Abstract
Routine testing of active pharmaceutical ingredients (APIs) for metal residues is an expectation of regulatory bodies such as the FDA (U.S. Food and Drug Administration). Sample preparation techniques are the rate-limiting step in the testing process and can be variable depending on the specific characteristics of the API under test. Simplification and standardization of the routine preparation of inductively coupled plasma spectroscopy sample solutions of organic compounds has been developed using a commercially available robotic workstation. Contamination from the metal components of the instrument and from sample tubes used in the methodology has been studied using a Design of Experiments approach. The optimized method described can be used for the measurement of trace metals in Pharmaceuticals at levels compliant with European and U.S. regulatory requirements.
Introduction
Inductively coupled plasma–atomic emission spectroscopy (ICP-AES) is routinely used in the pharmaceutical industry to measure trace metal content of active pharmaceutical ingredients (APIs) or intermediates against specification limits derived from external guidance. 1,2 Typically, the metals of interest are derived from catalysts used in the synthetic process used to manufacture the API and are tested for after the isolation and purification of the API but before final formulation of the drug product. Because there is no therapeutic benefit from these metals, toxicology data has been used to define a permitted daily exposure (PDE) for metallic residues. 1 PDE is defined as the maximum patient exposure (expressed as μg/kg/day) to an element, possibly on a chronic basis, that is unlikely to produce any adverse health effects. In our laboratories, we use a method based on dissolving samples in acidified dimethyl sulfoxide (DMSO) using sonication rather than the more usual aqueous acid digestion techniques. 3 The method complies with the requirements of United States Pharmacopoeia (USP) chapter 730 and is generally faster than digestion but some samples require extended sonication, so the time and effort required in preparing samples for an instrument run is unpredictable. We have therefore been looking for a method of sample preparation that is more predictable and controlled than the use of a sonication bath. Automation of ICP sample preparation is not a new concept 4,5 but no systems have previously been reported that meet our needs. We use Active Ingredient Processing Workstations (APW3) robots equipped with sonication probes for sample preparation in HPLC analyses 6 and therefore examined their application to ICP sample preparation. A successful automated generic method should require no further processing before analysis, be compliant with good manufacturing practice (GMP), and introduce no significant contamination with the trace metals requiring measurement.
Experimental
Reagents
Hydrochloric acid (HCl) high purity 35% w/v and AR grade DMSO were obtained from Fisher Scientific (Loughborough, UK). Diluent was prepared as 2:98 HCl: DMSO by volume.
Certified standard stock solutions of the test analytes were obtained from SPEX CertiPrep (Stanmore, UK), and working standard solutions were prepared from these stock solutions to contain 0.1 and 1 μg/mL of each element in diluent. Three-point calibrations were used in this work by including a blank.
Active Ingredient Processing Workstations
A Zymark APW3 (Sotax AG, Basel, Switzerland) with sonication probe and acid resistance upgrade was used for all automated sample preparation. Method parameters were system defaults unless specified in the text. Polystyrene 16 mm × 100 mm test tubes were obtained from VWR International (Lutterworth, UK).
Inductively Coupled Plasma–Atomic Emission Spectroscopy
A Vista Pro ICP-AES instrument equipped with a CETAC ASX-510 autosampler and AGM module for oxygen feed (Varian, Inc., Oxford, UK) was used for all ICP analyses. Operating conditions are listed in Table 1.
