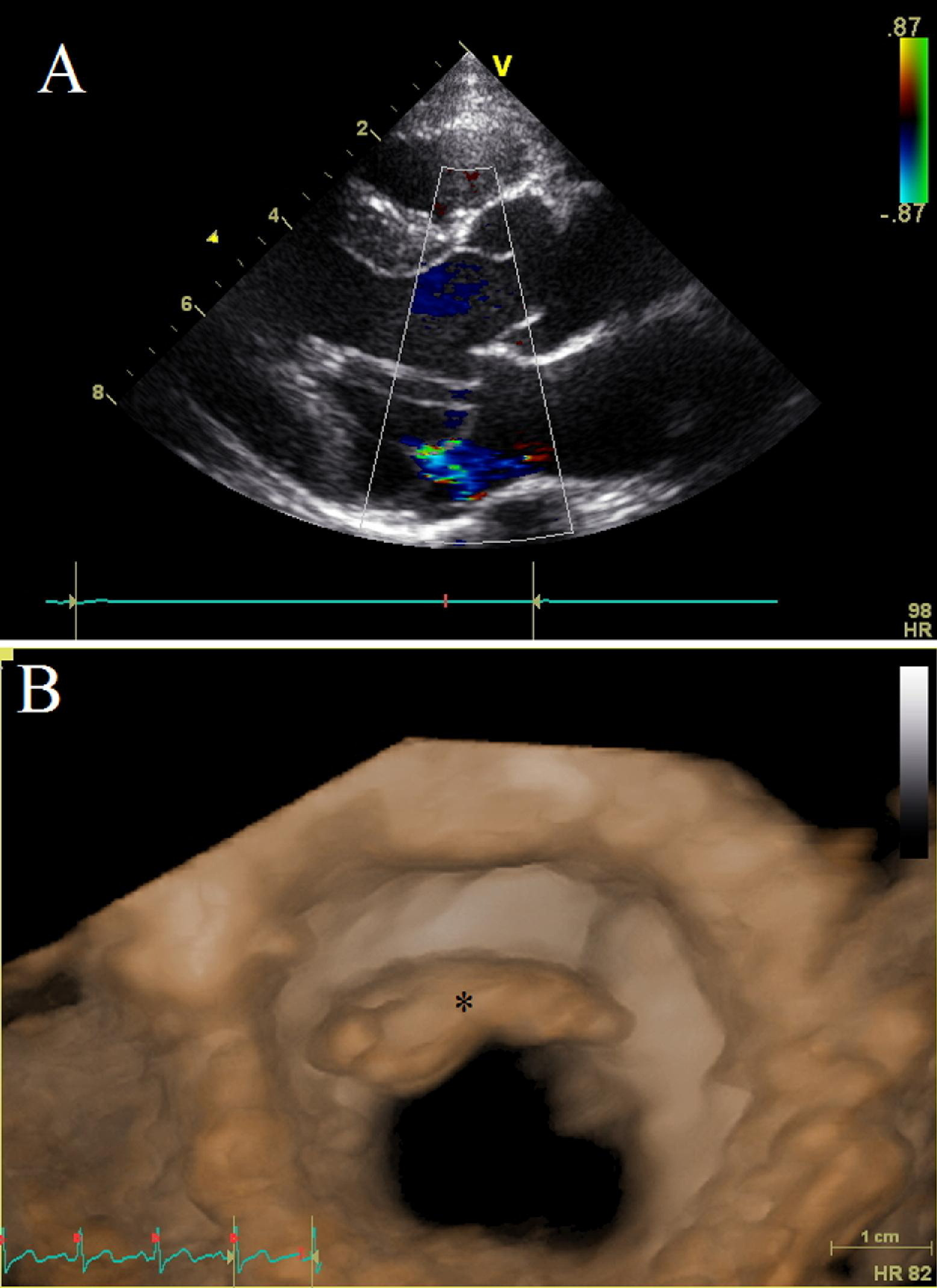
Echocardiographic images of mitral valve prolapse and myxomatous mitral valve. (A) Two-dimensional echocardiographic image of mitral valve prolapse with associated mitral regurgitation. (B) Three-dimensional echocardiographic image of myxomatous anterior mitral valve (shown with asterisk) as seen from left ventricle.
