Abstract
Carbon nanopipettes (CNPs), nanoprobes that integrate carbon nanotubes (CNTs) into larger easily maneuverable devices, provide a viable means of performing cell surgery with carbon-based nanostructures. Taking advantage of the nanoscopic tubular geometry and unique material properties of CNTs, CNPs facilitate minimally invasive cell probing, low-volume intracellular fluid injection, sensitive electrical measurements of cell signals, and other unique analytical abilities not possible with traditional glass-based cell probing technology. In this technology review, we highlight the cell probing applications where CNPs were used as nanoneedles for intracellular injection and nanoelectrodes for cell electrophysiology. Besides summarizing the proven capabilities of CNPs, the intent of this review is to encourage further development of CNT-based devices and related nanotechnology for novel cell probing and bioanalytical applications.
Introduction
Recently, there has been a growing interest across diverse fields of science, medicine, and engineering in devices capable of probing and manipulating materials at the nanoscale. 1 For instance, in cell biology, nano-sized devices can probe cells and subcellular environments with minimal intrusion to perform nanosurgery. 2 These activities include modifying cells (via material injection and removal), sensing cell processes (with optical, biochemical, mechanical, and electrical means), and studying cell structure (by manipulation of organelles and molecules).
Glass-based nanoprobes, such as glass micropi-pettes, are currently used to probe intracellular environments. Glass capillaries are pulled into glass micropipettes with predetermined taper geometry and tip dimensions depending on their intended application. For example, glass-based injectors have short tapers with micrometer-sized tip diameters to facilitate fluid transport, whereas glass-based, electrolyte-filled electrodes have long tapers with tip diameters in the hundreds of nanometers. However, there are several issues associated with glass-based cell probes, such as having fragile tips that break easily when encountering a hard surface, being limited to single functions (e.g., injector or electrode, not both), and having relatively large tips that tend to damage cells and organelles easily. These and other issues were reviewed in more detail. 3
The advent of nanotechnology has led to the development of new tools for cell surgery, such as nanoneedles for intracellular injection of material and nanoelectrodes for studying cell signaling. Carbon-based nanostructures, such as carbon nanotubes (CNTs), are particularly useful for probing cells because they are nanoscopic in size, have hollow geometry, and have tunable surface characteristics. CNTs are slender (high length to diameter ratio) and thin walled, possess remarkable mechanical and electrical properties,4, 5 and facilitate the transmission and storage of liquids.6, 7 Several groups have used a variety of methods to attach CNTs to macroscopic devices and used them to deliver surface-bound molecules into cells.8-11 However, these probes are difficult and time-consuming to assemble, hindering their widespread application as viable cell probes. 3
The carbon nanopipette (CNP) (Fig. 1), developed by our group, eliminates many of the obstacles that have prevented CNTs from being used as cell probes. CNPs integrate a carbon nanopipe at the tip of a pulled glass capillary without the need for assembly.12, 13 CNPs can be produced in large quantities with carbon tip diameters ranging from tens to hundreds of nanometers and exposed CNT lengths ranging from a few nanometers to hundreds of micrometers. The advantage of CNPs is their ability to transport fluids and carry out electrical measurements. Because glass capillaries are used as the substrate, CNPs readily fit standard cell physiology equipment, such as micromanipulators, microinjectors, and electrophysiology amplifiers.
CNP penetrating the membrane of an oral squamous carcinoma cell.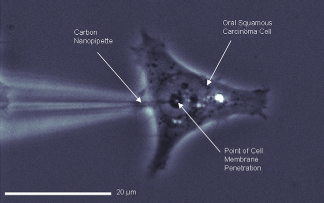
In this technology review, we summarize the development and applications of CNPs. Although CNPs have many bion-anotechnology applications, we focus on cell surgery, specifically on nanoneedles for intracellular injection and nanoelectrodes for cell electrophysiology. This review is an abridged version of our previously published Microfluidics and Nanofluidics article. 3
Fabrication
CNPs were fabricated from pulled glass capillaries without assembly, as depicted schematically in Figure 2A.