ICP operating conditions
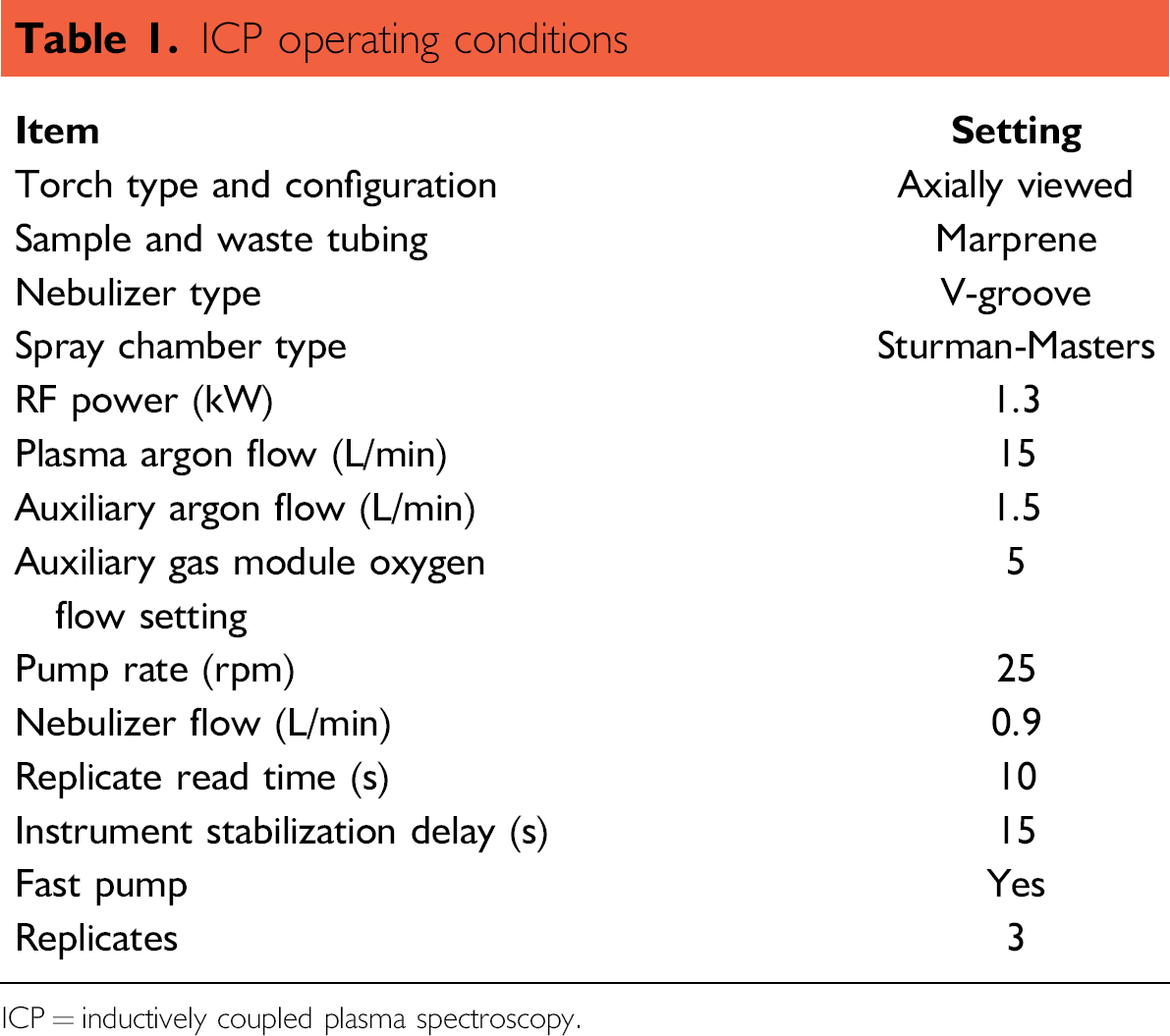
ICP = inductively coupled plasma spectroscopy.
Data Analysis
ICP data was acquired and results calculated using ICP Expert software for Vista Pro version 4.1 (Varian Inc., Oxford, UK). Design of Experiments (DOE) and data analysis were performed using Design Expert 7.1 for windows supplied by Stat-Ease Inc. (Minneapolis, MN).