Little is known about environmental effects that may cause or increase progression of MVP. A study conducted on Kyrgyz highlanders in the mid-1980s found an association between living at high altitude and occurrence of MVP, 4 although this association has not been validated with further studies.
In this study, we aimed to determine the prevalence of MVP in subjects living at moderately high altitude. In addition, we compared echocardiographic characteristics of the mitral valve in MVP patients living at moderate altitude and sea level.
Materials and Methods
This study was approved by the ethics committee of Kafkas University School of Medicine, and all participants gave their informed consent before participating in the study. For this study, a total of 494 persons living at moderately high altitude (Kars, Turkey, mean altitude 1750 m) and 442 persons living at sea level (Kartal district of İstanbul, Turkey, 7 m) who were admitted to cardiology departments in the study institutions were consequently enrolled. In both study institutions, enrolled subjects were born at that altitude and had not lived in a different altitude except for brief periods. All enrolled participants were of Caucasian origin and older than 15 years of age. Patients who were previously operated on for mitral valve disease were not included, as the etiology of their underlying mitral pathology could not be determined. Likewise, patients with identifiable causes of MVP, such as rheumatoid valve disease, severe aortic regurgitation with jet impingement on mitral valve, and ischemic mitral prolapse, were excluded. Those subjects who had traveled to sea level (for the moderate altitude group) or to high altitude (for the sea level group) 6 months before the study were also excluded from study.
Age and gender of all participants were noted, and further demographic and laboratory data were collected only in patients with echocardiographic MVP including height and weight of subjects, body surface area, body mass index, and complete blood count.
Echocardiographic Evaluation
Echocardiography was performed on the day of admission by using a GE Vivid 3 (GE Healthcare Systems, Piscataway, NJ) system equipped with a 2.5-MHz phased-array transducer at both institutions (Figure 2). Echocardiographic evaluation was performed by one cardiologist at each institution (TSG and YÇ). Mitral valve prolapse was defined as more than or equal to 2 mm of displacement of 1 or 2 leaflets of the mitral valve superior to the mitral annular plane, without an obvious disorder that may cause prolapse. Additional echocardiographic information regarding the amount of prolapse, leaflet thickness, dimensions of the left ventricular cavity and left atrial cavity, and the presence and degree of mitral regurgitation were acquired from patients diagnosed with MVP. M-mode and 2-dimensional measurements for the left ventricular cavity were performed from the parasternal long-axis view. From this view, the Teicholz formula was applied to calculate the left ventricular end-diastolic volume and ejection fraction. 5 Maximal protrusion of the affected leaflet(s) was measured from the parasternal long-axis view and defined as the distance between the annular plane and the point of maximal protrusion on the mitral leaflet. If both leaflets were protruding, then the leaflet with maximal protrusion was recorded. Maximal width of the affected leaflet(s) was measured at a still frame in which the leaflet is clearly observed apart from the chordae. If both leaflets were affected, the leaflet with maximal thickness was measured, as described above. Left atrial cavity volume was calculated using the anteroposterior dimension obtained from the parasternal view and the mediolateral and apicobasal dimensions obtained from the apical 4-chamber view. These dimensions were multiplied with each other and with 0.523 to obtain the left atrial volume. Mitral regurgitation was assessed using color Doppler interrogation from the parasternal long-axis and apical 4-chamber views. The severity of mitral regurgitation was determined with criteria defined by the American Society of Echocardiography. 5 According to these criteria, regurgitatant flow was further classified as mild and more-than-mild mitral regurgitation.