13
First, catalyst was deposited along the inner surfaces of quartz capillaries (1.0 mm OD × 0.7 mm ID × 75 cm Lg) by filling them with catalyst solution (Iron III Nitrate dissolved in iso-propanol) and allowing them to dry in air. Then, the capillaries were pulled to form fine-tipped micropipettes (Fig. 2A-i). Hundreds of micropipettes were placed together in a chemical vapor deposition (CVD) furnace (900 °C, methane/argon) where carbon was selectively deposited on the catalyzed inner surface of the pulled capillaries (Fig. 2A-ii). Finally, the tip of the carbon-coated micropipette was dipped in 5:1 buffered hydrofluoric acid (BHF) to remove the glass from the tip and expose the carbon nanopipe underneath (Fig. 2A-iii).
(A) CNP fabrication schematic showing (i) internal deposition of catalyst before micropipette pulling, (ii) deposition of carbon by CVD, and (iii) exposure of the carbon tip by wet etching. Images from ref 13. (B) Scanning electron micrograph of a carbon tip integrated into a quartz micropipette. Image from ref 14. (C) Scanning electron micrograph of several carbon tips integrated into a multibore quartz micropipette.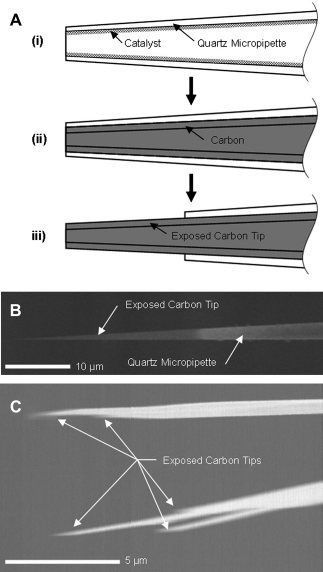
CNPs were produced with carbon tips ranging from tens to hundreds of nanometers in diameter and a few nanometers to hundreds of microns in length, as depicted in Figure 2B. The profiles and outer diameters of the carbon tips were controlled with the glass pulling parameters. The process conditions, such as CVD time and temperature and the type of precursor gas and catalyst, determined the thickness, morphology, and surface properties of the deposited carbon film.13, 15 The etching time and temperature control the length of carbon exposed. Using these methods, hundreds of CNPs of a predetermined and consistent size were fabricated in a single run. For example, 95% of the injection-style probes that underwent all the manufacturing processes had typical carbon tips with 200 ± 20 nm outer diameters, 30 ± 5 nm wall thicknesses, and 15 ± 2 μm lengths. Furthermore, the processes are amenable to scaled-up production for commercialization. Although CNPs can be manufactured using capillaries of various dimensions, quartz capillaries with 1 mm outer diameters were selected to maintain compatibility with standard cell physiology equipment.
The processes described above can also be used to produce CNPs with several independent hollow carbon conduits from capillaries with multiple bores, as depicted in Figure 2C. Optionally, conducting layers (with insulating intermediate layers) could be deposited on the outer surface of the micro-pipettes after the CVD process, but before the etching process, to form multielectrode probes (not shown). The additional electrodes could serve as counter and reference electrodes.
Properties
The carbon nanopipe tips of CNPs have similar properties 13 to carbon nanopipes produced with the alumina template method.16, 17 The carbon deposited along the inner surfaces of the quartz micropipette enables CNPs to conduct electricity from their nanoscopic tips to their larger macroscopic tails. Additionally, this hollow conducting channel enables fluid transport through the nanoscopic carbon tip. When their walls are sufficiently thin, the carbon tips of CNPs are transparent to light, electrons, and X-rays.18-20
CNPs have remarkable mechanical properties, as depicted in Figure 3. Figure 3A shows a series of images of a CNP tip (left, needle-like object) being pushed against the hard, non-deformable surface of a glass pipette (right). Once a sufficiently large axial load was applied, the carbon tip bent without breaking. The series illustrate the extent to which the carbon tip severely kinked several times, yet still returned to its original shape when backed away to remove the axial load. In contrast, the tip of a glass micropipette undergoing similar manipulation would shatter.
