Abstract
Objective
To evaluate the performance of the emOx emergency powdered oxygen portable nonpressurized delivery system. This device produces oxygen through chemical reaction and is marketed for emergency first aid use until professional medical assistance is available.
Methods
Seven unmanned trials were conducted under standard laboratory conditions. Measures included oxygen flow, reaction canister external wall temperature, delivered gas temperature, and delivered gas relative humidity.
Results
The mean oxygen flow was 1.75 ± 1.58 L · min−1 (mean ± SD) with a total yield of 40.4 ± 2.6 L. Oxygen flow increased slowly and with substantial variability between reactant groups, exceeding 2.0 L · min−1 after 15.7 ± 6.4 minutes of operation. Oxygen flow briefly peaked at 5.93 ± 0.56 L · min−1 at 17.8 ± 7.9 minutes before rapidly falling to zero. The mean oxygen fraction was 0.81 ± 0.28, exceeding 0.96 in 10.7 ± 2.9 minutes. The reaction canister external wall temperature reached 54.7 ± 7.4°C. Delivered gas temperature varied little from ambient. Delivered gas relative humidity surpassed 75% in 8 ± 3 minutes and 90% in 15 ± 5 minutes of operation.
Conclusions
A readily available, high concentration oxygen supply could have utility to manage many conditions in advance of the arrival of professional emergency medical services (EMS). Unfortunately, the highly variable activation time and low average oxygen flow rate make the rapid deployment value of the emOx equivocal. The limited total oxygen yield makes it inappropriate for conditions demanding significant oxygen resources. Advancement in oxygen concentrator systems likely holds far more promise than powdered chemical oxygen generation for first aid and emergency medical applications.
Introduction
Concentrated oxygen has great utility in remote or wilderness medicine applications. In addition to routine clinical care, it is used both prophylactically to avoid and as a first aid treatment of altitude-related disorders. Oxygen is also a primary first aid tool to manage decompression sickness following compressed gas diving. 1 Enhanced therapeutic efficacy can be achieved when oxygen is delivered at the highest concentration possible. With decompression sickness, for example, the inspired oxygen concentration is proportional to the gradient for inert gas elimination from the body.
Oxygen is typically stored in pressurized cylinders. Overland transportation of cylinders can be inconvenient and expensive for isolated locations. Air transportation of pressurized cylinders is also problematic, increasing the logistical considerations for many field environments. The potential utility of an easily transported oxygen source is attractive, particularly for remote applications.
Chemical oxygen-generating systems have been proposed as an alternative to pressurized gas supplies. Oxygen is produced through exothermic chemical reaction. The primary challenges of such devices are low oxygen flow rates and limited total oxygen yield. 2 Since supply volume limitations are a chief concern for most remote operations, the utility of such devices is practically restricted to settings in which relatively small volumes may be sufficient.
The emergency oxygen device (emOx) is a portable nonpressurized oxygen delivery system produced by Green Dot Systems, Inc (Miami, FL). The unit is advertised as being useful for first aid use until professional medical assistance is available. Promotion focuses on the absence of a pressurized storage container, the high purity of delivered oxygen, maximal flow duration (of multiple packs of reactants), and long shelf life of reactants. Our goal was to evaluate the performance of the emOx system under standard laboratory conditions. We addressed oxygen delivery; we did not evaluate shelf life.
System Description
The emOx system functions as an integrated canister composed of 3 elements (Figure 1). The largest is the 29 cm tall, 12.7 cm diameter polypropylene reaction chamber. A humidity chamber sits in an opening in the top of the reaction chamber. A 9 cm tall transparent cap covers the humidity chamber. An approximately 2.0 m long, 5 mm internal diameter polyvinyl chloride flexible supply line connects the top of the cap to a simple patient mask. There are 3 separate reactant containers. The first is filled with tap water before use. Sealed “White Powder” bottles contain sodium percarbonate. Sealed “Black Powder” bottles contain a proprietary manganese catalyst compound. The complete system comes in a carrying case that includes 1 empty bottle for water and 3 separately sealed bottles of each of the white and black powders. Refill packs include 4 separately sealed bottles of each of the white and black powders. The tubing and patient mask are designated as single use; the structural components are reusable. All components are robust with no obvious issues affecting field transportability.

