Abstract
Objective
This study aimed to compare 3 treatment modalities during sleep at an altitude of 5300 m to identify strategies for reducing the incidence of periodic breathing at high altitude.
Methods
Fifteen trekkers, with identical ascent profiles and no signs or symptoms of altitude illness, served as subjects. All study participants arrived at 5300 m after a gradual ascent from 1300 m. On their second night at 5300 m, subjects were randomly assigned (with a computer
Results
Upon comparing the 4 groups, there were no statistically significant differences between the variables. One-way analysis of variance indicated a trend toward statistical significance for Sa
Conclusions
These statistical trends found between control and treatment groups indicate that further study is warranted.
Introduction
On ascending to altitude, unacclimatized lowlanders typically complain of poor sleep. This is often characterized by a difficulty in getting to sleep, a restless night punctuated with frequent awakenings, and a feeling of tiredness the next day.
1
In most cases, this is due to cycles of periodic breathing that occur as a result of the fall in the partial pressure of oxygen (P
In this small study, our intention was to compare the effects of O2, O2/CO2 mixture, and acetazolamide vs placebo on nocturnal oxygenation and ventilation at 5300 m. We wished to identify a practical strategy for reducing the incidence of nocturnal periodic breathing at high altitude.
Methods
This system consisted of a nasal cannula attached to a battery-operated pulse dose meter and released oxygen into the nasal cavity when a fall in pressure was sensed along the nasal cannula. A dial on the regulator controlled the volume of oxygen delivered by each pulse. Throughout the study, the participants slept in a comfortable well-ventilated 2-man tent. The nasal demand system and oxygen cylinders are manufactured and tested to UK safety specifications. The accuracy of the pulse dose was confirmed by manufacturer's tests prior to departure and following the return of the system to the UK.
The LifeShirt system is an ambulatory, multi-sensor, continuous monitoring system for collecting, analyzing, and reporting physiologic data. The LifeShirt is able to collect continuous physiologic data throughout the study night via various sensors, including basic pulmonary function via respiratory inductive plethysmography (RIP) bands, electrical activity of the myocardium via a 3-lead echocardiogram, and arterial oxygen saturation via pulse oximetry.
The sensor array of the LifeShirt System is embedded in a sleeveless undergarment made of Lycra material that fits snugly and can be worn comfortably for extended periods. RIP sensors for monitoring a variety of pulmonary signals are embedded in this shirt. This ensures their correct and durable placement and allows multichannel recording. The RIP sensors consist of a sinusoidal arrangement of electrical wires that are excited through an extremely low current, electrical oscillator circuit. One sensor was sewn into the shirt at the level of the rib cage (fourth intercostal space) and one at the level of the abdomen (umbilicus). From these signals, a variety of calibrated respiratory pattern measures are extracted, such as minute ventilation, tidal volume, and respiratory rate. Additionally, apneas and hypopneas (identification and classification) can be automatically detected. An onboard personal digital assistant continuously encrypts and stores the patient's physiologic data on a compact flash memory card. VivoLogic, a proprietary personal computer-based software, decrypts and processes recorded data and provides viewing and reporting features.
Results
In the 4 groups enrolled into this study, there were no differences in HR, RR, SaO2, VT, v̇E, and AHI when compared to each other or the control group. Means with 95% CI for each of the 4 groups are listed in Table 1, and Figure 1 shows the raw data (VT, AHI, v̇E, and SaO2) for each group. One-way analysis of variance indicated a trend toward statistical significance for SaO2 between groups (F = 2.9, P = .08), but this analysis yielded no other results approaching statistical significance (HR, F = 1.2, P = .3; RR, F = 2.5, P = .1; Vt, F = 1.8, P = .2; v̇E, F = 1.1, P = .39; and AHI, F = 0.7, P = .6). More specifically, Tukey HSD post hoc tests indicated a statistical trend in the SaO2 difference between the O2 and control groups (P = .07) as well as in the RR difference between the O2 and O2/CO2 groups (P = .08).
Mean (95% CI). P values represent differences between groups from 1-way analysis of variance
HR, heart rate; RR, respiration rate; SaO2, blood oxygen saturation; Vt, tidal volume; v̇E, minute volume; AHI, apnea hypopnea index.
