Abstract
Objective
High altitude leads to an increase in sympathetic nervous system (SNS) activity and pulmonary arterial pressure (PAP). We assessed whether the SNS contributes to this increase in PAP.
Methods
Sympathetic discharge to the pulmonary vasculature was assessed by measuring plasma norepinephrine concentrations in central venous blood entering the lung and systemic arterial blood leaving the lung (arterial-central venous difference; a − cvdiff). Sympathetic activity in the adrenal gland was assessed by measuring systemic plasma epinephrine concentrations. The a − cvdiff of epinephrine was assessed to investigate its metabolism across the lung. The measurements were performed in 34 mountaineers during both rest and exercise at low altitude and after 20 hours at high altitude (4559 m). Norepinehrine and epinephrine concentrations were measured by high-performance liquid chromatography. Pulmonary blood flow was assessed by inert gas rebreathing, and systolic PAP (PASP) by transthoracic Doppler-echocardiography.
Results
Exercise and high altitude increased PASP and increased arterial and central venous plasma norepinephrine. In contrast, exercise but not high altitude increased arterial and central venous epinephrine. There was no significant a − cvdiff for norepinephrine and epinephrine during rest and exercise at low altitude, nor during rest at high altitude. However, during exercise at high altitude the a − cvdiff for norepinephrine was positive. There was no correlation between the a − cvdiff of both norepinephrine and epinephrine with PASP during exercise, high altitude or during a combination of both.
Conclusions
The degree of pulmonary hypertension that occurs upon high-altitude exposure is largely independent of the SNS activity in the pulmonary vasculature and adrenal gland.
Keywords
Introduction
Exposure to high altitude increases vascular resistance and pulmonary arterial pressure (PAP). 1 While there are numbers of explanations for this, we sought to explore the contribution of the sympathetic nervous system (SNS) to these phenomena.
High altitude is a stimulator of SNS activity as indicated by two lines of evidence: first, increased plasma levels of norepinephrine (indicator of sympathetic neurone discharge) and epinephrine (indicator of sympathetic activity in the adrenal gland) have previously been found upon high altitude exposure. 2 -4 Second, microneurographic recordings demonstrate an increased efferent sympathetic discharge to the skeletal muscle vascular bed upon high altitude exposure. 5 -7 It has been postulated that the increase in SNS activity accounts for the degree of high-altitude pulmonary hypertension. This hypothesis is based on the observation that increased systemic plasma levels of norepinephrine were associated with the increase in PAP at high altitude, 2 and that the degree of muscle sympathetic nerve activity positively correlated with the increase in PAP. 5 Moreover, the alpha-adrenergic blocker phentolamine has been shown to be more effective in decreasing PAP at high altitude than other non-specific vasodilators. 8
To investigate the role of the SNS in high altitude-induced pulmonary hypertension, we assessed sympathetic neurone discharge to the pulmonary vascular bed (1) by measuring differences in plasma norepinephrine concentrations between central venous blood entering the lung and systemic arterial blood leaving the lung (arterial-central venous difference; a − cvdiff) and (2) by measuring plasma epinephrine concentrations as indicator of sympathetic activity in the adrenal gland. We further assessed the a − cvdiff of epinephrine to characterize its metabolism across the lung. We correlated the changes of these parameters with changes in systolic PAP (PASP) in 34 mountaineers during rest and exercise at both low and high altitude (4559 m). We hypothesized that the increase in PASP during exercise and acute high altitude would be associated with increased transpulmonary production (indicated by a positive a − cvdiff) of plasma norepinephrine and with a decreased transpulmonary gradient (indicated by a negative a − cvdiff) of epinephrine due to adrenergic receptor binding.
Methods and Materials
Study Population
The study was conducted in accordance with the Declaration of Helsinki and its current amendments, and was approved by the Ethics Committee of the Medical Faculty of the University of Heidelberg. Thirty-four (29 male, 5 female) nonacclimatized healthy subjects (age: 37 ± 10 years; body weight: 77 ± 10 kg; height: 178 ± 9 cm) living at low altitude were included after written informed consent. Six subjects had a history of at least 1 documented episode of HAPE.