(A) From top to bottom: the carbon pipe tip of the CNP buckles when pushed against the wall of a glass pipette and recovers its initial shape once the force is removed. (B) From top to bottom: a CNP penetrates through the membrane of a smooth muscle cell. The cell is held in place by glass micropipette aspiration. Images from ref 13.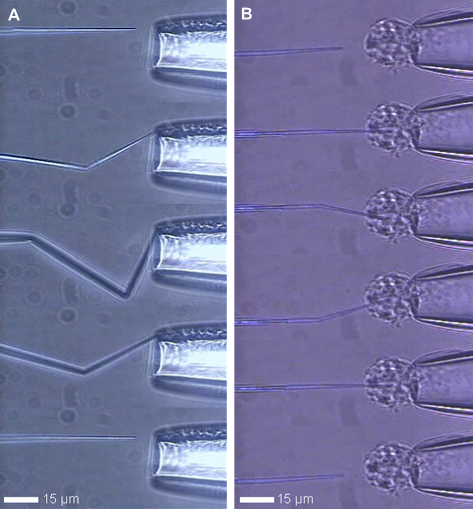
Although the CNP tips were flexible, they were sufficiently rigid to spear the membranes of cells. Figure 3B shows a series of images of the CNP tip (left) being pushed into a smooth muscle cell (∼15 μm outer diameter) held in place at the tip of a large glass pipette (right). Because of its nano-scopic size, the tip of the CNP easily penetrated the cell membrane. The series of images show that while the tip was still impaling the cell, the CNP could be maneuvered in such a way that it bent without breaking.
Nanoneedles for Cell Probing and Intracellular Injection
Recently, several studies have investigated the effects of CNTs on living cells.21-23 For example, Poland et al. 22 showed that CNTs could cause an asbestos-like inflammatory response in cells when the tubes are too large for the cell phagocytize. Liu et al. 23 reported nontoxic effects of intravenously injected CNTs in animal models. Other studies describing conditions where CNTs have toxic and nontoxic effects on cells have been reviewed. 21
In these and other in vitro and in vivo studies, cells are exposed to and remain in contact with CNTs for prolonged periods of time. In our work, however, the CNT at the tip of the CNP is removed from the cell after an experiment, thus avoiding any potential toxic effects caused by long-term expose to foreign material. For instance, CNPs can probe cells with minimal intrusion and inject fluids without causing any harm to cells. 13 In a set of experiments, not shown here, oral cancer cells were first probed with CNPs and then monitored for more than 2 weeks. The cells remained vital and continued to grow and proliferate after CNP probing, suggesting that the carbon material of CNPs was not toxic to cells. It was demonstrated that CNPs could inject fluorescent dye into oral cancer cells and neuron cells without causing cell death.
In another study (Fig. 4), CNPs were used to inject calcium-mobilizing secondary messengers into the SKBR3 breast cancer cell line to identify intracellular calcium signaling pathways.
14
Figure 4A-i shows a CNP, filled with inositol trisphosphate (IP3) and positioned tens of microns above a breast cancer cell. IP3 is a secondary messenger that gates IP3 receptors and mediates calcium release from the endoplasmic reticulum. This is an important and well-studied intracellular signaling mechanism in many cell types. Figure 4A-ii and -iii show, respectively, the intracellular calcium levels (as observed with fluorescent dye) immediately before and after the CNP injected IP3 into the cell on the right. A cell directly injected with the messenger released calcium (Fig. 4A-iii, white color), whereas cells that were not injected with IP3 (left cell) remained at basal calcium levels (Fig. 4A-ii and -iii, blue—green color).