emOx non-pressurized oxygen delivery system.
Basic Operation
Symbolized instructions are printed on the reaction chamber and on a separate instruction leaflet.
To activate, the cap and humidity chamber components are disassembled. The water bottle is filled to the bottom of the shoulder with tap water (∼500 mL). Water is poured in the humidity chamber to a line mark (∼50 mL). The remaining volume in the bottle is poured into the reaction chamber. The contents of Bottle 1 (sodium percarbonate) and Bottle 2 (manganese catalyst) are then emptied into the reaction chamber. The humidity chamber assembly is placed into the reaction chamber and the cap hand-tightened in place. The supply line is attached to the port on top of the system and the face mask. An instructional note printed on the reaction chamber canister (but not the instruction leaflet) indicates that in cold conditions the contents may be swirled briefly to accelerate the initial reaction. The reaction continues and gas flows as long as bubbles are visible through the transparent cap of the humidity chamber. When finished, the device can be disassembled, the reportedly nontoxic exhausted reactants poured out, and the components rinsed with freshwater before assembling another round of reactants.
Methods
Seven unmanned trials were conducted under standard laboratory conditions (121 m altitude). The device was operated in compliance with the manufacturer instructions provided with the device. The simple face mask was replaced with a monitoring assembly.
Reactant weights and liquid volumes were recorded prior to the start of a trial (Fisher Scientific model IT-1800 top loading balance, Pittsburgh, PA, and a 250 mL graduated cylinder). Tap water was equilibrated to room temperature for at least 30 minutes preceding each trial. (The temperature of the water was measured after 30 minutes of equilibration preceding the first trial and found to be equal to the room temperature; the temperatures of subsequent samples, some standing for as long as 70 minutes, were not measured.) Dry reactant containers were shaken prior to opening to minimize the possibility of clumping. Once the system was assembled, a single gentle swirling motion was applied and the device put on a stable, motionless flat surface for the duration of the trial.
Trial data were recorded on a Macintosh PowerMac 7600 computer (Apple, Inc, Cupertino, CA) using Maclab/8 data acquisition hardware and Chart software (version 3.3.4, ADInstruments, Castle Hill, New South Wales, Australia), set to capture 4 samples per second. Room temperature was measured within 2 m of the apparatus. Barometric pressure and room temperature were measured with a Perception II weather station (Davis Instruments, Haywood, CA).
Gas flow and volume were measured with a Hans Rudolf pneumotachometer (model 3813; Kansas City, MO) with Validyne carrier demodulator (model CD15; Northridge, CA) and Validyne pressure transducer (model DP103-10). Oxygen concentration was measured continuously with a BCI International Capnocheck Plus galvanic fuel cell sensor (Waukesha, WI). Gas flow was measured continuously and averaged over sequential 60-second periods until flow decreased to zero. Total volume was computed as the sum of the minute average flow readings.
Temperatures were measured using a Cole-Parmer Instrument Co temperature box (Chicago, IL) and Yellow Springs Instruments 400 series probes (Yellow Springs, OH). Canister temperature was measured continuously with a thermistor secured to the outside wall of the reaction chamber at approximately mid-reactant level. Delivered gas temperature and humidity were measured in the gas stream at the end of the flexible line (the approximate position of a patient mask) with a Dickson TH300 electronic temperature and humidity gauge (Addison, IL) in 4 of the 7 trials.
Values are reported as mean ± SD with ranges in parentheses.
Results
The laboratory conditions during testing included stable room temperature (23.5 ± 0.6°C) and relatively stable barometric pressure (750 ± 9 mm Hg).
The weight of system components (excluding reactants) was 1.75 kg. The additional weight for each set of reactants (including water and powder containers) was 0.9 kg. The total weight was therefore 2.65 kg complete with 1 set of reactants, 3.55 kg complete with 2 sets of reactants, etc.