General Procedures
Two to 4 weeks following baseline assessement at 110 m (Heidelberg, Germany), subjects ascended in less than 24 hours from 1130 to 4559 m (Capanna Regina Margherita), with an intervening overnight stay at 3611 m. Measurements were performed on the second day at high altitude, roughly 20 hours following arrival at 4559 m. Participants who developed HAPE were treated with nifedipine and supplemental oxygen, and headache was treated with acetaminophen (500 mg) or ibuprofen (400 mg). One mountaineer was excluded from the data analyses due to HAPE on the first night. For all other mountaineers requiring nifedipine or oxygen, the measurements were performed before treatment was initiated. In addition to the data reported in this paper, studies on pulmonary endothelial function and lung function were performed and reported separately.9,10
Diagnosis of HAPE and Acute Mountain Sickness
HAPE was clinically suspected at the appearance of dry cough, orthopnea, or pulmonary rales in at least 1 lung area. A posteroanterior thorax radiograph was then obtained as described previously. 11 Radiographs were scored retrospectively by a radiologist blinded to the study design. Interstitial or alveolar edema in at least 1 lung quadrant was judged as diagnostic of HAPE. Acute Mountain Sickness (AMS) was evaluated using the Lake Louise Score and the AMS-C score of the Environmental Symptom Questionnaire. Subjects were deemed to have AMS if they had a Lake-Louise Score ≥ 5 points and an AMS-C score ≥ 0.70 points. 12
Exercise Tests
Exercise tests were performed at low and high altitude on a bicycle ergometer in semisupine position while arterial blood pressure and 12-lead electrocardiography were recorded. Maximal individual workload was determined in normoxia as described previously. 13 Briefly, subjects performed an exercise test starting at 50 W with an increase of 25 W every 2 minutes until exhaustion. During the study day at low altitude, subjects exercised at 30% of maximal oxygen consumption (VO2max) for 15 minutes, and then for 10 minutes at 50% VO2max. The subjects repeated this protocol at high altitude. Because the sympathetic response is dependent on the relative exercise intensity, the level of exercise was adjusted such that exercise was performed at similar relative workloads during both low and high altitude. Therefore, the high altitude studies were performed at 21% and 35% of the individual VO2max, because VO2max declines with increasing altitude. The decrease in VO2max begins at about 700 m with a linear reduction of about 8% for every additional 1000 m up to approximately 6300 m. 14 Thus, at 4500 m VO2max is reduced by about 30%. Central venous and arterial blood was obtained before and during the last minute of exercise.
Assessment of Pulmonary Artery Systolic Pressure (PASP)
PASP was assessed by 2-dimensional and Doppler-echocardiography (Acuson Cypress, Siemens, Germany) with a 2.5 MHz duplex transducer according to the modified Bernoulli equation as described previously. 13 This method has previously been shown to closely correlate with invasive measurements of PASP at high altitude. 15 All measurements were recorded and evaluated offline in random order by a sonographer blinded to clinical and experimental data. The low and high altitude PASP measurements were performed at rest and during the last minute of exercise at a VO2max of 50% at low altitude and of 35% at high altitude. During exercise PASP could not be assessed in 12 subjects at low altitude and in 10 subjects at high altitude, as a result of insufficient Doppler profiles. Pulmonary vascular resistance (PVR) was calculated as mean PAP/pulmonary blood flow (PBF). Mean PAP was calculated as PASP × 0.6 + 2 as described previously. 16
Measurement of Plasma Norepinephrine and Epinephrine
A polyethylene catheter (RA 04020; Arrow International, Inc., Reading, PA, USA) was inserted into the radial artery under local anesthesia (2% lidocaine HCl, Braun Melsungen AG, Germany). A central venous catheter (Cavafix certodyn 375 catheter, Braun Melsungen AG, Germany) was inserted via an antecubital vein and advanced under electrocardiographic guidance into the superior vena cava. 17 Catheter patency was maintained with a pressurized flush system of normal saline.