Manual Sample Preparation
Samples were prepared by weighing 100 mg of the API into a polystyrene 16 mm × 100 mm test tube and adding 10mL of acidified DMSO. The test tubes were placed in a sonication bath and sonicated for 30 min. Any test tubes containing undissolved material were sonicated for a further 30 min and reexamined.
Results and Discussion
The automated method requires that the acidic diluent contacts a large number of components such as syringes, tubing, and sonication probe, which might be the source of leachable contaminants that are not involved in the manual process. There is also significant difference between the dissolution of a sample using a sonication bath, which may have variability in energy output dependent on where the sample tube is placed and dissolution using an insertion probe in contact with the diluent. This difference could lead to variation in temperature changes in the sample solutions and increased likelihood of contamination when using the robotic probe.
Therefore, it was necessary to not only demonstrate equivalence of result between the automated and manual methods for a range of samples, but also to establish the generic risk of introducing contaminants into any future test material.
Elements Analyzed
In API, it is necessary to test for catalyst residues from the chemical synthesis (e.g., palladium or platinum) and also the heavy metals covered by USP chapter 231, lead, mercury, bismuth, antimony, arsenic, tin, cadmium, silver, copper, and molybdenum. The allowable limit will typically be 0.1 μg mL−1 for all species. It is therefore necessary for any method for sample preparation to have minimal contamination from any of the species listed above. Our method uses a 10mg mL−1 solution of material under test and in the data discussed here we use the expression of ppm with respect to this concentration, hence our limit for palladium is 10ppm (equivalent to 0.1 μg mL−1).
Contamination from Tubes
To establish a baseline for the source of contamination, six test tubes had 9 mL of diluent manually added using a plastic dispensing pipette and were sonicated for 30 min in a conventional laboratory sonic bath. At the end of the sonication, the diluent temperature had risen from 20 to 34 °C. Data are shown in Table 2 for measurable elements in the test tubes. No significant contamination was observed.
Measured background from test tubes (ppm)

Contamination from APW3 Components
The automated method combines weighing of samples with addition of solvent and dissolution as shown in Table 3: Diluent is dispensed via a stainless steel cannula using a glass syringe and the sonicator probe is constructed from a titanium alloy containing aluminum and vanadium; otherwise all parts in contact with the diluent or sample are made from plastics.
Procedure for automated sample preparation

APW3 = Active Ingredient Processing Workstations; ICP = inductively coupled plasma spectroscopy.
To test for maximum levels of contamination from the sonication process, a tube was processed after the addition of 9 mL of diluent and sonication was applied using 10 pulses of 2-min duration (4-s pause between pulses) at 4.6 V (maximum power available).
These conditions demonstrated that contamination from titanium (19.8 ppm) and iron (5.6 ppm) were the only concerns for routine application of an APW3 method. After the optimization of the APW3 method, potential contamination was evaluated using six blank preparations (see Table 4). The elements detected in the blanks do not occur at levels, which affect the results of routine ICP analyses and the blanks are considerably cleaner than those from the manual procedure.
Measured background following APW3 sonication (ppm)
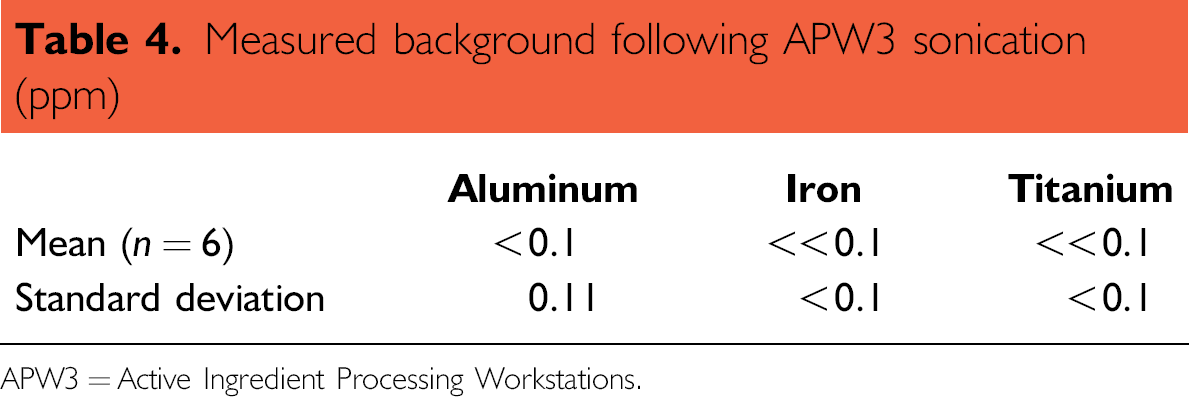
APW3 = Active Ingredient Processing Workstations.