(A-i) Bright field image of an IP3-filled CNP (dark feature above right cell) positioned tens of micrometers above a breast cancer cell before injection. (A-ii and A-iii) Images of intracellular Fura-2 fluorescence ratios 2 s before (A-ii) and I s after (A-iii) injection of IP3 (only the right cell is injected). (B) Time-course of changes in intracellular calcium concentration upon injection (arrow) of cADPr using CNPs and glass micropipettes. In some experiments, cells were pretreated with Ry (pink) or BAF (cyan). All results are means ± standard error of the mean of six cells. Adapted from ref
14
.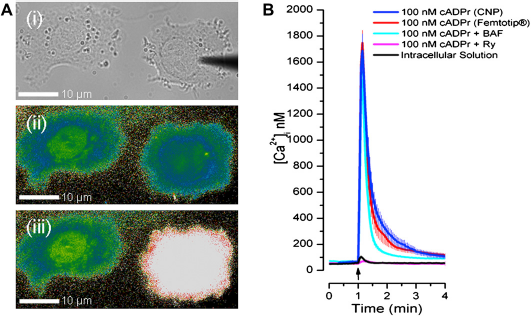
Using the above technique, cytosolic calcium levels were monitored over time in response to injections of the well-studied messenger, IP3, and the lesser-studied messengers, cyclic adenosine diphosphate ribose (cADPr) and nicotinic acid adenine dinucleotide phosphate (NAADP). As shown in Figure 4B, the injection of cADPr with CNPs (blue trace) evoked intracellular calcium release in breast cancer cells. The cADPr-mediated calcium release was transient and reproducible with high fidelity. In contrast, as shown in Figure 4B (black trace), basal intracellular calcium concentration was not significantly affected by penetration of the cell membrane with the CNP or the injection of neutral intracellular solution. These results and the transient nature of the calcium release upon CNP-mediated messenger injection indicate that the use of CNPs does not compromise cell viability or unexpectedly alter basal cytosolic calcium. The similarity between calcium signals elicited through injection with CNPs and commercially available glass injectors further signify that CNPs do not alter cells in any significant way.
To identify the mechanisms of messenger-evoked calcium release, cells were pretreated either with the ryanodine receptor antagonist, ryanodine (Ry, 10 μM for 15 min), or with the V-type ATPase inhibitor, bafilomycin-Al (BAF, 1 μM for 60 min). As shown in Figure 4B (cyan trace), calcium release was uninhibited when cADPr was injected into cells pretreated with BAF. In contrast (Fig. 4B, pink trace), calcium release was inhibited when cADPr was injected into cells pretreated with Ry. These findings indicate that the endoplasmic reticulum, and not the lysosomes, was solely responsible for cADPr-mediated calcium signaling in SKBR3 via the ryanodine receptor. In contrast, the same experiments carried out with NAADP (not shown) indicated that, in breast cancer cells, the lysosomes, with ryanodine-dependent participation of the endoplasmic reticulum, were responsible for the NAADP-mediated calcium signaling.
During our experiments, we observed significant advantages of CNPs for intracellular injection over glass pipettes. For example, as shown in Figure 3, the carbon tips of the CNPs were more likely to bend or buckle instead of shatter during cell probing. This structural behavior resulted in fewer broken CNP tips during normal experimental use (e.g., mishandling of the probes or accidentally manipulating the tip into coverslips) as compared with glass micropipettes and increased the overall experimental efficiency. Additionally, despite their smaller tip diameters, because of the carbon inertness, the CNPs were less prone to clogging than their glass counterparts. The dark color of carbon made the tips of CNPs more readily visible under light microscopy, which facilitated significant time-savings and more efficient injection. Overall, compared with glass injectors, CNPs injected four times as many cells and damaged half as many during injection.
Nanoelectrodes for Cell Electrophysiology
Because the CNPs are conducting, they can be used as nanoelectrodes for various electrochemical applications. Here, we describe the use of CNPs in cell electrophysiology. CNPs have been used to measure the electrical responses of the HT-22 mouse hippocampal cell line to chemical and pharmacological stimuli.
24
In this study, the CNP was affixed to an electrophysiology amplifier to assess the intracellular environment when the CNP penetrates the cell membrane with its conductive tip. As depicted in Figure 5A, when the CNP with a short carbon tip penetrated through the membrane of the cell, the lipid bilayer engulfed the quartz capillary to form a hermetic seal. In this configuration, the CNP measured the resting membrane potentials of cells and any variations in the cell potential resulting from external stimuli.