Dry chemical reactant weights were similar for all trials: sodium percarbonate 342.9 ± 6.8 (range 335.1–357.0) g and manganese catalyst 8.1 ± 0.6 (7.6–9.0) g.
The mean flow rate (measured to the last non-zero minute average) was 1.75 ± 1.58 (0.05–6.75) L · min−1 ambient temperature and pressure saturated (ATPS) (Figure 2). Oxygen was generated for 23 ± 6 (18–35) minutes. The oxygen flow exceeded 2.0 L · min−1 ATPS after 15.7 ± 6.4 (11–29) minutes of operation. The flow rate remained above 2.0 L · min−1 ATPS for 6.4 ± 1.0 (5–8) minutes. The oxygen flow rate peaked at 5.93 ± 0.56 (5.23–6.75) L · min−1 ATPS in 17.8 ± 7.9 (6–32) minutes. The flow remained above 2.0 L · min−1 for only 1 minute after reaching the peak rate. The total oxygen yield was 40.4 ± 2.6 (37.7–44.4) L.
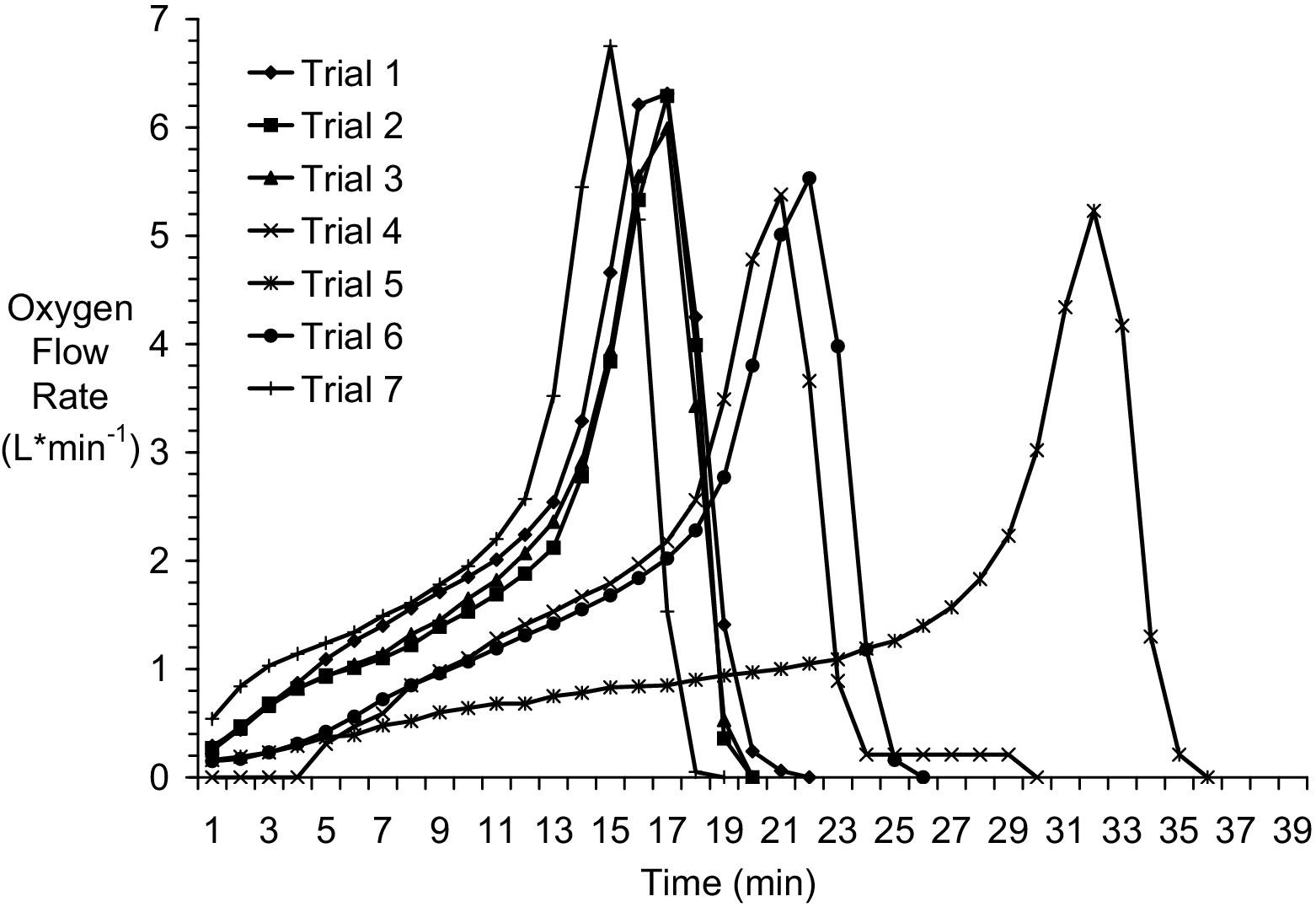
Oxygen flow produced by emOx nonpressurized oxygen delivery system in unmanned trials.