Blood samples were drawn simultaneously from the radial artery and superior vena cava. To minimize subject's risk, mixed venous blood samples were not drawn as this would have required pulmonary artery catheter placement. However, a previous study in resting subjects yielded no significant difference between the norepinephrine concentration in the superior vena cava and the pulmonary artery. 18 Blood samples were put on ice, centrifuged at 800 g for 15 minutes, and the plasma was immediately frozen in liquid nitrogen. Plasma norepinephrine and epinephrine were measured by high-performance liquid chromatography following return to low altitude as described previously. 19 In 2 mountaineers the catecholamine data set could not be completed due to problems with arterial blood sampling during exercise.
Pulmonary Blood Flow and Transpulmonary Differences of Plasma Norepinephrine and Epinephrine
Pulmonary blood flow (PBF) was determined noninvasively by inert gas rebreathing (Innocor, Innovision A/S, Odense, Denmark) using an oxygen-enriched mixture of 0.5% nitrous oxide and 0.1% sulfur hexafluoride measured with photoacoustic analyzers over a 5-breath interval. 20 Pulmonary plasma flow was calculated as [PBF × (1-hematocrit)] with hematocrit expressed as a fractional value. Transpulmonary plasma exchange of norepinephrine and epinephrine, respectively, was calculated as [arterial-central venous difference (a − cvdiff) × pulmonary plasma flow].
Statistics
Normality was tested using the Kolmogorov-Smirnov test. For normally distributed variables 1-way repeated measures analysis of variance (ANOVA) was used. Variables that were not normally distributed were compared using Friedman repeated measures ANOVA on ranks. Pairwise multiple comparison procedures were made using the Tukey-Test. The relationship between pairs of variables was expressed with the Pearson Correlation Coefficient for normally distributed variables and with the Spearman Rank Order Correlation for not normally distributed variables. Subgroup analysis for subjects with HAPE were not performed because there were only 4 cases. Data are expressed as mean values ± SD; p < 0.05 was considered statistically significant. Statistics were performed using the SigmaStat® software package (SPSS Inc., Chicago, IL).
Results
Observed Changes in Subjects With Acute Altitude Illness
During the stay at high altitude, 14 subjects developed AMS, and 4 subjects developed HAPE. Three of the subjects with HAPE had a previous history of at least 1 documented episode of HAPE. Three of the 4 subjects with HAPE also had AMS. Between the subjects with and without AMS there were no differences with regard to PASP, PVR, pulmonary blood and plasma flow, plasma norepinephrine and epinephrine concentrations, a − cvdiff, and transpulmonary plasma exchange of norepinephrine and epinephrine, respectively.
Effects of Exercise and Altitude on PASP and Hemodynamics
Exercise and high altitude decreased arterial PO2 (Table 1) and increased PASP (Figure 1) with a negative correlation between both parameters at high altitude (R = −0.28; p < 0.05). As summarized in Table 1, high altitude exposure was associated with an increase in PVR, heart rate, and systolic and diastolic blood pressure. In contrast, resting pulmonary blood and plasma flow were not affected after 20 hours at high altitude. During exercise, pulmonary blood and plasma flow increased, as did systolic blood pressure and heart rate (Table 1). After 5 minutes of exercise at a VO2max of 50% at low altitude, and of 35% at high altitude, respectively, heart rate had reached a steady state that was stable until the end of the exercise test (136 ± 15 versus 136 ± 16 beats/minute after 5 and 10 minutes at low altitude, and 141 ± 12 versus 143 ± 13 beats/minute after 5 and 10 minutes at high altitude).
Parameters of hemodynamics at low and high altitude
PVR = Pulmonary vascular resistance. Data are expressed as mean (±SD).
p < 0.05 versus low altitude.
p < 0.05 versus rest at the same altitude.