(A) Cross-section schematic of a CNP and of the interface between the CNP and the cell membrane. (B) CNP-recorded variations in membrane potential during the administration of GABA (12 mM, arrow) to HT-22 cells perfused in normal HBSS (black triangles), HBSS and 10 μM bicuculline (BIC) (red circles), and HBSS and 20 μM BIC (blue squares). Images from ref 24.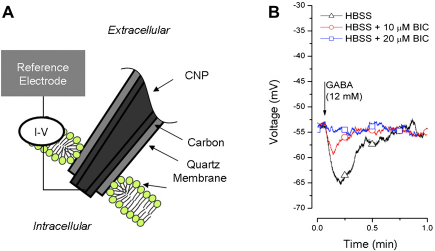
When left inside the cell for a prolonged period of time and in the absence of environmental perturbations, the CNPs recorded a reasonably stable membrane potential. When the CNP tip was brought into close proximity to the cell membrane, a small increase in potential was observed, followed by a large decrease in potential as the CNP penetrated into the cell. These potential variations associated with cell penetration potentially provide a means to sense cell penetration and automate the cell injection process. The CNPs recorded variations in the cell resting membrane potentials when the extracellular solution was enriched with potassium ions (not shown). The measured values matched well with predictions obtained with the Nernst equation.
The CNPs were also used to record changes in a cell's resting membrane potential because of extracellular administration of a pharmacological stimulant, γ-aminobutyric acid (GABA). GABA causes HT-22 cells to hyperpolarize by binding to GABA-A receptors and opening the ligand-gated Cl− ion channels. Figure 5B depicts the cell membrane potential as a function of time under various extracellular conditions. When the extracellular solution was not subjected to any perturbations, the CNP recorded a stable resting membrane potential. On the administration of GABA to the extracellular solution, the cell hyperpolarized (Fig. 5B, triangles). Once the stimulus was removed, the cell potential returned to its unperturbed value. The GABA-induced hy-perpolarization was forestalled by introducing bicuculline, a competitive antagonist for the GABA-A receptor (Fig. 5B, circles and squares).
Summary and Future Potential Applications
The major challenge that prevented the use of CNTs as cell probes was their difficult assembly. Fortunately, the integrated fabrication of CNPs eliminates the need for assembly and provides an easy way to interface between nanoscopic structures and macroscopic “handles.” The CNPs have a significant potential for cell nanosurgery applications, such as cell probing, intracellular injection, and cell electrophysiology. They have many advantages over the pulled glass capillaries, such as smaller size, robustness, and resistance to clogging. Furthermore, because, unlike glass pipettes, the electrically conductive path of CNPs is separate from the hydraulic path, the CNPs offer multifunctionality. Cell penetration can be sensed electrically to facilitate automated cell injection. Moreover, injection and electrical measurements can be conducted concurrently.
The carbon surface of CNPs can be functionalized with proteins, oligonucleotides, and particles, 15 offering a means for biosensing and detection of intracellular analytes. The multifunctionality of CNPs provides a means for biosensing concurrently with injection and electrical measurements. Alternatively, functionalized beads can be captured at the tips of CNPs for electrochemical detection in biological samples. 25
The CNPs has proven to be useful for other applications besides cell probing, such as sample holders for X-ray and electron microscopy. 26 The hollow conductive structure of CNPs can serve as nozzles, allowing one to print drops and lines with characteristic dimensions ranging from tens to hundreds of nanometers. 27 Additionally, CNPs may be used as manipulators to position magnetic beads or particles at desired locations. 28
Footnotes
Acknowledgments
This work was supported in part by the Nanotechnology Institute, Ben Franklin Technology Partners of Southeastern Pennsylvania, and the National Science Foundation's Nanoscale Interdisciplinary Research Team (NSF-NIRT) (CBET 0609062). This review is an abbreviated and updated version of our Microfluidics and Nanofluidics article. 3
Competing Interests Statement: The authors certify that all financial and material support for this research and work are clearly identified in the manuscript.