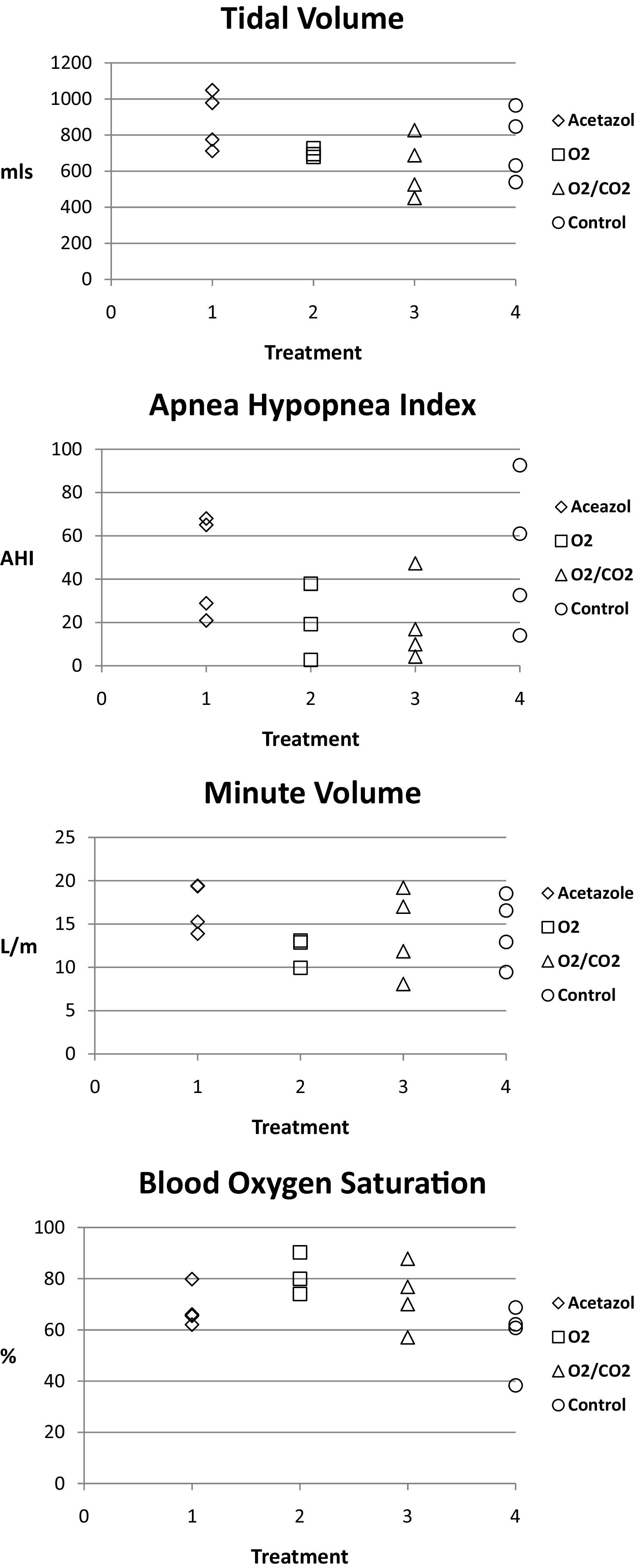
Raw data plotted for blood oxygen saturation (SaO2), tidal volume (Vt), minute volume (v̇E), and apnea hypopnea index (AHI). (Treatment: 1 = acetazolamide, 2 = O2, 3 = O2/CO2, and 4 = control).
Discussion
This study emphasizes the extraordinary effect that a low P
These results demonstrate the wide range of interindividual cardiopulmonary responses to sleep at high altitude. Perhaps the most prominent finding is that the subjects taking O2 had the highest percent SaO2 results (81.4 ± 4.7), yet the lowest v̇E (11.9 ± 1.0). The mean AHI results for the O2 and O2/CO2 groups, 20 (± 10.2) and 19.6 (± 9.6) respectively, were virtually identical values. These results should nevertheless be viewed and interpreted with the understanding that over 100 years ago, respiratory physiologists discovered that very small changes in PCO2 were sufficient enough to dramatically alter ventilation. Haldane and Priestley 9 showed that an increase in alveolar PCO2 of about 1.4 mm Hg was sufficient to double the alveolar ventilation at rest in healthy humans. Expanding on this work, Douglas and Haldane showed that hypocapnia caused by hyperventilation with an oxygen-enriched gas produced apneas, but not periodicity, in the apnea pattern. 10 This indicated that the development of hypoxia during nocturnal central apneas at high altitude was important in the subsequent Cheyne-Stokes-like periodicity.