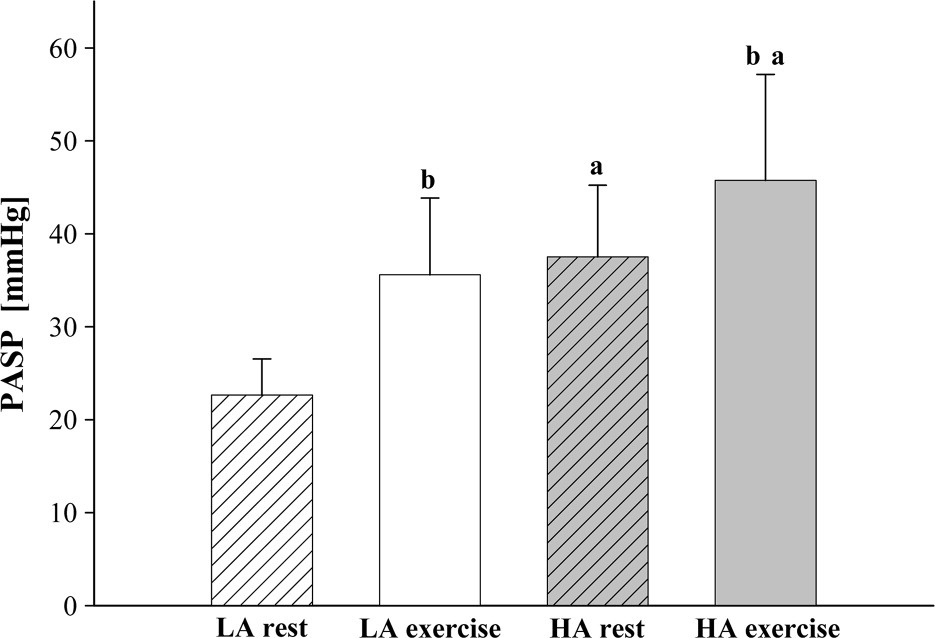
Pulmonary artery systolic pressure (PASP) at low (LA) and high altitude (HA) during rest and exercise. Data are expressed as mean (± SD). a p < 0.05 versus low altitude; b p < 0.05 versus rest at the same altitude.
Plasma Norepinephrine
Exercise and high altitude led to an increase in central venous norepinephrine plasma concentrations (Figure 2a). This increase was even more pronounced when both stresses were combined, ie, during exercise at high altitude (Figure 2a). The changes in central venous plasma norepinephrine were paralleled by comparable changes in arterial norepinephrine plasma concentrations (Figure 2b). Calculation of the transpulmonary exchange of plasma norepinephrine, which corrects for individual differences in pulmonary plasma flow, revealed no significant transpulmonary gradient at low-altitude rest and exercise, nor during rest at high altitude (Figure 2c). Only during exercise at high altitude was a significant transpulmonary gain of plasma norepinephrine seen (Figure 2c). As summarized in Table 2, there was no significant correlation between both PASP and PVR with the a − cvdiff and the transpulmonary exchange of plasma norepinephrine, neither during exercise nor during high altitude or during a combination of both. There was also no correlation between the exercise and altitude-induced changes of these parameters (ie, changes in the a − cvdiff and changes in the transpulmonary exchange of plasma norepinephrine) and the changes in PASP and PVR, respectively (not shown).
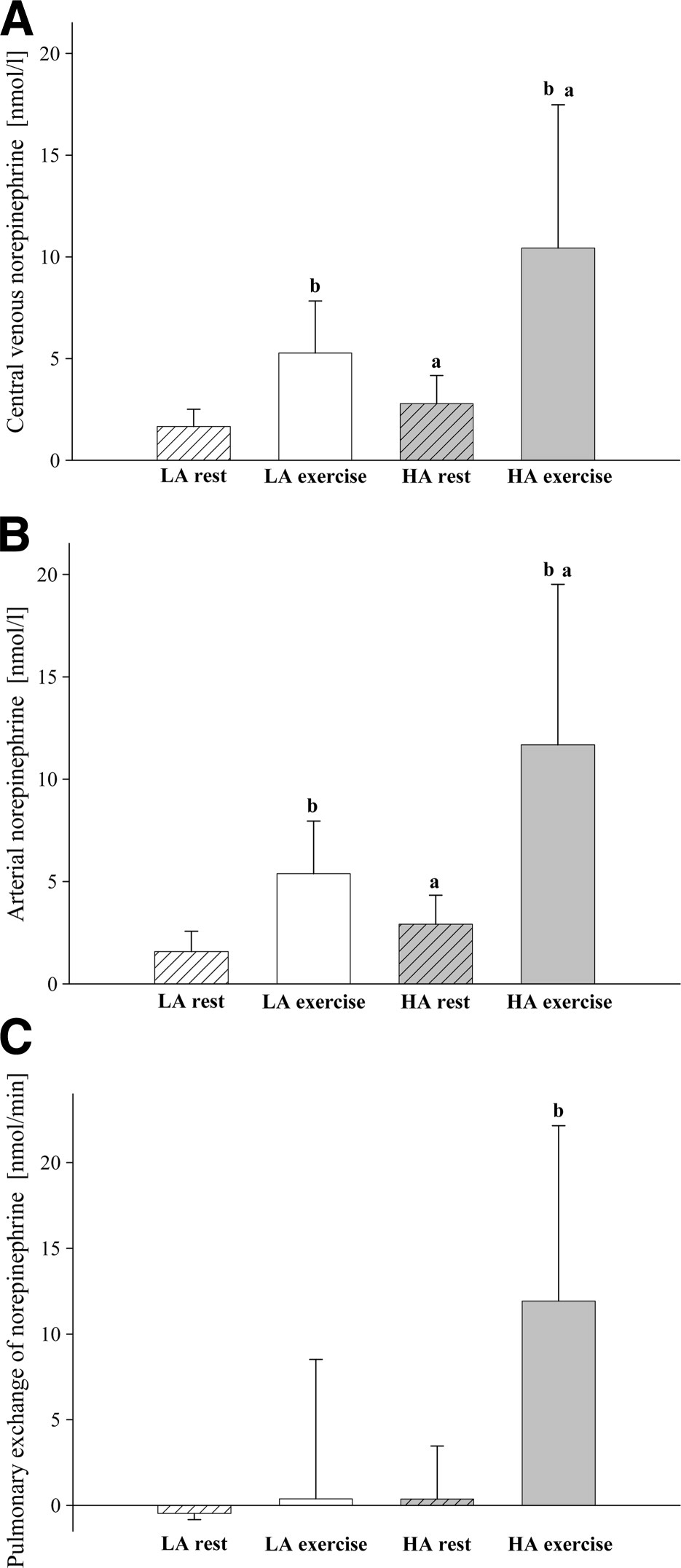
Plasma norepinephrine levels at low (LA) and high altitude (HA) during rest and exercise: (a) central venous blood, (b) arterial blood, (c) pulmonary exchange of plasma norepinephrine. Data are expressed as mean (±SD). a p < 0.05 versus low altitude; b p < 0.05 versus rest at the same altitude.