Optimization of Sonication Method
The sonication process uses an insertion probe capable of operation between 1.5 and 4.6 V either stationary or cycling vertically within the tube, with varying numbers of pulses that have user-defined duration and also a user-defined pause between pulses. The chosen sonication parameters will affect the overall experiment time, the temperature of the sample solutions (potentially causing evaporation), whether the sample is dissolved, and also the contamination levels from the metal sonication probe. To identify the optimum conditions for dissolution of our samples, we applied a DOE approach.
DOE is a structured, organized method for determining the relationship between the factors (sonication parameters) affecting the sample dissolution and the output of that process (measurable metals). The DOE experiments can be represented as a box (Fig. 1) where the data obtained can predict any combination of parameters falling within that box allowing a small number of experiments to be used to optimize an analytical methodology. In the cube shown in Figure 1, the circles represent measurements with E1 (blue) consisting of all low parameter values, E2 (pink) all high parameter values, and C (center point, green) median values for all parameters. The experimental space represented by the cube provides eight corners and six faces; therefore, a design using 18 conditions including 4 center points was sufficient to comprehensively study the sonication process. A preliminary scouting study using the full parameter range available on the APW3 but only measuring at points E1, E2, and C defined the ranges used in the full study. Voltage, pulse time, and number of pulses are the variables studied and the value ranges chosen are shown in Table 5.
DOE parameters
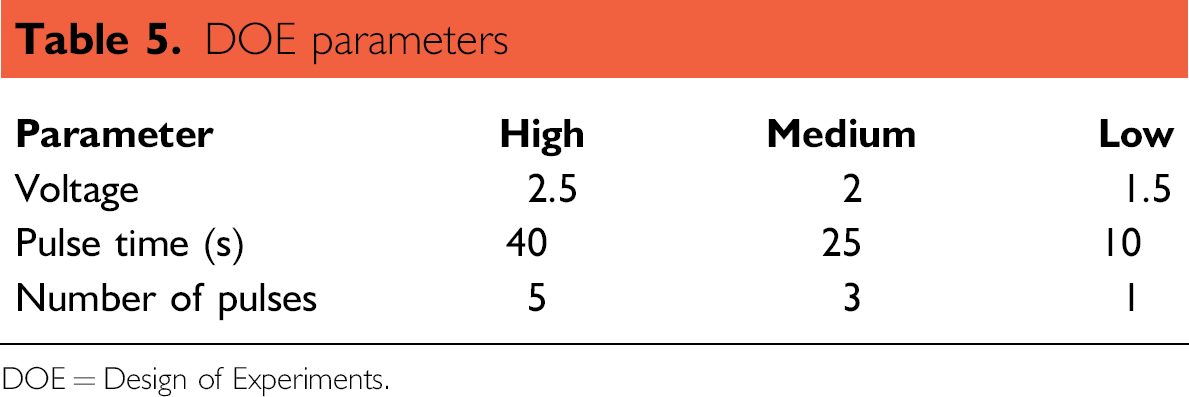
DOE = Design of Experiments.