The mean oxygen fraction was 0.81 ± 0.28 (0.21–0.99). The oxygen fraction exceeded 0.96 in 10.7 ± 2.9 (8–16) minutes. The fraction was 0.98 ± 0.01 (0.96–0.99) for the remainder of the oxygen generation period (Figure 3).
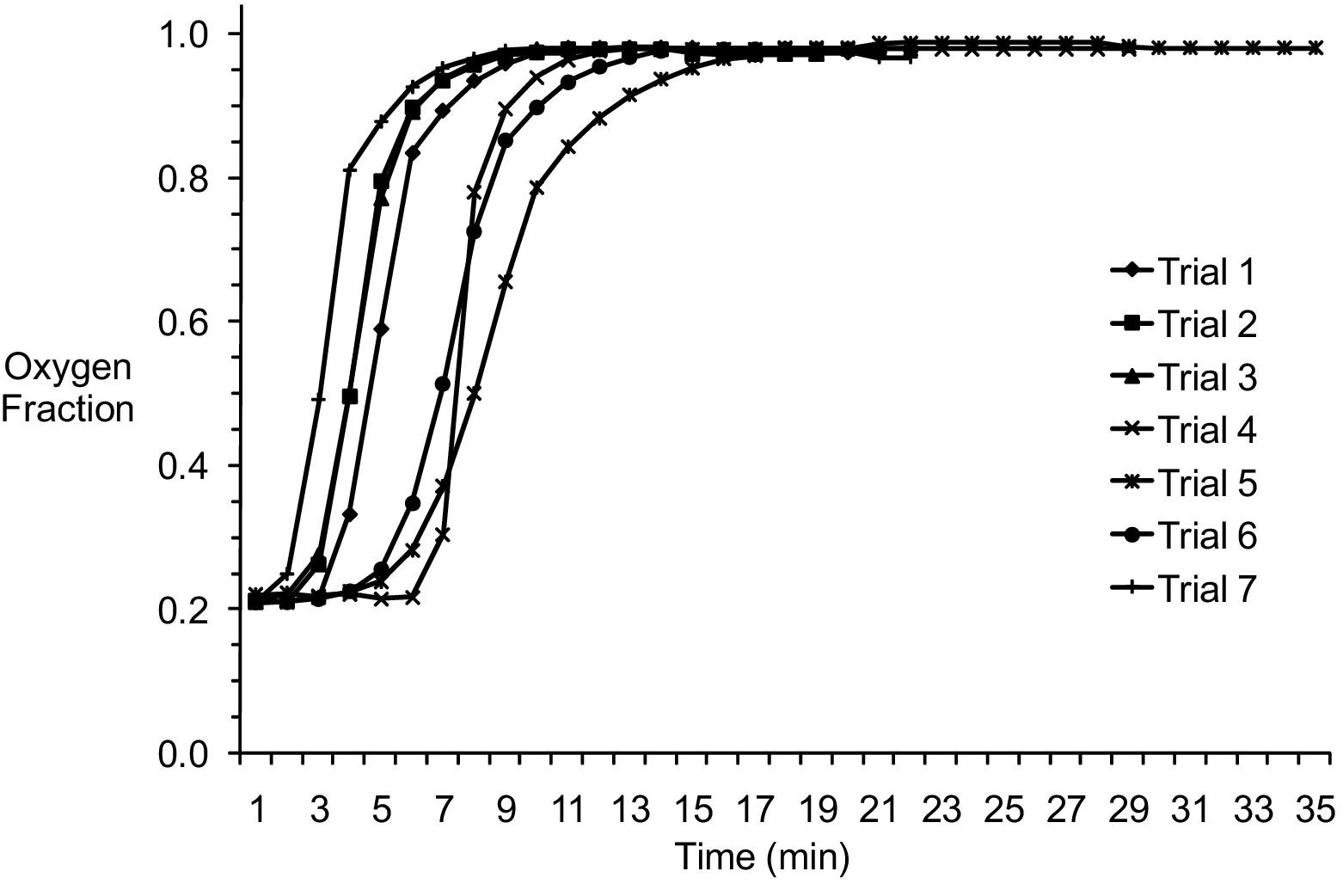
Oxygen fraction produced by emOx nonpressurized oxygen delivery system in unmanned trials.