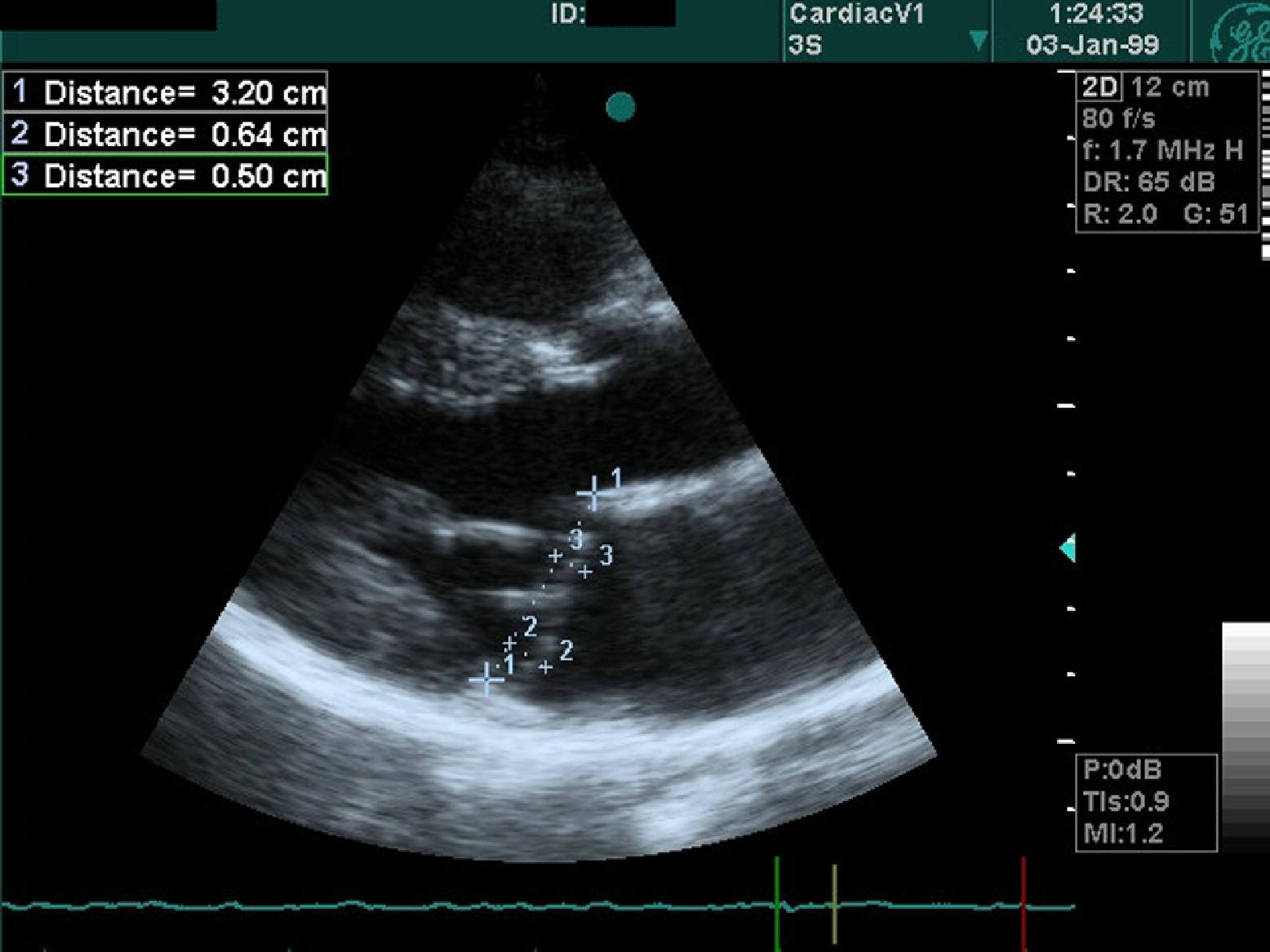
An example of nonclassical mitral valve prolapse that was included in the moderately high altitude group. In this example, maximal prolapse of both leaflets was recorded.
Blood Withdrawal and Analysis
Blood samples were collected on the morning of the echocardiographic examination into 7.5% edetic acid (EDTA). After overnight fasting, whole blood samples were obtained from patients in the supine position from the antecubital vein with a 20-gauge needle by applying minimal tourniquet force. Complete blood count measurement, including hemoglobin and hematocrit levels, was performed 1 hour after venipuncture using an autoanalyzer (Gen-S, Coulter Corporation, Miami, FL).
Statistical Analysis
Statistical analysis was performed using SPSS 16.0 (IBM, Armonk, NY). Quantitative data were given as mean ± SD, and categorical data were given as percentages. Normal distribution and group variances were determined using Kolmogorov-Smirnov and Levene tests, respectively. For parametric continuous variables, a Student's t test was performed, and the Mann-Whitney U test was used for nonparametric continuous parameters. For categorical variables, χ2 and Fisher's exact tests were used, as appropriate. A probability value of less than .05 was accepted as significant. For all data, exact probability values were provided.