Correlation coefficients between both the arterial central venous difference (a − cvdiff) and the plasma exchange of norepinephrine and epinephrine with pulmonary artery systolic pressure (PASP) and pulmonary vascular resistance (PVR), respectively, at low and high altitude
The central venous and arterial norepinephrine plasma concentrations were positively associated with systolic blood pressure (R = 0.71 and R = 0.69; each p < 0.001), heart rate (R = 0.78 and R = 0.80; each p < 0.001) and pulmonary blood flow (R = 0.69 and R = 0.66; each p < 0.001).
Plasma Epinephrine
Exercise led to an increase in central venous epinephrine plasma concentrations, that was about the same magnitude at low and high altitude (Figure 3a). In contrast, high altitude did not affect central venous epinephrine plasma concentrations (Figure 3a). As seen for plasma norepinephrine, the changes in central venous epinephrine were paralleled by comparable changes in arterial concentrations, inasmuch as exercise but not high altitude increased the arterial epinephrine content (Figure 3b). The transpulmonary exchange of plasma epinephrine showed no significant transpulmonary gradient during rest and exercise at low and high altitude (Figure 3c). As for norepinephrine there was no significant correlation between both PASP and PVR with the a − cvdiff and the transpulmonary exchange of plasma epinephrine, neither during exercise nor during high altitude or during a combination of both (Table 2). There was also no correlation between the exercise and altitude-induced changes of these parameters (ie, changes in the a − cvdiff and changes in the transpulmonary exchange of plasma epinephrine) and the changes in PASP and PVR, respectively (not shown).