Reaction canister outside wall temperatures increased throughout the trials, generally peaking within 1 minute of the end of gas flow. Temperatures reached 54.7 ± 7.4 (46.4–64.9)°C. Gas temperature measured at the approximate position of a delivery mask varied little from ambient temperature at any point in the reaction cycle. Ambient temperature at trial start was 23.5 ± 0.6 (22.8–24.3)°C. Temperature at the approximate position of a delivery mask at the end of the first minute of gas flow was 23.0 ± 0.2 (22.7–23.2)°C. Mask temperature peaked at 23.6 ± 0.1 (23.5–23.7)°C [25 ± 7 (17–34) minutes trial time] and fell slightly to 23.4 ± 0.1 (23.2–23.5)°C in the final minute of gas flow [31 ± 7 (23–40) minutes trial time] (Figure 4).
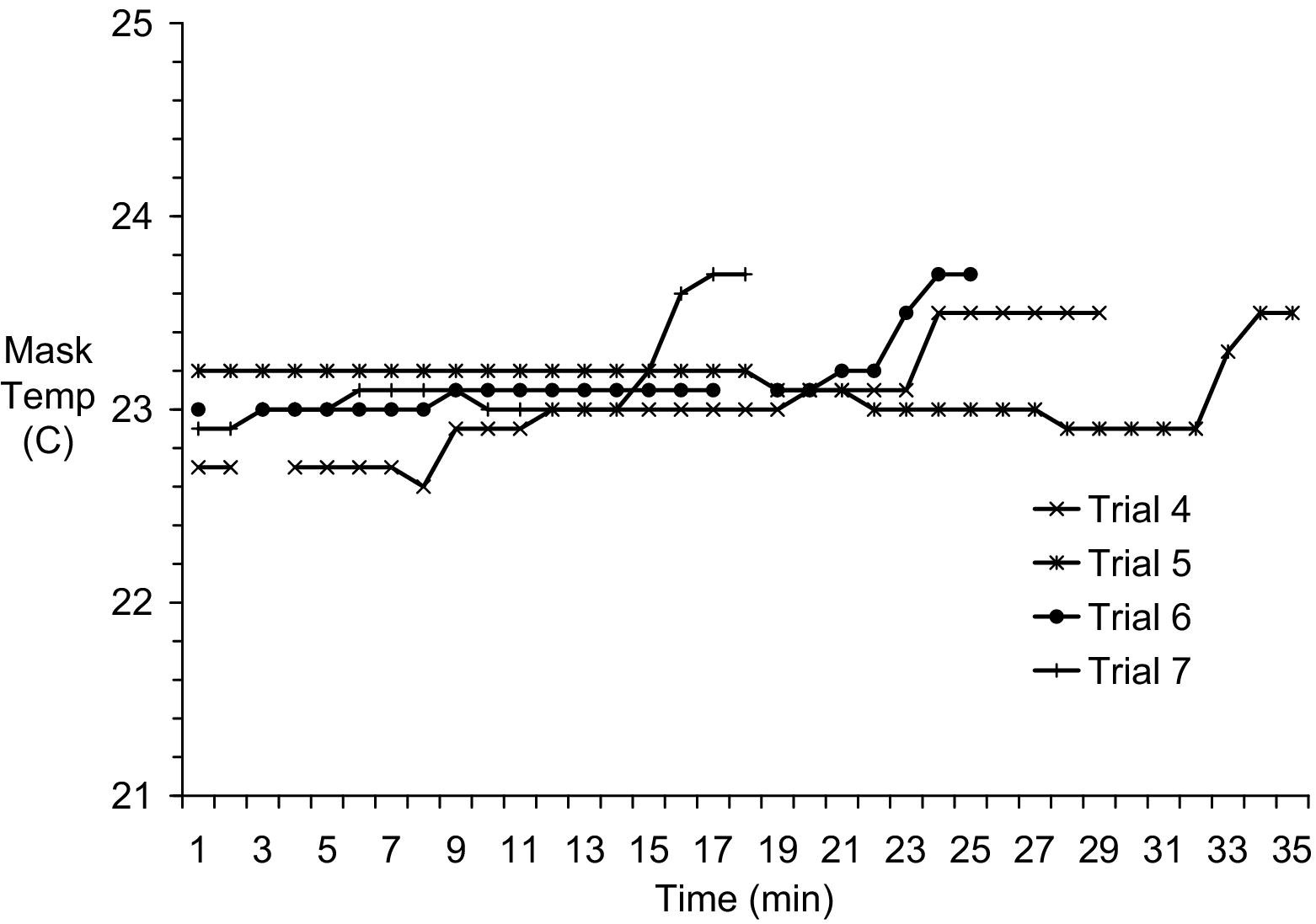
In-stream gas temperature at the approximate position of the delivery mask during unmanned trials of the emOx nonpressurized oxygen delivery system.
Relative humidity at the approximate position of a delivery mask increased over the course of the trial, surpassing 75% in 8 ± 3 (3–11) minutes and surpassing 90% in 15 ± 5 (8–19) minutes and for the rest of the trial (Figure 5).
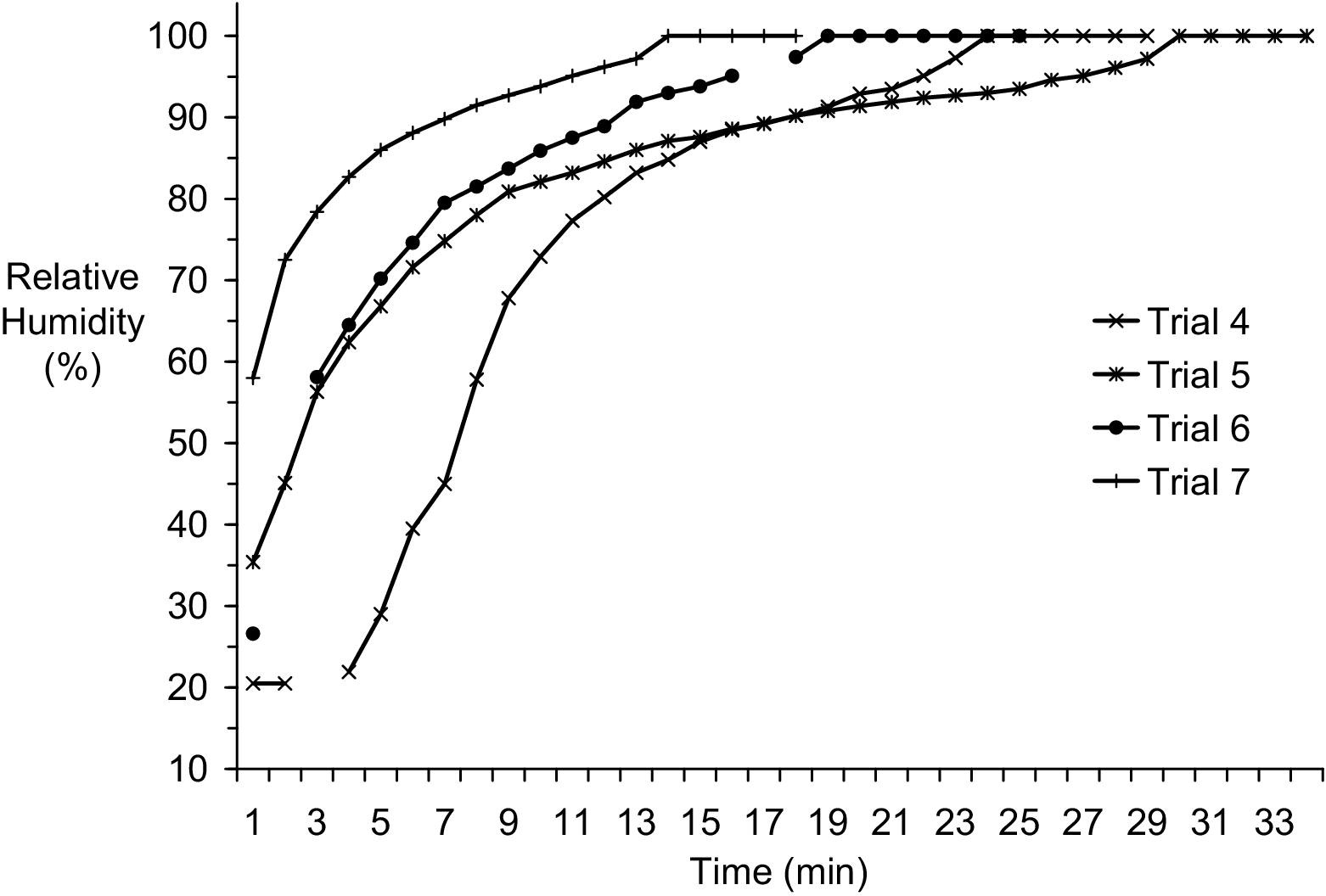