More recently, this finding was supported by a study conducted during the landmark American Medical Research Expedition to Mount Everest in 1981, when Lahiri and colleagues showed that carbon dioxide inhalation (without supplementary oxygen) eliminated central apneas during sleep at high altitude, but not the respiratory oscillations. 11 In agreement with our results, Lahiri and coworkers 11 also showed an increase in RR and a decrease in maximal Vt with carbon dioxide inhalation. Taken together, such background knowledge provides strong support for O2/CO2 mixtures (rather than O2 or CO2 alone) as an aid to sleep-breathing problems at high altitude. However, the results from our study could be interpreted to suggest that the extra work of the heart and lungs when using this particular O2/CO2 mixture (compared to O2 alone) is of no physiological advantage. Additionally, when considering the use of supplemental gas delivered from a metal tank vs pharmaceuticals in pill form, it must be kept in mind that the weight of the former may in some cases be a factor in its effective utilization. This is especially true in circumstances such as alpinism or high altitude trekking where every kilogram must be scrutinized.
Although certain trends are visible from our results, a number of study limitations are present. The small number of subjects in each arm of the study, the relatively large variation in subject response, the limited number of physiological parameters measured, and the relatively low doses of gas and acetazolamide may have led to our inability to identify differences between our groups. While a larger sample size may have resulted in a smaller CI within each group, the use of a higher dose of acetazolamide or supplemental gas may have resulted in a greater increase in oxygenation and significant changes in the other physiological variables we measured. It should also be noted that our results are not necessarily in line with other reports as reviewed in Weil 2 that suggest a 125 mg dose of acetazolamide is effective at reducing nocturnal periodic breathing at high altitude. We were intrigued by these findings and, while we have no definitive explanation for this, it is possible that the respiratory and metabolic acclimatization gained from a gradual 13-day ascent to 5300 m prior to the study (during the night of day 14) may have influenced the subject's response to acetazolamide. In addition, sleep staging was not measured, since the equipment needed to record EEG activity was not available and no subjective data on participant perception of sleep quality was collected.
It should also be noted that the well-known phenomenon of night-to-night respiratory variability may have produced somewhat different results if these studies had been repeated with the same subjects on multiple consecutive nights. For instance, in studies examining multiple study nights in the same subjects for comparison, there is evidence of broad interstudy variation in the percentage of subjects having discordant findings in AHI between study nights—for example, from 43% to 8%.12,13 Therefore, the case has been made that a negative first-night study is insufficient to exclude the diagnosis of sleep apnea in patients with clinical markers of this disease because of the potential for night-to-night variability.14,15 As such, the results of our single study night may have been somewhat different if it had been repeated on a second night. In line with the repeated-measures “ideal” for a study such as this, the investigation would have been strengthened if all subjects had acted as their own control on a preceding or subsequent study night.
To summarize, in this small pilot study we were unable to identify differences between those in our control and treatment groups. However, larger studies using similar or higher doses of acetazolamide, O2, and O2/CO2 mixtures may, in the future, be able to identify key differences between these treatments at high altitude. In addition, we can confidently report that the novel equipment used to deliver supplemental gas and measure cardio-respiratory parameters in this study are well suited to the remote high altitude environment.
Footnotes
Acknowledgments
The research was funded from a variety of sources, none of which are public. The entrepreneur John Caudwell, whose name the expedition carries, donated £500,000 specifically to support the research. BOC Medical, now part of the Linde Group, generously supported the research early on and continues to do so. Eli-Lilly Critical Care, Smiths Medical, Deltex Medical, The London Clinic (a private hospital), and Rolex have also donated money to support the research and logistics. All monies were given as unrestricted grants.
Funding
Specific research grants were awarded by the Association of Anaesthetists of Great Britain and Ireland, the Halley Stuart Foundation, and the UK Intensive Care Foundation. The CXE volunteers who trekked to Mount Everest Base Camp also kindly donated to support the research.