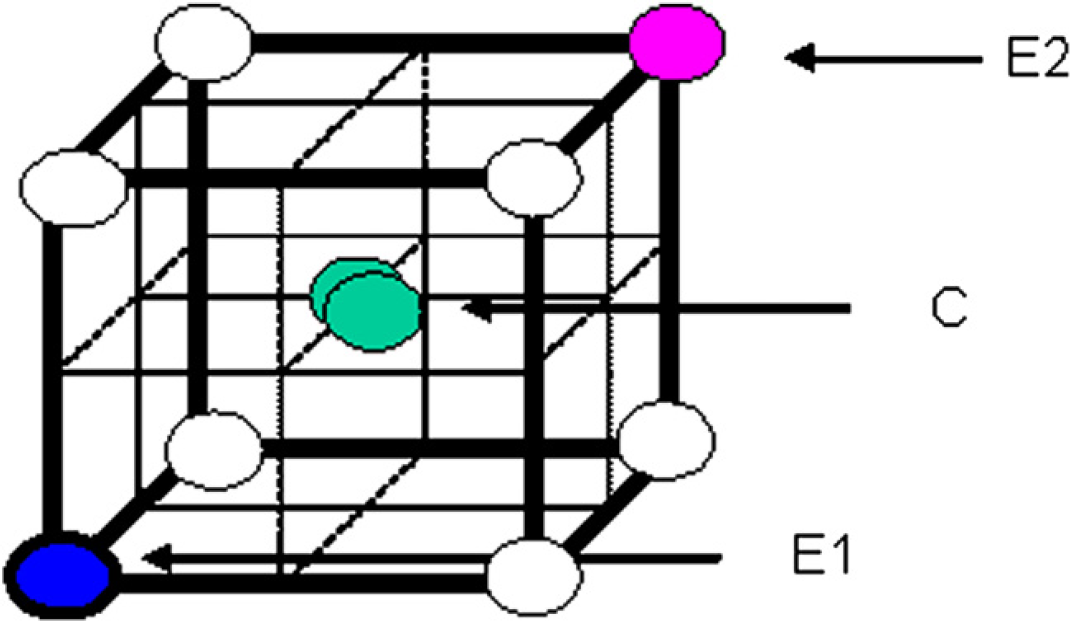
Representation of DOE.
Under the conditions studied, Ti was controlled to <1 ppm. Temperature increase postsonication was not greater than 14 °C in all the samples, which was consistent with the manual method. Within the examined parameter space, we identified 2 V, 2 pulses of 30 s with a 5-s pause, and a probe set at 5 mm from the bottom of a tube as optimal to maximize dissolution capability while minimizing sonicator contamination. The sonicator probe is in contact with the analytical solution and therefore requires washing with 10mL of diluent between sample preparations. A crude sample containing approximately 1000 ppm of palladium provided no detectable carryover in the subsequent blank. The optimized conditions provided a cycle time of 8 min per sample and are listed in Table 6.
APW3 final method

Accuracy of Automated Sample Preparation
Accuracy of the automated procedure was evaluated against the results obtained for our test APIs using the established manual sample preparation procedure. Ten current APIs were tested in duplicate and recoveries were calculated based on the manual analysis representing 100% of the available metal. Data were acquired in a sequence where each sample was bracketed by blanks to ensure that no carryover was present between samples. A three-point calibration was used for this study covering the range 0–100 ppm with an uncertainty of ±0.5 ppm. These data showed recoveries for automated samples in the range of 96–106% for all the measurable metals.
Conclusions
The use of APW3 for the routine preparation of solutions for ICP analysis has been demonstrated to produce results in good agreement to those observed using manual sample preparation. Using the automated approach, every sample receives the same energy input because the sonication parameters are closely controlled and each sample is fully dissolved within the 8-min procedure. The previous manual method required a sonication time dependent on the sample and the particular bath used, which varies from minutes to hours.
The APW3 reduces the effort required in sample preparation, provides improved control over the sample solution, and maintains the quality of data generated.
Footnotes
Acknowledgments