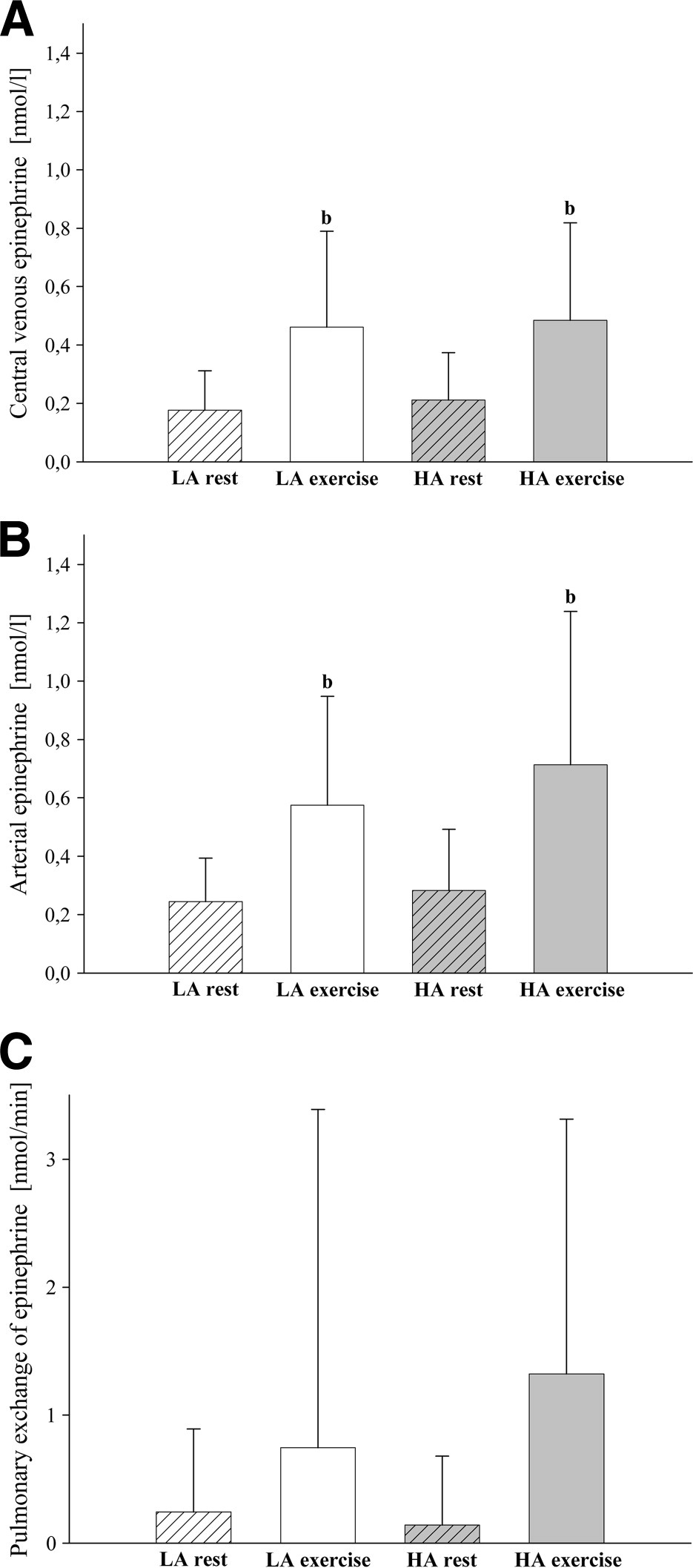
Plasma epinephrine levels at low (LA) and high altitude (HA) during rest and exercise: (a) central venous blood, (b) arterial blood, (c) pulmonary exchange of plasma epinephrine. Data are expressed as mean (±SD). b p < 0.05 versus rest versus rest at the same altitude.
Central venous and arterial epinephrine positively correlated with systolic blood pressure (R = 0.40; p < 0.001 and R = 0.43; p < 0.001, respectively), heart rate (R = 0.51 and R = 0.52; each p < 0.001), and pulmonary blood flow (R = 0.48, p < 0.001 and R = 0.42, p < 0.01).
Discussion
This study documents that the degree of pulmonary hypertension that occurs upon high altitude exposure is largely independent of the SNS activity in the pulmonary vasculature and adrenal gland.