Intraobserver Variability
To test intraobserver variability, 50 random patients from sea level and moderate altitude groups were recalled 1 week after the end of study. All recalled patients were subjected to echocardiography for the presence of MVP as defined previously. Percentage of intraobserver variability and Cohen's κ values were calculated as a measure of intraobserver agreement.
Results
Demographic data for study and control groups are summarized in Table 1. Mean age was similar in sea level (50 ± 17.7 years) and moderate altitude (51 ± 16.6 years) groups (P = .24); however, women were more frequently enrolled in the sea level group compared with the moderate altitude group (65% and 56%; P = .007). The prevalence of MVP was threefold higher in the moderate altitude group compared with the sea level group (Figure 3), which was statistically significant (6.1% and 2%, respectively; P = .007). Demographic data regarding MVP patients were similar in both groups, with no difference in age, height, weight, systolic blood pressure, and diastolic blood pressure (Table 1), although systolic blood pressure was insignificantly higher in the moderate altitude group (125 ± 11.9 mm Hg and 116 ± 21.2 mm Hg; P = .077). Hemoglobin levels were significantly higher in the moderate altitude MVP patients compared with patients at sea level (15.00 ± 1.33 g/dL and 12.8 ± 1.16 g/dL; P < .001; Table 2).
Demographic variables of general population and mitral valve prolapse patients living at sea level and moderately high altitude
MVP, mitral valve prolapse.
Data are given as mean ± SD.
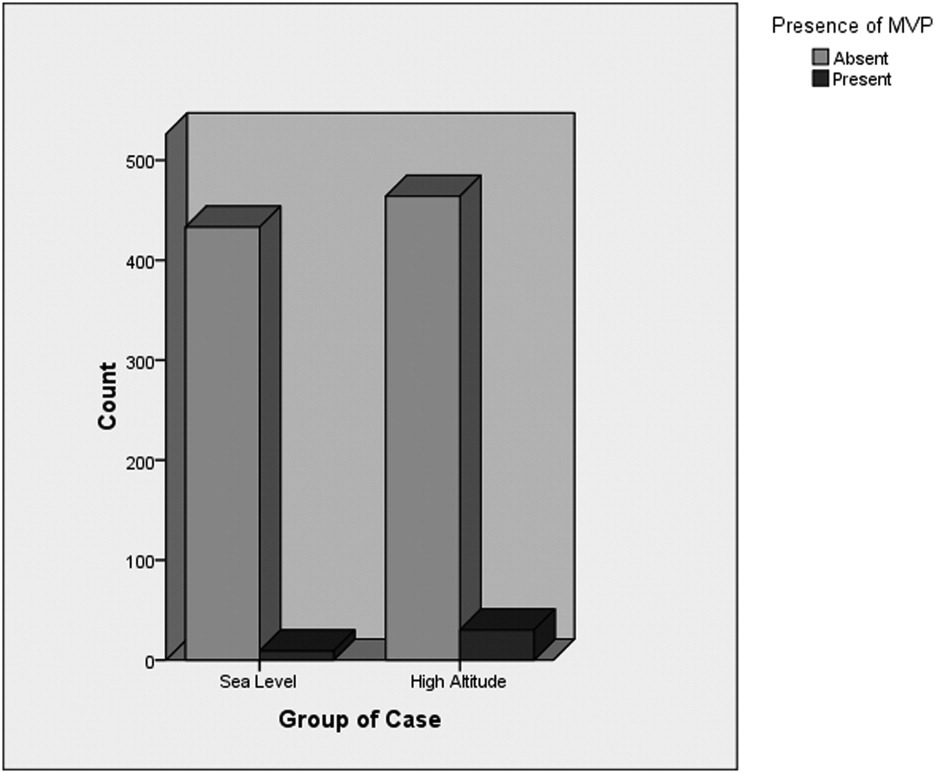
Histographic illustration of total study population and persons diagnosed as mitral valve prolapse (MVP) at sea level and moderately high altitude.
Echocardiographic data and hemoglobin count of patients with mitral valve prolapse living at sea level and moderately high altitude
Data are given as mean ± SD.