In-stream relative humidity at the approximate position of the delivery mask during unmanned trials of the emOx nonpressurized oxygen delivery system.
Discussion
Oxygen delivery systems appropriate for first aid use must be reliable, easy to use, easy to transport, and able to provide sufficient volume and flow rates for the conditions being treated. Rapidly deployable but limited oxygen supplies could be appropriate for some urban or suburban settings with readily available emergency medical services (EMS) support. Remote settings or situations in which rapid EMS response could not be relied upon demand greater emergency oxygen resources.
Pressurized sources of oxygen are standard for conditions requiring substantial supply volumes. The major practical challenge in supporting mobile activities is in transporting pressurized cylinders, particularly by air. Commercial air travel restrictions can make it impossible to transport pressurized vessels on passenger aircraft. The need for oxygen to meet emergency readiness plans during travel makes it compelling to consider alternatives to pressurized sources of gas.
Compact oxygen concentrators have gained popularity for remote facility and portable use. 3 -7 Key challenges to true field utility are oxygen yield, system weight, and power requirements.
We have previously recommended that an alternative to chemical oxygen-generating systems in replacing pressurized oxygen for remote operations would be to combine low-flow oxygen concentrators with high-efficiency closed-circuit rebreathers. 2 We independently described the performance of a closed-circuit device capable of delivering very high inspired gas fractions with just over 1.0 L · min−1 mean oxygen flow. 8
Preliminary test use of portable oxygen concentrators by healthy subjects in a field study at 3750 m altitude has now been reported. 9 While the light weight of a battery-powered concentrator was compelling (3.8 kg), the increase in arterial oxygen saturation indicated by pulse oximetry (SpO2) from unassisted breathing (79 ± 6%) was substantially less for the battery-powered oxygen concentrator and simple, non-rebreather facemask alone (82 ± 6%) than with the battery-powered concentrator plus semi–closed-circuit system (90 ± 3%). Battery power remains a problem for the portable concentrator for both charge duration (reportedly only 30 minutes) and delivery capability. The enhanced performance of an externally powered mobile oxygen concentrator (33 kg weight) resulted in SpO2 of 95 ± 1% with the standard mask alone.