Transpulmonary Plasma Norepinephrine
The vascular endothelium of the lung participates in norepinephrine metabolism by norepinephrine release and uptake. 21 By measuring both the pulmonary plasma flow and the differences in plasma norepinephrine concentrations in central venous blood entering the lung and in systemic arterial blood leaving the lung, we calculated the transpulmonary net exchange of plasma norepinephrine. Our data document no significant transpulmonary gradient at low altitude and during rest at high altitude. Only during exercise at high altitude was the transpulmonary gradient positive, indicating that the balance between pulmonary norepinephrine clearance and norepinephrine production had shifted in favor of net production. Whether the positive transpulmonary gradient resulted from an increased sympathetic norepinephrine release into the pulmonary circulation, from a decreased clearance, or from a combination of both, remains unknown; radiotracer dilution methodology would be needed to sort this out, allowing separate measurement of pulmonary norepinephrine spillover and clearance from plasma. 22
The transpulmonary production of plasma norepinephrine during high altitude exercise was paralleled by an increase in PASP raising the possibility that the increased availability of pulmonary plasma norepinephrine caused the pulmonary hypertension. Therefore, we calculated the correlation coefficient between the transpulmonary norepinephrine gradient and both PASP and PVR. This analysis revealed, however, that during none of the tested conditions was there a correlation between the transpulmonary spillover of plasma norepinephrine and the pulmonary vascular responses, suggesting that SNS activity in the pulmonary vasculature did not determine the degree of pulmonary hypertension during exercise and high altitude exposure.
This finding is consistent with previous data indicating that hypoxic pulmonary vasoconstriction to a great degree occurs independent of the SNS. In dogs, hypoxic pulmonary vasoconstriction, is unaffected by chemical sympathectomy, and surgical denervation of the carotid and aortic chemoreceptors increased hypoxic pulmonary vasoconstriction. 23 In cats, a species that has a well-developed hypoxic pulmonary vasoconstriction, as shown by von Euler and Liljestrand, 24 the pulmonary sympathetic discharge remained unchanged until a hypoxic arterial PO2 of about 45 mmHg was achieved, and only increased below this level. 25 Of note, the PO2 in our subjects at high altitude decreased just below this threshold, which might account for the unchanged transpulmonary gradient of norepinephrine we observed. Moreover, in human heart-lung transplant recipients, the intensity and length of hypoxic pulmonary vasoconstriction were preserved despite the loss of sympathetic input to the denervated transplanted lung. 26 These results, in conjunction with our findings, suggest that the sympathetic innervation of the pulmonary vascular bed does not account for the degree of hypoxic pulmonary vasoconstriction and high altitude pulmonary hypertension. However, to what extent vasodepressor responses elicited by a hypoxia-induced activation of the parasympathetic nervous system 27 might affect the complex regulation of pulmonary vascular tone during hypoxia remains unclear.
Systemic Plasma Norepinephrine
The elevated central venous norepinephrine concentrations we observed upon exercise and high altitude exposure can be interpreted as increased sympathetic nerve activity at the systemic vasculature, which is consistent with previous studies. 2 ,4,28,29 A heightened sympathetic response resulting in neural norepinephrine release is known to elicit vasoconstriction via alpha-adrenergic receptors, thus producing an elevation in vascular resistance. 30 Therefore, it was possible that the cardiovascular adjustments in our subjects were caused by enhanced SNS activity. Indeed, the positive correlation between central venous plasma norepinephrine and systolic blood pressure, heart rate, and pulmonary blood flow indicates that the elevated SNS activity in the systemic circulation resulted in these cardiovascular adaptions.
Transpulmonary and Systemic Plasma Epinephrine
Previous reports about the epinephrine response to acute high altitude exposure are conflicting. Some investigators reported no change in systemic plasma epinephrine after 2–4 days at high alitude,31,32 whereas others found increased levels. 3 ,4,33 In the present investigation, 20 hours at 4559 m did not affect central venous and arterial epinephrine concentrations and also did not affect the transpulmonary gradient of plasma epinephrine. When viewed in conjunction with the fact that central venous plasma norepinephrine increased, this finding suggests that sympathetic nerve, rather than adrenal medullary activity, increases in subjects acutely exposed to high altitude. The lack of a transpulmonary epinephrine gradient further indicates that the pulmonary vasculature does not contribute to epinephrine clearance by either uptake or adrenergic receptor binding. This finding further argues against a pivotal role of the SNS in regulating PVR and PASP upon high altitude exposure.