Echocardiographic characteristics of MVP in patients living at moderately high altitude and at sea level are given in Table 2. Posterior leaflet prolapse was encountered significantly more at sea level (66% vs 10%; P = .002); however, anterior leaflet prolapse was encountered more frequently in the moderate altitude group (50% vs 11%; P = .056) as a not significant trend. There was no statistically significant difference between groups concerning the frequency of bileaflet prolapse. Maximal leaflet protrusion was higher in the sea level group (4.6 ± 2.08 mm) than in the moderate altitude group (3.9 ± 0.91 mm), but this finding did not reach statistical significance (P = .093). Maximal leaflet width was similar in both groups (4.1 ± 0.80 mm, sea level group; 3.6 ± 0.66 mm, moderate altitude group; P = .169). Mitral regurgitation was present in 89% of patients enrolled at sea level and in 73% of patients enrolled at moderate altitude (P = .65). There were no differences in terms of left ventricular end-diastolic volume (87.5 ± 17.99 mL and 98.1 ± 18.46 mL; P = .15) or ejection fraction (0.59 ± 0.04 and 0.60 ± 0.04; P = .33).
Intraobserver agreement for the moderate altitude group was 94% (Cohen's κ = .788; P < .001) and 96% (Cohen's κ = .728; P < .001) for the sea level group.
Discussion
In this study, we have demonstrated an increase in MVP prevalence in residents of moderately high altitude compared with people living at sea levels. Echocardiographic characteristics, in terms of leaflet width, maximal systolic prolapse, and the presence or severity of mitral regurgitation, were similar in both groups, with the majority of cases having “nonclassic” MVP (width of leaflet <5 mm) and presenting with mild mitral regurgitation that had no immediate clinical impact on the patients.
Etiology of MVP remains poorly understood despite the fact that it is the most common cause of severe mitral regurgitation in the developed world. 6 Mitral valve prolapse is observed with congenital connective tissue disorders including Marfan syndrome, Ehlers-Danlos disease, Sticler syndrome, and adult type polycystic disease. 7 Primary MVP, which is not associated with other diseases that affect the valve or the subvalvular apparatus, is mostly associated with myxomatous degeneration.8,9 Myxomatous degeneration is macroscopically associated with leaflet thickening and redundancy, chordal elongation, and annular dilatation.10,11 Histopathologically, the spongiosa layer is expanded with proteoglycan accumulation, along with altered collagen structure and abnormal chordae. 10 ,12,13 An increased expression of proteolytic enzmes and matrix metalloproteinases, as well as mutations in the filamin A gene, is observed in patients with MVP. 14 –16 Leaflet thickening appears to be the most important clinical determinant of the disease course, as it is correlated with the risk of sudden death, endocarditis, and mitral regurgitation. 17
An environmental effect on valve prolapse is an inadequately studied subject, as this disorder is mainly considered to have genetic origins. In a series consisting of 1734 newborns, only 19 had a prolapsing valve, and none of them had leaflet thickening or pathologic mitral regurgitation. 18 This study demonstrated that postnatal environmental factors may have a role either in formation or progression of myxomatous degeneration and MVP. In a landmark study conducted on mountain-dwelling Kyrgyz people, the authors showed the prevalence of MVP is affected by altitude, with a higher prevalence found in higher altitudes, although this study did not provide valve features such as leaflet thickness or the degree of prolapse. 4 In that study, the incidence of MVP was found to be twice as great at 2000 m as that found at 760 m, which is similar to our findings. Aldasheva et al 4 concluded that the association between MVP and altitude could be clinically important, because high altitude residents were also observed to have more severe symptoms. Our results are in accordance with this study. Patients with MVP and living at sea level or moderate altitude had similar valve characteristics, with similar prevalence of mitral regurgitation, and the severity of mitral regurgitation is mostly mild in both groups. Likewise, thickness of the prolapsing leaflets or maximal protrusion of the prolapsing leaflets was similar in both groups. In the majority of cases, leaflet thickness was less than 5 mm, and therefore classified as nonclassic MVP.