More effort is required to improve the capability of portable oxygen concentrators. Ultimately, the flexibility for remote applications may be best served by the integration of solar-charged, battery-powered, high-efficiency oxygen concentrators with compact semi-closed- or closed-circuit breathing loops.
The concept of chemical oxygen generation is intuitively attractive since it avoids issues of pressurized vessels and power supply. High-purity oxygen can be generated by stable and safe reactants. The problems, however, are limited oxygen flow rate and total yield.
The emOx portable non-pressurized oxygen delivery system is compact, robust, and easy to use as long as a modest source of water is available. Unfortunately, the total oxygen yield for a set of reactants dramatically limits the utility of the device. Both flow rate and total volume are extremely limited. It is noteworthy that, in our trials, the time between device activation and achieving an oxygen flow exceeding 2.0 L · min−1 was both long and highly variable and that the oxygen flow remained over 2.0 L · min−1 for only a short time. While multiple sets of reactants are included, potential treatment time would be lost to changing over and reactivating the system. Practically, the extremely limited supply would likely be ineffective in treating most conditions. The substantial variability in the early rate of oxygen production, despite careful standardization of activation steps, also brings into question any benefit of rapid deployment in advance of EMS arrival. Ultimately, the time spent dealing with the device and not spent paying attention to other needs of the patient do not seem justified for the limited benefits delivered.
The written information provided by the manufacturer of the emOx was circumspect, focusing on the high purity of the oxygen generated and the total yield of multiple reactant packs. No reference to flow rate was found on the corporate website (
We have previously evaluated a similar chemical oxygen generating system, the SysO2. 2 The SysO2 device was later subject to a warning letter issued by the US Food and Drug Administration (FDA) for several deficiencies, including unrealistic performance claims. 11 The FDA subsequently issued a medical device recall of the product. 12 The reason for recall was stated as “System O2 oxygen flow rate found to be inadequate and RejuvO2 Home Oxygen Bar contains ‘Adulterated Labeling’.”
In our trials, the emOx and SysO2 systems achieved similar maximum flow rates, but the emOx device delivered an even lower mean flow rate (1.75 ± 1.58 vs 2.98 ± 1.52 L · min−1) and substantially lower total oxygen yield (40.4 ± 2.6 vs 62.9 ± 6.6 L). The volume limitation is undoubtedly due to the substantially smaller reactant mass provided per reactant dose with the emOx. Practically, the low oxygen yield per reactant set for the emOx fares poorly in comparison with the greater than 400 L yield for a single ‘D’ size oxygen cylinder. The reason for the marked inconsistency in the timeline of oxygen generation with the emOx device is unclear, but this character increases the uncertainty of even short term benefit to a recipient. Taking a minimum of 11 minutes and a mean of almost 16 minutes to achieve a 2.0 L · min−1 flow obviates much of the benefit of fast response. Overall, the performance of the emOx system tested does not warrant a positive recommendation for first aid or emergency medical applications.
Conclusions
Increasing the number of alternatives to pressurized oxygen sources for the effective delivery of first aid oxygen is desirable. Unfortunately, our testing of the emOx system indicates an extremely limited mean oxygen flow rate, an extremely limited total oxygen yield, and a problematically inconsistent timeline of oxygen generation. Based on these results, we conclude that the emOx device does not provide an adequate source of emergency oxygen. The limited supply offers almost trivial value for remote applications. The inconsistency in the timeline of oxygen generation makes the benefit questionable even for situations in which emergency medical support is likely to be rapidly available. The practical benefits of powdered chemical oxygen-generating systems for first aid or emergency medical use remain marginal at best. We expect that future efforts to replace compressed gas sources will be more productively directed at improving oxygen concentrator technology, including reductions in the energy requirements, creating opportunities for solar charging, and integration of semi–closed- or closed-circuit breathing loops.
Footnotes
Acknowledgment
The emOx equipment and materials tested were provided by Green Dot Systems, Inc., Miami, FL.