In contrast to high altitude, exercise at both low and high altitude increased the systemic plasma concentration of epinephrine, which is consistent with previous results.3,34 This increase can be interpreted as an increased SNS activity in the adrenal glands.
The absolute workload at high altitude was reduced such that the subjects were exercising at about the same relative workload as reflected by comparable cardiac outputs at low and high altitude, and by only slightly higher heart rates at high altitude. This might account for the comparable increase in central venous epinephrine at both altitudes.
One limitation of our study is the small numer of subjects which developed HAPE. Our findings do not exclude the possibility that subjects with HAPE have a higher SNS activity that might account for the exaggerated increase in PASP that typically preceeds HAPE. Because only 4 mountaineers developed HAPE, we could not perform subgroup analyses for these subjects. However, excluding the HAPE subjects from the data analysis did not significantly alter the results. Another limitation of our study is that we did not perform pulmonary artery catheterization for sampling mixed venous blood for the determination of the transpulmonary norepinephrine and epinephrine gradient. Instead, central venous blood was drawn, realizing that there may be differences in regional venous catecholamine concentrations. However, a previous study demonstrated no significant difference between the norepinephrine concentration in the superior vena cava and the pulmonary artery at rest. 18 In contrast, it remains unclear to what extent the exercise test may have caused regional differences between the catecholamine concentrations in superior caval and mixed venous blood. Given that the major source of plasma catecholamines is the exercising muscle, it is possible that, during exercise on a bicycle ergometer, the catecholamine concentration in the superior vena cava underestimates the catecholamine influx into the lung and thus overestimates the transpulmonary spillover of these catecholamines. However, we would like to point out that our subjects exercised for the last 10 minutes at a VO2max of 50% at low altitude and 35% at high altitude, respectively, which is below the individual anaerobic threshold. 35 Exercise at the individual anaerobic threshold on a cycle ergometer has previously been shown to result in constant systemic norepinephrine concentrations within the first 10 minutes of exercise. 36 This finding is in line with the observation that during prolonged dynamic knee-extensor exercise limb blood flow demonstrates stable levels after 6 to 10 minutes. 37 It is, therefore, conceivable that during the steady state exercise test that our subjects performed, the plasma levels of norepinephrine and epinephrine had also reached a stable concentration in the systemic circulation at the time of blood drawing, and that thus the catecholamine concentrations in the superior vena cava and the pulmonary artery were similar. However, to the best of our knowledge data comparing the catecholamine concentrations in the superior and inferior vena cava or the pulmonary artery during leg exercise are not available.
In summary, we found that the rise in PASP that occurs with acute high altitude exposure and exercise is largely independent of the SNS activity in the pulmonary vasculature and adrenal gland. The SNS activity at high altitude primarily elicits cardiac and systemic vascular adaptions without significantly affecting PVR and PASP. This finding might in part be attributable to beta-adrenergic receptors present in the pulmonary vascular bed, because stimulation of pulmonary vascular beta-2-receptors by norepinephrine causes vasodilation,38,39 thus counteracting the norepinephrine-induced vasoconstriction via alpha-adrenergic receptors. This effect seems to be even more pronounced when the vasconstrictor tone in the pulmonary vascular bed is elevated, eg, by hypoxia. 40
Footnotes
Acknowledgments
We gratefully acknowledge the mountaineers who took part in the study; the hut keepers and the Sezione Varallo of the Club Alpino Italiano for providing an excellent research facility at the Capanna Regina Margherita; Martina Haselmayr, Sonja Engelhardt, and Christiane Herth for expert technical assistance; Semanta Greppi, and Chantal Imesch for taking the chest radiographs at high altitude.
The study was performed at the University Hospital, Heidelberg, Germany and at the Capanna Regina Margherita, Italy (4559 m).