High altitude has profound effects on cardiovascular physiology, both acutely and after acclimatization. A decrease in plasma volume, which is persistent after acclimatization, leads to a decrease in left ventricular preload. 19 A drop in preload consequently translates into a reduction of left ventricular dimensions and stroke volume, without impairment in contractile function. Reduction in left ventricular dimension may partly explain the increased prevalence of MVP seen at moderately high altitude, as reduced left ventricular volume relative to the length of the chordae may result with valve prolapse, so-called ventriculoannular disproportion. This condition is considered to be benign and self-limiting, and disappears with advanced age. However, this may not be the case at high altitude, as a chronic decrease in preload keeps left ventricular dimensions lower than expected during life, and ventriculoannular disproportion may be sustained.
Neurohormonal derangement, particularly concerning the renin-angiotensin-aldesterone system and the sympathetic nervous system, were studied in patients with isolated MVP and MVP syndrome. Perturbations in the renin-angiotensin-aldosterone system, including abnormal plasma renin activity and absence of changes in plasma aldosterone levels, were demonstrated in dogs with MVP, as well as in those with MVP syndrome.6,20 Sympathetic activation, as demonstrated with higher 24-hour urinary vanilmandelic acid excretion, increased plasma epinephrine and norepinephrine levels, and excessive α-adrenergic activation, was observed in patients with MVP syndrome, as well as in those with asymptomatic isolated MVP. 6 ,21,22 This is of particular interest, as sympathetic activation and decreased vagal stimulation were observed in normal subjects at high altitudes. 23 This increase in activity is present even after acclimatization, 24 but is absent in natives living at very high altitudes. 25 This increase in sympathetic stimulation could be associated with the increased MVP prevalence observed at high altitudes.
Study Limitations
This study was conducted on people admitted to a cardiology department, and therefore does not represent the true population. However, we conducted echocardiography consequently to all applicants who gave informed consent, therefore minimizing potential bias originating from patient selection. A study with random selection design and application of echocardiography to this sample would be clearly preferable to our study design.
As our study was observational in design, our results show an increase in the prevalence of MVP, although it lacks giving an explanation that might underlie such an association. As stated in the preceding discussion, ventriculoannular disproportion may in part explain this increased prevalence. Unfortunately, as we did not collect information regarding ventricular and mitral annular dimensions from all participants, our data are inadequate to show the presence of ventriculoannular disproportion in the study population. It should also be considered that a decrease in preload becomes more manifest at extremely high altitudes, 22 so ventriculoannular disproportion may not be the sole cause of the increased MVP prevalence observed at moderately high altitude.
Another possible explanation is activation of the sympathetic nervous system that is observed in people living at high altitude. However, our study was not designed to measure sympathetic activity, and it is not possible to conclude that increased sympathetic drive is present in subjects with MVP living at moderately high altitude. It should also be mentioned that direct evidence linking the adrenergic system to the development of MVP is lacking, and heightened sympathetic activity per se might be an associated condition rather than a causative factor for MVP.
Our results indicate that more women were recruited at sea level compared with the number recruited at moderately high altitude, which may be a confounding factor that may affect results. Studies reported in the literature have shown that either MVP is seen more frequently in women 26 or there is no gender difference at all with regard to MVP. 27 Therefore, the gender difference in our results is either not expected to influence our results or there could be a bias favoring the sea level group. In both situations, however, neither the prevalence of MVP in the moderately high altitude group nor the statistical difference between groups should be altered.
Diagnosis of MVP is best accomplished using 3-dimensional echocardiography, as the mitral valve is saddle-shaped and 2-dimensional images could inappropriately show physiologic movement of the mitral valve as valve prolapse. 2 This is especially true when the apical 4-chamber view is used to diagnose valve prolapse. This effect is minimized when parasternal long-axis views are used, with a cutoff value of 2 mm protrusion superior to the mitral annular plane. In our study, we used the parasternal long-axis view with the aforementioned criteria, so we expect minimal false-positive cases.
Conclusions
Although our study indicates a possible relationship between the prevalence of MVP and long-term residence at moderate altitude, valve characteristics and the frequency of mitral regurgitation were similar to those of patients living at sea altitude. Further studies are required to identify the cause of this phenomenon and the implications this has for those living at moderate altitude.
Footnotes
Disclaimer: No conflicts of interest to disclose.
Presented at the 8th International Congress of Update in Cardiology and Cardiovascular Surgery, March 1–4, 2012, Antalya/Turkey.
